Introduction
Materials and Methods
Plant Materials and Growth Environment
Pathogen Preparation
Disease Scoring
Data Analysis
Results and Discussion
Screening of Pepper Cultivars for Resistance to Two Isolates of P. capsici
Evaluation of Pepper Cultivars for Resistance to Phytophthora Root Rot and Foliar Blight
Introduction
Hot peppers (Capsicum spp.) of the nightshade family Solanaceae are economically important and widely cultivated horticultural vegetable crops. Hot peppers are characterized by their production of capsaicinoids, the compounds responsible for the hot pungent taste of peppers. Hot peppers are also rich in phenolic-derived compounds with unique physiological and pharmacological properties, some of which provide human health benefits (Howard and Wildman, 2007; Loizzo et al., 2015). According to the Food and Agriculture Organization of the United Nations, 40 million tons of fresh and dried hot pepper were produced on –3.76 Mha worldwide in 2018, which consistently increased over the last decade (FAOSTAT, 2018).
Phytophthora capsici (Leonian) is a soil-borne oomycete plant pathogen that is the most devastating pathogen affecting hot pepper production. P. capsici has a wide host range of more than 15 plant families, including Solanaceae, Fabaceae, and Cucurbitaceae, and can cause severe diseases such as foliar blight, crown blight, root rot, and fruit rot (Lamour et al., 2012; Yeom et al., 2012; Barchenger et al., 2018; Kim et al., 2019b). Significant yield losses due to P. capsici infection in open production and greenhouse cultivation have been reported in many countries, accounting for a worldwide economic loss of over one billion dollars per year (Hausbeck and Lamour, 2004). In the sequential production systems commonly used for pepper cultivation, soil-borne diseases such as Phytophthora blight often become a recurring issue, consistently impacting the yield and quality of the produce.
P. capsici can survive in the soil and host plant debris, spreading rapidly by wind-blown rain through the air and water movement in the soil under warm (25°C–28°C) and wet conditions. Thus, when P. capsici infection occurs in the field, it is often difficult to control (Lamour et al., 2012; Barchenger et al., 2018). Chemical control and cultural practices are used to manage P. capsici; however, these management practices are not completely effective. To date, the development of a disease-resistant cultivar is one of the best approaches to control P. capsici infection, but the availability of resistant breeding resources for disease management programs are limited in the pepper industry (Hausbeck and Lamour, 2004; Foster and Hausbeck, 2010; Granke et al., 2012).
Plants have developed a range of defense mechanisms to combat diverse environmental conditions, which provide tolerance and/or resistance to abiotic and biotic stresses (Lee and Yeom, 2015; Kang and Yeom, 2018; Kang et al., 2020; Lee et al., 2020). In peppers, the expression of resistance against P. capsici is determined by variable environmental factors including the variety, growth stage, temperature, inoculation method, and isolate. The development of functional gene-based markers and characterization of the genes that confer resistance against P. capsici are essential to facilitate efforts to breed disease-resistant pepper cultivars (Kim et al., 2017a, 2017b; Chunthawodtiporn et al., 2018; Kim et al., 2019b). Numerous molecular markers have been identified within a major quantitative trait locus (QTL) region; however, little progress has been made in the development of functional gene-based markers of P. capsici resistance, and no P. capsici resistance genes (R gene) have been characterized. In addition, the plant host defense responses to different strains of P. capsici are not well understood (Glosier et al., 2007). It appears that foliar blight and root rot are different P. capsici disease symptoms that should be studied independently (Foster and Hausbeck, 2010). Furthermore, resistance to Phytophthora-induced diseases such as root rot and foliar blight showed different patterns of complex inheritance (Reifschneider et al., 1992; Oelke et al., 2003; Thabuis et al., 2004; Ogundiwin et al., 2005; Yeom et al., 2011; Kim et al., 2019a, 2019b).
In this study, we conducted a thorough screening to identify sources of P. capsici disease resistance in Capsicum spp. that could be incorporated into new commercial pepper cultivars. The severity of root rot caused by different P. capsici isolates and correlations between root rot and foliar blight were evaluated in several Capsicum accessions. Further genetic studies to verify our results are warranted, with the overall aim to provide information on the important characteristics of pepper germplasm by revisiting classical genetic studies and demonstrating the contribution of genomics to the understanding of the molecular basis of resistance.
