Introduction
Materials and Methods
Plant materials, growth conditions, and treatments
Growth parameters determination
Chlorophyll and carotenoid content
Gas exchange measurements
Determination of proline and soluble sugar contents
Determination of reactive oxygen species
Antioxidant activity assessment
Expression analysis of stress-responsive genes in grapevine rootstocks under waterlogging stress
Statistical analysis
Results
Effect of waterlogging on grapevine rootstock leaves and roots phenotypes
Effect of waterlogging on growth indicators of grapevine rootstock
Effect of waterlogging on photosynthetic pigments and gas exchange characteristics in grapevine rootstocks
Effect of waterlogging on proline and soluble sugar content in roots of grapevine rootstocks
Effect of waterlogging on oxidative stress markers in grapevine rootstocks
Effect of waterlogging on antioxidant activities in grapevine rootstocks
Expression analysis of stress-responsive genes in grapevine rootstocks under waterlogging stress
Discussion
Conclusions
Introduction
Grapes (Vitis spp.) represent one of the most valuable and economically important horticultural fruit crops, with significant cultural and economic value across different regions of the world (Elatafi and Fang 2022; Iqbal et al. 2026). The viticulture sector plays an increasingly crucial role in global agriculture and in the global economy. China has emerged as a major player in the global grape industry, with its vineyard surface area ranking second in the world and its grape production ranking first globally. Additionally, China occupies the fifth position worldwide in terms of wine consumption and imports. The grape industry in China has not only contributed significantly to economic development but has also played an important role in poverty reduction in rural areas (Hu et al. 2021). In recent years, the Chinese wine industry has seen substantial growth, with an increasing number of wineries and vineyards established across various regions, particularly in Shandong, Hebei, and Ningxia provinces (Jian et al. 2023). This growth has positioned China as a significant contributor to the global wine market, despite its relatively recent entry into commercial wine production compared to traditional wine-producing countries.
Abiotic forms of stress represent major environmental challenges that significantly impact plant growth, development, and productivity in horticultural crops worldwide (Elatafi et al. 2025a; Elatafi et al. 2025b; Hakeem et al. 2025a; Hakeem et al. 2025b; Mustapha et al. 2025). Regardless of whether plants grow in rainfall-dependent or irrigated environments, waterlogging has emerged as a particularly damaging phenomenon among these types of stress. Waterlogging occurs when soil becomes saturated with water, creating oxygen-deficient conditions in the root zone. This hypoxic environment disrupts normal physiological processes not because of the water itself but due to the oxygen deficiency caused by water saturation. The lack of oxygen triggers a cascade of detrimental effects, including the forcing of the plant roots into anaerobic respiration, weakening respiratory processes, impairing vital physiological functions, altering hormone synthesis, and disrupting nutrient absorption. Under prolonged waterlogging conditions, plants exhibit morphological changes and typically form adventitious roots and develop aeration tissues to enhance oxygen transport as adaptive responses to hypoxic stress (Zhang et al. 2025; Yan et al. 2026).
Waterlogging presents a significant threat to grapevine cultivation, especially in regions prone to excessive rainfall or inadequate drainage. In China and other areas around the world, grape production is frequently affected by heavy rains during the late spring and early summer period, commonly known as the “plum rain season”, which continues for nearly two months in the southern regions. Excessive precipitation events often lead to waterlogged conditions that negatively impact grapevine growth and development. Under waterlogging stress, grapevines exhibit decreased photosynthetic rates, chlorophyll degradation, and impaired physiological functions (Pandey et al. 2021; Wang et al. 2021; Yan et al. 2026). Studies have shown that waterlogging significantly affects enzyme activities in grapevines, with superoxide dismutase (SOD) and peroxidase (POD) activities initially increasing and then decreasing, while catalase (CAT) and ascorbate peroxidase (APX) processes follow an opposite trend (Calabritto et al. 2025). Additionally, waterlogging leads to the increased accumulation of malondialdehyde (MDA) and hydrogen peroxide (H2O2), indicating oxidative stress, while the chlorophyll content decreases (Wang et al. 2021).
Grapevine rootstocks are vital for enhancing plant resilience against various types of biotic and abiotic stress, which are becoming more severe with the impacts of climate change (Hafez et al. 2024; Elatafi et al. 2025b). Although initially developed to combat phylloxera, the role of rootstocks has expanded significantly, with modern breeding efforts now targeting a wider range of environmental challenges, such as drought, salinity, poor nutrient availability, soil compaction, and, more recently, tolerance to waterlogging (Elatafi et al. 2025a; Hakeem et al. 2025a; Iqbal et al. 2025a).
Susceptibility to waterlogging varies significantly among different rootstocks, with some showing greater tolerance than others. Rootstocks serve as the foundational part of grafted plants and play a critical role in plant adaptation to abiotic stress, including waterlogging. The mechanisms employed by rootstocks are crucial in determining the plant’s endurance and resilience under waterlogging (Zhang et al. 2023). Rootstock adaptations in grapevines have a profound impact on the overall plant health by modulating the physiological, molecular, and biochemical processes that influence the vine’s growth, stress resilience, nutrient uptake, and fruit quality (Delrot et al. 2020; Iqbal et al. 2025b). For instance, Schwarzmann rootstock exhibits high tolerance to waterlogging and moderate tolerance to fungal root rot, while the 101–14, 3309, and Richter 99 rootstocks demonstrate poor tolerance to waterlogged conditions (Longbottom and Richard Fennessy 2022). Rootstock adaptations modulate grapevine health by enhancing tolerance to abiotic types of stress such as salinity and drought, optimizing nutrient uptake and metabolism, shaping beneficial microbial communities, and improving physiological processes such as photosynthesis and water use. These adaptations are critical for sustaining vine growth, fruit yields, and quality, especially in the face of climate change challenges (Prinsi et al. 2021).
