Introduction
Materials and Methods
Materials
Preparation of the seed MC gradient
Germination test
Determination of physiological and biochemical indices and analysis of the antioxidant system
Determination of proteins, seed-protective, and toxic compound levels
Data analyses
Results
Effects of the seed moisture content and seed storage duration under different temperatures on the seed germination parameters
Effects of the seed moisture content and seed storage duration under different temperatures on the seed antioxidant enzyme contents
Effects of the drying treatment on the protein content of A. mongolicum seeds at different storage temperatures
Effects of the drying treatment and storage temperature on protective and osmoregulatory compounds and superoxide (O2‐) levels in A. mongolicum seeds
Analysis of the correlations between the various indices of A. mongolicum seeds after drying at different temperatures
Discussion
Conclusions
Introduction
Allium mongolicum Regel is a perennial xerophytic plant in the family Liliaceae, primarily distributed in the arid regions of the Mongolian Plateau (Song et al. 2023). Due to its unique habitat, A. mongolicum has a well-developed root system, making it an excellent sand-fixing species whose structure is adapted to harsh climatic conditions. It is also a resource plant of high research value (Jin et al. 2022). In recent years, however, severe soil desertification and the overexploitation of wild A. mongolicum have led to a drastic population decline, seriously impeding its conservation. Therefore, effective strategies for its preservation are urgently needed to prevent extinction and safeguard this germplasm resource (Jin et al. 2022).
The seed moisture content (MC) and storage temperature are considered the main factors in maintaining the viability of seeds (Endoh et al. 2021). Currently, the International Commission on Plant Genetic Resources (IPGR) recommends a seed-storage MC of 5 ± 1% and temperature of –18°C. However, because seed types and habitats vary widely, some seeds are desiccation-sensitive while others are desiccation-tolerant. Accordingly, storage methods must be tailored: desiccation-sensitive seeds are commonly preserved at low temperatures, whereas desiccation-tolerant seeds typically undergo an ultra-dry treatment (Telewski and Jan Rijn 2002; Visscher et al. 2016).
Dehydrating certain seeds to moisture contents below 5% at room temperature may have more pronounced effects on germination and biochemical activity than similar treatments at lower temperatures (Buitink and Olivier 2004). Consequently, efforts have been made to optimize storage methods for A. mongolicum seeds to enhance germination, protect germplasm resources, and facilitate seedling management. Dry storage involves reducing the seed MC to < 5% using desiccants or other physical methods to extend the shelf life at room temperature, offering an economical, convenient, and effective approach to maintain genetic stability (Xie et al. 2021). This technique relies on drying or cooling to increase the intracellular viscosity, slowing chemical and physical reactions to shift the system from a fluid–liquid state—where intermolecular interactions are transient and molecules move freely—to a viscoelastic–solid state—where structural formation and molecular migration are restricted (Walters et al. 2010). Dry storage reduces the water content and converts any remaining water into a glassy matrix, resulting in limited reductions in respiratory metabolism and suppressed O2 consumption, thereby reducing susceptibility to free radical attacks. Moreover, drying treatments preserve the activities of superoxide dismutase (SOD), catalase (CAT) and other free-radical-scavenging enzymes, which are rapidly activated upon water imbibition during germination to inhibit lipid peroxidation and preserve membrane integrity (Ballesteros and Walters 2011). Consequently, many species’ seeds are currently preserved via dry storage. However, excessive drying can disrupt the continuous water–membrane interface at cell membranes, exposing macromolecules at vulnerable sites to reactive oxygen species (ROS), promoting lipid peroxidation, and ultimately leading to cell senescence and death (Zhang et al. 2018). Therefore, further studies across species are needed to explore the mechanisms and feasibility of ultra-dry storage.
The present study aimed to develop a method of storing A. mongolicum seeds that ensures high germination percentages and robust seedling growth. Seeds were dried to various MC levels and stored at different temperatures to establish optimal drying conditions. In addition, we analyzed seed germination vigor and physiological changes under ultra-dry storage, providing valuable insights for future research.
Materials and Methods
Materials
The A. mongolicum seeds used in this study were harvested in 2020 from Ordos City, Inner Mongolia, China. All experimental seeds originated from the same batch to ensure consistency. To establish different moisture content treatments, the initial MC was determined by weighing 100 seeds before and after drying at 130°C for 3 h. For dehydration treatments, seeds were placed in Eppendorf tubes containing silica gel and dried to target MC levels of 6.0%, 4.5%, 3.5%, 2.5% and 1.5% (Ballesteros et al. 2020). The dried seeds were then sealed in three-layer aluminium-foil bags and stored in incubators at –20°C, 20°C, 35°C and 50°C (Ellis and Hong 2006). Because seed aging is effectively arrested at –20°C, those samples were retrieved only once after 60 d. Seeds stored at 20°C, 35°C and 50°C were sampled at 60‐d intervals over 420 d to measure the germination percentage and physiological indices.
Preparation of the seed MC gradient
Seed samples, weighed for their initial mass, were placed in a nylon mesh bag and buried in silica gel in a desiccator at a gel-to-seed ratio of 10:1 (w/w). The desiccator was tightly sealed and maintained at 25°C. Every 24 h, the silica gel was regenerated by drying at 120°C and cooling, after which it was replaced to ensure continuous moisture absorption. At each interval, seeds were removed and weighed on an electronic balance, and the mass recorded.
MC was calculated as follows: MC (%) = (initial seed mass − final seed mass) / initial seed mass × 100%. Measurements were carried out in triplicate and averaged (e.g., moisture contents between 1.45% and 1.55% were recorded as 1.5%). Once seeds reached one of the five target moisture contents, they were sealed in three-layer aluminium-foil bags for storage (Gao 2017).
Germination test
After dry dehydration and rehydration, A. mongolicum seeds were placed on two layers of filter paper moistened with deionized water in Petri dishes. Each dish was weighed daily, and deionized water was added to restore the initial weight, maintaining a constant moisture level. The dishes were then incubated in darkness at 20°C for germination trials. Each treatment comprised three replicates of 50 seeds. The germination potential was recorded on day 3, and final germination was assessed on day 10 after each storage period. The following calculations were used:
Germination percentage (GP, %) = (n / N) × 100, where n is the number of seeds with normal germination and N is the total number of seeds.