Materials and Methods
Plant Materials and Growth Environment
Thirty pepper accessions consisting of six landrace and 24 commercial hybrid cultivars were screened for resistance to Phytophthora diseases. C. annuum “CM334” with known resistance to P. capsici and C. annuum “ECW30R” with known susceptibility to P. capsici were used as negative and positive controls for disease development, respectively. Pepper seeds were sown in a Petri dish lined with a wet tissue layer for 2 weeks. After germination, the seedlings were transplanted into a 32-cell plug seedling tray in a plant growth chamber at 25°C with 16 h of light per day.
Pathogen Preparation
Two pathogen accessions (P. capsici Leon “KACC 40470” and “KACC 40476”) were obtained from the Korea Agricultural Culture Collection (KACC) of Rural Development Administrations (Table 1). P. capsici inoculum preparation was described previously (Yeom et al., 2011). P. capsici was grown in potato dextrose agar medium in the dark for 7 d at 28°C, and then 8-mm diameter mycelial plugs were cut and cultured on V8 juice agar media (20% V8 juice, 0.3% CaCO3, and 1.8% agar) for 5 d. Next, the mycelia were scraped and incubated under light for 2 d to promote sporangium formation. Chilling stress was induced by flooding the plate with sterile water and incubating at 4°C for 90 min; then, plates were incubated at 28°C for 30 min to stimulate the release of zoospores. The zoospores were counted using a hemocytometer, and the concentration was adjusted to 1 × 105 zoospores per ml with distilled water. Seedlings at the four-true-leaf stage were inoculated by root drenching with P. capsici “KACC 40470” (hereafter, KACC 40470) and “KACC 40476” (hereafter, KACC 40476) at a density of 2.0 × 105 zoospores per plant. To induce Phytophthora foliar blight, P. capsici “KACC 40476” suspension concentrated at 1.0 × 105 zoospores per ml was infiltrated on the marked leaf region on the same four-true-leaf stage plant. For spray inoculation, 500 µl of a zoospore suspension ofKACC 40476 (1.0 × 105 zoospores per ml) was sprayed on each pepper leaf (Oh et al., 2010). For dropping inoculation, a 0.5-cm-diameter paper disc of Whatman filter paper (Sigma-Aldrich, St Louis, MO) was placed on the surface of each leaf, and then 20 µl (approximately 2000 zoospores) of KACC 40476 inoculum was dropped on each paper disc (Monroy-Barbosa and Bosland, 2010). Next, we prepared additional inoculum for plug inoculation of P. capsici (Bouwmeester and Govers, 2009). A mycelial plug (1 cm diameter) of KACC 40476 was placed on the abaxial side of leaf. Growth conditions were maintained at 25°C under a 16:8-h light/dark cycle.
Table 1.
Characteristics of the Phytophthora capsici strains used in this study
| KACCz No. | Location of isolation | Source | Pathogenicity | |
| Reportedy | Tested in this study (root rot) | |||
| 40470 | Buyeo, Chungnam, Korea | Solanum lycopersicum | Unknown | Strong Pathogenicity |
| 40476 | Goesan, Korea | Capsicum annuum | Strong Pathogenicity | Strong Pathogenicity |
Disease Scoring
Evaluations of foliar blight and root rot were performed when 80% of the “ECW30R” positive control plants were diseased, which occurred 7 days after inoculation (DAI). A disease score for foliar blight was modified based on previous research (Monroy-Barbosa and Bosland, 2010; Jo et al., 2014). Foliar blight assessment was performed using a 0–3 scale in which 0 = no disease symptoms observed or small necrosis tissue; 1 = water-soaked lesion is less than 30% of leaf area; 2 = 30% to 60% of leaf area is wilted (scalded or necrotic); 3 = 60% or more of the leaf is wilted or the leaf has dropped off from the plant (Fig. 1A). Wilt induced by P. capsici was assessed using the following root rot severity scale. The plants were scored from 0–3 based on the proportion of defoliation symptoms observed on the entire plant following a previous study (Jo et al., 2014), in which 0 = no symptoms; 1 = stem lesion, defoliated first and second leaves, or less than 30% of entire plant diseased; 2 = stem lesion extending to second leaves, yellowing or defoliation of some upper leaves, or 30% to 60% of entire plant diseased; 3 = all leaves except uppermost leaf defoliated, more than 60% of entire plant diseased, or the plant was dead (Fig. 1B).