The SO4 (Vitis berlandieri × Vitis riparia) and 101–14 (Vitis riparia × Vitis rupestris) rootstocks are two important grapevine rootstock options with distinct characteristics and adaptations. The SO4 rootstock, developed in Germany, has gained popularity due to its moderate vigor and outstanding adaptation to various soil conditions. It demonstrates outstanding compatibility with many scion varieties and provides reasonable resistance to phylloxera (Chen et al. 2024). However, SO4 has also been observed to have a notable adverse effect on berry color, with berries showing higher h° values, and this rootstock shows lower levels of total soluble solids and improved titratable acidity in grafted vines (Wang et al. 2025). The 101–14 rootstock, developed in France by Millardet and De Grasset in 1882, features medium-sized, wedge-shaped leaves and long, well-branched, elastic woody shoots that are purple-dark brown in color. This rootstock demonstrates a favorable ability to promote fruit setting but has slow system regeneration. In terms of adaptability, 101–14 shows acceptable performance in compact soils but poor adaptation to wet, dry, or stony conditions. It is particularly sensitive to acidic soils and exhibits sensitivity to various nutrient deficiencies, including nitrogen, potassium, iron, magnesium, zinc, and boron. Despite these limitations, 101–14 offers good resistance to root phylloxera, although it shows poor resistance to leaf phylloxera and high sensitivity to anthracnose. SO4 is known for being moderately vigorous and able to grow in a variety of soil types. 101–14, on the other hand, is known for how it reacts to different levels of soil moisture, as it does not do well in wet soils (Longbottom and Richard Fennessy 2022). These rootstocks hold significant relevance for sustainable viticulture under changing environmental conditions.
As climate change intensifies, extreme weather events that involve excessive rainfall and flooding are becoming more frequent in many viticultural regions. Understanding how different rootstocks respond to waterlogging stress is crucial for developing resilient grape production systems. Both SO4 and 101–14 are widely grown in vineyards globally, with SO4 being particularly popular in regions with moderate to high rainfall levels and 101–14 being utilized in areas where its specific characteristics align with local growing conditions. While previous studies have explored waterlogging tolerance and general adaptability of grapevine rootstocks, detailed comparative analyses of specific rootstocks under prolonged waterlogging conditions are still lacking. SO4 and 101–14 both exhibit distinctive characteristics of resilience under waterlogged conditions. However, limited research has been conducted to evaluate and compare the physiological, biochemical, and molecular responses of these rootstocks under sustained waterlogging stress. Thus, the research objectives of the present study are as follows: a) evaluate and compare the morphological and physiological responses of SO4 and 101–14 rootstocks under different durations of waterlogging stress; b) determine the biochemical changes, including those linked to antioxidant enzyme activities and stress indicators, in both rootstocks during waterlogging conditions; c) identify and validate the molecular biomarkers of waterlogging tolerance by characterizing the expression patterns of key stress-responsive genes in both rootstocks; and d) provide recommendations for rootstock selection in grape-growing regions prone to waterlogging based on the comparative performance outcomes of SO4 and 101–14.
Materials and Methods
Plant materials, growth conditions, and treatments
The study was conducted in summer of 2024 at Baima Base, Nanjing Agricultural University, China, using SO4 (Vitis berlandieri × Vitis riparia) and 101–14 (Vitis riparia × Vitis rupestris) rootstocks. Virus-free seedlings were grown in pots (50 × 30 × 18 cm) containing orchard soil, compost, and vermiculite (1:2:3, v/v) under greenhouse conditions (temperature; 20–25°C and relative humidity; 70–80%). After 45 days, 20 uniform plants per rootstock were subjected to waterlogging (WL) by maintaining 4 cm of standing water above the soil surface, while controls (CK) were kept in well-drained soil. The experiment followed a completely randomized design with three replicates. Morphological traits and leaf samples were collected every seven days until 28 days, with leaves frozen in liquid nitrogen and stored at ‒40°C for the subsequent analyses.
Growth parameters determination
Morphological responses to waterlogging stress were evaluated by measuring the root length, stem diameter, number of roots, and plant height and by assessing the leaf count and node count at 0, 7, 14, 21, and 28 days after treatment. Plant height was determined from the base at the water level to the apex using a 120-cm ruler. Stem diameter was measured using calipers approximately 5 cm above the water surface. Plant nodes were counted from the base to the apex of each plant, while fully expanded leaves were counted, excluding any senescent or partially developed leaves. Root count, length, and diameter measurements were similarly recorded using a ruler and calipers for both control and waterlogged plants.
Chlorophyll and carotenoid content
The chlorophyll (chlorophyll a, chlorophyll b) and carotenoid contents were determined spectrophotometrically according to the protocols of Lichtenthaler and Wellburn (1983). Fresh leaf samples were collected and grounded immediately in liquid nitrogen using a mortar and pestle. We weighed 0.2 mg of ground leaf tissue (in a 10 mL tube) and added 10 mL of methanol. To ensure the complete extraction of the pigments, the mixture was incubated in the dark at room temperature for 48 hours. After incubation, the samples were centrifuged at 4000 rpm at 4°C for 10 min. Absorbance was measured with a UV-Vis spectrophotometer for each sample after the supernatant was extracted. Chlorophyll a (665 nm), chlorophyll b (562 nm) and carotenoids (470 nm) were quantified. The pigment concentrations were computed as mg g-1 FW.
Gas exchange measurements
The gas exchange parameters measured were the stomatal conductance (gs), transpiration rate (Tr), intercellular CO2 concentration (Ci), and the photosynthetic rate (Pn) per unit grapevine leaf area, which were measured in leaves using a portable photosynthetic system (LI-6400; Li-COR, Lincoln, NE, USA) following the manufacturer’s instructions. Morning measurements of gs, Tr, Ci, and Pn were taken from 9:00 A.M. to 12:00 P.M., and the gas exchange parameters were assessed at 60% relative humidity, a CO2 concentration of 390 µmol CO2 mol-1, and with a flow rate to the chamber of 300 ml min-1. Following Salazar-Tortosa et al. (2018), the ratio of Pn to Tr was used to assess water use efficiency (WUE).
