Introduction
Materials and Methods
Plant Material and Experimental Setup
Water Uptake and Relative Fresh Weight Calculations
Evaluation of the Senescence Rate and Vase Life
Petal Color
Statistical Analysis
Results and Discussion
Introduction
The vase life of a cut flower is considered the most critical factor affecting consumers’ preferences and growers’ production (Shi et al., 2021). Postharvsest senescence is a limiting factor in marketing many species of cut flowers related to water quality, water deficit stress, ethylene, environmental (e.g., temperature, light, and humidity), and biological factors during the harvest and transportation (Roh et al., 2020; Ha et al., 2021). Preservatives are applied to the vase water to reduce postharvest deterioration and premature senescence of cut flowers and hinder the development of microorganisms on the stem that block water passage, causing dehydration (Ha et al., 2020; García-González et al., 2022). Water uptake is a significant factor that affects the quality and vase life of cut flowers (Ha et al., 2021). Low water uptake of cut flowers is mainly caused by blocked vascular tissues and stem xylems, and loss of turgor due to microorganisms that prevent the uptake of water and nutrients from the vase solution (van Doorn, 2012; Lee and Kim, 2018; Manzoor et al., 2022).
Ozone (O3) is a strong oxidizing molecule used for food and seed sterilization due to its ability to kill microorganisms such as viruses, protozoa, fungi, and bacteria (Kwack et al., 2014). The vase life of cut rose (Rosa hybrida L.‘Pascha’) treated with 5.5 mg·L-1 of O3 was extended 3-fold relative to roses treated with deionized water, due to a reduction in the number of bacteria in the solution (Robinson et al., 2009). Cut flowers of Mokara ‘Calypso Jumbo’ and Dendrobium ‘Darren Glory’ treated with aqueous O3 (5.2 nL·L-1) had prolonged vase life and postharvest quality (Almasi et al., 2015). Ozonated water must be used within the first 5–10 min after production to ensure potency due to its short half-life of 20 min, in spite of sterilization ability (Hayakumo et al., 2014).
A nanobubble (NB) is a small gas bubble in liquids of ≤ 0.2 µm in diameter, which is more stable in water due to less buoyancy than macrobubbles (≥ 50 µm) or microbubbles (10–50 µm) (Ebina et al., 2013; Minamikawa et al., 2015; Jeong et al., 2017). The collapse of microbubbles after an electrical discharge produces NBs in the solution (Hayakumo et al., 2014). The free radicals generated by collapsing NBs and microbubbles prevent odors in the solution, degrade organic carbon in wastewater, and inactivate microorganisms (Marui, 2013; Minamikawa et al., 2015). NBs can be produced from various gases, such as oxygen, air, nitrogen, and carbon dioxide, which remain stable in liquids for a long time and replenish dissolved oxygen (DO) (Ahmed et al., 2018). Increasing DO is required in agriculture to purify water and promote plant and root growth (Noh et al., 2022). The root length of radish (Raphanus sativus L.) and wheat (Triticum aestivum) increased in plants treated with oxygen NBs (21.0 mg·L-1 DO) compared to the control (9.2 mg·L-1 DO) (Lee et al., 2021). The number of leaves in tomato (Solanum lycopersicum) increased by 8.7 leaves when exposed to oxygen NBs compared to 5.9 leaves of tap water (TW) treatment (Ahmed et al., 2018). Oxygen NBs are used for plant cultivation; however, oxygen NBs are rarely applied for the postharvest preservation of cut flowers, except hydrogen NBs. Hydrogen NBs in the vase solution delayed petal senescence and prolonged the vase life of cut carnation (Dianthus caryophyllus L.) by reducing reactive oxygen species accumulation and initial activities of senescence-associated enzymes (Li et al., 2021). The half-life of O3 is longer when O3 is generated in the form of NB (ozone nanobubble, O3NB) because mass transfer efficiency is improved to supply O3 constantly (Hu and Xia, 2018). The O3NBs reduce pathogenic bacteria in aquaculture systems and help reduce the risk of bacterial disease outbreaks in farmed fish (Jhunkeaw et al., 2021), however, there have been few studies on treating cut flowers with NBs and O3NBs to extend vase life and improve postharvest quality.
