Introduction
Potential and Limitations of Plant Cell Culturing
Production of functional materials via plant cell Culturing
Limitations and challenges in the industrial utilization of plant cell culturing
Biological Characteristics and Significance of CMCs and Calli with Stem Cell-like Features for the Large-scale Production of Functional Natural Products
Production of Functional Materials through calli with stem cell-like features of Three Korean Native Plants: A Four-Stage Framework
Step 1: Optimization of solid culture conditions for the induction of calli with stem cell-like features
Step 2: Biochemical and morphological approaches for the characterization of calli with stem cell-like features
Step 3: Establishment of liquid culture systems for the mass production of functional compounds derived from calli with stem cell-like features
Step 4: LC-MS/MS-Based quantification of functional compounds in production systems of calli with stem celll- like features
Plant Stem Cell and Bioprocess Research: Recent Advances in Single-Cell, CMCs, and AI Applications
Recent advances in single-cell–based plant stem cell research
Emerging directions in cambial meristematic cells (CMCs) platforms and AI utilization
Future Perspectives : AI-Guided Optimization of Natural Product Biosynthesis in Plant Stem Cell Culturing for Industrial Applications
Conclusion
Introduction
Plant secondary metabolites are vital for plant defense against both environmental challenges and biological forms of stress, thereby improving plant resilience and adaptability. In addition to their ecological functions, these compounds have significant economic value, supporting extensive applications in the food, pharmaceutical, and cosmetic sectors (Ravishankar and Ramachandra 2000; Yun et al. 2012; Kim 2013; Pagare et al. 2015; Bapat et al. 2023). The increasing preference for naturally sourced functional ingredients highlights their growing industrial importance.
Traditional extraction techniques from whole plants are hampered by limitations such as low yields, variable quality levels and environmental impacts related to overharvesting (Yun et al. 2012), highlighting the necessity for more sustainable and scalable production approaches. Plant cell and tissue culture methods have emerged as effective alternatives (DiCosmo et al. 1989; Canter et al. 2005), with cambial meristematic cell (CMC) cultures—often called plant stem cell cultures—demonstrating especially notable potential (Ye 2002; Lee et al. 2010; Yun et al. 2012).
CMCs, found in the meristematic regions including the shoot apices, root tips, and vascular cambia, maintain pluripotency, indefinite division, and the ability to generate genetically uniform cultures (Laux 2003; Heidstra and Sabatini 2014; Sozzani and Iyer-Pascuzzi 2014; Greb and Lohmann 2016). These properties directly address the challenges associated with dedifferentiated cells (DDCs), such as cellular heterogeneity, decreased metabolite production, and cell clumping in suspension cultures (Lee et al. 2010; Ochoa-Villarreal et al. 2015). Despite their advantages, fully authenticated CMC cultures have only been produced in a limited number of species; in practice, cultures that possess merely stem cell-like qualities are obtained (Yun et al. 2012; Ochoa-Villarreal et al. 2015; Kaňuková et al. 2024).
Research efforts since 2020 have broadened the application of cambial meristematic cell (CMC) cultures across a variety of plant taxa. Studies have improved triterpenoid accumulation in Ocimum basilicum, demonstrated metabolomic and transcriptomic confirmation of enhanced catalpol biosynthesis in Rehmannia glutinosa, and the use of CMCs alongside calli with stem cell-like features in Fraxinus mandshuricaandCalendula officinalis (Mehring et al. 2020; He et al. 2021; Zhou et al. 2023; Kaňuková et al. 2024; Partap et al. 2023).
This review summarizes recent progress in the development of culture systems utilizing CMCs and calli with stem cell-like features for the production of secondary metabolites, while presenting specific case studies conducted in our laboratory. Using three native Korean species—Artemisia capillaris (Park et al. 2023a), Ixeridium dentatum (Park et al. 2023b), and Perilla frutescens (Park et al. 2024; Lee et al. 2024)—we outline a practical workflow consisting of four key stages: (1) initiating calli with stem cell-like characteristics through optimization of solid media, (2) confirming these features using catalase assays and TEM imaging, (3) refining liquid culture conditions to enable large-scale metabolite synthesis, and (4) conducting comprehensive qualitative and quantitative analyses of metabolites. This approach establishes a robust foundation for the scalable production of functional metabolites using calli with stem cell-like features.
Potential and Limitations of Plant Cell Culturing
Production of functional materials via plant cell Culturing
Plant tissue culture methodologies—including callus, suspension, and organ cultures—offer sustainable in vitro platforms for the generation of natural bioactive compounds while minimizing the ecological impact (Choi et al. 2002; Yun et al. 2012; Efferth 2019; Bapat et al. 2023). These approaches serve as enabling tools for both research and industrial applications by supporting the standardized and controlled synthesis of metabolites, independent of environmental factors such as climate or soil variabilities (Rao and Ravishankar 2002; Efferth 2019). They facilitate the long-term preservation of cell lines from a wide range of species, including those threatened by extinction (Roberts 2007; Efferth 2019). The extraction processes are simplified, use fewer hazardous solvents, and comply with GMP requirements, thereby enhancing their suitability for pharmaceutical and cosmetic uses (Davies and Deroles 2014).
A widely cited example is paclitaxel, an anticancer compound originally isolated from Taxus brevifolia, whose wild populations were threatened by excessive harvesting. While semisynthetic methods using yew leaves are still limited by low yields and complex extraction processes, employing the plant cell culture methodology has enabled efficient, scalable, and sustainable production (Zhu and Chen 2019). In another case, shikonin—a red pigment from Lithospermum erythrorhizon with antimicrobial and wound-healing activities—faces the challenges of slow plant growth and low yields (1–2%). Plant cell culture techniques have overcome these barriers by significantly improving production efficiency rates (Zhang et al. 2010; Malik et al. 2016; Yazaki 2017; Sargazi Moghaddam et al. 2025).