Determination of proline and soluble sugar contents
The proline content was determined using a method in the literature (Iqbal et al. 2025a). Fresh leaf tissue (0.5 g) was crushed in 5 mL of 95% ethanol before centrifugation. The residue was re-extracted with 5 mL of 70% ethanol, and the combined extracts were centrifuged at 15,000 rpm for 15 min. One milliliter of extract was diluted with 9 mL of distilled water before reacting with 5 mL of a freshly made ninhydrin reagent (0.125 g ninhydrin in 2 mL 6 M phosphoric acid and 8 mL acetic acid). This mixture was incubated in a water bath at 90°C for 45 min under gentle shaking. After cooling, 4 mL of toluene was added to the samples, which were violently shaken for 30 min. The absorbance of the toluene phase was measured at 515 nm with a spectrophotometer. The soluble sugar concentration was measured as follows (Iqbal et al. 2025a). An aliquot (0.1 mL) of the alcoholic extract was combined with 3 mL of anthrone reagent (150 mg anthrone in 100 mL 72% sulfuric acid) and incubated at 90°C for 10 min. After cooling to room temperature, the absorbance was measured at 625 nm.
Determination of reactive oxygen species
The MDA concentration was assessed with the thiobarbituric acid (TBA) method as outlined by Patterson et al. (1984). Fresh leaf samples were homogenized in 0.1% trichloroacetic acid (TCA) and subjected to centrifugation at 12,000 × g for 10 min. The supernatant (reaction mixture) was combined with 20% TCA containing 0.5% TBA and incubated at 100°C for 30 min. After cooling on ice, the mixture underwent centrifugation at 12,000 × g for 10 min. The absorbance of the supernatant was quantified at 530 nm and adjusted by subtracting the absorbance value measured at 600 nm.
The concentration of H2O2 was assessed using the methodology established by Patterson et al. (1984). Leaf tissue was homogenized using 2 mL of cold acetone and subsequently centrifuged at 10,000 × g for 15 min at 4°C. A 0.5 mL aliquot of the supernatant was combined with 0.05 mL of a 20% titanium tetrachloride (TiCl4) solution and 0.1 mL of a concentrated ammonia solution, with this followed by centrifugation at 3,000 × g for 10 min. The supernatant was removed, and the precipitate was rinsed three times with cold acetone. Three milliliters of 2 M sulfuric acid were added to dissolve the precipitate, and the absorbance was recorded at 415 nm. The concentration of H2O2 was determined using a standard curve and is expressed as µmol g-1 of fresh weight.
Antioxidant activity assessment
Composite leaf samples (200 mg) were homogenized in 1.6 mL of cooled phosphate buffer (pH 7.8) on ice. The homogenate underwent centrifugation at 12,000 × g for 20 min at 4°C, and the supernatant was utilized for enzyme tests. The activity of SOD was evaluated according to a method by Hakeem et al. (2025a). The reaction mixture (3 mL) comprised 30 µM EDTA-Na2, 0.05 M phosphate buffer (pH 7.8), 2.25 mM nitro blue tetrazolium (NBT), 14.5 mM methionine, 60 µM riboflavin, and 40 µL of the enzyme extract. Samples were incubated at 4000 lux for 20 min, and the absorbance was recorded at 560 nm. The activity of POD was assessed according to the method in Scalet et al. (1995). The experiment comprised 3 mL of 0.2 M phosphate buffer (pH 6.0), 2% H2O2, 50 mM guaiacol, and 40 µL of the enzyme extract. An increase in absorbance was noted at 470 nm. The activity of CAT was assessed according to the methodology established by Dhindsa et al. (1982). The 3 mL reaction mixture comprised 0.3% H2O2 and 0.1 mL of the enzyme extract in a phosphate buffer at pH 7.0. A reduction in absorbance was observed at 240 nm. The activity of APX was evaluated by homogenizing 0.1 g of frozen leaf tissue in 1 mL of a sodium phosphate buffer (0.5 M, pH 7.8) containing 1 mM EDTA and 2% (w/v) PVP-40. This was followed by centrifugation at 10,000 × g for 20 min at 4°C. The assay combination comprised 50 µL of the enzyme extract, 3 mL of an extraction buffer (0.1% Triton X-100, 1.0 mM ascorbate), and 4.5 µL of 30% H2O2. The reduction in absorbance was quantified at 290 nm over a duration of 1 min. One unit of APX activity signifies the oxidation of 1 µmol of ascorbate per min (Nakano and Asada 1981). All enzyme activity was measured in units of mg-1 FW.
Expression analysis of stress-responsive genes in grapevine rootstocks under waterlogging stress
The expression levels of key stress-responsive genes were analyzed to investigate the molecular mechanisms underlying grapevine adaptation to waterlogging stress. Target genes were selected based on their established roles in critical physiological processes, in this case signal transduction, redox regulation, and cellular defense. The following genes were analyzed: KEEP ON GOING (KEG; VIT_09s0002g07110), a serine/threonine protein kinase involved in abscisic acid (ABA)-mediated stress signaling; Stress-Induced Protein (VIT_05s0020g01990), implicated in cellular protection through the stabilization of proteins and membranes; a calmodulin-like protein (CML; VIT_01s0011g03620), a calcium sensor involved in stress signal perception and reactive oxygen species (ROS) regulation; alcohol dehydrogenase 1 (ADH1; VIT_10s0071g01160), a key enzyme in anaerobic respiration that facilitates ethanol fermentation under low-oxygen conditions such as waterlogging; peroxidase (POD; VIT_05s0020g00600), which functions in ROS scavenging to mitigate oxidative damage. A respiratory burst oxidase homolog (RBOH; VIT_16s0039g02010), responsible for generating ROS as part of stress signaling cascades; and a late embryogenesis abundant protein (LEA; VIT_16s0050g02670), known for its role in cellular protection and stabilization under dehydration stress (Liu and Stone 2010; Danquah et al. 2014; Wang et al. 2017; Chen et al. 2019).