Cymbidium is a popular cut flower because of its longevity, various colors, size, and unique floral structure and ranks in the sixth position among the top ten cut flowers in international trade (An et al., 2015; Park et al., 2021). Cymbidium had the highest price at $3.60 per stem among the top 19 species sold in 2018 at the Royal Flora Holland, the world’s largest flower market (Faust and Dole, 2021). Cymbidium is an important ornamental plant and has the largest cultivation area of any orchid in Korea, accounting for 47.4% of the orchid cultivation area (RDA, 2021). Most studies of Cymbidium focused on the production of potted plants despite the expansion in cut flower production (An et al., 2015). Many studies of cut Cymbidium have been conducted to improve postharvest quality using ethylene inhibitors such as 1-methylcyclopropene (1-MCP) and aminoethoxyvinylglycine (AVG) or application of preservatives, which were applied at various levels and durations (Heyes and Johnston, 1998; Lee et al., 2010; An et al., 2015; Kim et al., 2016; Kim et al., 2017; An et al., 2021).
The various senescence symptoms of cut Cymbidium,such as lip coloration, tip burn, and wilting, are caused by decreased water uptake (Kim et al., 2017). We hypothesized that NB and O3NB treatments in the vase solution extend the vase life of cut Cymbidium by maintaining water uptake. Therefore, we investigated the vase life and postharvest quality of cut Cymbidium treated with different concentrations of NBs and O3NBs in the vase solution.
Materials and Methods
Plant Material and Experimental Setup
Cut flowers of Cymbidium ‘Spring Pearl’ were obtained from the Yangjae flower market (Seoul, Republic of Korea) on April 28, 2021. The flowers were transported to Seoul Women’s University (Nowon, Seoul, Republic of Korea). The flowers were recut to 35 cm in air and individually placed in a glass bottle with 500 mL of vase solutions. The experimental environment was kept at 20.3°C, 60 ± 5% relative humidity, and 10 µmol·m-2·s-1 in 16/8 h (light/dark) cycles.
The cut flowers were placed into the vase solutions with various concentrations of nanobubble (NB) and ozone nanobubble (O3NB) treatments (Table 1). Cut flowers held in tap water (TW) were used to control. Four treatments were configured with two levels of NBs and O3NBs as follows: NB:TW = 20:80 (NB 20%), NB:TW = 100:0 (NB 100%), O3NB:TW = 20:80 (O3NB 20%), and O3NB:TW = 100:0 (O3NB 100%). The ozone (O3) and dissolved oxygen (DO) concentrations were 0.0 and 10.0 mg·L-1 (TW, control), 0.0 and 16.3 mg·L-1 (NB 20%), 0.0 and 38.3 mg·L-1 (NB 100%), 0.7 and 16.4 mg·L-1 (O3NB 20%), and 3.8 and 38.3 mg·L-1 (O3NB 100%), respectively. The NBs and O3NBs were generated with an oxygen concentrator (CR-P3W, Fawoo Nanotech Co., Ltd., Bucheon, Gyeonggi, Republic of Korea) and a NB generator (SNT-03, Fawoo Nanotech Co., Ltd., Bucheon, Gyeonggi, Republic of Korea). The O3 is not separate because O3NBs are simultaneously produced when NBs are generated. The NB concentrations were monitored using research-grade meters (HI 5421, HANNA Instruments, Seoul, Republic of Korea), and O3 concentrations were monitored using a portable color reader (Q-03-2, Shenzhen Sinsche Technology Co., Ltd., Shenzhen, Guangdong, China). The vase solution in each glass was renewed daily.
Table 1.