Fig. 1.
The severity of Phytophthora blight on pepper plants rated using a scale of 0 to 3. (A) Foliar blight assessment of pepper plants. Evaluations were performed 6 days after inoculation (DAI): 0 = no symptoms or hypersensitive response, 1 = water-soaked lesion is less than 30% of leaf area, 2 = 30% to 60% of leaf area is wilted (scalded or necrotic), 3 = 60% or more of the leaf is wilted or the leaf has dropped off from the plant. (B) Root rot scale of pepper plants. Scoring was established 6 DAI using a scale of 0 to 3: 0 = no symptoms, 1 = stem lesion defoliated first and second leaves, or less than 30 % of entire plant diseased, 2 = stem lesion extending to second leaves, yellowing or defoliation of some upper leaves, or 30%–60% of entire plant diseased, and 3 = all leaves except uppermost leaf defoliated, more than 60% of entire plant diseased, or plant was dead.
The lines were classified as resistant or susceptible based on the average severity of each line. The root drenching inoculation assay was measured by the average degree on 16–20 plants per cultivar; the leaf injection assay was measured by the average degree on 8–10 plants per cultivar. The trait classifications for peppers were evaluated as follows: 0 to less than 1 = resistance; 1 to less than 2 = moderate resistance; 2 to 3 = susceptible (Monroy-Barbosa and Bosland, 2010; Jo et al., 2014).
Data Analysis
The statistical analyses were performed using SAS software (version 9.4; SAS Inc., Cary, NC). The differences among average values of disease scores on the pepper cultivars were analyzed using Duncan’s multiple range tests. There were no significant (p < 0.05) interactions between cultivar and biological replicate. Means across two biological replicates are reported.
Results and Discussion
Screening of Pepper Cultivars for Resistance to Two Isolates of P. capsici
In this study, 30 chili pepper accessions were tested for resistance and susceptibility to Phytophthora blight caused by P. capsici, including four C. annuum species, one C. chinense species, one C. baccatum species, and 24 Korean commercial pepper cultivars. Among the 30 accessions of pepper germplasm, the resistance level against Phytophthora blight of 23 accessions was previously characterized (Jo et al., 2014), and the remaining 7 accessions had not been previously scored.
To determine disease resistance to P. capsici isolates by root inoculation, we investigated the severity of disease caused by root drenching with KACC 40470 and KACC 40476 in 30 Capsicum germplasms (Table 2). C. annuum “CM334” is reported to have the highest resistance level against multiple strains of P. capsici (Yeom et al., 2011; Kim et al., 2019b); thus, it was used as a negative control in this study. C. annuum “CM334” was confirmed to be resistant against both strains of P. capsici, with an average root rot severity score of 0. The positive control C. annuum “ECW30R” was confirmed to be susceptible to P. capsici, with average root rot severity scores of 2.8 and 3.0 when inoculated with KACC 40470 and KACC 40476, respectively.
Table 2.
