Introduction
Materials and Methods
Plant Material and Growing Conditions
Chlorophyll Fluorescence Measurement
Measurement of Chlorophyll Content
Measurement of Proline Concentration
Measurements of Growth Parameters
Authentic Standards and Chemicals
Statistical Analysis
Results and Discussion
Variation in Photosynthetic Parameters
Variation in Chl and Proline Content, and Growth Parameters
Introduction
Abiotic stress, such as temperature, salinity, drought, or inadequate crop management, may reduce crop yield by as much as 80% (Boyer, 1982; Anjum et al., 2011). Among stress conditions, temperature is an important abiotic factor that can severely limit morphological, physiological, and biochemical performance of plants, thus inhibiting plant growth and development, and significantly reducing crop productivity (Wahid et al., 2007; Ortiz et al., 2008; Feller and Vaseva, 2014). Temperature regimes above or below the optimal range of a species may significantly affect and even interrupt metabolic processes, including photosynthesis, respiration, and development, ultimately affecting final yield. The photosynthetic process is temperature-sensitive and genotype-dependent (Berry and Bjorkman, 1980; Wang et al., 2009; Mathur et al., 2011). Carbonfixation and the photosystem II (PSII) complexes are regarded as the most heat-sensitive components of the photosynthetic apparatus (Ogweno et al., 2009). Plants experiencing high or low temperature stress respond by increasing the photo-oxidation capacity and reducing the CO2 assimilation capacity. The primary target of thermal damage in plants is the PSII oxygen-evolving complex, along with the associated cofactors (Allakhverdiev et al., 2008). Measurement of chlorophyll (Chl) fluorescence, a non-invasive technique, can provide insight into the ability of a plant to tolerate environmental stress and the extent of damage to the photosynthetic apparatus by adverse conditions (Maxwell and Johnson, 2000; Baker and Rosenqvist, 2004; Gorbe and Calatayud, 2012; Zhou et al., 2018); therefore, Chl fluorescence can be used as a parameter to detect and quantify temperature-induced changes in the photosynthetic apparatus (Wang et al., 2009).
Many studies have reported on the effect of environmental stress on Chl fluorescence parameters, Chl content, and several other physiological responses in different plant species (Weng and Lai, 2005; Partelli et al., 2009; Zribi et al., 2009; Yang et al., 2011; Olvera-Gonzalez et al., 2013; Toomey, 2013; Feng et al., 2014). The use of Chl fluorescence to determine the photosynthetic response to temperature stress has been reported for many plants, such as coffee (Coffea sp.) (Partelli et al., 2009), fingered citron (Citrus medica var. sarcodactylis) (Chen et al., 2012), chrysanthemum (Chrysanthemum sp.) (Janka et al., 2013), wheat (Triticum sp.) (Sharma et al., 2014), basil (Ocimum basilicum L.) (Kalisz et al., 2016), tomato (Solanum lycopersicum) (Nankishore and Farrell, 2016), and rice (Oryza sativa) (Phyo and Chung, 2017). However, studies concerning the analysis of Chl fluorescence in paprika (Capsicum annuum L.), a popular vegetable that contains beneficial phytochemicals of medicinal value such as carotenoids, capsaicinoids, flavonoids, vitamins (C and E), and phenolics (Kim et al., 2011; Kim et al., 2015), have not yet been performed. Furthermore, there is limited information about the effect of cold temperature on fluorescence parameters in plants (Partelli et al., 2009) and little is known about the effect of long-term high or low temperatures on Chl fluorescence parameters.
In this study, we evaluated the effect of temperature stress (low and high) on the photosynthetic parameters, growth status, and proline and Chl content in paprika plants grown under different temperature conditions in a closed plant production system at Chonbuk National University, Jeonju.