Mean concentrations of ozone (O3) and dissolved oxygen (DO) in vase solutions treated with tap water (TW) and with various levels of nanobubbles (NBs) and ozone nanobubbles (O3NBs)
| Treatments | O3 (mg·L-1) | DO (mg·L-1) |
| TW (Control) | 0.0 | 10.0z |
| NB 20% | 0.0 | 16.3 |
| NB 100% | 0.0 | 38.3 |
| O3NB 20% | 0.7 | 16.4 |
| O3NB 100% | 3.8 | 38.3 |
Water Uptake and Relative Fresh Weight Calculations
The water uptake was determined by weighing the vases daily without the cut flowers. The water uptake was calculated by the formula (St -1–St), where St and St- 1 are the weight of the vase solution (g) at t = day 1, 2, 3, etc., and the weight of the vase solution (g) on the previous day, respectively (He et al., 2006).
The relative fresh weight (RFW) of cut flowers was estimated daily from RFW (%) = (Wt/W0) × 100, where Wt is the weight of the stem (g) at t = day 1, 2, 3, etc., and W0 is the weight of the same stem (g) at t = day 0 (He et al., 2006).
Evaluation of the Senescence Rate and Vase Life
The senescence rate was assessed by visual observation of petal wilt and color change of the floret lip and petal. The senescence stage of flowers was classified into five stages as follows: half-open (stage 1), open (stage 2), full-bloom (stage 3), senescence progression (stage 4), and senescence (stage 5) (Fig. 1). When the floret reached stage 4, it was defined as having no ornamental value. The following senescence symptoms were detected: bending of the stem (bent-neck; neck angle greater than 90°), the lip of the labellum turning red, browning, floret abscission, and wilting (Lee et al., 2010; Kim et al., 2017). The senescence rate was calculated using the formula: Senescence rate (%) = Ff/Nn × 100, where Ff refers to the number of florets that entered stage 4 of the senescence stage, and Nn means the number of total florets of each stem. Vase life was defined as when 60% of florets on the inflorescence entered senescence stage 4, and the fresh weight fell below 70% (Kim et al., 2017).
Petal Color
Flower color was measured using a color reader (CR-10 Plus, Minolta Co., Ltd., Tokyo, Japan) on day 9. The center of the petal of the last floret on the inflorescence was measured per treatment. Petal color measurement was based on the uniform color space’s CIE (L*, a*, and b*) scale. The L* scale for brightness ranged from 0 (black) to positive 100 (white), the a* scale ranged from negative 100 (green) to positive 100 (red), and the b* scale ranged from negative 100 (blue) to positive 100 (yellow) (Yeon and Kim, 2020).
Statistical Analysis
The experimental design was a completely randomized block with five replications for each treatment. Statistical analyses were conducted using the R software for Windows (version 4.3.0, R Core Team, Vienna, Austria). Analyses of variance were conducted to test the data and Duncan’s multiple range test at p < 0.05 was used to compare the data. Graph modules were performed using Sigma Plot, version 10.0 (Systat, Software Inc., San Jose, CA, USA).
Results and Discussion
The water uptake increased 1.4-fold in cut Cymbidium treated with ozone nanobubble (O3NB) 20% relative to the control on day 1 (Fig. 2). The water uptake was higher in cut Cymbidium treated with nanobubble (NB) 20% than in control on day 2. The germination rate of barley (Hordeum vulgare L. ‘Hyegang’) seeds treated with oxygen NBs was increased by approximately 40% compared to treatment with pure water (Oh et al., 2016). Increasing dissolved oxygen (DO) has the advantage of the removing residual pesticides and purifying contaminated water, which is required in agriculture (Noh et al., 2022). The water uptake of cucumber (Cucumis sativus L. cv. Chojitsu-Ochiai) increased under the highest oxygen levels of 0.20 mM oxygen relative to 0.01 or 0.10 mM oxygen (Yoshida et al., 1996). The water uptake was higher in cut rose treated with the highest level of O3 (5.5 mg·L-1) than those treated with deionized water due to a reduction in bacterial growth and prevention of vascular occlusions in the xylem (Robinson et al., 2009). Our results were different from previous studies. The concentration of hydrogen NBs on the cut carnation flower was effective in a specific range (Li et al., 2021). In radish and wheat treated with various concentrations of oxygen NB (including NB 20%, 33%, and 100%), only treatment with NB 33% increased the RFW (Lee et al., 2021). The 1% hydrogen-rich water (HRW) increased the vase life of the cut rose ‘Movie Star’ because it had more beneficial bacteria than other treatments, including distilled water (control), and 0.5%, 25%, and 50% HRW (Fang et al., 2021). The senescence rate of cut Cymbidium treated with NB 20% (16.3 mg·L-1 DO) was lower than that in plants treated with NB 100% (38.3 mg·L-1 DO) (Fig. 4). Therefore, NB 20% and O3NB 20% treatments were more effective than NB 100% and O3NB 100% in the vase solution to maintain the ornamental quality of cut Cymbidium.
