Introduction
Materials and Methods
Plant Materials, Limited Irrigation Treatment, and Investigation of Fruit Growth and Quality
Pretreatment of Sampling for Measuring Antioxidant Activity
DPPH Free-Radical Scavenging Activities Test
Analysis of Total Flavonoid Content
Experimental Design Statistical Analyses
Results and Discussion
Introduction
Jeju is located about 100 kilometers south of the Korean Peninsula. Its mean annual temperature and precipitation is about 15°C and 1,800 mm respectively. Citrus cultivation in Korea is limited to Jeju Island and the southern coast of the Korean Peninsula (Kim et al., 2008). Therefore, Korean farmers cultivate the most popular early-maturing ‘Miyagawa’ Satsuma mandarin in open fields because of its stronger cold tolerance compare to other varieties (Jeju Citrus Cooperative, 2000). The cultivar adapts well to humid conditions and shows high resistance to cold weather. Productivity is high and fruit quality is excellent. The Fruit weighs 100-120 g, and has an oblate to subglobose shape. The total soluble solids (TSS) content of the juice is 10-12% and acidity is 0.8-1.0% (Mizutani, 2006).
The average TSS of the juice in the fruit produced on Jeju Island is less than 10%, and the acidity is more than 1.0% (Jeju Special Self-governing Province Citrus Marketing & Shipping Association, 2015). Since fruit with high TSS is obtained from water-stressed trees, growers mulch the ground
with a special plastic film that prevents water supply to the roots. The film has the property of transporting water vapor unilaterally from the ground to the air but not from the air to the ground.
The Jeju provincial government and the mandarin industry plan to strengthen efforts to increase TSS by using cultivation methods and a non-destructive optical sensor fruit-sorting system. In years of heavy precipitation, the TSS content is not constant because most of the soil is volcanic ash and highly permeable to water so that rainwater flows into the roots. In greenhouse cultivation, the ground is covered with vinyl; therefore, it is possible to control soil moisture because it is only necessary to prevent water inflow from the outside of the greenhouse. Fruit quality, fruit weight, and tree performance are greatly affected by soil management (Hirose, 1988). The Satsuma mandarin early-maturing ‘Harye’ was selected as a nucellar seedling of early-maturing ‘Tachima’ × ‘Natsudaidai’ at the Jeju Citrus Research Institute (Yun et al., 2008). The characteristics of the varieties are a higher TSS and lower acidity than the early-maturing ‘Miyagawa’, a popular cultivar in Jeju (Lee et al., 2016).
Sprouting and flowering in citrus occur after a period of adverse environmental conditions, usually related to cold temperatures in temperate climates and water deficiency in tropical and subtropical regions (Iglesias, 2007). Antioxidants could be used as a potential growth regulator to improve plant growth and yield and increase plant resistance to such environmental stress conditions as newly reclaimed sandy soils. In an analysis of the influence of the timing of deficit irrigation on the fruit quality of grapefruit, trees under moderate water stress showed increased levels of flavonoids (Navarro et al., 2015). Hirose (1988) recommended periods of limited irrigation from 60 days to increase the fruit sugar contents during green house cultivation of the Satsuma mandarin. To our knowledge, no previous publications have provided detailed descriptions of anti-oxidant and flavonoid content in accordance with limited irrigation of citrus fruits grown under plastic greenhouse conditions. Therefore, we investigated the characteristics of fruit quality, anti-oxidant activity, and flavonoid content in accordance with limited irrigation from after full bloom of early-maturing ‘Harye’ Satsuma mandarin in a plastic greenhouse.
Materials and Methods
Plant Materials, Limited Irrigation Treatment, and Investigation of Fruit Growth and Quality
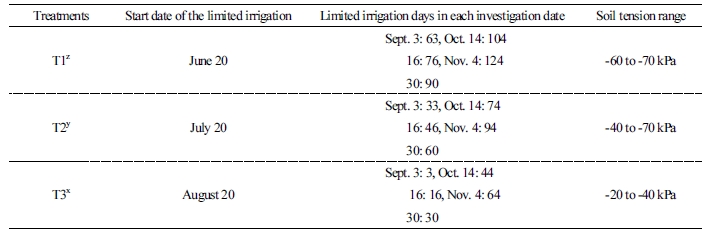
The study was conducted in a plastic greenhouse planted with seven-year-old early-maturing ‘Harye’ Satsuma mandarin (C. Unshui Marc.) trees on a farm in Seogwipo City. The trees were planted in silty clay loam and irrigated with a springkler for one hour with a 1-day interval for 5 days until 30, 60 and 90 days after full bloom, in about late May. The limited irrigation was done 10 trees at the times from 30 days [(T1) Jun 20th], 60 days [(T2) July 20th] and 90 days [(T3) August 20th] to fruit harvest season after full bloom but, the auxiliary irrigation for about 20 min in 1 day was done at the time of apparent wilting of the leaves by a vinyl hose connected with tap water. The soil tension in the greenhouse was measured at hourly intervals from June to October with a tension meter (ZF-3023, Usem, Korea). The irrigations were maintained from -20 to -40 kPa, and the limited irrigations were from -50 to -70 kPa. The start dates of treatment and days in each investigation period are shown in Table 1.