Germination potential on day 3 (GP3, %) = (m / N) × 100, where m is the number of seeds germinated by day 3.
Germination index (GI) = Σ(Gt / Dt), where Gt is the cumulative number of seeds germinated by day t and Dt is the corresponding day number.
Vigor index (VI) = GI × Fw, where Fw is the fresh weight of the seedlings.
Determination of physiological and biochemical indices and analysis of the antioxidant system
To assess antioxidant enzyme activities, 0.2 g of intact germinated seeds from each moisture-content treatment were collected. CAT and ascorbate peroxidase (APX) activities were measured by UV spectrophotometry; SOD activity was assayed using the nitroblue tetrazolium method; guaiacol peroxidase (GPX) activity was determined via the guaiacol assay; and polyphenol oxidase (PPO) and glutathione reductase (GR) activities were quantified colorimetrically (Han et al. 2021).
Determination of proteins, seed-protective, and toxic compound levels
Malondialdehyde (MDA) content was determined by the thiobarbituric acid method (Keeble and Rayner 2021). The rate of superoxide anion (O2‒) production was measured colorimetrically (Europe 2016). Vitamin C (VC) content was quantified spectrophotometrically. Soluble protein (SP) and heat-stable protein (HP) levels were assayed by Bradford’s method, and proline (Pro) content was determined using the sulfosalicylic acid method.
Data analyses
Seed germination and physiological data were organized in Excel 2019 (Microsoft, Redmond, WA, USA) and analyzed in SPSS 22.0 (IBM, Armonk, NY, USA). One-way and two-way ANOVA were performed, followed by Duncan’s multiple range test to evaluate statistical significance. Graphs were produced in Origin 2021 (OriginLab Corporation, Northampton, MA, USA).
Results
Effects of the seed moisture content and seed storage duration under different temperatures on the seed germination parameters
Under 20°C storage, a univariate analysis showed that reducing MC from 6.0% to 1.5% caused a significant decline in the GI and DW parameters (p < 0.05), with no differences among the 3.5%, 4.5% and 6.0% treatments (Table 1). Multiple comparisons by one-way ANOVA revealed that the GI of the low-MC group (1.5–2.5%) was significantly lower than that of the mid-to-high MC group (3.5–6.0%) at 0, 60 and 120 d (p < 0.05), highlighting a persistent reduction under extreme desiccation, in contrast to the stability observed at higher MC levels. Over 420 d, mid-to-high MC (3.5–6.0%) exhibited a slight mid-term increase in GI and DW values before an eventual decline, whereas low MC (1.5–2.5%) trended downward. Two-factor ANOVA confirmed highly significant main effects of MC and the storage time on GI and DW (p < 0.05) and significant MC×storage time interactions (p < 0.05)—with multiple comparisons at each interval revealing a consistent “low < medium < high” stratification (1.5–2.5% < 3.5–4.5% < 6.0%) that shifted dynamically as storage progressed. Notably, at 240 d, the 1.5% MC group exhibited a transient rebound in GI, likely reflecting stress-induced protective responses and survivorship of the most robust seeds under prolonged desiccation. Seed vigor index (VI) remained relatively stable across MC, with only a weaker but still evident increase over time, dependent on MC. Together, these results demonstrate that maintaining MC at 3.5–6.0% under 20°C storage consistently preserves superior germination performance and seed biomass over 420 d, whereas extreme desiccation (1.5–2.5%) leads to persistent declines punctuated by a brief mid-term recovery in GI.
Table 1.
Seed germination indexes of A. mongolicum with various seed moisture content under different storage duration at the 20°C of storage temperature
Note: At the same storage duration and with varying moisture contents, the values represented by different lowercase letters indicate statistically significant differences based on a univariate analysis (p = 0.05), while uppercase letters reflect differences identified through the interaction of MC * Storage days according to a two-factor analysis (p = 0.05). Different letters signify statistically significant variations.
Under 35°C storage, a univariate analysis revealed significant differences in germination percentage, GI, and dry weight parameters among the MC treatments (Table 2). Seeds at 1.5% MC consistently exhibited low germination, while those at 3.5% and 4.5% MC showed a marked improvement by 120 days, followed by a gradual decline. By 360 d, seeds at 4.5% and 6.0% MC maintained germination levels comparable to pre-storage values, but all MC groups showed a decline by 420 d, with no significant differences among them. Dry weight increased slightly up to 60 d and subsequently declined, with 3.5% and 4.5% MC seeds maintaining higher values at 120 d. A two-factor analysis corroborated these findings and identified significant MC × storage time interactions. At 120 d, seeds at 3.5% and 4.5% MC demonstrated enhanced germination, while those at 1.5% MC remained consistently low. Germination performance peaked at 120 d and declined thereafter, particularly for the range from 3.5% to 6.0% MC. Dry weight exhibited a similar trend, with maximal values at 120 d for 3.5% and 4.5% MC. Overall, these results suggest that drying A. mongolicum seeds to approximately 4.5% MC and storing at 35°C for 120 d offers optimal germination performance. However, extended storage beyond 120 days leads to a general decline in seed vigor across all MC levels.
Table 2.
Seed germination indexes of A. mongolicum with various seed moisture content under different storage duration at the 35°C of storage temperature
Note: At the same storage duration and with varying moisture contents, the values represented by different lowercase letters indicate statistically significant differences based on a univariate analysis (p = 0.05), while uppercase letters reflect differences identified through the interaction of MC * Storage days according to a two-factor analysis (p = 0.05). Different letters signify statistically significant variations.