Materials and Methods
Plant Material and Growing Conditions
Paprika (Capsicum annuum L.) cultivar ‘Nagano’ was used for this study. Seeds were sown on rockwool plugs and 30-day-old seedlings were transplanted into plastic pots filled with peat. Seedlings were grown for 3 d in a closed plant production system (Synersys Co. Ltd., Jeonju, Korea) for optimization of the plants under more controlled conditions. Temperature and humidity were controlled by a proportional-integral-derivative (PID) controller (Honeywell Co. Ltd., Seoul, Korea) and a humidity controller (M-Tech Corp, Gyeonggi-do, Korea), respectively. The conditions were set as: a 25/20°C (day/night) temperature, a 16/8 h light/dark photoperiod, a photosynthetic photon flux density (PPFD) of 200 µmol·m-2·s-1, and 70% relative humidity. Forty seedlings for each treatment were then grown in a controlled closed plant production system under three temperature conditions, 10, 25, and 35°C with a 10/5, 25/20, and 35/30°C day/night temperature, respectively, for 32 d, where the 25°C temperature treatment was considered a moderate condition (control). Plants were irrigated every day with schematic nutrient solution [Proefstation voor Bloemisterij en Glasgroente (PBG) nutrient solution (N 17.25, P 3.75, K 8.0, Ca 8.0, and Mg 2.75 me·L-1); EC 2.0 dS·m-1, pH 5.7].
Chlorophyll Fluorescence Measurement
Chlorophyll (Chl) fluorescence parameters from the upper surface of all leaves of intact plants were assessed using an open FluorCam 800-O/1010 (Photon System Instruments, Drasow, Czech Republic) using the automatic integration mode. Measurements were taken at 0, 1, 2, 4, 8, 16, 24, and 32 d after the initiation of the temperature treatments after a 3-h illumination of light. On each day of measurement, 5 plants (from the 40 plants of each treatment) were randomly selected and used. Plants were adapted to dark conditions for 15 minutes before the assessment of Chl parameters. Minimal (F0) and maximal Chl a fluorescence (Fm) were assessed; the maximum quantum efficiency of PSII photochemistry was calculated as Fv/Fm = (F0-Fm)/F0. Non-photochemical quenching (NPQ), coefficient of non-photochemical quenching (qN), excitation transfer efficiency from antenna pigments to the reaction center of PSII in the light-adapted state (F’v/F’m), quantum yield of non-regulated energy dissipation in PSII (ϕNO), and the ratio of Chl fluorescence decrease (Rfd) were also assessed. All the leaves were then freeze dried, ground into a fine power, and stored at -80ºC for further analysis of Chl and proline content.
Measurement of Chlorophyll Content
Chl a and b contents were measured using a micro-plate reader (Multiskan GO; Thermo Scientific Inc., Waltham, MA, USA) according to the previously described method (Warren, 2008). The freeze-dried powdered leaf sample (0.1 g) was extracted in 50 mL MeOH (100%); the aliquot was then centrifuged at 3000 rpm for 10 min, and absorbance was measured at 652 and 665 nm. Chl a and b contents were calculated according to a 1-cm corrected path length formula as follows:
Chl a (µg·mL-1) = -8.0962*A652,1 cm + 16.5169*A665,1 cm
Chl b (µg·mL-1) = 27.4405*A652,1 cm-12.1688*A665,1 cm
Measurement of Proline Concentration
Freeze-dried powdered leaf tissue (0.1 g) was extracted in distilled water by shaking for 30 minutes. Next, the extract was centrifuged, filtered through a 0.2-µm syringe filter, injected (10 µL) into a 1260 Infinity high-performance liquid chromatography (HPLC) system (Agilent Technologies, Santa Clara, CA, USA), and analyzed according to Bhandari et al. (2018). Briefly, pre-column automatic derivatization of proline was performed using borate buffer, 9-fluorenylmethyl chloroformate (FMOC), and o-phthalaldehyde (OPA) at first. Then, the proline peak was separated using a Zorbax Eclipse AAA (4.6 × 150 mm, 5 µm, Agilent Technologies, Santa Clara, CA, USA) column at 40°C and a diode array detector (DAD) set at 262 nm for peak detection. The mobile phase was composed of 40 mM sodium phosphate dibasic with solvents 0.1% phosphoric acid (A) and acetonitrile:methanol:water (45:45:10) (B) used for elution of proline at a flow rate of 1.5 mL·min-1. The gradient consisted of a constant flow of 60% solvent B for 3.0 min followed by a linear stepwise increase from 60 to 90% within 5 min. Then, solvent (B) was maintained at the same conditions for 8 min, quickly dropped to 60% for 10 min, and maintained at an isocratic condition of 60% for 15 min. Proline was identified and quantified using commercial standard L-proline with a linear range of 0.0-50 µg·mL-1.