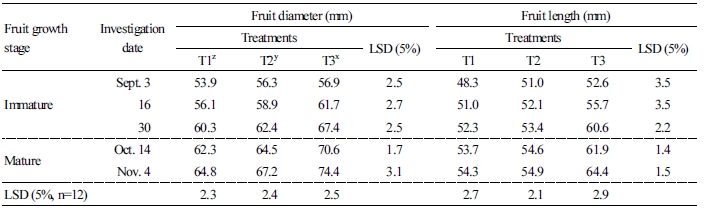
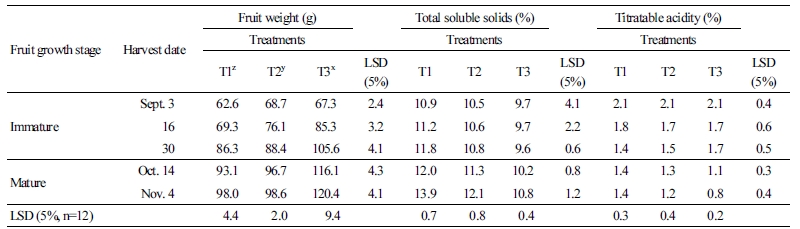
The diameter and length of labeled fruit on five trees were measured with Vernier calipers (CFC-G, Mitutoyo, Japan) from September to harvest season and about two weeks after physiological fruit drop and before fruit tinning. The 12 fruits were harvested in September and November 2016, peeled, and squeezed to extract juice. The extracted juice was measured for TSS with a digital refractometer (PR-101, Atago, USA) and for titratable acidity using the neutralization titration reaction.
Pretreatment of Sampling for Measuring Antioxidant Activity
Peel and flesh samples were sliced and kept in a freezer at -80°C until analysis for various substances. The frozen samples were freeze-dried and crushed in a blender. Then, 400 mg of dried powder was put in a 50-mL tube, and the tube was filled with 30 mL of 80% ethanol solution. Ethanol extraction was carried out for 10 hours at 60°C with slight shaking. The extracts were centrifuged and the supernatant was completely dried with a rotary evaporator (Laborota 4000 Heidolph, Germany). The dried materials were dissolved in 2 mL of methanol and used as samples in the material analysis.
DPPH Free-Radical Scavenging Activities Test
The electron-donating ability (EDA) in each sample of the ethanol extracts was tested with a slightly modified method of Blois (1958). Four milliliters of methanol was added, and then 0.15 mM 1,1-diphenyl-2-picryhydrazyl (DPPH) was added, and the reaction was stabilized at room temperature. After that, absorbance was measured with a UV/Vis spectrophotometer (V-570 Shimazu, Japan) at 725 nm. We indicated the percentage difference of the absorbance value between the experimental sample and the control and then measured the electron-donating ability by the following calculation:
EDA (%) = (1-ABS sample/ABS control) ×100
where ABS sample is the absorbance of the experimental sample, and ABS control is the absorbance control.