Under 50°C storage, a univariate analysis revealed significant differences in the germination potential, germination percentage, GI, and dry weight across various MC treatments (Table 3). Seeds at 1.5% and 2.5% MC consistently exhibited lower values for all indices compared to seeds at 3.5% and 4.5% MC across all time points. Seeds at 6.0% MC performed similarly to those at 3.5% and 4.5% MC until 120 days, after which all indices for 6.0% MC seeds sharply declined. Germination potential increased for all MC up to 120 days, followed by a decrease with prolonged storage. Notably, seeds at 6.0% MC experienced a pronounced decline in germination percentage post-120 days, ending the storage period below their initial levels. Dry weight peaked at 60 days and then decreased significantly, with seeds at 3.5% and 4.5% MC maintaining the highest values. Although the dry weight increased with higher MC values, the differences were not statistically significant for a given storage interval. A two-factor analysis further confirmed these findings and highlighted significant interactions between MC and the storage time for both germination percentage and GI. The interaction was particularly notable for seeds at 6.0% MC, which showed high germination potential up to 120 d but experienced a sharp decline thereafter. Seeds at 3.5% MC exhibited the highest germination potential and GI, demonstrating the greatest resistance to aging under the 50°C storage condition. Although dry weight differences across MC treatments were not statistically significant, higher MC levels were associated with slightly higher dry weight values, particularly at 60 d, followed by a decrease. These results suggest that drying A. mongolicum seeds to approximately 3.5% and 4.5% MC provides optimal resistance to aging when stored at 50°C for 120 d. Seeds at 6.0% MC can be reliably stored only for 120 d, after which all indices show a sharp decline. In contrast, seeds with lower MC levels (1.5% and 2.5%) consistently exhibited poorer performance throughout the storage period.
Table 3.
Seed germination indexes of A. mongolicum with various seed moisture content under different storage duration at the 50°C of storage temperature
Note: At the same storage duration and with varying moisture contents, the values represented by different lowercase letters indicate statistically significant differences based on a univariate analysis (p = 0.05), while uppercase letters reflect differences identified through the interaction of MC * Storage days according to a two-factor analysis (p = 0.05). Different letters signify statistically significant variations.
Under–20°C storage, a univariate analysis revealed that seeds at 2.5%, 3.5%, 4.5%, and 6.0% MC showed no significant changes in germination indices after 60 d, and their performance generally improved compared to that on day 0 (Table 4). In contrast, seeds at 1.5% MC displayed diminished germination ability, with all indices consistently lower than those at higher moisture contents. The germination potential, germination percentage, and GI for seeds at 2.5%, 3.5%, 4.5%, and 6.0% MC remained stable and, in some cases, slightly improved, indicating that low-temperature storage at –20°C effectively preserves seed vigor at these MC levels.
Table 4.
Seed germination indexes of A. mongolicum with various seed moisture content under different storage duration at the ‒20°C of storage temperature
Note: At the same storage duration and with varying moisture contents, the values represented by different lowercase letters indicate statistically significant differences based on a univariate analysis (p = 0.05), while uppercase letters reflect differences identified through the interaction of MC * Storage days according to a two-factor analysis (p = 0.05). Different letters signify statistically significant variations.
A two-factor analysis further confirmed these results, showing no significant interaction between MC and the storage time for the higher MC treatments (2.5%, 3.5%, 4.5%, and 6.0%) over 60 d, suggesting stable seed performance during the storage period. However, seeds at 1.5% MC consistently showed a decline in all indices (denoted by lowercase letters), highlighting the poor storage potential of seeds at very low MC levels under –20°C storage. These findings suggest that the optimal MC range for low-temperature storage of A. mongolicum seeds is between 2.5% and 6.0%, as these treatments demonstrated the most consistent or improved performance over time. The storage period at –20°C was limited to 60 d because low temperatures significantly slow down seed aging. As a result, storage at –20°C effectively preserved seed vigor in the short term, with minimal changes observed within 60 d.
Effects of the seed moisture content and seed storage duration under different temperatures on the seed antioxidant enzyme contents
As the MC level was increased at 20°C, the LOX activity followed a “rise–fall–rise” pattern, peaking at 8.75 mg·min‒1 in seeds at 2.5% and 7.11 mg·min‒1 at 6.0%, while 4.5% yielded the lowest rate (3.20 mg·min‒1). Under 35°C, LOX initially declined and then rebounded (max. 3.58 mg·min‒1 at 1.5%; min. 0.96 mg·min‒1 at 3.5%), with a similar trend appearing at 50°C (highest 12.71 mg·min‒1 at 1.5%; lowest 4.45 mg·min‒1 at 4.5%). At –20°C, LOX rose only slightly with MC, stabilizing around 4.79 mg·min‒1. A two-factor analysis showed that seeds at 1.5% MC at 50°C exhibited the highest LOX activity, with significant differences compared to the other treatments, as indicated by different letters (Fig. 1A).
CAT activity showed an “increase–decrease–increase” pattern with an increase in MC at both 20°C and 50°C, with the highest value (~0.27 µmol·mg‒1 FW·min‒1) found in seeds at 2.5% MC stored at 50°C and the lowest (~0.065 µmol·mg‒1 FW·min‒1) in 2.5% MC seeds at 35°C. At 50°C, seeds at 2.5% MC had the highest CAT activity, significantly higher than at the other temperatures, as denoted by distinct letters in the analysis results (Fig. 1B).
SOD activity remained relatively stable across MC levels at –20°C, 20°C, and 50°C, with seeds at 1.5% showing a slight elevation (~0.74 U·mg‒1). At 35°C, SOD activity followed a “rise–fall–rise” pattern, with the highest activity observed in seeds at 2.5% MC (0.31 U·mg‒1). At 35°C, all MC levels are marked with “b” to indicate lower SOD activity compared to the other temperatures. This suggests that 35°C may impose temperature-induced stress, negatively affecting SOD activity. In contrast, at 20°C, SOD activity remained relatively stable, with moderate temperatures supporting its function, likely stimulating antioxidative responses. Similarly, at 50°C, higher temperatures induced an increase in SOD activity, although with some fluctuations across different MC levels. At –20°C, SOD activity was stable across all MC levels, showing no significant variations. However, compared to higher temperatures (20°C and 50°C), the SOD activity at –20°C remained lower, indicating that low temperatures do not stimulate as much antioxidative enzyme activity as moderate or high temperatures (Fig. 1C).