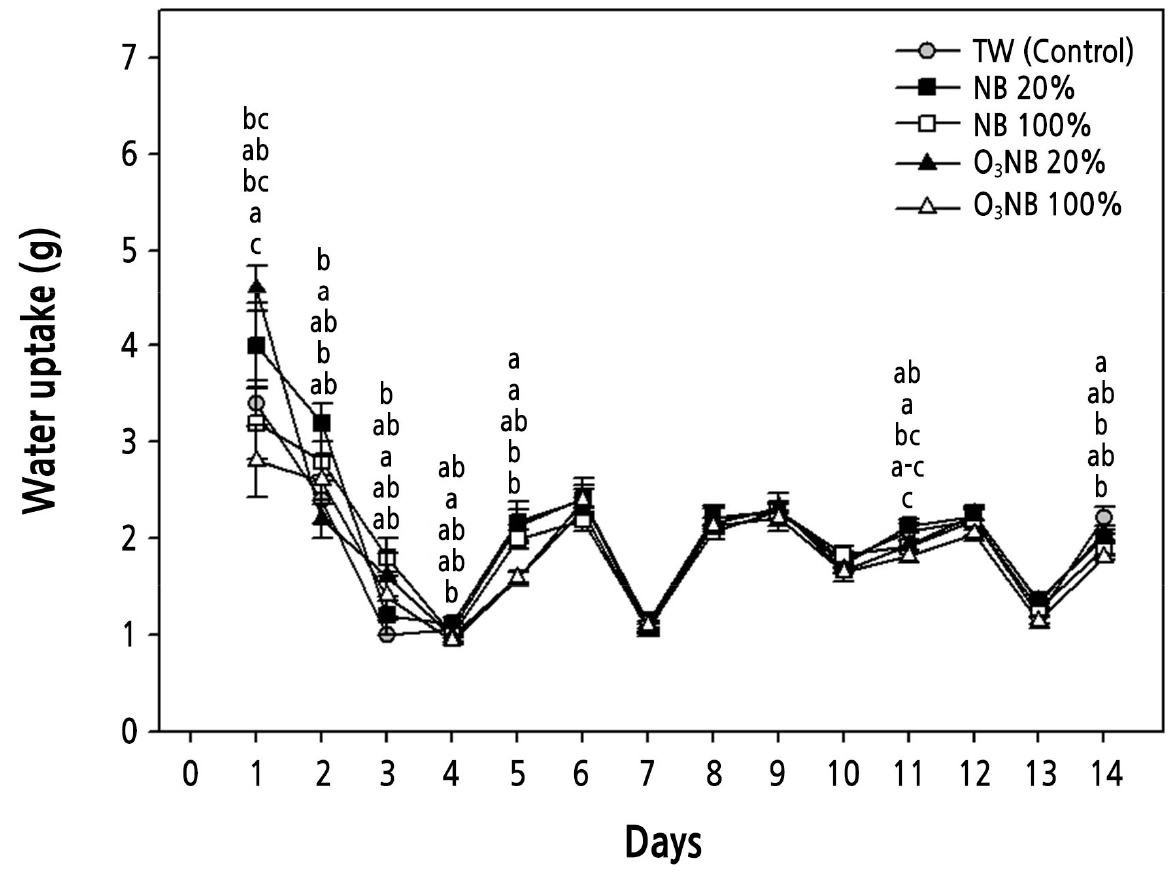
Fig. 2.