Measurements of Growth Parameters
At the end of the experiment (32 d after the initiation of the treatments), plant height and shoot and root fresh weight were also measured from the five respective plants of each temperature treatment. Plant height was measured from the soil surface to the top of the plant. The shoot and root fresh weight were measured using a digital scale (UX420H, Shimadzu Corporation, Kyoto, Japan).
Authentic Standards and Chemicals
Methanol and acetonitrile were purchased from Avantor Performance Materials Co. (Center Valley, PA, USA). L-proline, phosphoric acid, and sodium phosphate dibasic were obtained from Sigma-Aldrich (St. Louis, MO, USA). Borate buffer, 9-fluorenylmethyl chloroformate (FMOC), and OPA (o-phtalaldehyde) were purchased from Agilent Technologies (Santa Clara, CA, USA).
Statistical Analysis
For the Chl fluorescence and growth parameters, the results are reported as means ± standard deviation (SD) of five biological replicates. The results of Chl and proline contents are reported as means ± SD of three replicates. Data were tested by analysis of variance (ANOVA) and Duncan’s multiple range test was performed to assess statistical differences among the means at p < 0.05 using SPSS version 20 (IBM Corp., Armonk, NY, USA).
Results and Discussion
Variation in Photosynthetic Parameters
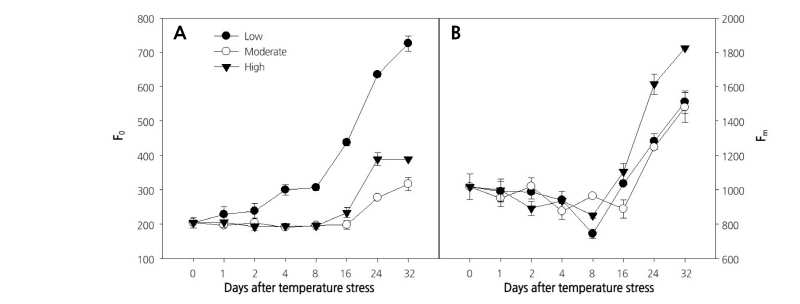
Different photosynthetic parameters were evaluated to study the effect of temperature on the photosynthetic activities in paprika plants. Initial fluorescence intensity (F0) measured in the dark-adapted state, when all PSII reaction centers are open, has been used as a thermo-injury index. A sudden change in F0 means that photosynthetic membranes have suffered irreversible injury (Georgieva and Yordanov, 1993). In this study, seedlings grown at low temperature showed a continuous increase in F0 from the first day (203.5) until the end of the experiment (726.5), with a sharp increase at 8 d after the initiation of treatment, which was caused by the physical separation of the PSII reaction centers from their pigment antennae and alternations in the capacity to trap radiation energy (Briantias et al., 1996). Although seedlings grown at moderate or high temperature also showed an increase in F0 after 8 d of treatment, the effect was lower than that reported for the low temperature treatment (Fig. 1A). The highest increase in maximum Chl fluorescence (Fm) was recorded for leaves of seedlings grown at the higher temperature treatment (Fig. 1B).