Limitations and challenges in the industrial utilization of plant cell culturing
DDCs, derived from reprogrammed plant cells, frequently display both genetic and epigenetic variability, which can result in diminished proliferation rates and inconsistent metabolite production (Baebler et al. 2005; Law and Suttle 2005; Grafi et al. 2007; Pauwels et al. 2009). Unlike microbial platforms, plant cell cultures are characterized by marked heterogeneity, with productivity fluctuations reaching up to tenfold during successive subcultures (Ketchum et al. 1999; Thorpe 2000; Chattopadhyay et al. 2002). As an illustration, in Camellia sinensis callus lines, catechin concentrations declined from 1625 to 633 mg/L over 2.5 years, accompanied by coefficients of variation between 33 and 48% (Esteban-Campos et al. 2024).
The presence of large vacuoles and rigid cell walls in plant cells imparts structural robustness but also heightens their vulnerability to osmotic and mechanical stress. Exposure to such stress can cause vacuole leakages, by which sequestered metabolites or toxins are released, ultimately compromising cell viability (Jiang et al. 2021). Sensitivity to shear stress poses major challenges in stirred-tank bioreactors; although wave-type systems reduce some of these issues, they do not fully address the issues of culture instability, inhibited growth, or reduced metabolite accumulation (Eibl and Eibl 2007; Eibl et al. 2018; Wilson and Roberts 2020). To improve the resilience of cultures and limit performance variability, selective cultivation of shear-tolerant lines has been advocated (Ochoa-Villarreal et al. 2016; Verdú-Navarro et al. 2023; Titova et al. 2024).
Cell aggregation constitutes an additional significant challenge. Incomplete cell separation, coupled with polysaccharide-mediated adhesion, results in size-heterogeneous aggregates that give rise to sedimentation, inadequate mixing, and restricted nutrient transfer (Joshi et al. 1996; Naill and Roberts 2004). Experimental results on Ficus deltoidea suspensions demonstrated that the aggregate size, initial cell density, and medium pH strongly impacted both biomass and flavonoid productivity (Haida et al. 2019). Conventional methods—including enzymatic treatments, osmotic adjustment, and medium replacement—have achieved only limited and highly species-dependent effectiveness. Recently, engineering-based solutions such as population balance modeling have been implemented to characterize aggregate distributions and refine shear regimes, highlighting that robust management of aggregation is critical for achieving uniform large-scale production of valuable metabolites (Wilson and Roberts 2020; Verdú-Navarro et al. 2023).
Biological Characteristics and Significance of CMCs and Calli with Stem Cell-like Features for the Large-scale Production of Functional Natural Products
Plant cell culturing offers a sustainable approach for synthesizing functional natural products; however, delete industrial implementation of this method is frequently limited by low yields, instability, and difficulties that impede scale-up efforts. CMCs, the stem cells present in vascular tissues, present clear benefits such as a high proliferation capacity, reduced aggregation, enhanced stress tolerance, and robust responsiveness to elicitors (Lee et al. 2010; Ochoa-Villarreal et al. 2015; Partap et al. 2023). When cultured, they produce genetically stable and homogeneous cell lines with consistent metabolite yields (Ye 2002; Yun et al. 2012). Nevertheless, authentic CMC cultures have thus far been established for only a limited number of taxa.
As a pragmatic solution, calli with stem cell-like features can mimics many of the advantages of CMCs while being suitable for a wider range of plant species. In Calendula officinalis, optimized protocols yielded calli suspensions containing 72–93% stem cell-like cells (Kaňuková et al. 2024). Therefore, both CMCs and calli with stem cell-like features constitute complementary approaches for addressing the instability and heterogeneity characteristics of dedifferentiated cell (DDC) cultures.
CMCs are localized within meristematic regions such as shoot and root apices, where they exhibit theoretically limitless division and preserve pluripotency necessary for regeneration across the plant's lifespan (Laux 2003; Scheres 2005; Umeda et al. 2021). The presence of mitochondria-rich cytoplasm facilitates rapid cellular proliferation and efficient metabolite biosynthesis. Differentiated cells (DDCs) can also be induced to re-enter the cell cycle through hormonal stimulation, with auxin–cytokinin treatments promoting callus formation and organogenesis (Birnbaum and Alvarado 2008; Ikeuchi et al. 2019). Critical characteristics of CMCs include their high proliferation potential, low sensitivity to shear stress minimal cell aggregation, small vacuole size, and thin primary cell walls (Greb and Lohmann 2016).
Despite these advantages, achieving fully verified CMCs in practical applications continues to be challenging. In our studies, we similarly obtained calli with stem cell-like features rather than true CMCs. While molecular verification of full stemness was lacking, these calli with stem cell-like features were characterized by their small size and numerous vacuoles, clearly paralleling the CMC morphology and serving as a viable alternative for metabolite production.
Production of Functional Materials through calli with stem cell-like features of Three Korean Native Plants: A Four-Stage Framework
Plant stem cell technology holds considerable promise; however, its application to native species is limited due to unique physiological characteristics and challenges in vitro systems. Therefore, developing protocols tailored to specific species is essential for the industrial utilization of native flora. Our laboratory has established optimized methodologies for three Korean species—Artemisia capillaris Thunb., Ixeridium dentatum, and Perilla frutescens—to generate calli with stem cell-like features for the enhanced production of functional metabolites (Park et al. 2023a, 2023b, 2024; Lee et al. 2024). These research outcomes were systematically organized into a four-step workflow, focusing on the industrial applicability of stem cell–based technologies for native plant species (Fig.1) (Lee et al. 2024).