The resistance response of 30 Capsicum pepper varieties to root rot disease after inoculation with two isolates of Phytophthora capsici
| No. | Pepper accession | “KACC 40470” | “KACC 40476” | Known Traitz | ||||
| P1 | C. annuum “CM334” (NC)y | 0 | ± 0.00x | Rw | 0 | ± 0.00 | R | R |
| P2 | C. annuum “ECW30R” (PC)v | 2.8 | ± 0.11 | S | 3 | ± 0.00 | S | S |
| P3 | C. annuum “PR-Manidda” | 0.5 | ± 0.14 | R | 0 | ± 0.00 | R | MR/R |
| P4 | C. annuum “Dokyacheongcheong” | 0.2 | ± 0.09 | R | 0 | ± 0.00 | R | S/MR/R |
| P5 | C. annuum “Muhanjilju” | 0.1 | ± 0.07 | R | 0 | ± 0.00 | R | R |
| P6 | C. annuum “Ilsongjung” | 0.3 | ± 0.21 | R | 0 | ± 0.00 | R | MR/R |
| P7 | C. annuum “PR-Kstar” | 0 | ± 0.00 | R | 0 | ± 0.00 | R | R |
| P8 | C. annuum “PR-Shindaejang” | 0.2 | ± 0.27 | R | 0 | ± 0.00 | R | R |
| P9 | C. annuum “Asia-jumbo” | 0 | ± 0.00 | R | 0.1 | ± 0.08 | R | R |
| P10 | C. annuum “Dokjuyeokgang” | 2.3 | ± 0.61 | S | 0.6 | ± 0.60 | R | R |
| P11 | C. annuum “PR-Gugupalpal” | 0.8 | ± 1.06 | R | 0 | ± 0.00 | R | R |
| P12 | C. annuum “PR-Oganmanjok” | 0.8 | ± 0.07 | R | 0 | ± 0.00 | R | R |
| P13 | C. annuum “PR-Jeonguktongil” | 0.9 | ± 0.08 | R | 0 | ± 0.00 | R | R |
| P14 | C. annuum “PR-Daechon” | 0.2 | ± 0.14 | R | 0.1 | ± 0.07 | R | S/MR/R |
| P15 | C. annuum “Yeokganghongjanggun” | 0.4 | ± 0.05 | R | 0 | ± 0.00 | R | MR/R |
| P16 | C. annuum “PR-Mujeok” | 0.7 | ± 0.25 | R | 0.1 | ± 0.07 | R | MR |
| P17 | C. annuum “Yeokgangsumunjang” | 0 | ± 0.00 | R | 0 | ± 0.00 | R | MR |
| P18 | C. baccatum “PBC81” | 2.9 | ± 0.18 | S | 2.2 | ± 0.08 | S | S |
| P19 | C. chinense “PI159236” | 1.7 | ± 0.41 | MR | 1.7 | ± 0.52 | MR | S |
| P20 | C. annuum “Daedulbo” | 2.9 | ± 0.16 | S | 1 | ± 1.40 | MR | S |
| P21 | C. annuum “Bukang” | 3 | ± 0.06 | S | 2.4 | ± 0.28 | S | S |
| P22 | C. annuum “Supermanidda” | 2.5 | ± 0.30 | S | 0.8 | ± 0.14 | R | S |
| P23 | C. annuum “Nokgwang” | 2.5 | ± 0.48 | S | 2.7 | ± 0.21 | S | S |
| P24 | C. annuum “Tanegang” | 0.8 | ± 0.53 | R | 0 | ± 0.00 | R | NDu |
| P25 | C. annuum “AT-Shinhotan” | 1.7 | ± 0.08 | MR | 0.2 | ± 0.35 | R | ND |
| P26 | C. annuum “Jeoktoma-plus” | 1.4 | ± 0.11 | MR | 0.2 | ± 0.31 | R | ND |
| P27 | C. annuum “Mawuntan” | 2.7 | ± 0.21 | S | 1.5 | ± 0.64 | MR | ND |
| P28 | C. annuum “Meojjinsanaie” | 0.3 | ± 0.44 | R | 0 | ± 0.00 | R | ND |
| P29 | C. annuum “Subicho” | 2.9 | ± 0.14 | S | 2.6 | ± 0.64 | S | ND |
| P30 | C. annuum “MC4” | 0 | ± 0.00 | R | 0 | ± 0.06 | R | ND |
The results of the inoculation with KACC 40470 revealed that 18 cultivars (60%), including “CM334”, had a mean root rot severity score of 0 to 1 and were considered resistant, while 9 cultivars (30%) including “ECW30R” had an average score between 2 and 3 and were considered susceptible. The results of the root inoculation with KACC 40476 revealed that 22 (73%) pepper cultivars showed resistance and 5 (16.7 %) showed susceptibility. Although we did not measure the severity of pathogenicity for KACC 40470, the root inoculation results showed that comparatively, the pathogenicity of KACC 40470 was slightly higher than that of KACC 40476. Based on this result, we assumed that both KACC40470 and 40476 would have strong pathogenicity (Table 1). Comparing the root rot results between the two strains, 6 out of 30 pepper accessions showed different resistance levels to the two P. capsici isolates. Four pepper cultivars, “Dokjuyeokgang”, “Daedulbo”, “Supermanidda”, and “Mawuntan”, were scored as susceptible to KACC 40470 but showed resistance against KACC 40476. Two cultivars, “AT-Shinhotan” and “Jeoktoma-plus”, showed moderate resistance against KACC 40470 and resistance against KACC 40476 (Table 2). According to previous