GR activity increased and then decreased as MC rose at 20°C and 50°C, peaking at 4.5% MC (0.00225 and 0.01075 µmol·mg‒1·min‒1, respectively). At 50°C, seeds at 4.5% MC showed the highest GR activity, with significant differences compared to the other treatments, as indicated by the different letters (Fig. 1D).
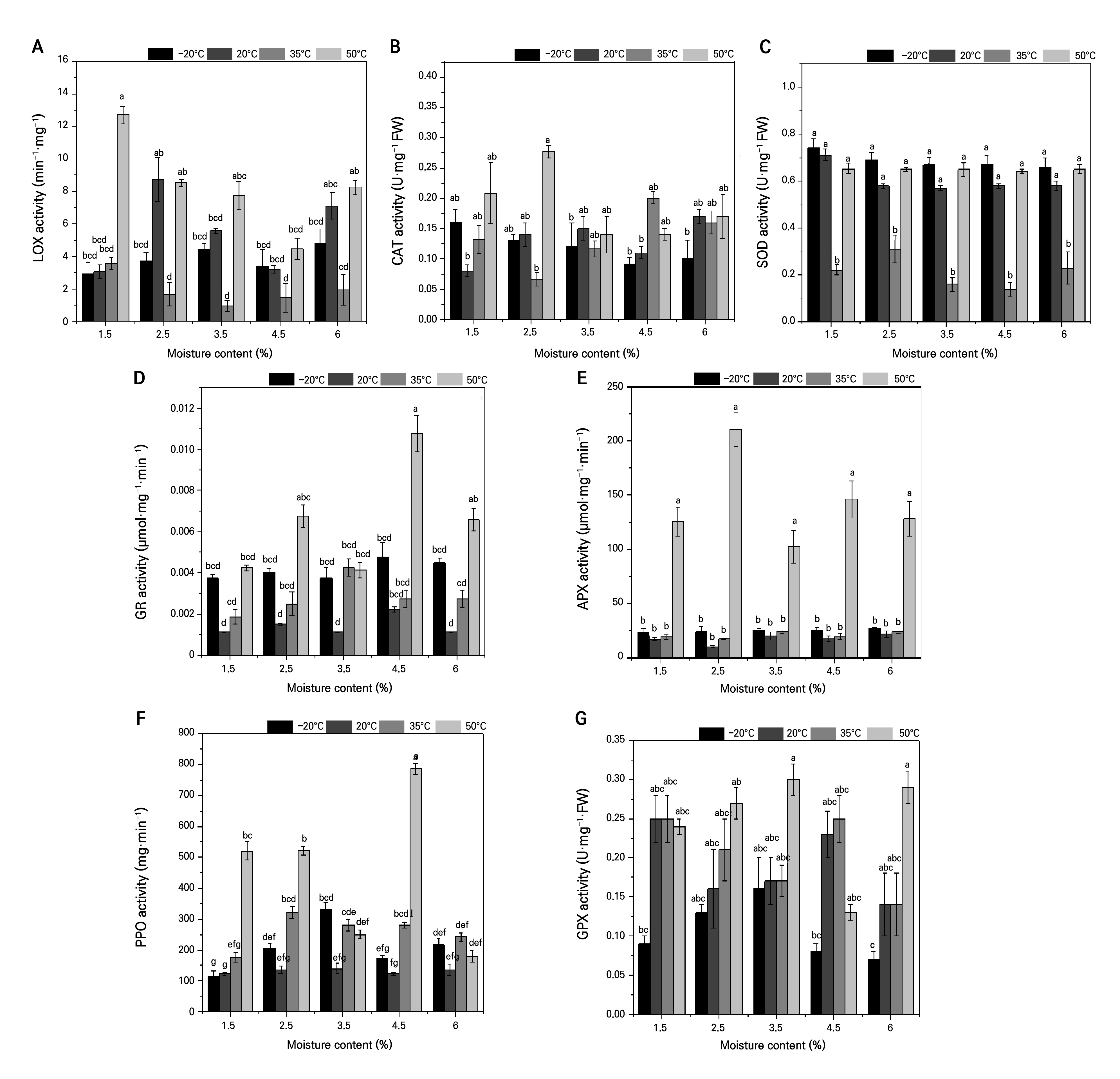
Fig. 1.
Effects of drying treatments and different storage temperatures on the antioxidant enzyme system of A. mongolicum seeds: (A) effects of dry storage and different temperatures on the activity of LOX enzymes; (B) effects of dry storage and different temperatures on the activity of CAT enzymes; (C) effects of dry storage and different temperatures on the activity of SOD enzymes; (D) effects of dry storage and different temperatures on the activity of GR enzymes; (E) effects of dry storage and different temperatures on the activity of APX enzymes; (F) effects of dry storage and different temperatures on the activity of PPO enzymes; (G) effects of dry storage and different temperatures on the activity of GPX enzymes. Different lowercase letters indicate statistically significant variations under the interaction of MC * temperature at the P = 0.05 level.
APX activity remained low (~0.020–0.030 µmol·mg‒1 FW·min‒1) at 20°C and 35°C, showing no clear trend. At 50°C, APX activity was significantly higher across all MC levels, with the highest activity observed at 2.5% MC (0.210 µmol·mg‒1 FW·min‒1), whereas the other treatments showed considerably lower levels of APX activity (Fig. 1E).
PPO activity showed minimal variation with MC at 20°C. At 35°C, PPO activity peaked at 322.22 mg·min‒1 at 2.5% and then decreased at 1.5%. At 50°C, it followed a “decrease–increase–decrease” profile (max. 786.11 mg·min‒1 at 4.5%; min. 180.56 mg·min‒1 in controls), whereas at –20°C, it exhibited the opposite trend (max. 330.56 mg·min‒1 at 3.5%; min. 113.89 mg·min‒1 at 1.5%). The two-factor analysis showed significant MC × temperature interactions for PPO activity, especially at extreme temperatures (Fig. 1F).