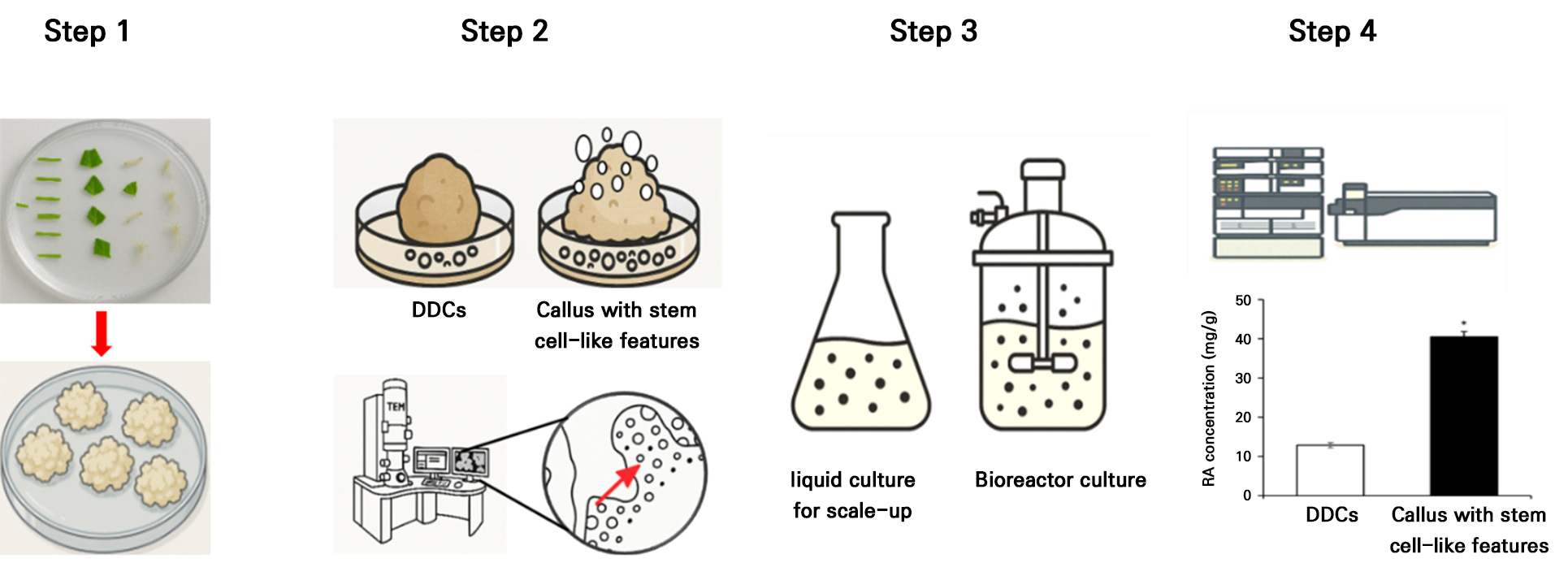
Fig. 1.
Schematic illustration depicting the procedure for establishing calli with stem cell-like properties using Perilla frutescens as a representative model.
Step 1, Initiation of callus from various explants, followed by isolation and serial subculturing of calli exhibiting stem cell-like properties. Step 2, Verification of the stem- cell-like callus by assessing catalase activity and performing microscopic evaluation. Step 3, Optimization of the liquid culture parameters for the high-efficiency, large-scale production of targeted metabolites. Step 4, Quantitative analysis of rosmarinic acid levels in DDCs and calli with stem cell-like features by LC-MS/MS. Data are presented as the mean ± SE (n=3); statistical significance (*p < 0.05) was established using Student’s t-test (Lee et al. 2024).
Step 1: Optimization of solid culture conditions for the induction of calli with stem cell-like features
Step 1 involves selecting suitable explants and cultivars for the induction of calli with stem cell-like features and exploring optimal induction conditions under solid culture systems (Fig.1 Step 1).
Commonly used explants include stems, leaves, petioles, and roots; the success of induction is affected by both the inherent properties of the species and the culture conditions, such as the medium composition, light, pH, and oxygen supply (Efferth 2019; Bojko et al. 2024). Among basal media, MS, White, and WPM are predominantly utilized (White 1939; Murashige and Skoog 1962; Salvo et al. 2014). Sucrose acts as both a carbon source and an osmotic regulator, facilitating the accumulation of secondary metabolites (Martínez et al. 2023; Walker et al. 2021). Among plant growth regulators (PGRs), 2,4-D is extensively applied for callus induction, but its efficacy diminishes at elevated concentrations, necessitating the integration of natural auxins or combinatory PGR strategies (Mahood et al. 2022; Bansal 2025; Abdelazeez et al. 2025).
Shoot and root apical meristems have traditionally been utilized, though their fragility restricts their practical scalability. Alternatively, the vascular cambium is recognized as a reliable and scalable source of CMCs (Lee et al. 2010; Loake and Ochoa-Villarreal 2017), with recent research also demonstrating cambial activity throughout dicot veins and petioles (Lev-Yadun 2025).
In our laboratory, Step 1 involved optimizing the solid-culture conditions to promote the formation of calli with stem cell-like features. Explants (leaves, petioles, stems, and roots) collected from greenhouse-grown plants underwent surface sterilization before being placed on solid media containing selected PGRs. Subsequently a callus with stem cell-like features—white or light yellow, soft, and composed of readily dissociable microgranules—was selected and subcultured. The optimal culture conditions identified across these studies are summarized in Table 1 (Park et al. 2023a, 2023b, 2024; Lee et al. 2024).
Table 1.
Optimal solid and liquid culture media for stem cell induction in various plants (units: mg·L-1) (Park et al. 2023a, 2023b, 2024; Lee et al. 2024)
To distinguish calli with stem cell-like features from dedifferentiated cells (DDCs), multiple criteria were applied, as summarized in Table 2. These included color, morphological characteristics, catalase activity assays, and microscopic observations. Color and morphology were employed as visual indicators that may reflect cellular status, while the catalase assay was included to indirectly evaluate stress response capacity and stem cell-like states through antioxidant enzyme activity. In addition, microscopic observations focusing on vacuolar characteristics were used as a criterion to more precisely examine structural differences between the two cell populations. Figure 2 visually presents the process of inducing calli with stem cell-like features from selected plant species (Fig.2). Using this approach, calli with stem cell-like features was induced from leaf explants of the source plant, Artemisia capillaris (Fig. 2A-1 and A-2). Repeated subculturing enabled the selective enrichment of callus populations maintaining these characteristics (Fig. 2A-3) (Park et al. 2023a). Similarly, calli with stem cell-like features were induced from petiole explants of Ixeridium dentatum (Fig. 2B-1 and B-2), and callus populations maintaining these characteristics were selected through successive subculturing (Fig. 2B-3) (Park et al. 2023b). Meanwhile, in Perilla frutescens, calli with stem cell-like features were also induced from leaf explants (Fig. 2C-1 and C-2), and repeated subculturing enabled the selection of callus retaining the same characteristics (Fig. 2C-3) (Park et al. 2024; Lee et al. 2024).