studies, there was a large difference in pathogenicity between P. capsici among isolates, and P. capsici isolates showed varying levels of virulence to pepper cultivars (Silvar et al., 2006; Candole et al., 2010; Jo et al., 2014). Consistent with previous studies, our results revealed that these two P. capsici isolates showed different pathogenicity against pepper cultivars. Eighteen cultivars including “CM334” were consistently resistant to both KACC 40470 and 40476. We also compared the root rot results in this study with those of previous studies (Table 2); differences in disease severity to P. capsici strains were detected in “PR-Manidda”, “Dokyacheongcheong”, “Ilsongjung”, “PR-Daechon”, and “Yeokganghongjanggun” cultivars. The majority of P. capsici-resistant pepper cultivars were derived from “CM334” by breeding programs in Korea. However, the P. capsici resistance in “CM334” is controlled by a QTL, and no pepper cultivars have shown complete resistance to all strains of P. capsici. These findings may provide relevant information about the differences in pepper resistance related to the pathogenicity of P. capsici, depending on the number of superior resistance genes introduced through the breeding of diverse pepper landraces (Foster and Hausbeck, 2010; Jo et al., 2014).
Evaluation of Pepper Cultivars for Resistance to Phytophthora Root Rot and Foliar Blight
To investigate the resistance response of pepper to root rot and foliar blight caused by P. capsici, we infiltrated KACC 40476 onto the 3rd and 4th true leaves of pepper plants. Disease resistance levels of the inoculated leaves of 30 pepper cultivars were observed 6 DAI (Fig. 2). Assessment of foliar blight severity was performed based on scores from 0 to 3, with 0 indicating no symptoms and 3 indicating the most severe symptoms. The pepper cultivars showed significant differences in resistance responses to foliar blight and root rot. Among the 30 pepper cultivars, “CM334” and “ECW30R” were consistently resistant and susceptible to both root inoculation and leaf inoculation, respectively. Only five cultivars (16.7%) [P1 (CM334), P6 (Ilsongjung), P26 (Jeoktoma-plus), P14 (PR-Daechon), and P9 (Asia-jumbo)] were resistant to foliar blight, with scores between 0 and 1. Seventeen cultivars (56.7%) were deemed susceptible, with scores ranging from 2 to 3. Even though the leaf infiltration method by syringe is a commonly used method for plant pathogen inoculation (Kwon et al., 2021; Kang et al., 2022), this method could affect artificially effects during infection of zoospore compared to the root drench method. Therefore, to determine the effect of artificial inoculation by syringe on the phenotypic response of pepper against P. capsici, we used three different leaf inoculation methods, including leaf spray, dropping, and plug inoculation. These methods have been used to inoculate leaves of P. capsici in previous studies (Bouwmeester and Govers, 2009; Monroy-Barbosa and Bosland, 2010; Oh et al., 2010). We tested five cultivars; three cultivars [P11 (PR-Gugupalpal), P16 (PR-Mujeok), and P30 (MC4)] showed large differences in disease severity between root rot and foliar blight, and two cultivars were used as positive (P2) and negative control (P1). The phenotypic results of three leaf inoculation methods were consistent with leaf infiltration results, which showed that the responses of three tested cultivars (P11, P16, and P30) were susceptible to KACC 40476 with average disease scores of 3.0 displaying wilted leaves (Suppl. Fig. 1 and Suppl. Table 1). These results suggest that the leaf infiltration method could provide reliable results for screening of foliar blight resistance.