Maximum quantum yield (Fv/Fm) represents the capacity for using the energy of radiation absorbed by PSII in a photochemical process in the dark-adapted state (Oxborough, 2004). The Fv/Fm decreased continuously from 0.81 at zero days to 0.518 at the end (32 d) of the experiment (~35% decrease) under the low temperature treatment (Fig. 2A). In contrast, the Fv/Fm remained at a similar level throughout the experiment (~0.80) in plants grown under moderate or high temperature. These results suggest that high temperature had less impact on photosynthetic activity than low temperature in paprika seedlings. The significant decrease in Fv/Fm at low temperature from the 4th day of treatment indicates that seedlings were under severe stress (Baker and Rosenqvist, 2004), and that the photochemical efficiency of PSII was severely impaired, revealing that low temperature has significant effects on the photochemistry of PSII and results in photoinhibition. The photochemistry of PSII and photoinhibition are associated with the structural modification of PSII, especially involving protein D1 (Asada et al., 1998). Furthermore, the sharp decrease in the Fv/Fm at low temperature was due to the increase in F0 under the stress condition. The decline in Fv/Fm involves an increase in F0 (Yamada et al., 1996). Our result is consistent with those reported by Partelli et al. (2009), who recorded a continuous reduction of Fv/Fm in coffee grown at low temperature. Our results also agreed with those of Hogewonning and Harbinson (2007), who observed photoinhibition in variegated leaves of Calathea makoyana during chilling stress. On the other hand, the higher temperature in our experiment might not have been high enough to cause the changes in Fv/Fm that were reported by many other researchers, which generally occurred above 40°C (Haldimann and Feller, 2004; Chen et al., 2012; Janka et al., 2013; Phyo and Chung, 2017). In addition, the results of this study did not agree with a previous report by Ogweno et al. (2009) who observed a continuous decrease in Fv/Fm at high temperature but a stable Fv/Fm at low temperature in tomato leaves. Janka et al. (2013) also reported a decreased Fv/Fm in chrysanthemum at high temperature. These discrepancies might be due to the differences in plant genotypes, as heat susceptibility is dependent on plant genotype (Camejo et al., 2005; Wahid et al., 2007). Thus, further research under high temperature conditions is required to better understand the effect of high temperature on Chl fluorescence parameters in the paprika plant.
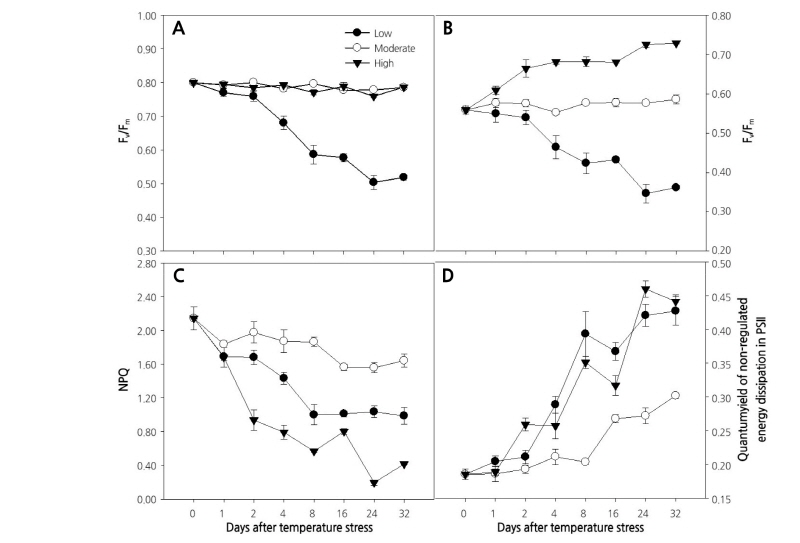
Fig. 2. Changes in maximum quantum yield (Fv/Fm) [A], excitation transfer efficiency from antenna pigments to the reaction center of PSII in the light-adapted state (F’v/F’m) [B], non-photochemical quenching (NPQ) [C], and quantum yield of non-regulated energy dissipation in PSII (ϕNO) [D] in paprika plants grown under different temperature conditions for 32 days. Values are means ± standard deviation (SD) of five biological replicates.
The operating photon efficiency of PSII photochemistry in the light-adapted state (F’v/F’m) remained unaltered throughout the experiment in plants grown at moderate temperature, whereas the value for F’v/F’m decreased from the beginning to the end of the experiment at low temperature (0.56-0.35), which was in agreement with the observations by Partelli et al. (2009) in coffee. In contrast, plants grown at high temperature showed a continuous increase in F’v/F’m until the second day of the treatment, then it remained unchanged until the 16th day and again increased (Fig. 2B). The results were inconsistent with those by Ogweno et al. (2009), who found a continuous reduction in F’v/F’m in detached tomato leaves treated at high or low temperature for 5 d. Heat stress inhibits the protection mechanism of PSII, which is reflected by a decrease in non-photochemical quenching (NPQ) (Schreiber and Klughammer, 2008). NPQ also decreased from the beginning to the end of the experiment at the low and high temperatures compared to the moderate temperature (Fig. 2C), suggesting that the plants were under stress at the high and low temperatures. After 8 d of treatment, NPQ was stable until the end of the experiment at the low temperature, while it showed an inconsistent decreasing pattern at the high temperature. In contrast to NPQ, the quantum yield of non-regulated energy dissipation in PSII (ϕNO) showed an increasing pattern and peaked (0.30-0.44) at the end of the experiment in all three temperature treatments tested (Fig. 2D). Plants grown at high and low temperature showed a higher increase in ϕNO than those grown at moderate temperature. The increase of ϕNO, even at the moderate temperature, was probably due to the age of the plant and leaves, as the age of the plants and leaves are reportedly responsible for the increase in ϕNO (Bielczynski et al., 2017).