Total RNA was isolated from grapevine rootstock leaf tissues utilizing the RNeasy Plant Mini Kit (Qiagen, Shanghai, China) in accordance with the manufacturer’s guidelines. The concentration and purity of RNA were assessed using a NanoDrop 2000 spectrophotometer (Thermo Fisher Scientific, USA), whereas integrity was evaluated by means of electrophoresis on a 1.0% agarose gel. First-strand cDNA was generated from 1 µg of total RNA utilizing the RevertAid First Strand cDNA Synthesis Kit (Thermo Fisher Scientific, USA) in accordance with the specified methodology. Quantitative real-time PCR (qRT-PCR) was performed in a 20 µL reaction including 1 µL of cDNA, 10 µL of SYBR Green PCR Master Mix (Applied Biosystems, USA), and 0.5 µM of each forward and reverse primer built using Primer3 software (Rozen and Skaletsky 2000; Wang et al. 2021) (Supplementary Table S1). Primer efficiency varied between 90% and 110%. All reactions were conducted in triplicate using a Step One Plus Real-Time PCR system (Applied Biosystems, USA). The cycling technique comprised initial denaturation at 95°C for 10 min, succeeded by 40 cycles of 95°C for 15 s, 60°C for 30 s, and 72°C for 30 s. Gene expression was standardized to the grapevine actin gene, and relative expression levels were determined using the 2−ΔΔCT method (Livak and Schmittgen 2001).
Statistical analysis
The intervention was designed utilizing two grapevine rootstocks and two treatments (CK and WL). Each treatment comprised three duplicates. This research utilized a factorial randomized design. The data were analyzed using CoStat-Statistics (version 6.3, CoHort Software, Birmingham, UK). The means and values of two trials were analyzed by means of a two-way ANOVA, and significant differences between treatment means from repeated experiments were evaluated with Tukey’s HSD range test at the p < 0.05 probability level. We provide the ANOVA results for analysis in Supplementary Table S2.
Results
Effect of waterlogging on grapevine rootstock leaves and roots phenotypes
During the 28 days of our study of waterlogging, the SO4 and 101–14 rootstocks both displayed distinct phenotypic responses in the leaves (Fig. 1). SO4 maintained healthy leaves until day 14, with visible damage occurring after day 21, while 101–14 showed early stress symptoms, including chlorosis, wilting, and leaf drop, by day 28. Additionally, the root responses reflected a similar pattern (Fig. 1). SO4 retained structural integrity until day 28, whereas 101–14 roots showed early discoloration, progressive necrosis, and severe disintegration. The individual effects of the grapevine rootstock type and the waterlogging treatment are detailed in Supplementary Tables S3 and Supplementary Tables S4, respctively.
Effect of waterlogging on growth indicators of grapevine rootstock
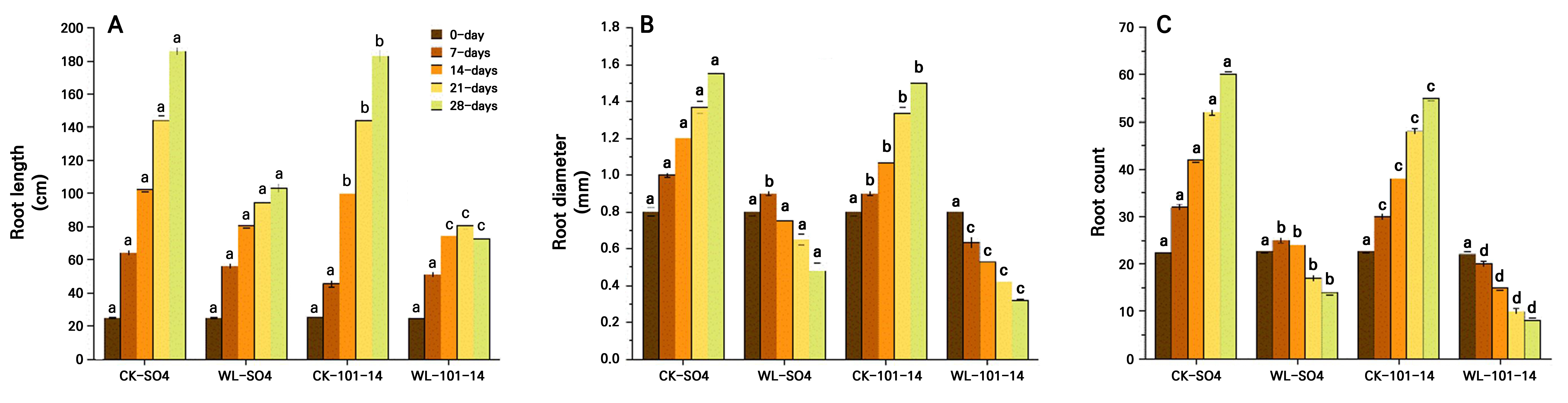
The study found that waterlogging stress significantly inhibited growth in both grapevine rootstocks, but with clear differences in tolerance. Plant height progressively declined, with reductions of 24.60% in SO4 and 35.13% in 101–14 by day 28 (p < 0.05) under waterlogging stress compared with the CK groups (SO4 and 101-14) (Fig. 2A). Stem diameter decreased by 31.41% in SO4 and by 45.04% in 101–14 , with SO4 maintaining significantly thicker stems (Fig. 2B). Leaf numbers fell sharply, by 56.47% in SO4 and 81.60% in 101–14 (Fig. 2C), while node numbers decreased by 23% and 49.44%, respectively (p < 0.05) (Fig. 2D). Root length was similarly suppressed, declining by 34.64% in SO4 and 54.84% in 101–14, with SO4 sustaining significantly longer roots (Fig. 3A). Both rootstocks were negatively affected by prolonged waterlogging, but SO4 exhibited significantly greater resilience, whereas 101–14 was more sensitive to stress.