Table 2.
Experimental approaches and characterization of calli with stem cell-like features and DDCs, involving morphological and biochemical assessments (Lee et al. 2010; Yun et al. 2012; Kaňuková et al. 2024; Lee et al. 2024)

Fig. 2.
Images depicting the derivation of calli with stem cell-like features from selected plant species.
A-1, Artemisia capillaris (source plant); A-2, induction of callus from leaf explants; A-3, subculture to enhance of a callus with stem- cell-like characteristics; B-1, Ixeridium dentatum (source plant); B-2, induction of calli from petiole explants; B-3, subculture to promote enrichment of a callus with stem- cell-like characteristics; C-1, Perilla frutescens (source plant); C-2, induction of callus from leaf explants; C-3, subculture to enrich a callus with stem- cell-like characteristics (71Park et al. 2023a, 722023b, 732024; 52Lee et al. 2024).
Step 2: Biochemical and morphological approaches for the characterization of calli with stem cell-like features
The second step involved verifying the properties of calli with stem cell-like features. Catalase activity was confirmed by detecting the formation of oxygen bubbles upon the application of H2O2 while ultrastructural characteristics were assessed using transmission electron microscopy (TEM) (Fig. 1 Step2) (Lee et al. 2024).
Catalase activity test
Figures 3 shows the results of the catalase activity assay conducted to verify the presence of calli with stem cell-like features (Fig. 3). To determine the presence of calli with stem cell-like features, both compact and friable callus types were analyzed by means of a catalase activity assay. Catalase, an iron-porphyrin enzyme commonly found in CMCs, decomposes hydrogen peroxide (H2O2) into water and oxygen, resulting in visible bubble formation that signals protection against oxidative stress (Du et al. 2008).
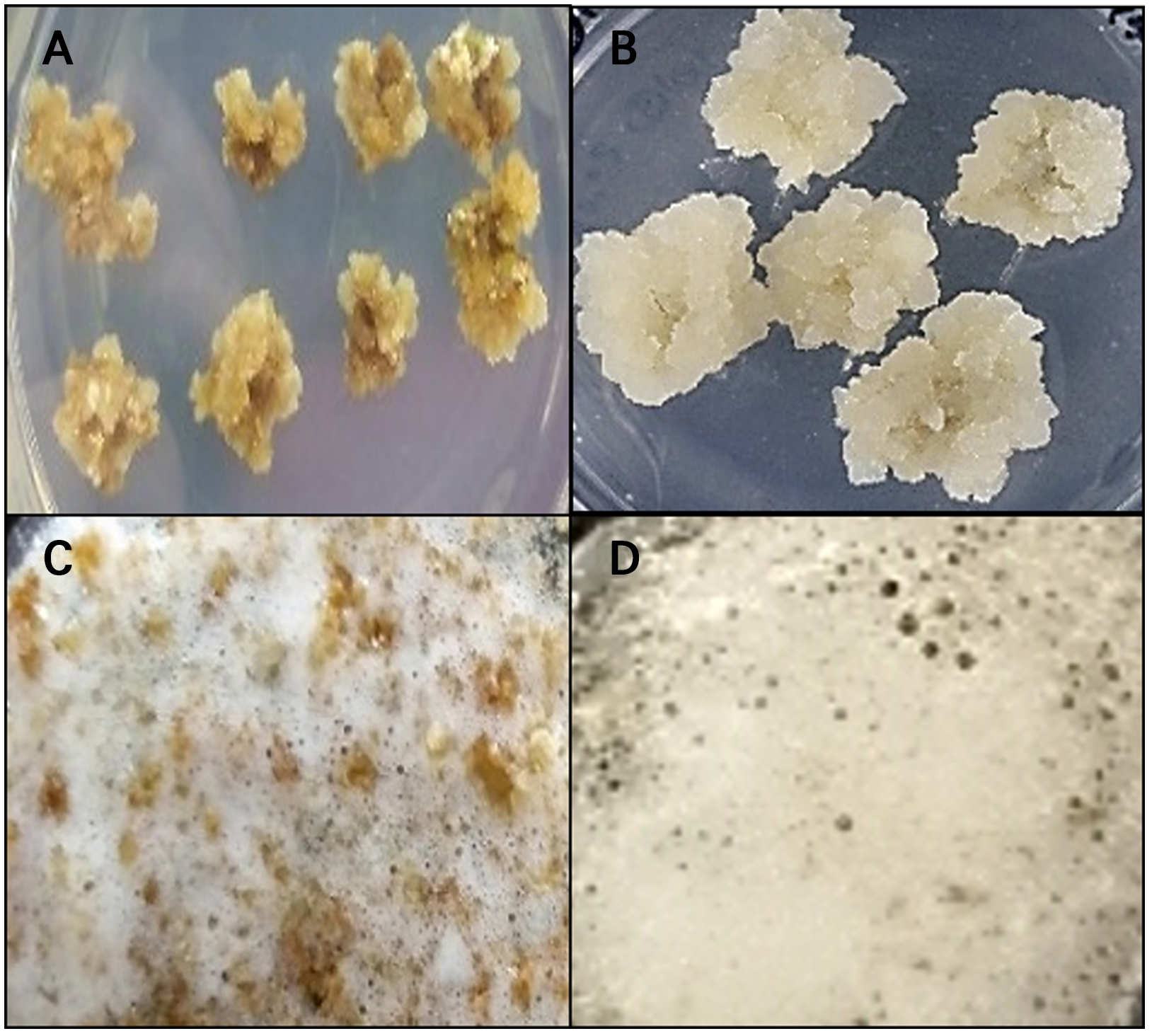
Fig. 3.