Changes in water uptake of cut Cymbidium ‘Spring Pearl’ treated with tap water (TW) and with various levels of nanobubbles (NBs) and ozone nanobubbles (O3NBs). The ozone and dissolved oxygen concentrations were 0.0 and 10.0 mg·L-1 (TW, control), 0.0 and 16.3 mg·L-1 (NB 20%), 0.0 and 38.3 mg·L-1 (NB 100%), 0.7 and 16.4 mg·L-1 (O3NB 20%), and 3.8 and 38.3 mg·L-1 (O3NB 100%), respectively. The vertical bars represent the standard error of the mean value (n = 5). The different letters represent statistically significant differences as determined by Duncan’s multiple range test (p < 0.05).
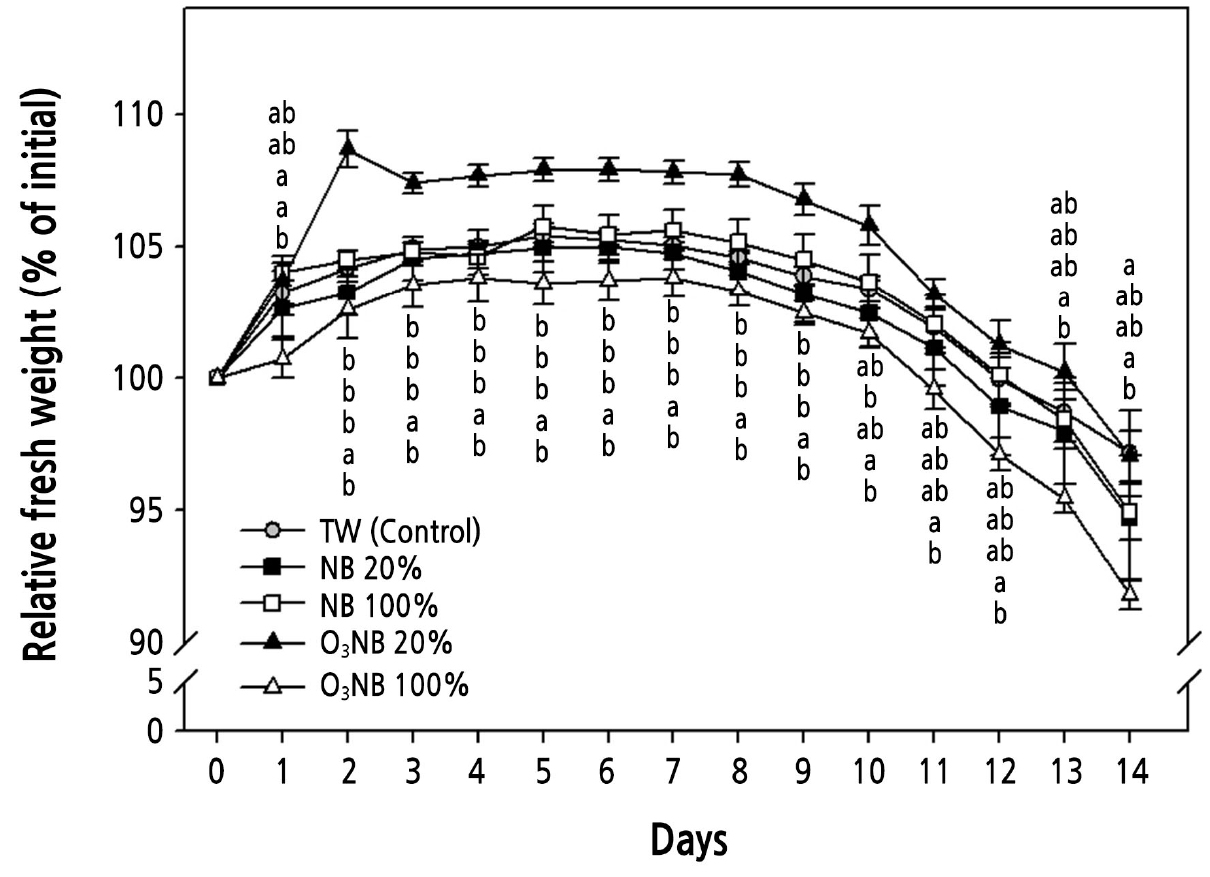
Fig. 3.