Finally, GPX activity showed a “fall–rise–fall” response to increasing MC levels at 20°C and 35°C, with controls at approximately 0.14 µmol·mg‒1 FW·min‒1. Under 50°C, it peaked at 0.27 µmol·mg‒1 FW·min‒1 in 2.5% seeds and dipped to 0.13 µmol·mg‒1 FW·min‒1 at 4.5%. At –20°C, GPX rose to 0.16 µmol·mg‒1 FW·min‒1 at 3.5% before declining, with no significant deviation from the control (Fig. 1G). The two-factor analysis indicated significant interactions, where high heat promoted GPX activity, while low temperatures suppressed it.
Effects of the drying treatment on the protein content of A. mongolicum seeds at different storage temperatures
The soluble protein content remained largely unchanged across moisture contents at –20°C, 20°C and 35°C, but at 50°C it rose to a maximum of 0.45% at 3.5% MC before falling to 0.22% at 6.0%, suggesting that only high-temperature storage promotes protein degradation (Fig. 2A).
Heat-stable protein levels were similarly stable at –20°C, 20°C and 50°C; at 35°C they dipped to 0.28% at 2.5% MC and peaked at 0.45% in the control, yet differences among treatments were not significant, indicating minimal impact of the storage temperature on thermostable proteins (Fig. 2B).
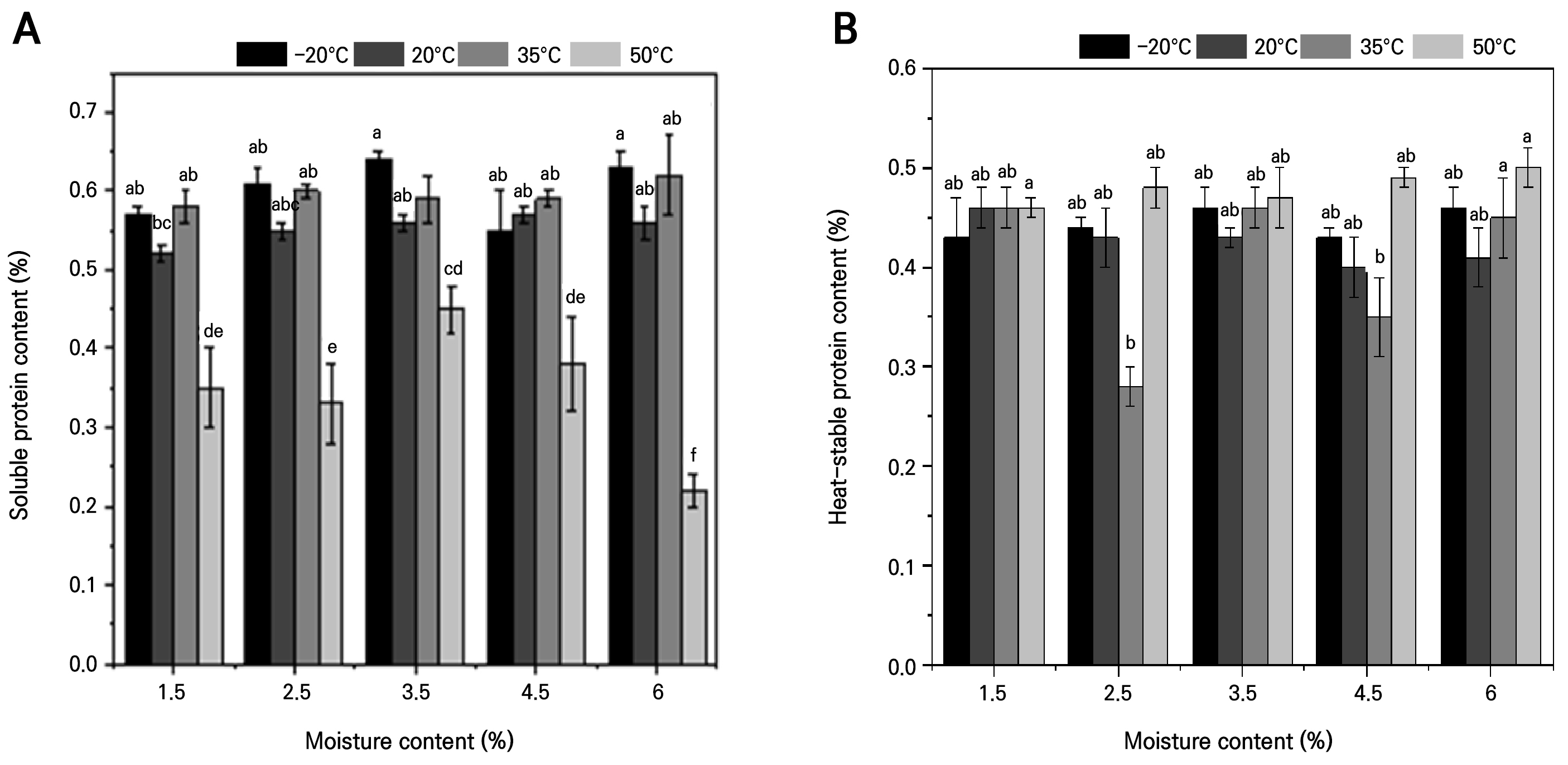
Fig. 2.
Dynamic changes in the contents of soluble and heat-stable proteins in dried A. mongolicum seeds stored at different temperatures: (A) effects of different temperatures on the soluble protein content under dry storage; (B) effects of different temperatures on the heat-stable protein content under dry storage. Different lowercase letters indicate statistically significant variations under the interaction of MC * temperature at the p = 0.05 level.
Effects of the drying treatment and storage temperature on protective and osmoregulatory compounds and superoxide (O2‐) levels in A. mongolicum seeds
The VC content did not vary significantly with MC or storage temperature, suggesting that neither drying nor storage conditions markedly affect its level (Fig. 3A). In contrast, proline exhibited a bell-shaped response to an increase in MC at both –20°C and 20°C: at –20°C, proline decreased from 0.016% to 0.0111%, whereas at 20°C it increased from 0.0055% to 0.0086%, with overall greater accumulation at the warmer temperature. At 35°C, proline rose steadily from 0.0022% to 0.011%, while at 50°C it declined from 0.0058% to 0.0040% (Fig. 3B). These findings indicate that moderate storage temperatures promote osmolyte accumulation, whereas extreme heat inhibits it.