Analysis of Total Flavonoid Content
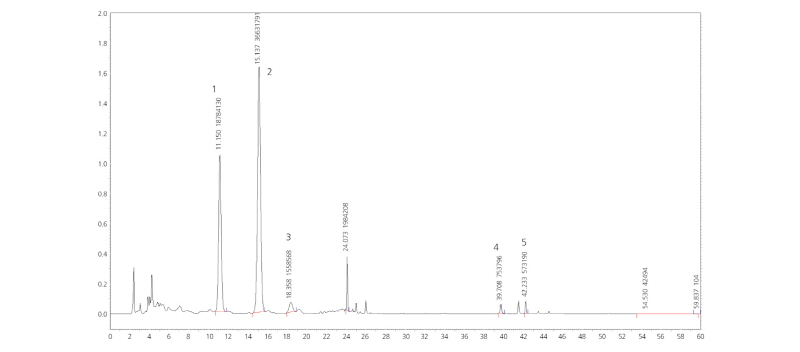
Reconstituted samples were filtered through a membrane filter (0.45 µm). A 10-µL aliquot of filtered samples was injected into a high-performance liquid chromatography system (HPLC, LC-10AD VP, Shimadzu, Japan) equipped with a pump, UV/Vis detector, column oven, and injector through an auto-sampler. A Luna C18 (2) column (100 A° 250 × 46 mm, Phenomenex, USA) was used for the HPLC system. The mobile phase for the HPLC system was acetonitrile with 0.5% acetic acid added (A) and water with 0.5% acetic acid added (B) with a flow rate of 1 mL·min-1. The mobile phase program consisted of four periods: 0-14 min, 20% A; 14-33 min, 40% A; 33-38 min, 70% A; and 47-60 min, 20% A. The column was operated at 40°C, and the eluent was monitored with a single-channel UV detector at a wavelength of 280 nm. The flavonoids were identified by comparing their retention times and UV spectra with those of authentic standards stored in a data processor. The content of each flavonoid was calculated from the integrated peak area of the sample and corresponding standard. The flavonoid standard solutions consisted of flavones (narirutin, hesperidin, and neohesperidin) and polymethoxylated flavones (nobiletin and tangeretin). Most of the standard flavonoid materials were purchased from Sigma (St. Louis, MO, USA), while nobiletin and tangeretin were extracted and purified from Citrus depressa at the Okinawa Research Center in Japan.
Experimental Design Statistical Analyses
Each tree in a block was treated as an experimental unit and was randomly assigned to limited irrigation. All treatment combinations were replicated 5 and 12 times. Statistical analyses were conducted using least significant difference (LSD) and Duncan’s multiple range test (Sigma Plot 13, SYSTAT, USA).
Results and Discussion
Changes to fruit growth and quality with limited irrigation treatments from after full bloom in the greenhouse are shown in Tables 2 and 3. For fruit diameter, there was a significant difference in the first investigation date on September 3rd with limited irrigation treatment during fruit growth, and there were significant differences between T1, T2, and T3 on the last investigation date on November 4th. In the case of fruit length, there was a significant difference between T1 and T2 treatments and T3 from the first investigation date to the last investigation date. Although there were no differences in fruit weight at the beginning, T3 fruits were heavier than T1 and T2 at the last harvest. Regarding the fruit quality, the highest TSS, 13.9%, was recorded for T1 at the fruit harvest date. Also, titratable acidity was the highest for T1, 1.4%, but there was no significant difference with T2 (1.2%), and it was lower than T3, at 0.8%
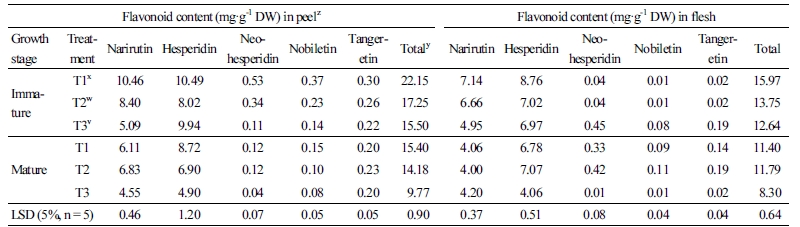
Change in the active oxygen inhibition ratio of the ethanol extract from peel and flesh in the immature season (Fig. 1) showed that inhibition ratios increased as the content of the ethanol extract increased. In the case of the peel, there were no significant differences among T1, T2, and T3 except for the content 0.05 mg (Fig. 1A). On the other hand, for the flesh there were some inhibition ratios with significant differences in extracts less than 2.5 mg (Fig. 1B). As such, inhibition ratios increased for immature season with the increasing content of the extracts. In contrast, the inhibition ratios for the mature season (Fig. 2) for T1 and T2 showed differences compared with T3 for extract contents less than 1.0 mg in the peel. However, there were no differences for T2 and T3 when compared with T1 for extract contents in the flesh.