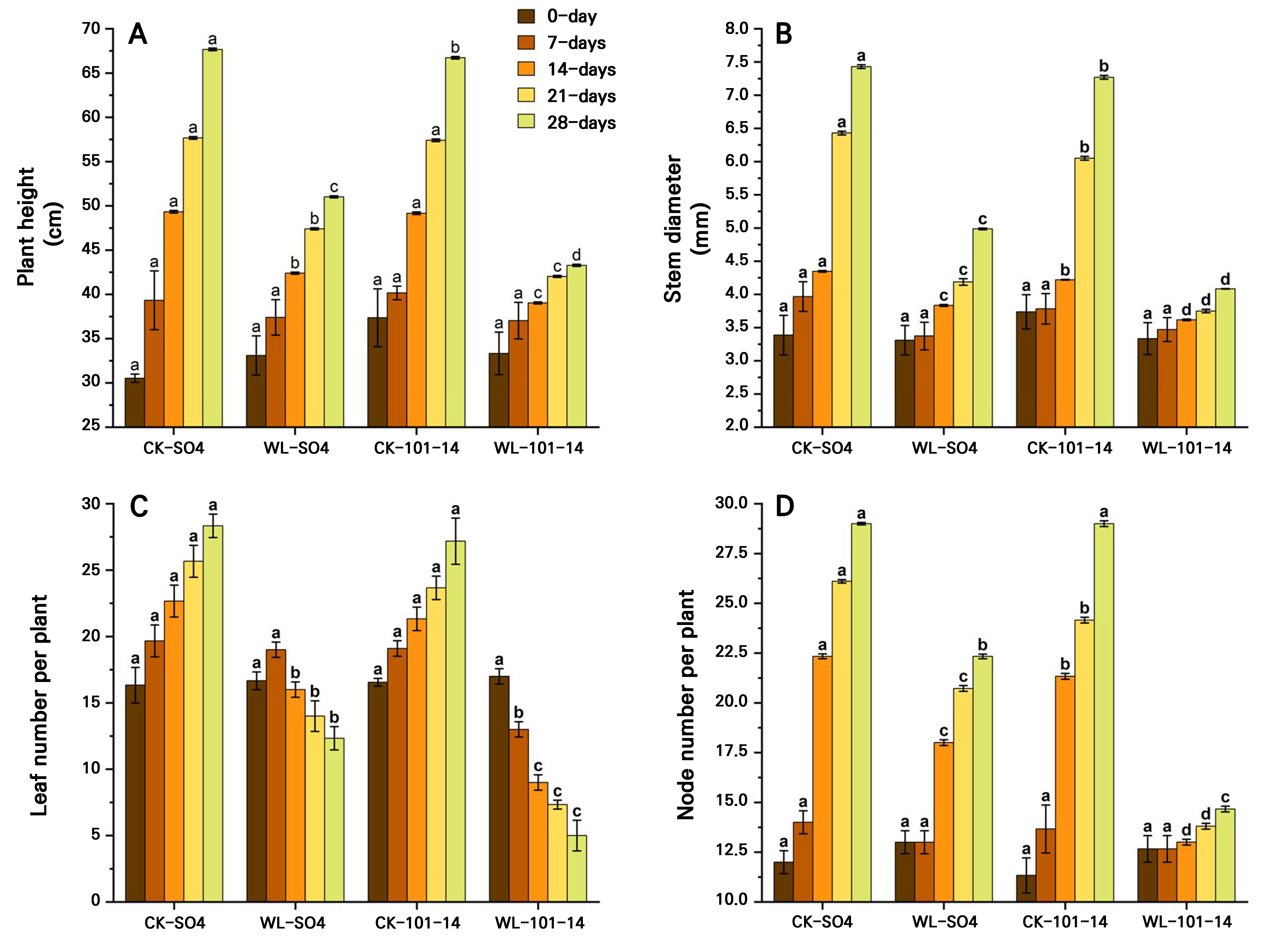
Fig. 2.
Effects of waterlogging (WL) on the plant height (A), stem diameter (B), leaf number (C), and node number (D) in two grapevine rootstocks (SO4 and 101–14) during 28 days of stress. Significant differences between treatments are indicated by different letters (p < 0.05). WL: waterlogging, CK: control.
The grapevine rootstock morphology was significantly affected by the waterlogging stress treatment compared to the CK treatment, featuring reductions in the root diameter and root number in both rootstocks (Fig. 3B–3C). No differences were observed during the early stages (0–7 days), but from day 14 onward reductions became evident. By day 28, root diameter decreased by 20.41% in SO4 and 43.43% in 101–14, with SO4 maintaining a 36.73% larger diameter than 101–14 under stress. Similarly, root count declined progressively under waterlogging. On day 28, SO4 and 101–14 showed reductions of 45.76% and 66.90%, respectively, compared with the controls. Notably, SO4 retained 53.19% more roots than 101–14, indicating a stronger capacity for lateral root development. Overall, SO4 demonstrated greater stability with regard to root thickness and greater root proliferation than 101–14, reflecting its superior resilience to hypoxic conditions. The individual effects of the grapevine rootstock type and the waterlogging treatment are detailed in Supplementary Tables S3 and Supplementary Tables S4, respectively.
Effect of waterlogging on photosynthetic pigments and gas exchange characteristics in grapevine rootstocks
Waterlogging significantly reduced photosynthetic pigments, specifically chlorophyll a, chlorophyll b, total chlorophyll, and carotenoids, in both grapevine rootstocks, SO4 and 101–14, over 28 days (Fig. 4A–4D). Total chlorophyll declined by 47.2% in SO4 and by 53.95% in 101–14 relative to the controls, with SO4 retaining 10.56% more chlorophyll and higher carotenoid levels. Additionally, the results showed that the waterlogging condition significantly reduced key gas exchange parameters in both the SO4 and 101–14 rootstocks over 28 days (Fig. 5A–5D). The Pn, Tr, and gs levels declined progressively under stress, particularly from day 14 onward. In contrast, the Ci level increased under waterlogging, suggesting that non-stomatal limitations, such as inhibited photosynthetic enzymes and the chloroplast function, also contributed to the reduced Pn level. Waterlogging significantly reduced WUE in both rootstocks, with a greater decline observed in the sensitive 101–14 compared to the more tolerant SO4 (Fig. 5E). This reduction reflects genotype-dependent differences in the ability to convert water into biomass under stress. The individual effects of the grapevine rootstock type and the waterlogging treatment are detailed in Supplementary Tables S3 and Supplementary Tables S4, respectively.