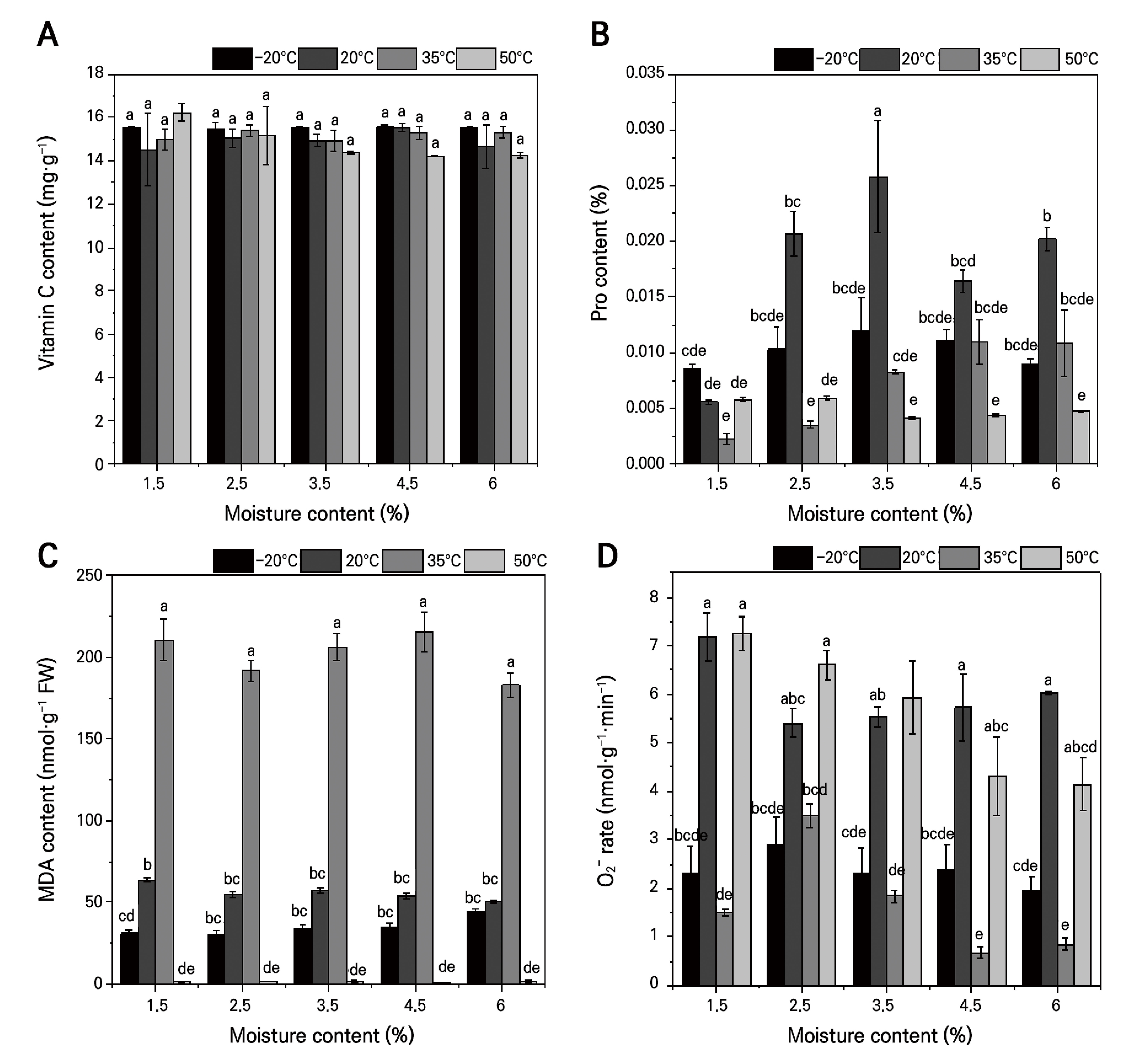
Fig. 3.
Effects of drying treatments and different storage temperatures on the contents of protective components, osmo-modulating compounds, and O2- levels in A. mongolicum seeds: (A) effects of different temperatures on the VC content under dry storage; (B) effects of different temperatures on the Pro content under dry storage; (C) effects of different temperatures on the MDA content under dry storage; (D) effects of different temperatures on the O2‒ rate under dry storage. Different lowercase letters indicate statistically significant variations under the interaction of MC * temperature at the p = 0.05 level.
MDA peaked at 0.06344 µmol·g‒1 FW at 1.5% MC and 20°C, was consistently highest under 35°C, lowest at –20°C (minimum 0.03091 µmol·g‒1 FW at 1.5% MC), and reached its lowest overall level at 50°C—likely reflecting suppressed metabolism or cellular collapse rather than reduced oxidative damage (Fig. 3C).
Superoxide-anion production at 20°C decreased then increased with a decrease in MC (0.0054 → 0.00717 µmol·mg‒1·min‒1 FW); at 35°C the trend inverted, with a minimum of 0.00068 µmol·mg‒1·min‒1 FW at 4.5% MC, and it was generally suppressed at both 50°C and –20°C (Fig. 3D). These patterns suggest that extreme temperatures dampen ROS generation and that ROS production rises as the seed moisture content decreases.
Analysis of the correlations between the various indices of A. mongolicum seeds after drying at different temperatures
Under 20°C storage, the correlations among MC, the germination parameters, antioxidant enzymes and related compounds were generally weak (Fig. 4A). The only significant positive associations were between the germination potential and both the germination percentage and APX activity, while MC correlated negatively with heat-stable proteins. No other enzyme–compound or MC–enzyme relationships reached significance.