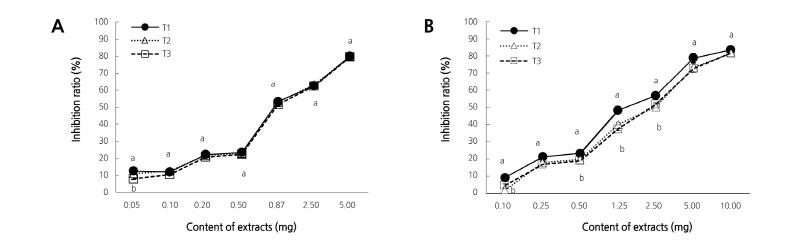
Fig. 1. Changes in active oxygen inhibition ratio in ethanol extracts of peel (A) and flesh (B) with limited irrigation of T1, T2, and T3 after full bloom of early-maturing ‘Harye’ Satsuma mandarin in the immature season in a plastic greenhouse. Vertical bars indicate standard error. Each value is the mean obtained from replicates and columns with the same letter are not significantly different by Duncan’s multiple range test at p < 0.05 (n=5). T1: limited irrigation treatment from 30 days after full bloom (-60 to –70 kPa soil tension maintained from June to October), T2: treatment of limited irrigation from 60 days after full bloom (-40 - to -70 kPa soil tension maintained from June to October), T3: treatment of limited irrigation from 90 days after full bloom (-20 to -40 kPa soil tension maintained from June to October).
Fig. 2. Changes in active oxygen inhibition ratio in ethanol extracts of peel (A) and flesh (B) with limited irrigation from 30, 60, and 90 days after full bloom of early-maturing ‘Harye’ Satsuma mandarin in a plastic greenhouse in maturation season. Vertical bars indicate standard error. Each value is the mean obtained from replicates and columns with same letter are not significantly different by Duncan’s multiple range test at p < 0.05 (n=5). T1: limited irrigation treatment from 30 days after full bloom (-60 to -70 kPa soil tension maintained from June to October), T2: treatment of limited irrigation from 60 days after full bloom (-40 to -70 kPa soil tension maintained from June to October), T3: treatment of limited irrigation from 90 days after full bloom (-20 to –40 kPa soil tension maintained from June to October).
The flavonoids of the ethanol extracts were separated by flavonoids (narirutin, hesperidin, and neohesperidin) and polymethoxylated flavones (nobiletin and tangeretin) through HPLC (Fig. 3). The content of each was added and assumed as total flavonoid content. In the immature season, there were significant differences among T1, T2, and T3 in the peel, and there were differences in the flesh. On the other hand, the mature season, T1 and T2 showed a higher content differences compared with T3; for flesh contents, the T1 and T2 showed a higher content than the T3 (Table 4).
It was reported that fruit growth is restrained by soil water stress treatment as stress increases (Chae et al., 2007; Huang et al., 2000; Hyun et al., 1993; Mukai et al., 1996), while growth recovers with irrigation after dry treatment (Maotani and Machida, 1980). In this study, fruit growth showed significant differences in the limited irrigation for T1 and T2 compared with T3. The most important factor for increasing fruit quality in the plastic greenhouse cultivation of Satsuma mandarin is initiates of water stress with dry soil treatment. The mechanism for increasing TSS in fruit is thought to be related to the following factors. The levels of water stress will rise when fruit growth is retarded if the sugar content of the fruit is constant. The other factor is the change in the translocation patterns of photosynthesis assimilates. Although these assimilates decrease under water stress, leaf, branch, and root growth decreases at the same time, while the fruit TSS increases because of the increasing prevalence of the translocation into the fruit. The mechanism for the increase in TSS is not a simple action, such as for the concentration of fruit juice, but it is considered to be the result of a physiological reaction of water-absorbing tree nutrients that increase the amount of soluble substances, such as sugar (Hirose, 1988). Kaufman (1970), Kadoya et al. (1973), and Yakushiji et al. (1996, 1998) reported that the accumulation of carbohydrates in juice sacs of ‘Navel’oranges and Satsuma mandarin was caused by osmotic control work corresponding to lower water potential. The monosaccharides, glucose and fructose, were involved in active osmotic control, and sugar accumulation was produced by the increasing translocation of assimilates into the juice sac. In the studies, the T1 showed the highest level of TSS, and TSS was lower as the period of limited irrigation became shorter. For example, the limited irrigation in T1 and T2 from Jun 20th and July 20th to November 4th, for 124 and 94 days, -50 to -70 kPa soil tension, respectively, may have caused serious water stress in the canopies compared with T3, from August 20th to November 4th, for 64 days, -20 to -40 kPa soil tension. The water stress in the canopies may have increased the TSS, acidity, anti-oxidant activity and flavonoid content even thought there was a reduction of fruit growth. The flesh weight on last harvest date on the November 3rd, showed a difference