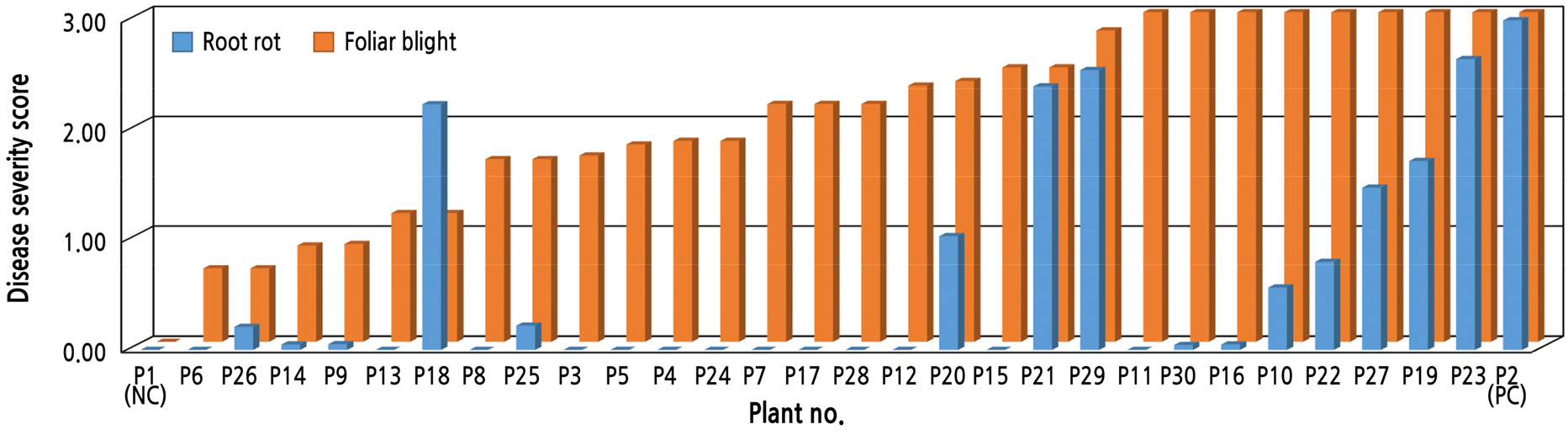
Fig. 2.
Phytophthora capsici “KACC 40476”-induced root rot and foliar blight severity derived from the screening of 30 Capsicum spp. accessions. The values are means of two biological replicates with 16 plants each. Disease severity of the seedlings was rated on a scale of 0 to 3 at 6 days after inoculation: 0 to less than 1 = resistance; 1 to less than 2 = moderate resistance; 2 to 3 = susceptibility. On the x-axis, each pepper cultivar is numbered as shown in Table 2. The disease severity score is presented on the y-axis. PC, positive control; NC, negative control.