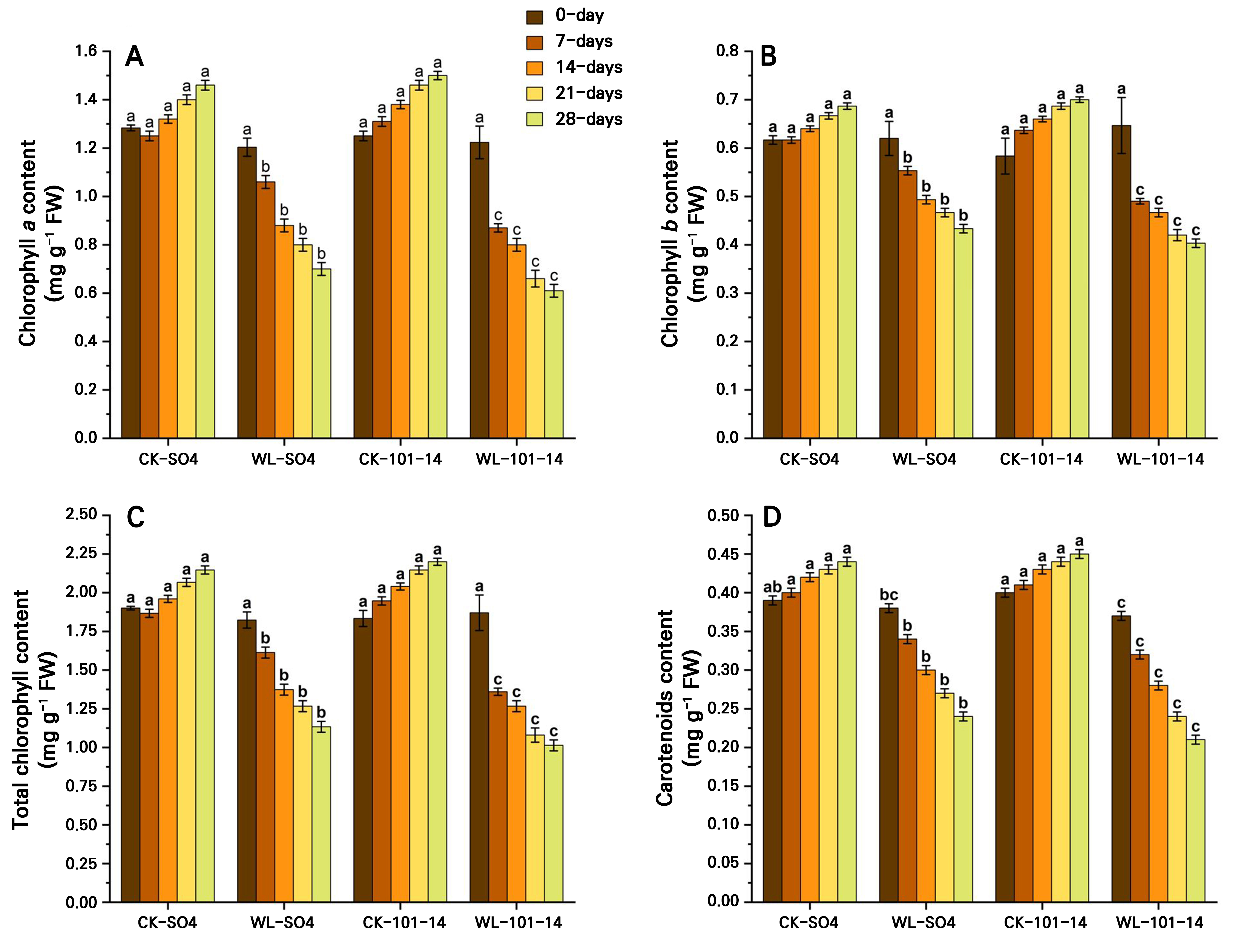
Fig. 4.
Effects of waterlogging on chlorophyll a (A), chlorophyll b (B), total chlorophyll (C), and carotenoids (D) in two grapevine rootstocks (SO4 and 101–14) during 28 days of stress. Significant differences between treatments are indicated by different letters (p < 0.05). WL: waterlogging, CK: control.