Changes in the relative fresh weight (% of initial) of cut Cymbidium ‘Spring Pearl’ treated with tap water (TW) and with various levels of nanobubbles (NBs) and ozone nanobubbles (O3NBs). The ozone and dissolved oxygen concentrations were 0.0 and 10.0 mg·L-1 (TW, control), 0.0 and 16.3 mg·L-1 (NB 20%), 0.0 and 38.3 mg·L-1 (NB 100%), 0.7 and 16.4 mg·L-1 (O3NB 20%), and 3.8 and 38.3 mg·L-1 (O3NB 100%), respectively. The vertical bars represent the standard error of the mean value (n = 5). The different letters represent statistically significant differences as determined by Duncan’s multiple range test (p < 0.05).
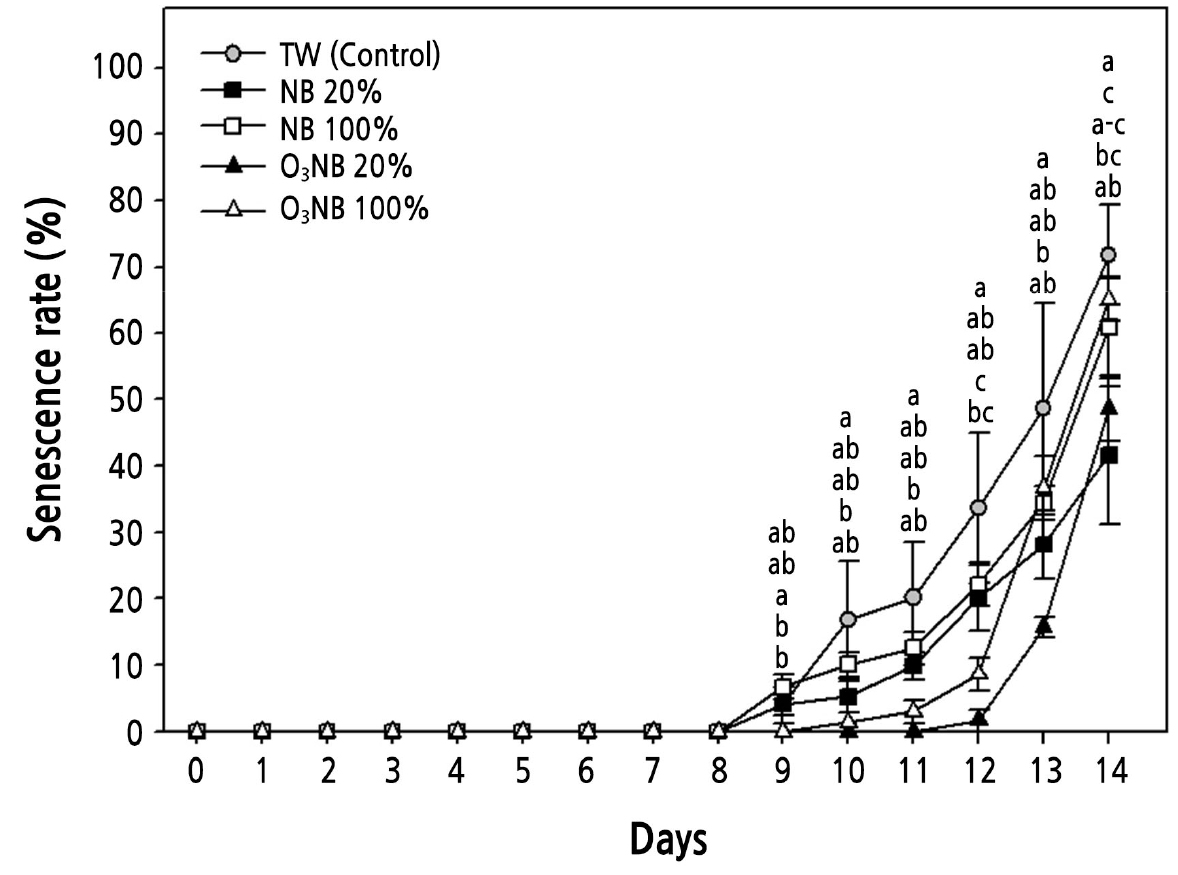
Fig. 4.