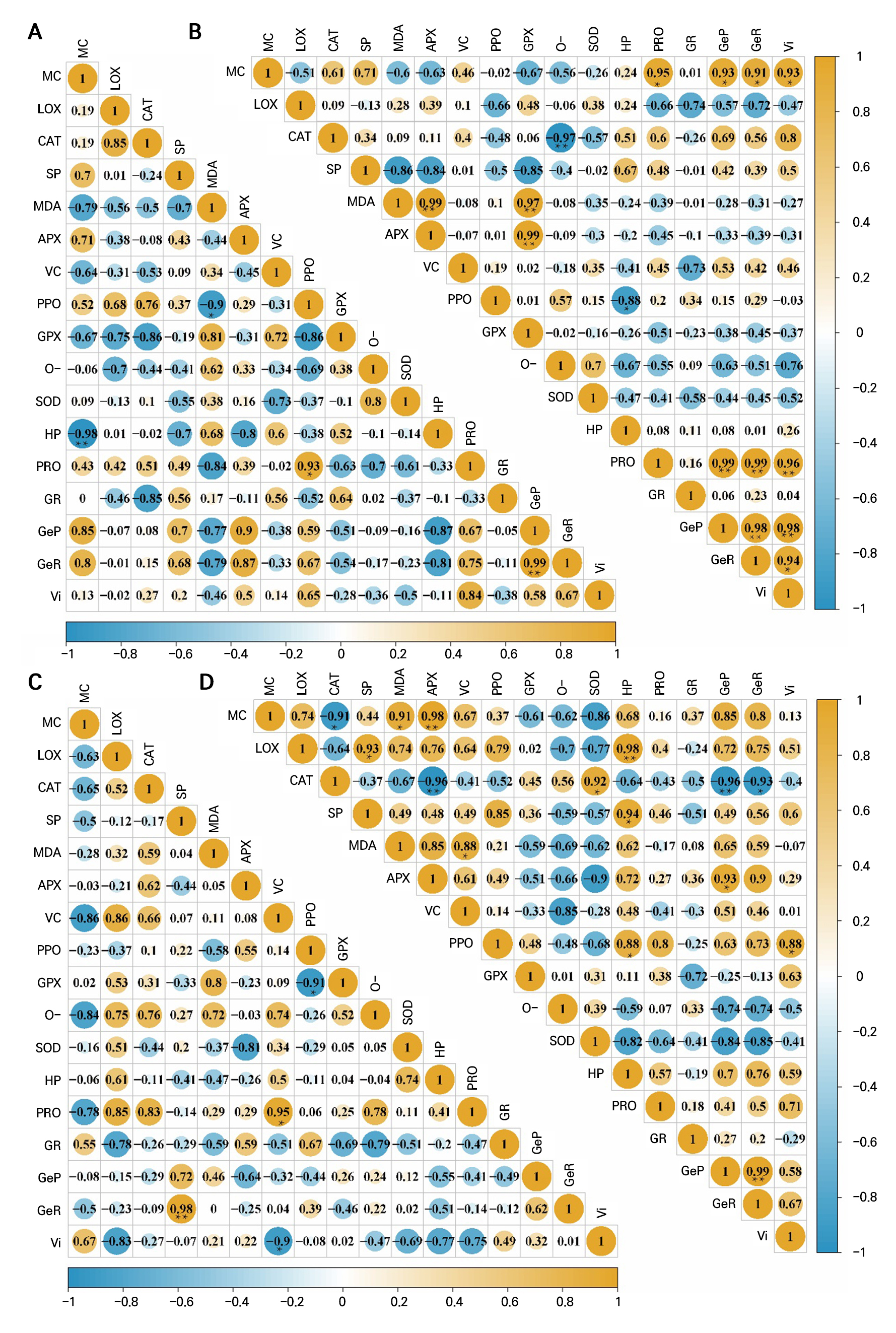
Fig. 4.
Correlations among germination indicators, activities of the antioxidant system, and proteins in dry-treated A. mongolicum seeds: (A) correlation analysis of various indexes of A. mongolicum seeds stored at 20°C; (B) correlation analysis of various indexes of A. mongolicum seeds stored at 35°C; (C) correlation analysis of various indexes of A. mongolicum seeds stored at 50°C; (D) correlation analysis of various indexes of A. mongolicum seeds stored at ‒20°C. *Significant correlations at the 0.05 level, r = 0.8783. **Significant correlations at the 0.01 level, r = 0.9587.
At 35°C, MC showed a strong positive correlation with the germination potential and percentage, and these two indices were highly interrelated with the vigor index (Fig. 4B). Germination parameters also correlated significantly with antioxidant enzymes and protective substances—especially proline. Among enzyme–enzyme relationships, CAT and LOX activities were positively linked, as were GPX and APX. Finally, CAT activity correlated negatively with superoxide-anion production, whereas APX and GPX correlated positively with MDA levels.
At 50°C, all germination indices were significantly intercorrelated (Fig. 4C). The germination percentage was correlated positively with the soluble protein content, as was the vigor index with LOX activity. A notable positive correlation emerged between PPO and GPX activities; conversely, VC and proline were negatively associated.
Under –20°C storage, MC correlated positively with CAT and highly with APX activities (Fig. 4D). The germination potential, percentage and vigor index were again highly intercorrelated, but each correlated negatively with SOD activity. Additionally, APX and SOD activities were positively linked; LOX activity correlated with soluble proteins; PPO with heat-stable proteins; and heat-stable proteins with soluble proteins. These patterns highlight that even at low temperatures, specific enzyme–compound and MC–enzyme relationships can influence seed vigor.