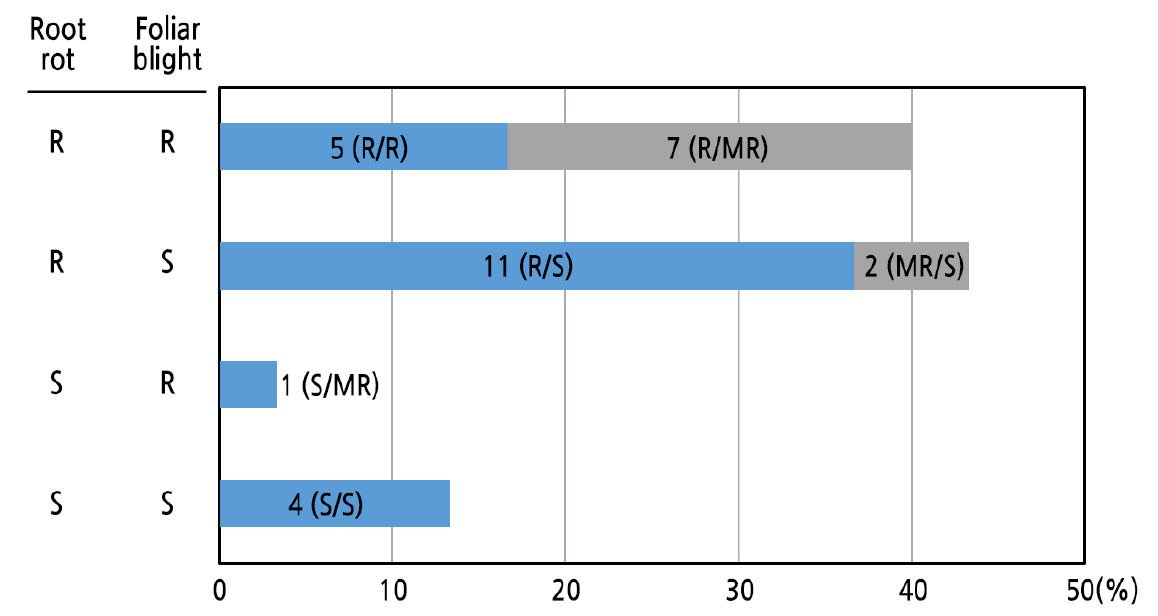
To examine the relationship between root rot resistance and foliar blight resistance, we compared the results of the root drenching inoculation and leaf infiltration of KACC 40476 (Fig. 3) and found that the resistance responses to Phytophthora root rot and Phytophthora foliar blight were inconsistent among Capsicum accessions. Twelve accessions (40%) showed resistant phenotypes (resistance or moderated resistance) to both root rot and foliar blight. Four accessions [P2 (ECW30R), P21 (Bukang), P23 (Nokgwang), and P29 (Subicho)] showed susceptibility to both inoculation methods (Figs. 2 and 3). The other 14 pepper accessions (46.7%) exhibited different disease resistance responses depending on the inoculation site (Fig. 3). Among these 14 cultivars, most (13 accessions) showed resistance or moderate resistance to root rot and susceptibility to foliar blight. Only one pepper accession (P18; C. baccatum “PBC81”) was susceptible to root rot and moderately resistant to foliar blight (Figs. 2 and 3). When comparing root rot response and foliar blight response, 66% of pepper accessions showed lower levels of resistance to leaf inoculation than root inoculation (i.e., R/MR, R/S, or MR/S). The major QTL that contributes to P. capsici resistance is identically expressed among plant organs, but resistance levels could differ depending on the presence or absence of minor QTL introduction (Liu et al., 2014; Rehrig et al., 2014; Kim et al., 2019b).
In this study, the severity of Phytophthora-induced diseases from P. capsici isolates KACC 40470 and KACC 40476 was determined in 30 Capsicum accessions. In addition, we compared the defense responses to root rot and foliar blight and found that some accessions showed different resistance responses depending on the inoculation method. Our findings suggest that the resistance to blight in peppers differs according to the isolate of P. capsici and the inoculation site, even within the same pepper cultivar. Therefore, pepper resistance is related to the pathogenicity of P. capsici isolates as well as the number of resistance genes introduced from resistant pepper germplasms during the breeding process. The major resistance genes could be identically expressed in all organs; however, resistance levels were different depending on the presence or absence of minor QTL in pepper accessions. The findings of this study contribute to our understanding of the molecular basis of resistance to P. capsici in peppers and can be applied to improve breeding methods.
Supplementary Material
Supplementary materials are available at Horticultural Science and Technology website (https://www.hst-j.org).
- HORT_20220009_Table_1s.pdf
The responses of pepper varieties by inoculation methods of KACC 40476
- HORT_20220009_Figure_1s.pdf
Confirmation of the phenotype through leaf-inoculation methods against KACC 40476. (A) Foliar blight symptoms of 5 pepper accessions through spray, dropping, and plug inoculation methods at 4 days after inoculation (DAI). The white scale bar in photo indicates 2 cm. (B) Foliar blight assessment of pepper plants at 4 DAI. NC: negative control; PC: positive control.