Confirmation by catalase activity assessment comparative evaluation of catalase activity between DDCs (A and C) and calli with stem cell-like features (B and D).
DDCs produced bubbles more slowly and in smaller amounts (A, C), whereas calli with stem cell-like features generated bubbles more rapidly and abundantly (B, D) (52Lee et al. 2024).
Consistent with previously reported catalase test results from our laboratory, DDCs exhibited slower and less abundant oxygen bubble formation (Fig. 3A and 3C), whereas calli with stem cell-like features generated bubbles more rapidly and abundantly, indicating higher catalase activity (Fig. 3B and 3D). (Arora et al. 2002; Lee et al. 2010; Ransy et al. 2020; Lee et al. 2024). The catalase activity test results related to these characteristics are summarized in Table 2.
Ultrastructural characterization via transmission electron microscopy (TEM)
TEM analysis revealed clear ultrastructural differences between DDCs and calli with stem cell-like features (Fig. 4). TEM was chosen because it allows high-resolution observation of intracellular structures, enabling precise comparison and evaluation of calli with stem cell-like features and cellular states. DDCs typically formed a single prominent central vacuole, indicating a relatively more advanced state of differentiation. In contrast, calli with stem cell-like features exhibited a friable structure and a pale appearance, along with elevated catalase activity and numerous small vacuoles uniformly distributed throughout the cytoplasm. These characteristics are consistent with features commonly reported for CMCs or calli with stem cell-like features. (Yun et al. 2012; Lee et al. 2024).
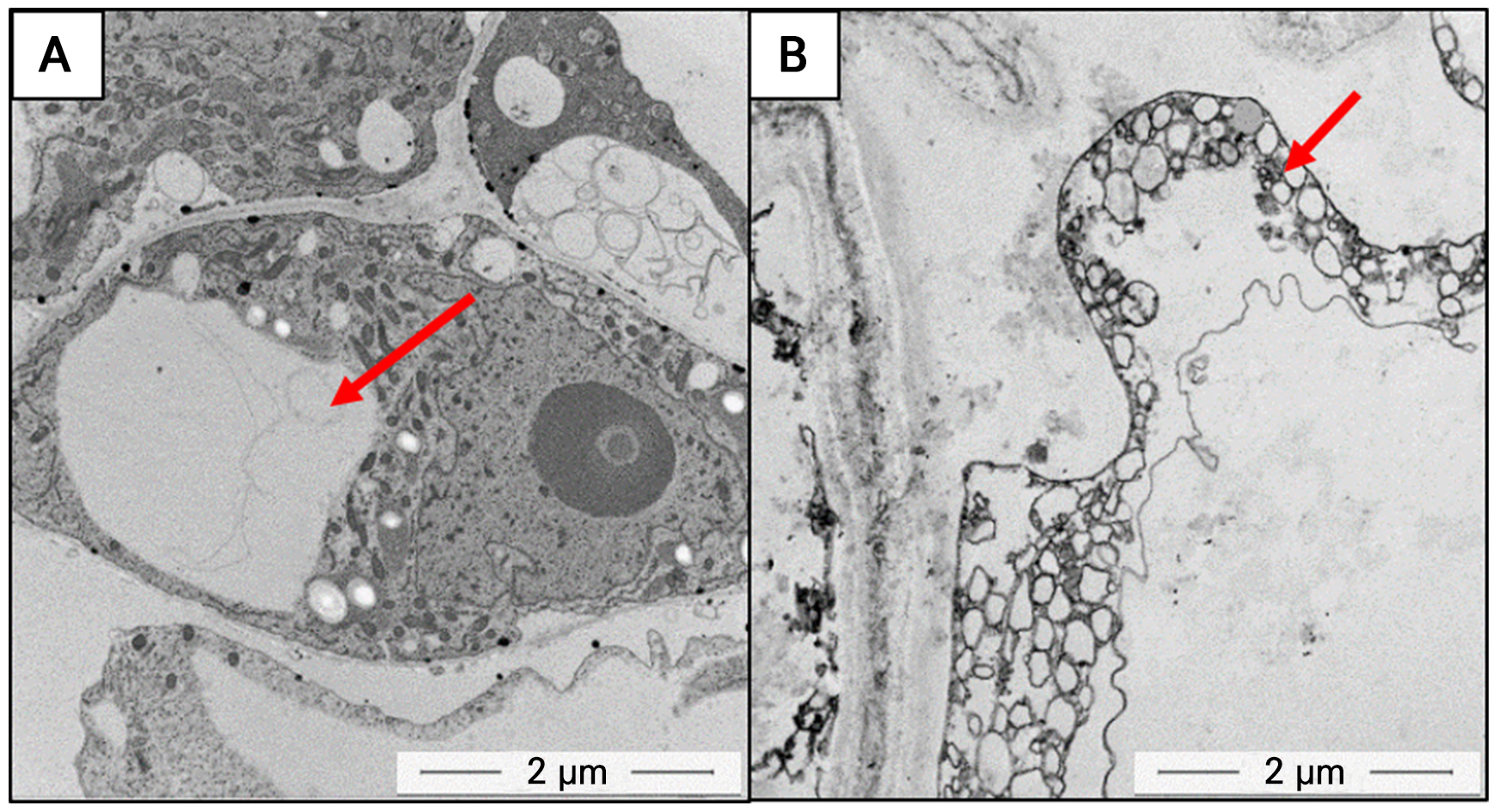
Fig. 4.
Microscopic analysis of DDCs and callus displaying stem cell-like features.
A, DDCs observed at 5000× magnification, displaying large vacuoles. B, calli with stem cell-like features observed at 8000× magnification, showing an abundance of small vacuoles. Vacuoles are marked by red arrows. Figure reproduced with permission from the Korean Journal of Medicinal Crop Science (52Lee et al. 2024).