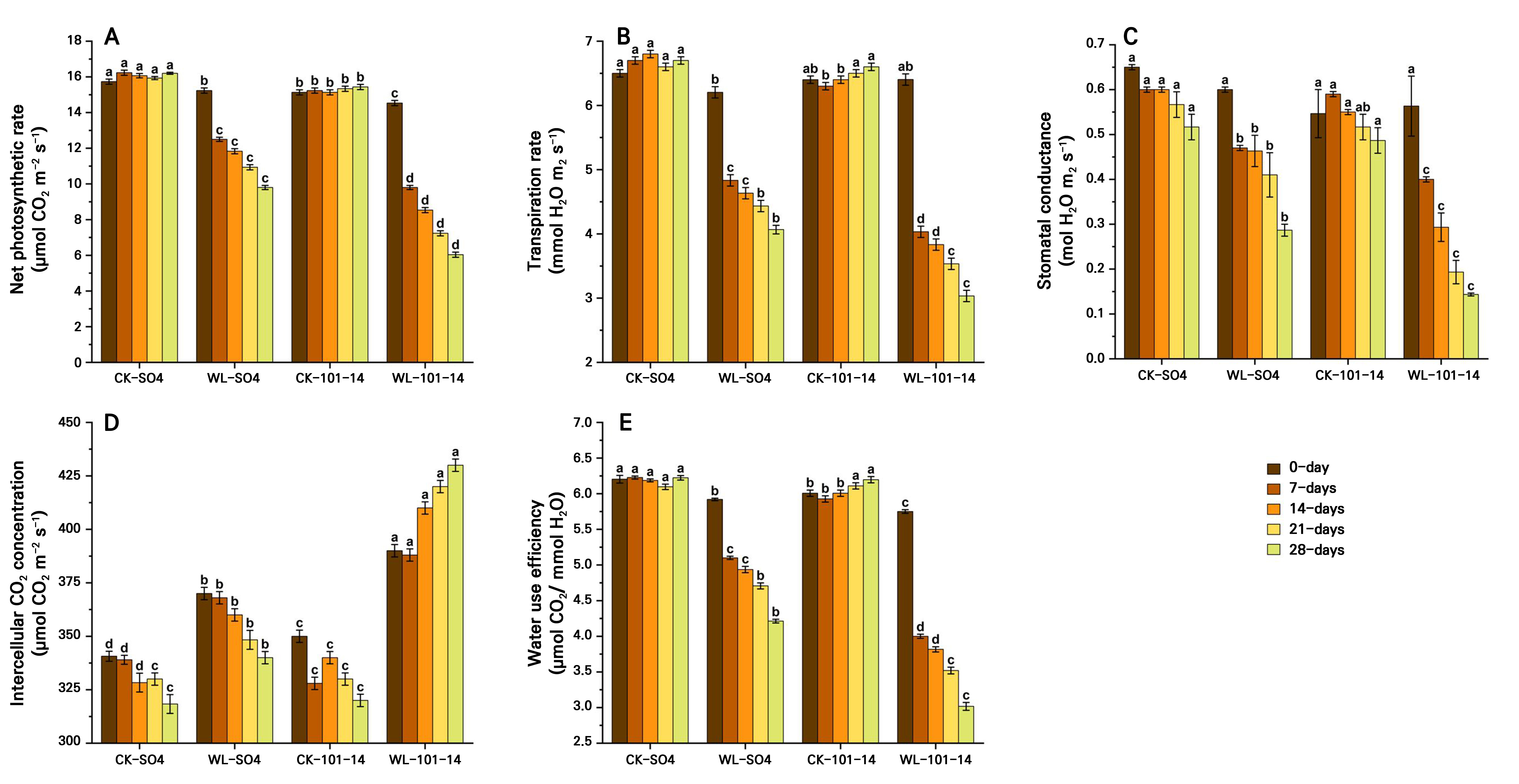
Fig. 5.
Effects of waterlogging on the gas exchange characteristics, specifically the net photosynthesis rate (A), transpiration rate (B), stomatal conductance (C), intercellular CO2 concentration (D), and water use efficiency (E) in two grapevine rootstocks (SO4 and 101–14) during 28 days of stress. Significant differences between treatments are indicated by different letters (p < 0.05). WL: waterlogging, CK: control.
Effect of waterlogging on proline and soluble sugar content in roots of grapevine rootstocks
Waterlogging progressively increased oxidative stress in both grapevine rootstocks over 28 days, as indicated by elevated proline and soluble sugar levels (Fig. 6A–6B). By day 28, the proline content increased by 60% in SO4 and by 65.62% in 101–14, while the soluble sugar levels rose by 48.5% and 56.66%, respectively, compared with the CK groups. The higher accumulation of proline in 101–14 indicates greater oxidative damage relative to SO4. Significant differences were observed between WL and CK plants across the time points set here, highlighting genotype-dependent sensitivity to waterlogging-induced oxidative stress. The individual effects of the grapevine rootstock type and the waterlogging treatment are detailed in Supplementary Tables S3 and Supplementary Tables S4, respectively.
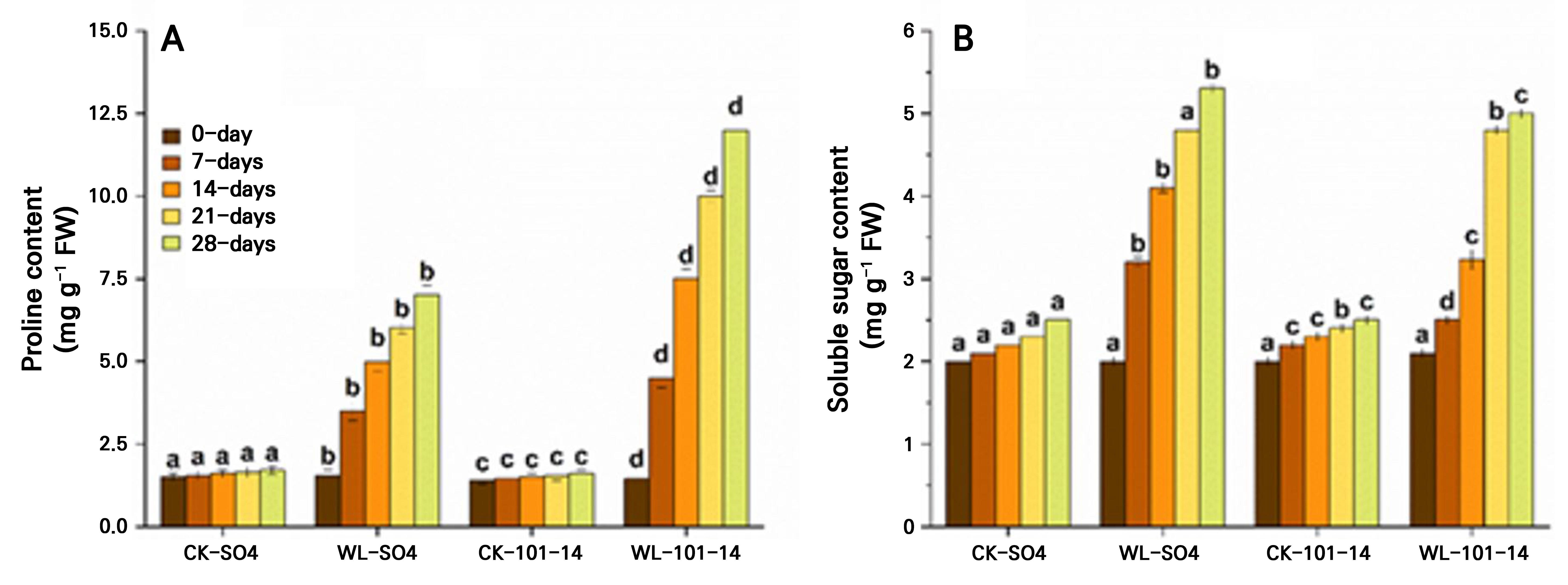
Fig. 6.