among T1, T2 and T3, and this was attributed to severe water stress in the fruit. Also, TSS and acidity of the fruit juice after 3-4 months of limited irrigation showed differences among T1, T2 and T3. The early-maturing ‘Harye’ Satsuma mandarin is the first breeding cultivar, and its characteristics are a higher TSS and lower acidity than the early-maturing ‘Miyagawa’ cultivar in Jeju. A study on the effect of various mulching periods of the porous film on fruit quality showed that the TSS of ‘Harye’ was higher than that of ‘Miyaga’, but the acidity in the two cultivars was similar (Lee et al., 2016). Water stress caused the accumulation of citric acid, lower fruit quality, and changes in relative gene expression for citric synthesis and degradation (Zhang et al., 2014). TSS and acidity were 11.1% and 1.4%, respectively, in the last harvest date after the use of Tyvek (porous film) mulching treatment at the end of August for early-maturing ‘Miyagawa’ Satsuma mandarin with increasing TSS attributed to a reduction of water and osmotic potential in juice vesicles and water content of the flesh because of drought stress (Han et al., 2004). TSS in the juice vesicles in T1 in this study was higher at 13.9% than with the Tyvek treatment, but the acidity was similar.
Antioxidants could be used as potential growth regulators to improve plant growth and yield and increase plant resistance to such environmental stress conditions as newly reclaimed sandy soils. Antioxidants such as amino acids and phenolic acids are regulators, that when added in small quantities maintain the normal growth and proper development of plants (Sadark et al., 2015). Citrus fruit, in particular, is valued because of its nutritional benefits, good flavor, and other intrinsic attributes. They and their products provide a number of nutrients, such as ascorbic acid, vitamin B6, and flavonoid (Kim et al., 2008). The peel of citrus fruit is a rich source of flavanones, as well as many polymethoxylated flavones, which are rare in other plants (Nagata et al., 2006). These compounds not only play important physiological and ecological roles, but they are also of commercial interest because they have a multitude of applications in the food and pharmaceutical industries. For example, naringin and hesperidin may act as anti-inflammatory agents (Chen et al., 1990; Berkarda et al., 1998; Emim et al., 1994; Kobayashi et al., 2006). Polymethoxylated flavones are also of interest for their various pharmaceutical potentials, the most important of which are antitumor, anti-inflammatory, antimutagenic, and antiallergic properties (Murakami et al., 2000a; Murakami et al., 2000b; Kawaii et al., 1999; Kandaswami et al., 1991; Miyazawa et al., 1999; Ishiwa et al., 2000). In general, the content and distributions of flavonoids in different Citrus species are highly variable and depend on genetic and environmental factors (Nagata et al., 2006). Deficit irrigation influence fruit quality in grapefruit, with trees under moderate water stress producing more flavonoid (Navarro et al., 2015).
Changes in anti-oxidant activity and flavonoid content were different in the peel and flesh at the immature season. The flesh tissues of citrus consist of juice vesicles containing a large quantity of moisture, and water loss is easier than from peel tissues. There were differences in anti-oxidant activity and flavonoid content in the flesh between T1, T2 and T3 through the immature season. On the other hand, there were differences in changes in anti-oxidant activity in the 0.05 mg extract and flavonoid contents in peel at the mature season. The decrease in water supply with limited irrigation in the mature season may be a trigger that increases anti-oxidant activity related to flavonoid content in the peel. Also, the difference in activity was the lowest in, the 0.05 mg extract among the treatments based on the antioxidants concentration dilution sufficient for limitation in the DPPH free-radical scavenging activities test.
The flavonoid content in citrus peel was shown to be higher in the immature season than the mature season (Choi et al., 2007). In this study’s experiment, the content was higher in the immature season than at the mature season. Flavonoid content, which is closely related to antioxidant activity, increased with limited irrigation, and this may be because of an increase in the content of the main constituents as a survival response to serious water deficiency. Our results, suggests that limited irrigation from before 60 days after full bloom in greenhouse cultivation of the early-maturing ‘Harye’ Satsuma mandarin would be useful for improving fruit quality by increasing TSS and anti-oxidants in the mature season.