Cytological observations in our laboratory showed that DDCs typically formed a single prominent central vacuole (Fig. 4A), whereas calli with stem cell-like features displayed numerous small vacuoles evenly distributed throughout the cytoplasm (Fig. 4B). These characteristics suggest that the callus may be classified as CMCs and as calli with stem cell-like features, and are generally consistent with previously reported biochemical and imaging analyses. (Lee et al. 2024). Based on TEM cytological observations, the comparative characteristics between calli with stem cell-like features and dedifferentiated cells (DDCs) are summarized in Table 2.
Molecular identification of plant stem cells: marker genes and recent advances
Molecular validation of plant stem cells has predominantly depended on analysis of established marker genes. In the shoot apical meristem (SAM), a regulatory network involving WUS, CLV1, and CLV3 governs stem cell specification, with CLV3 notably serving as a key molecular marker due to its restricted expression in undifferentiated cells (Fletcher et al. 1999; Fuchs and Lohmann 2020; Han et al. 2020). Within vascular tissues, the cambium acts as a bifacial stem cell layer that produces both the xylem and phloem; however, there are few definitive molecular markers for cambial stem cells. HD-ZIP III transcription factors, TDIF signaling, and auxin signaling peaks have each been linked to the regulation of the fate of cambial stem cells (Hirakawa et al. 2008; Donner et al. 2009; Etchells and Turner 2010; Ursache et al. 2014; Smetana et al. 2019; Smit et al. 2020; Mäkilä et al. 2023), yet a universally accepted marker gene remains elusive. This highlights a significant challenge of current molecular methods for plant stem cell identification in tissue culture and emphasizes the need to expanded the screening and validation of potential candidate genes.
Recently, significant progress has been made in elucidating the identity of cambial stem cells. A study published in Science identified CAIL (Cambium-Expressed AINTEGUMENTA-Like) as a transcription factor activated by the TDIF–PXY pathway, functioning as a key regulator of cambial stem cell identity and ensuring the spatial restriction of TDIF signaling to maintain accurate stem cell positioning (Eswaran et al. 2024). In addition, lineage tracing studies provided direct evidence that a single cell layer in radial tissues carries out authentic stem cell roles (Wybouw et al. 2024), and the sequential changes marking the transition from procambium to cambium have been thoroughly detailed (Wang et al. 2023). Collectively, these studies represent substantial advancements in determining the specific location and characteristics of cambial stem cells and lay the groundwork for precise molecular studies of these cells in upcoming research.
In our laboratory, we also conducted molecular marker analyses, but failed to identify any unequivocal molecular signatures. Therefore, we assessed calli with stem cell-like features using two different methods: a catalase activity test that monitors bubble generation in response to H2O2 treatment, and a detailed ultrastructural investigation using TEM. While these experiments support the presence of calli with stem cell-like features, they do not constitute sufficient evidence to confirm bona fide stem cell identity at the molecular level. Consequently, we have chosen not to classify these cells as stem cells, referring to them instead as calli with stem cell-like features. Future studies will prioritize validation using molecular markers specific to cambium.
Step 3: Establishment of liquid culture systems for the mass production of functional compounds derived from calli with stem cell-like features
Step 3 involves optimizing suspension-culture conditions (Fig.1 Step 3). To do this, the medium composition (such as MS or ½ MS salts), hormone levels (notably 2,4-D), and sucrose concentrations are methodically adjusted to improve both cell growth and metabolite accumulation.
Characteristics of CMCs and calli with stem cell-like features in a suspension culture
Cell aggregation is a persistent challenge that limits oxygen and nutrient diffusion, resulting in heterogeneous microenvironments. As a result, minimizing the aggregate size is crucial, particularly for large-scale bioreactors (Ochoa-Villarreal et al. 2016). Compared to DDCs, CMCs demonstrate reduced aggregation, improved uniformity, and greater tolerance to mechanical stress. In Taxus cuspidata, CMCs continued to proliferate in stirred tanks where DDCs could not, and in Tripterygium wilfordii, CMCs yielded higher amounts of terpenoids following elicitation (Ochoa-Villarreal et al. 2016; Song et al. 2019). Likewise, Calendula officinalis suspension cultures were composed of 72–93% stem cell-like cells, emphasizing their capability for metabolite synthesis capabilities (Kaňuková et al. 2024). Overall, both CMCs and calli with stem cell-like features exhibit key features, including decreased aggregation, increased resistance to shear stress and robust biosynthetic potential, positioning them as suitable candidates for industrial-scale processes.
Key factors for optimization in a suspension culture
Optimizing the suspension culture is critical to realize a cost-effective metabolite synthesis method. The composition of the culture medium—including salts, sucrose, and growth regulators—profoundly shapes metabolite output. In Lavandula vera, refined medium conditions resulted in a 27-fold increase in rosmarinic acid production (Pavlov et al. 2000). Typically, a half-strength MS medium with an intermediate sucrose levels (2–4%) supports better cell propagation and metabolite biosynthesis in liquid systems (Simões et al. 2009; Ayub et al. 2019; Raikar et al. 2024).
Elicitation can further amplify metabolite accumulation, employing abiotic factors (UV, jasmonates, SA, nanoparticles) or biotic stimuli (microbial extracts, polysaccharides) as inducers (Zhao et al. 2005; Matkowski 2008; Singh et al. 2023). Innovations include exploiting the nanoparticle-induced modulation of ROS (Lala 2021) and the application of CRISPR/Cas-mediated metabolic pathway engineering (Sargazi Moghaddam et al. 2025).
Bioreactor performance is also a key determinant of culture outcomes. Ensuring sufficient oxygen transfer and deploying suitable feeding regimes are essential: fed-batch approaches can boost metabolite levels, while perfusion and wave-type reactors lower shear exposure (Georgiev et al. 2009; Eibl and Eibl 2007; Eibl 2018). The stirring intensity must maintain an appropriate balance between adequate mixing and minimizing damage to cells.