Effective quantum yield of photochemical energy conversion in PSII (ϕPSII) represents the capacity for use of the energy of radiation captured by PSII in a photochemical process by light-adapted plants (Oxborough, 2004). Effective quantum yield increased on the first day of temperature treatment and continuously decreased until the end of the experiment, regardless of temperature (data not shown). The coefficient of non-photochemical quenching of variable fluorescence (qN) showed a similar trend as the maximum quantum yield, remaining stable throughout the experiment at the moderate temperature treatment (0.74-0.79) (Fig. 3A). Similarly, plants grown at low temperature also showed a stable qN during the experiment, while it decreased from 0.79 to 0.21 under the high temperature condition. The ratio of Chl fluorescence decrease (Rfd) under moderate temperature remained stable throughout the experiment, although a slight decrease was observed at the end (Fig. 3B). On the other hand, the Rfd decreased continuously under the low and high temperature treatments, with a higher decrease recorded at the high temperature (2.34 to 0.71) indicating a decline in photosynthetic quantum conversion (Lichtenthaler et al., 2005).
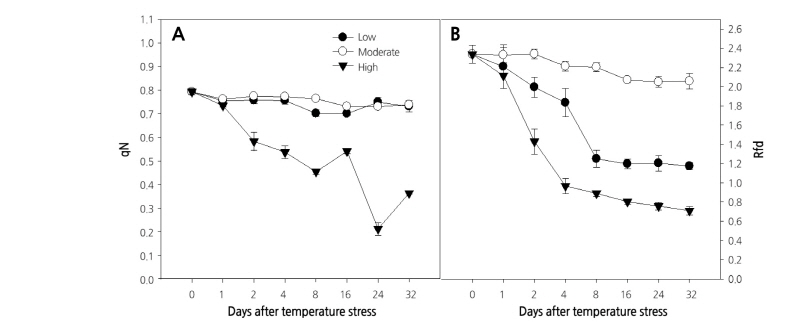
Fig. 3. Changes in the coefficient of non-photochemical quenching of variable fluorescence (qN) [A], and the ratio of the fluorescence decrease (Rfd) [B] in paprika plants grown under different temperature conditions for 32 days. Values are means ± standard deviation (SD) of five biological replicates.
Overall, Chl fluorescence parameters, such as F0, Fv/Fm, and F’v/F’m, were significantly affected under low temperature, whereas NPQ, ϕNO, and Rfd, were all significantly affected at low and high temperatures. Further, qN and Fm had the greatest change under the high temperature condition. Inconsistencies in some parameters in this experiment were probably due to the longer duration of the experimental period and the age of the plants. All these results imply that the photosynthetic activities in paprika plants are affected by high and low temperature stress in different ways; thus, special consideration should be given to the use of the appropriate fluorescence parameters for stress detection in the paprika plant.