Changes in the senescence rate of cut Cymbidium ‘Spring Pearl’ treated with tap water (TW) and with various levels of nanobubbles (NBs) and ozone nanobubbles (O3NBs). The ozone and dissolved oxygen concentrations were 0.0 and 10.0 mg·L-1 (TW, control), 0.0 and 16.3 mg·L-1 (NB 20%), 0.0 and 38.3 mg·L-1 (NB 100%), 0.7 and 16.4 mg·L-1 (O3NB 20%), and 3.8 and 38.3 mg·L-1 (O3NB 100%), respectively. The vertical bars represent the standard error of the mean value (n = 5). The different letters represent statistically significant differences as determined by Duncan’s multiple range test (p < 0.05).
The concentration of dissolved O3 kept higher and the half-life to maintain the maximum concentration of dissolved O3 was longer when O3 is combined with micro-bubbles (Kwack et al., 2014), and O3NB can used for the high stability of O3. In cut Cymbidium treated with O3NB 20% solution, the RFW was 1.7-fold higher on day 2 than on day 0, and the highest RFW value was maintained throughout the experiment except on day 14 (Fig. 3). The RFW was increased more in cut roses treated with ozonated water than with de-ionized water (Robinson et al., 2009). The O3NB 20% treatment remarkably extended the RFW and slowed down the weight loss. The senescence rate of cut Cymbidium treated with O3NB 20% was significantly lower than in control from days 10 to 14 (Fig. 4), which may be related to the stability of O3. Excessive concentrations of O3 in water adversely affected bean sprout and chrysanthemum growth, caused visible injury to eggplant, and the freedom of motion in biological membranes is lost after oxidative stress (Kim et al., 2005; García et al., 2014; Li et al., 2019). A high level of O3 generates more hydroxyl radicals producing reactive oxygen species (ROS) that have a vital role in the senescence of cut flower petals (Jing and Li, 2015). High levels of various oxygen free radicals and hydrogen peroxide (H2O2) have been reported in petal tissues and plants after exposure to higher than usual oxygen concentrations during senescence of flowers, including Gladiolus, Hemerocallis, and Chrysanthemum (Halliwell, 2006; Ahmad and Tahir, 2016). The single O3 concentrations in the vase solution cannot be measured due to technical reasons in our study. However, the combination of O3 and NBs especially 20%, in the vase solution maintained the quality of cut Cymbidium.
The a* value was significantly lower in cut Cymbidium treated with O3NB 20% on day 9, which means that the flower color of cut Cymbidium under the O3NB 20% treatment remained greener than in the other treatments (Table 2). The b* value was significantly higher in cut Cymbidium treated with O3NB 20% than in the NB 100% and O3NB 100% treatments on day 9. This means that the color of cut Cymbidium under the O3NB 20% treatment remained yellower than in the plants treated with NB 100% and O3NB 100%. Vivid coloration is a critical factor for high-quality cut flowers, and the onset of senescence is often readily visible as a color change, petal rolling, and wilting (Jiang and Reid, 2012; An et al., 2015; Ma et al., 2018). Senescence symptoms are wilting, abscission, or organ shedding of cut flowers related to physiological disorders such as early wilting of flowers and leaves and bending of the peduncle (In et al., 2015; Ha et al., 2020). The petals of cut Cymbidium shrunk inside or outside and changed color as senescence progressed (Suh et al., 1998). The petals of florets in cut Cymbidium treated with NB 100% and O3NB 100% wilted and shrunk more than those treated with NB 20% and O3NB 20%, as determined by visual observation (Fig. 5).