Preserving stem cell properties remains a significant hurdle, as both CMCs and calli with stem cell-like attributes are susceptible to stress and frequently lose their stemness at scale (Lee et al. 2010; Greb and Lohmann 2016). Strategies such as reducing agitation, optimizing hormone levels, incorporating antioxidants, or implementing encapsulation provide some benefit, yet they are inadequate for sustained preservation (Partap et al. 2023).
In summary, the successful optimization of suspension culturing demands a holistic approach, integrating considerations of medium formulation, elicitation, bioreactor configuration, and maintenance of stemness. Prospective improvements will likely involve 3D scaffolds, hydrogel-based immobilization, omics-driven insights, synthetic biology tools, and AI-based modeling for efficient and scalable metabolite production.
Collectively, calli with stem cell-like features serves as an important resource for the large-scale synthesis of functional compounds. Stable callus lines are usually developed through repeated subculturing, with subsequent refinement of the liquid culture conditions. Among the critical factors the concentrations of the MS medium and 2,4-D have the greatest impact; in liquid culture systems, lower concentrations are generally more beneficial than in solid culture systems (Maruyama et al. 2020; Linh et al. 2021; Raikar et al. 2024; Rybin et al. 2024). This observation is well documented in existing literature and is further corroborated by our own laboratory data, with a summary of optimized parameters provided in Table 1 (Park et al. 2023a, 2023b; Lee et al. 2024). Suspension culture systems facilitate efficient large-scale propagation and continuous biosynthesis of metabolites, serving as the foundation for industrial bioreactor technology (Ravishankar and Ramachandra 2000).
Step 4: LC-MS/MS-Based quantification of functional compounds in production systems of calli with stem celll- like features
Step 4 represents the phase of metabolite quantification (Fig. 1, Step 4). The concentrations of target compounds were determined using LC–MS/MS, and scale-up parameters were chosen based on the highest measured titers.
Cambial meristematic cell (CMC) cultures provide key benefits over dedifferentiated cell (DDC) cultures for improving target metabolite yields. For example, CMCs derived from Taxus cuspidata show significantly higher paclitaxel accumulation than DDCs (Lee et al. 2010). Comparable enhancements have also been documented in other taxa: in Taxus spp., CMC cultures led to increased production of Taxamarin A and C (Yang et al. 1999), and in Panax ginseng, stem cell cultures enhanced ginsenoside concentrations, which are noted for their neuroprotective and antioxidant activities (Lee et al. 2010; Leung and Wong 2010). A further significant benefit of CMC cultures lies in their capacity to release functional metabolites into the culture medium rather than retaining them within cells. This significantly streamlines downstream separation processes and decreases total production costs (Wilson and Roberts 2012). As demonstrated in a 3-L airlift bioreactor, CMCs generated much greater paclitaxel yields compared to DDCs (Lee et al. 2010).
This trend was similarly observed in our laboratory. We verified that root cells derived from calli with stem cell-like features of Artemisia capillaris exhibited approximately fourfold higher artemisinin content than intact plants (Park et al. 2023a). In calli with stem cell-like features originating from Ixeridium dentatum, the chrysoeriol-7-O-glucoside concentration was more than tenfold higher compared to plant tissue, and secretion of this compound into the culture medium was more than twice that observed in plant tissue (Park et al. 2023b). These findings indicate that stem cell–based culture systems can greatly enhance the species-specific production of functional metabolites. Furthermore, calli with stem cell-like features developed from Perilla frutescens leaves generated nearly threefold more rosmarinic acid relative to DDCs (40.4 ± 1.4 vs. 12.9 ± 0.7 mg·g-1) (Lee et al. 2024). Every experiment was conducted at least three times, with the results presented as mean ± standard deviation. Statistical analysis was conducted with SPSS software (Statistical Package for the Social Sciences, version 12.0; SPSS Inc., Chicago, IL, USA). Statistical significance was determined using Student’s t-test at the 5% level (p < 0.05).
Accordingly, the four-step platform developed in our laboratory—the induction of calli with stem cell-like features, biochemical and ultrastructural verification, optimization of the suspension culture, and metabolite quantification—offers an efficient strategy for the large-scale, stable, and economically viable production of high-value secondary metabolites from plant stem cells. Nevertheless, achieving fully validated CMC lines remains an ongoing challenge.
The cultures generated in our laboratory did not constitute authentic CMCs but were callus lines displaying stem cell-like features, with incomplete validation of stemness at the molecular level. However, these callus cultures exhibited morphological attributes closely resembling those of CMCs, including a small cell size and multiple vacuoles. As such, calli with stem cell-like features serves as a practical and promising approach for functional metabolite production and offers further potential for industrial applications through improved cross-species adaptability and long-term stability.
Plant Stem Cell and Bioprocess Research: Recent Advances in Single-Cell, CMCs, and AI Applications
Recent advances in single-cell–based plant stem cell research
The combination of single-cell/single-nucleus transcriptomics (sc/snRNA-seq) with spatial transcriptomics has now become the prevailing technique in plant stem cell research. Nuclear–spatial atlases in Arabidopsis enable the reconstruction of cell states and lineages across different organs and developmental stages, revealing both universal and organ-specific markers as well as the axes that govern cell-state transitions (Shahan et al. 2022; Sun et al. 2024; Lee et al. 2025). This methodology has also been applied to reconstruct root developmental trajectories, compare mutants, and identify new leaf markers.
At single-cell resolution, investigations into callus-to-shoot regeneration and meristem development have shown that auxin–cytokinin signaling and transcription-factor networks are organized both spatially and temporally within the stem cell niche, leading to more refined models of fate determination and regeneration mechanisms (Cho et al. 2025). Implementing snRNA-seq could help to minimize protoplasting bias, while declining costs and increasingly standardized workflows are allowing its application to crops and non-model species (Chen et al. 2023). The continued progress in spatial transcriptomics and the integration of multi-omics approaches further drive improvements in mapping cell types and regenerative processes across diverse plant species (Yoon et al. 2024; Lee et al. 2025).