Discussion
Seed dehydration resistance correlates with the type and concentration of storage compounds: protein-rich seeds are most susceptible to water loss, starch-rich seeds are at an intermediate level, and oil-rich seeds are most resistant. A. mongolicum seeds, being starch-based, contain 19.09% sugar, 11.79% fat and 0.49% protein. Their broad ranges of stored water content and starch dominance render them less temperature-sensitive during dehydration than protein- or fat-rich seeds (Afzal et al. 2019; Bakhtavar and Afzal 2020). In this study, peak germination percentage and vigor occurred at 50°C with MC levels of 3.5% and 4.5%, at 20°C and 35°C with MC between 3.5% and 6.0%, and at –20°C with MC between 2.5% and 6.0%. The optimal MC range narrowed as the storage temperature rose—3.5% to 6.0% at 20°C and 2.5% to 6.0% at –20°C—indicating that even excessively dehydrated seeds can remain viable at low temperatures. At 0 d, seeds at 1.5–2.5% MC exhibited lower GI values likely due to excessive membrane rigidification that impairs imbibition, whereas at higher MC levels the balance between hydration and metabolic suppression remained optimal. By 60 d, progressive desiccation stress in low-MC seeds further reduced enzyme functionality and antioxidant capacity levels, whereas the mid-to-high MC treatments maintained membrane integrity and ROS-scavenging activity. At 120 d, cumulative oxidative damage overwhelmed protective systems in both low and high MC extremes, but seeds at 3.5–6.0% MC still retained partial vigor due to sustained cellular homeostasis. A transient rebound in the germination index at 240 d under 1.5% MC likely reflects a combination of stress-induced protective mechanisms and selective survival rather than a true restoration of vigor across the entire seed lot. After prolonged extreme desiccation, seeds may have gradually entered an adaptive phase, triggering the activation of protective proteins and antioxidants, which could temporarily stabilize cellular functions and repair some damage. This “survivorship” effect means that only the more robust seeds remained viable, leading to the observed short-term improvement in germination performance. However, due to the cumulative damage from prolonged dehydration, this rebound was not sustained, and as storage continued (360 d and 420 d), the germination ability declined again. Thus, while the 240 d rebound suggests some degree of physiological recovery, it was not enough to overcome the long-term effects of desiccation.
Most researchers agree that seed deterioration is driven primarily by lipid peroxidation induced by free radicals (Afzal et al. 2019). Excessive drying leads to radical over-accumulation, upsetting the balance between generation and scavenging and causing the peroxidation of unsaturated membrane lipids and damage to proteins, DNA and RNA (Møller 2001; Apel and Hirt 2004). While most seeds tolerate drying below 5% MC—and oilseeds can reach < 1% MC (Huo et al. 2015)—their survival depends on efficient detoxification, toxin sequestration and antioxidant defenses. During desiccation, active mechanisms (antioxidant enzymes such as SOD, CAT, GR, APX and glutathione reductase, plus dehydroascorbic acid) and passive mechanisms (low-molecular-weight antioxidants such as GSH/GSSG, tocopherols and ascorbic acid) collaborate to ROS scavenging.
In these experiments, SOD activity did not differ significantly among the MC treatments at a given temperature, although when MC was held constant, SOD activity at 35°C was notably lower than at the other temperatures. Both the highest and lowest CAT activities occurring at 2.5% MC highlight strong MC×temperature interaction: moderate moisture under severe heat stress (50°C) maximally induces protective CAT responses, whereas under milder heat (35°C) the same moisture level fails to trigger—and may even partially inhibit—enzyme activity. GR activity was significantly higher at 50°C than at other temperatures. APX activity was also markedly elevated at 50°C, although varying the MC level had a subtler effect. Conversely, PPO, GPX, and LOX activities were strongly influenced by the storage temperature, with higher temperatures enhancing these enzyme activities. Overall, these findings indicate that both MC level and the temperature modulate the antioxidant enzyme system during storage, with temperature exerting the dominant influence.
Seed desiccation induces physiological changes that stimulate protective enzymes and cause the accumulation of osmoprotectants. Beyond a critical desiccation threshold, however, these responses wane (Walters 2015). We observed no significant variation in the VC content across MC or temperatures. At 20°C, seeds with 3.5% MC exhibited the highest proline levels, suggesting that moderate dehydration at this temperature optimizes osmolyte accumulation. This likely reflects the activation of both enzymatic and non-enzymatic ROS-quenching pathways, thereby limiting lipid peroxidation.
Lipid peroxidation that occurs as seeds dry generates harmful byproducts such as O2‒ and MDA. O2‒ damages macromolecules and membranes, triggering reactions that culminate in seed deterioration (Guo et al. 2012). In this study, the MDA content did not vary significantly among MC levels at 35°C but was significantly higher than at the other temperatures. Although MDA was lowest at 50°C, this likely reflects rapid cellular damage or metabolic suppression rather than reduced oxidative stress. At 20°C and –20°C, MDA levels were similar across MC levels, indicating that the temperature rather than the drying intensity governs MDA accumulation. O2‒ production was highest at 20°C and 35°C, with seeds at 6.0% MC showing the lowest rates, suggesting that both dehydration and high temperatures promote ROS generation.
During seed maturation, storage proteins undergo dynamic changes (Bewley and Marcus 1990; Kermode 1990; Choi et al. 2009), and late embryogenesis abundant (LEA) proteins accumulate to confer desiccation tolerance in transgenic plants (Baker et al. 1988; Yan 2016). In soybeans, dehydration tolerance correlates with specific mature protein accumulation (Blackman et al. 1991). Drying alters soluble-protein conformation and enhances thermal stability via interactions with cellular components, preserving membrane integrity (Rajjou and Debeaujon 2008). Thus, protein stability underpins desiccation tolerance (Xie et al. 2021). In this study, soluble and thermostable protein contents did not differ significantly across MC levels at the same temperature, indicating that these proteins alone may not determine drying tolerance, although they may still contribute to macromolecular stabilization or seed–water interactions (Blackman et al. 1991; Rajjou and Debeaujon 2008). Notably, the soluble protein content was lower at 50°C—likely due to heat-induced degradation—whereas thermostable (heat-shock) proteins remained constant, suggesting that they impart greater resistance to high temperatures than general soluble proteins. Overall, protein-related mechanisms of dehydration tolerance in A. mongolicum seeds were not clearly evident.
Conclusions
This study confirms that the seed moisture content and the storage temperature are the key determinants of A. mongolicum viability under dry storage. Seeds at 4.5–6.0% MC stored at 20–35°C for 180–360 d consistently showed the highest germination vigor, elevated CAT and PPO activities, increased proline and protein contents, and the lowest MDA and O2‒ levels. In contrast, both lower MC levels (1.5–3.5%) and prolonged exposure to 50°C resulted in diminished germination and the accumulation of toxic compounds. The germination percentage correlated positively with protective biochemical markers and negatively with oxidative-stress indicators. Taken together, maintaining seeds at 4.5–6.0% MC and 20–35°C offers the most reliable strategy for the long-term dry storage of A. mongolicum germplasm.