Table 2.
The L*, a*, and b* values in the petals of cut Cymbidium ‘Spring Pearl’ treated with tap water (TW) and with various levels of nanobubbles (NBs) and ozone nanobubbles (O3NBs)
| Treatmentsz | Petal colory | ||
| L* | a* | b* | |
| TW (Control) | 63.8 ax | 0.7 a | 34.9 ab |
| NB 20% | 64.8 a | 0.4 a | 34.5 ab |
| NB 100% | 63.4 a | –0.1 a | 33.3 b |
| O3NB 20% | 64.0 a | –1.6 b | 35.7 a |
| O3NB 100% | 64.2 a | 0.2 a | 32.7 b |

Fig. 5.
Morphological changes of cut Cymbidium ‘Spring Pearl’ treated with tap water (TW) and 20% or 100% of nanobubbles (NBs) and ozone nanobubbles (O3NBs) on day 3, 6, 9, 12, and 15. The ozone and dissolved oxygen concentrations were 0.0 and 10.0 mg·L-1 (TW, control), 0.0 and 16.3 mg·L-1 (NB 20%), 0.0 and 38.3 mg·L-1 (NB 100%), 0.7 and 16.4 mg·L-1 (O3NB 20%), and 3.8 and 38.3 mg·L-1 (O3NB 100%), respectively. The vertical bars represent the standard error of the mean value (n = 5). The different letters represent statistically significant differences as determined by Duncan’s multiple range test (p < 0.05).
Since vase life is an essential consideration when people purchase cut flowers, delaying flower senescence and prolonging ornamental life are important (Zhao et al., 2018). The vase life of cut Cymbidium treated with NBs and O3NBs was increased relative to the control regardless of the NB percentage in each treatment (Table 3). The vase life of cut Cymbidium treated with NB 20% and O3NB 20% was significantly prolonged by 1.4 days compared to the control. In previous studies, aqueous O3 of 5.5 mg·L-1 was used to prolong the vase life of cut rose by 13.0 days relative to the control, and the vase life of Dendrobium and Mokara was extended when treated with 5.2 nL·L-1 of O3 (Robinson et al., 2009; Almasi et al., 2015). The vase life of cut Cymbidium treated with NB 100% and O3NB 100% was increased relative to the control, but there was no significant difference. The 5% hydrogen nanobubble water (HNW) has been shown to increase the vase life of cut carnation relative to other treatments, including 1%, 5%, 10%, and 50%, by reducing ROS accumulation, suggesting that HNW is effective at increasing cut flower vase life within a specific range (Li et al., 2021). The O3NB and NB were effective at 20% to increase the vase life of cut Cymbidium compared to the control.
Table 3.
Vase life of cut Cymbidium ‘Spring Pearl’ treated with tap water (TW) and with various levels of nanobubbles (NBs) and ozone nanobubbles (O3NBs)
| Treatmentsz | Vase life (days)y |
| TW (Control) | 13.60 bx |
| NB 20% | 15.00 a |
| NB 100% | 14.40 ab |
| O3NB 20% | 15.00 a |
| O3NB 100% | 14.20 ab |
zThe ozone and dissolved oxygen concentrations were 0.0 and 10.0 mg·L-1 (TW, control), 0.0 and 16.3 mg·L-1 (NB 20%), 0.0 and 38.3 mg·L-1 (NB 100%), 0.7 and 16.4 mg·L-1 (O3NB 20%), and 3.8 and 38.3 mg·L-1 (O3NB 100%), respectively.
Here, we examined the effects of NBs and O3NBs for extending vase life and improving the visual quality of cut Cymbidium ‘Spring Pearl’. The water uptake and RFW were increased and, petal coloration was maintained by delaying senescence of cut Cymbidium ‘Spring Pearl’ when O3NB 20% was added to the vase solution consisting of O3 and DO levels of 0.7 and 16.4 mg·L-1, respectively.