In summary, the single-cell approach avoids signal loss associated with tissue averaging, retains rare stem cell signatures, and concurrently distinguishes markers, maps differentiation trajectories, and elucidates spatial organization (niches), thus facilitating the rigorous optimization of culture conditions and scalable production strategies.
Emerging directions in cambial meristematic cells (CMCs) platforms and AI utilization
CMC culturing has gained recognition as a sustainable foundation for metabolite biosynthesis. Recent investigations delineate induction protocols, key growth determinants, and strategies for process transfer, highlighting advantageous characteristics—homogeneity, reduced cell aggregation, shear resistance, and long-term stable productivity—that collectively endorse the scalability and suitability of this strategy for industrial applications (Partap et al. 2023; Khalafalla 2025).
This progress is most evident in Taxus paclitaxel bioproduction, where use of elicitors such as coronatine, salicylic acid, and methyl jasmonate in conjunction with omics-based approaches enhances regulation and product yields (Yin et al. 2024). CMC-derived lines demonstrate superior performance compared to DDCs through less clumping, increased secretion, and enhanced tolerance to hydrodynamic stress, resulting in improved recovery during processing (Santoyo-García et al. 2023). On the genetic front, TcJAMYC5 has been shown directly to activate paclitaxel biosynthetic genes, establishing transcription-factor modulation as a viable metabolic engineering approach (Cai et al. 2025).
The application of CMCs extends to non-model plant species: in Ocimum basilicum, CMCs have demonstrated robust elicitor-mediated responses leading to increased yields, while stem-cell-like suspensions in Calendula officinalis have expanded the range of possible applications (Zayed et al. 2023; Kaňuková et al. 2024). Process development workflows now incorporate media optimization, elicitor treatments, and omics-based monitoring strategies to could minimize the gap between laboratory and pilot-scale operations. Collectively, the integration of transcription-factor engineering and process intensification establishes CMCs as promising next-generation platforms for the sustainable production of target metabolites (Partap et al. 2023; Zayed et al. 2023; Santoyo-García et al. 2023; Yin et al. 2024; Cai et al. 2025; Kaňuková et al. 2024; Khalafalla 2025).
Furthermore, AI-based process optimization techniques are being developed concurrently. ML/DL soft sensors and digital-twin/hybrid modeling approaches are being integrated with PAT, MPC, and reinforcement learning frameworks to decrease operational delays, enhance control over quality and yield, reduce batch-to-batch fluctuations, and strengthen scale-up predictability and continuous optimization outcomes (Mondal et al. 2023; Isoko et al. 2024; Kwon et al. 2024).
Future Perspectives : AI-Guided Optimization of Natural Product Biosynthesis in Plant Stem Cell Culturing for Industrial Applications
Natural products are considered to serve as essential materials in pharmaceuticals, pigments, agriculture, and fragrances/cosmetics, and the heightened demand for environmentally sustainable alternatives is expected to potentially drive the steady growth of the plant-derived compounds sector. Despite this, conventional farming methods may be constrained by environmental uncertainty, suboptimal metabolite yields, and the challenges associated with purifying active constituents from heterogeneous biomass. As a viable solution, plant cell culturing—particularly cambial meristematic cell (CMC) culturing—could offer advantages under well-controlled environments, potentially enabling accelerated proliferation rates, genetic consistency, and more uniform productivity (Daniell et al. 2001), with improved yields having been reported for ginsenosides in Panax ginseng and additional bioactive compounds in Taxus and Ginkgo. However, the transition to industrial application appears to remain hindered by high physicochemical sensitivity, limited availability of universal molecular markers, and operational stability challenges; therefore, additional studies focusing long-term culture management, scalable bioprocess engineering, and robust diagnostic standardization may be required.
Within this framework, the implementation of sensor technologies, AI, and closed-loop control is increasingly regarded as a highly effective approach for advancing plant stem cell culture. AI-based non-invasive monitoring could enhance the reliability and reproducibility of CMC-based platforms (Wu et al. 2025). Multisensor arrays operating in real-time—hyperspectral, pH/DO, Raman—can be used to monitor physiological conditions throughout cultivation. Spectral data may also enable predictions of metabolite concentrations, such as rosmarinic acid, without necessitating destructive assays (Yoon et al. 2024; Jakovljević et al. 2025).
Supervised ML models (e.g., SVMs, CNNs, random forests) trained on sensor inputs have the potential to provide accurate classification of stem cell states and reasonably reliable predictions of metabolite yields. When incorporated into feedback control, these models could facilitate the real-time modulation modulation of hormone administration and agitation rates; correspondingly, AI-based biofeedback in vertical farming has been suggested to contribute to continuous optimization with reduced manual input (Bethge et al. 2023; Kernbach 2024).
AI-driven platforms are expected to show considerable promise in expediting the synthesis of bioactive compounds from native Korean plants. The integration of their unique phytochemical characteristics with CMC-based systems and AI-optimized processes may represent a compelling strategy for the biosynthesis of high-value natural products. Nevertheless, achieving generalizability across species and environments will likely require the development of multi-species training datasets and the adoption of standardized sensor methodologies; additionally, effectively mitigating sensor noise, condensation, and calibration drift, as well as addressing data scarcity/overfitting, is expected to remain critical to ensuring robust, long-term system performance (Bethge et al. 2023; Wu et al. 2025).
Conclusion
CMCs and calli with stem cell-like features serve as scalable, sustainable platforms that surpass DDCs in terms of homogeneity, reduced aggregation, shear tolerance, and yields. Building upon our research involving Artemisia capillaris, Ixeridium dentatum, and Perilla frutescens, we developed a four-step workflow—induction, verification, suspension-culture optimization, and LC–MS/MS quantification—which was validated across these three native species. The integration of single-cell/spatial omics technologies with AI (real-time monitoring, digital twins) is accelerating the translation to industrial applications. Looking forward, robust stemness markers, AI with cross-species generalizability, and stable bioreactor operation will be essential, empowering AI-enabled biofactories to achieve the reliable large-scale production of high-value natural products.