Variation in Chl and Proline Content, and Growth Parameters
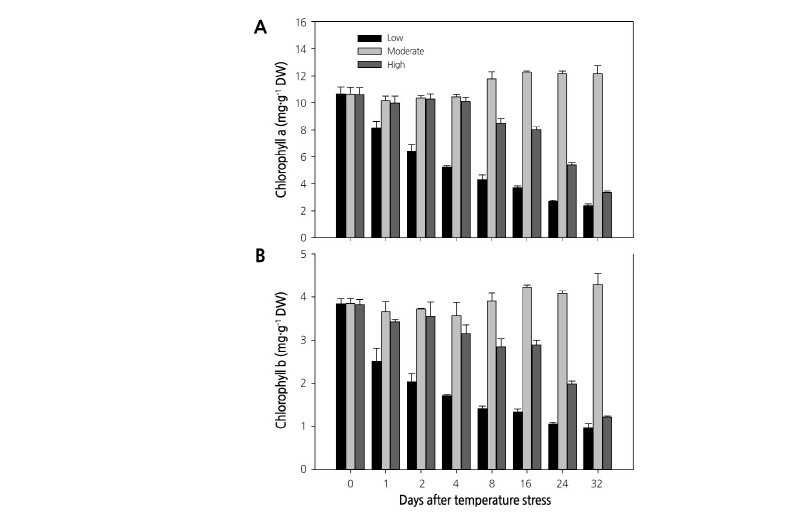
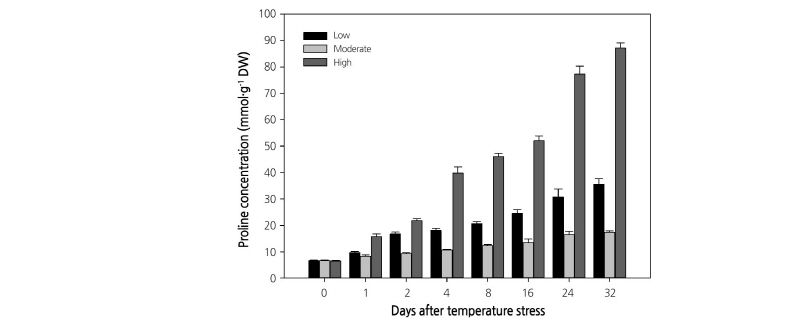
Temperature stress also altered the Chl content. The Chl a and b content in leaves showed a different accumulation pattern throughout the experiment. Plants grown at moderate temperature showed a similar Chl content throughout the experiment, while the Chl content decreased continuously from the start to the end of the experiment in plants growing at the high or low temperatures (Fig. 4). A similar decrease in Chl content was previously observed at low and high temperatures in Coffee (Partelli et al., 2009) and Chenopodium album (Haq et al., 2013). Among the three treatments, plants growing at the low temperature showed the lowest Chl content (Chl a: 10.7 to 2.4 mg·g-1, and Chl b: 3.8 to 1.0 mg·g-1) throughout the experiment. Plant height and fresh shoot and root weights on the 32nd day of treatment also showed significant changes (Table 1). The maximum plant mean height was recorded at moderate temperature (13.64 cm), which was followed by the height at high (11.34 cm) and low temperature (7.6 cm). Shoot and root fresh weight showed a similar trend, with the highest value reported for plants grown under the moderate temperature and the lowest value for plants grown under low temperature. Proline, a major compatible solute (Szabados and Savoure, 2010), is generally found in large amounts under any stress conditions (Trovato et al., 2008; Verslues and Sharma, 2010). In this experiment, proline was accumulated in all the temperature treatments (Fig. 5) as it plays a significant role in plant development processes by different ways and found in the plants even in the non-stressed conditions (Trovato et al., 2008). However, the highest proline concentration was observed at the high temperature, followed by the low and moderate temperatures. These results indicate that paprika plants grown at high or low temperature experienced high stress levels, as accumulation of proline is considered a strong indicator of biotic and abiotic stress (Trovato et al., 2008).
In conclusion, the photosynthetic responses, and ultimately the morphological characteristics, proline concentration, and Chl content in paprika seedlings were differentially affected by low, moderate, and high temperature conditions. Fluorescence parameters, such as F0, Fv/Fm, and F’v/F’m, plant growth, and Chl content were more significantly affected under low temperature than under the other two temperature conditions tested here; furthermore, NPQ, ϕNO, Rfd, and proline accumulation showed the highest response to low and high temperature. Therefore, selection of appropriate fluorescence parameters is necessary to identify the changes in plant performance for the successful application of Chl fluorescence measurement in monitoring stress incidence in paprika. Research for a shorter duration using a higher range of temperature treatments may be useful to identify quick responses to temperature by paprika photosynthetic performance.