Effects of waterlogging on osmoprotectant accumulation in the grapevine rootstocks SO4 and 101–14 over 28 days. Proline content (A) and soluble sugar content (B). Values represent the means ± SE (n = 3). A two-way ANOVA (factors: rootstock and waterlogging) followed by Tukey’s HSD test was used to determine significances (p < 0.05); different letters indicate significant differences. WL: waterlogging, CK: control.
Effect of waterlogging on oxidative stress markers in grapevine rootstocks
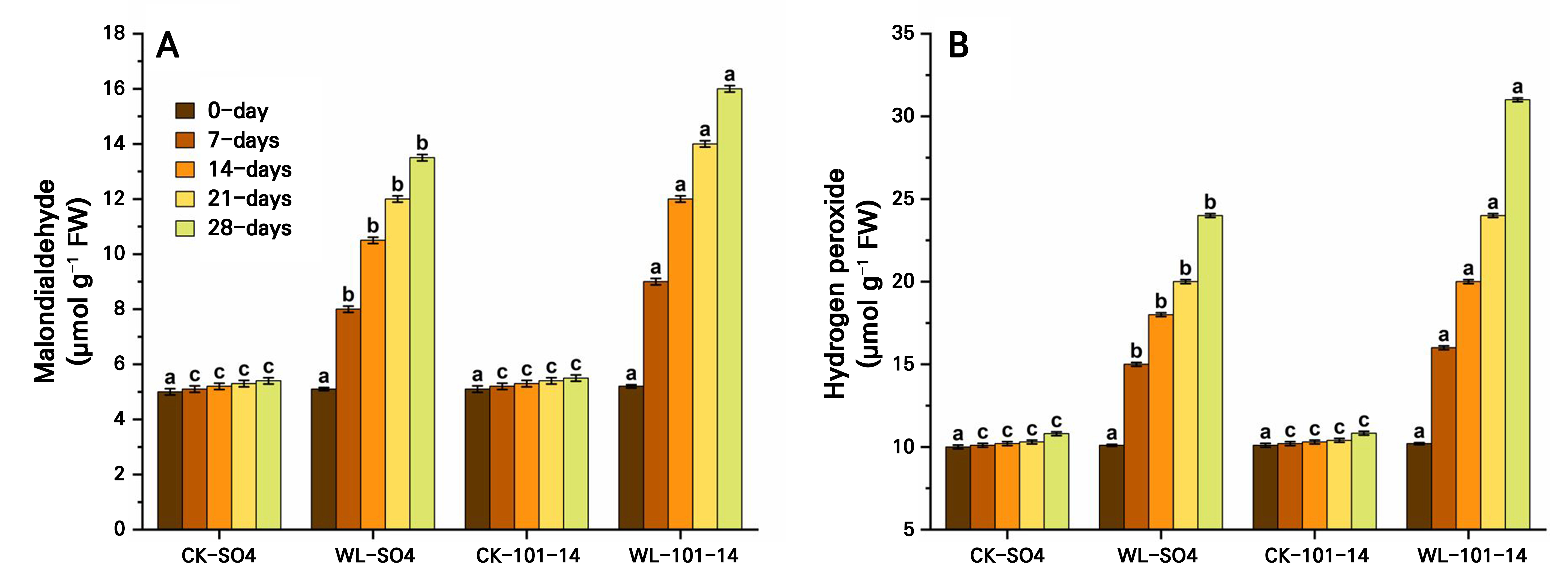
Waterlogging stress progressively increased oxidative damage in both grapevine rootstocks, as indicated by rising MDA and H2O2 levels over 28 days (Fig. 7A–7B). Both SO4 and 101–14 showed significantly higher values under waterlogging compared to the controls, with the greatest increases recorded on day 28. Overall, 101–14 accumulated more MDA and H2O2 than SO4, suggesting greater oxidative injury. Significant differences were observed between the treatments and rootstocks, though the control values remained similar across both genotypes. Again, the individual effects of the grapevine rootstock type and the waterlogging treatment are detailed in Supplementary Tables S3 and Supplementary Tables S4, respectively.
Effect of waterlogging on antioxidant activities in grapevine rootstocks
The antioxidant enzyme activities (POD, SOD, CAT, and APX) significantly increased in the grapevine rootstocks SO4 and 101–14 over 28 days, with clear genotype-dependent differences. SO4 exhibited significantly higher CAT (63.5%) and APX (41.42%) activity levels than 101–14, which showed only modest increases, while POD and SOD increased in both rootstocks (Fig. 8A–8D). The results indicated that antioxidant enzyme activities, in this case POD, SOD, CAT, and APX activities, were significantly elevated in the grapevine rootstocks SO4 and 101–14 under waterlogging stress over 28 days. These enzymes exhibited clear genotype-dependent variations, with SO4 showing considerably higher CAT and APX activity levels, with corresponding 63.5% and 41.42% increases, compared to the more modest enhancements observed in 101–14. Both rootstocks showed increased POD and SOD activity levels, but the magnitude of the increase was greater in SO4. Conversely, the attenuated response in 101–14 suggests a weaker antioxidant defense, correlating with its increased susceptibility to stress. The individual influences of the grapevine rootstock type and the waterlogging treatment are also detailed in Supplementary Tables S3 and Supplementary Tables S4.
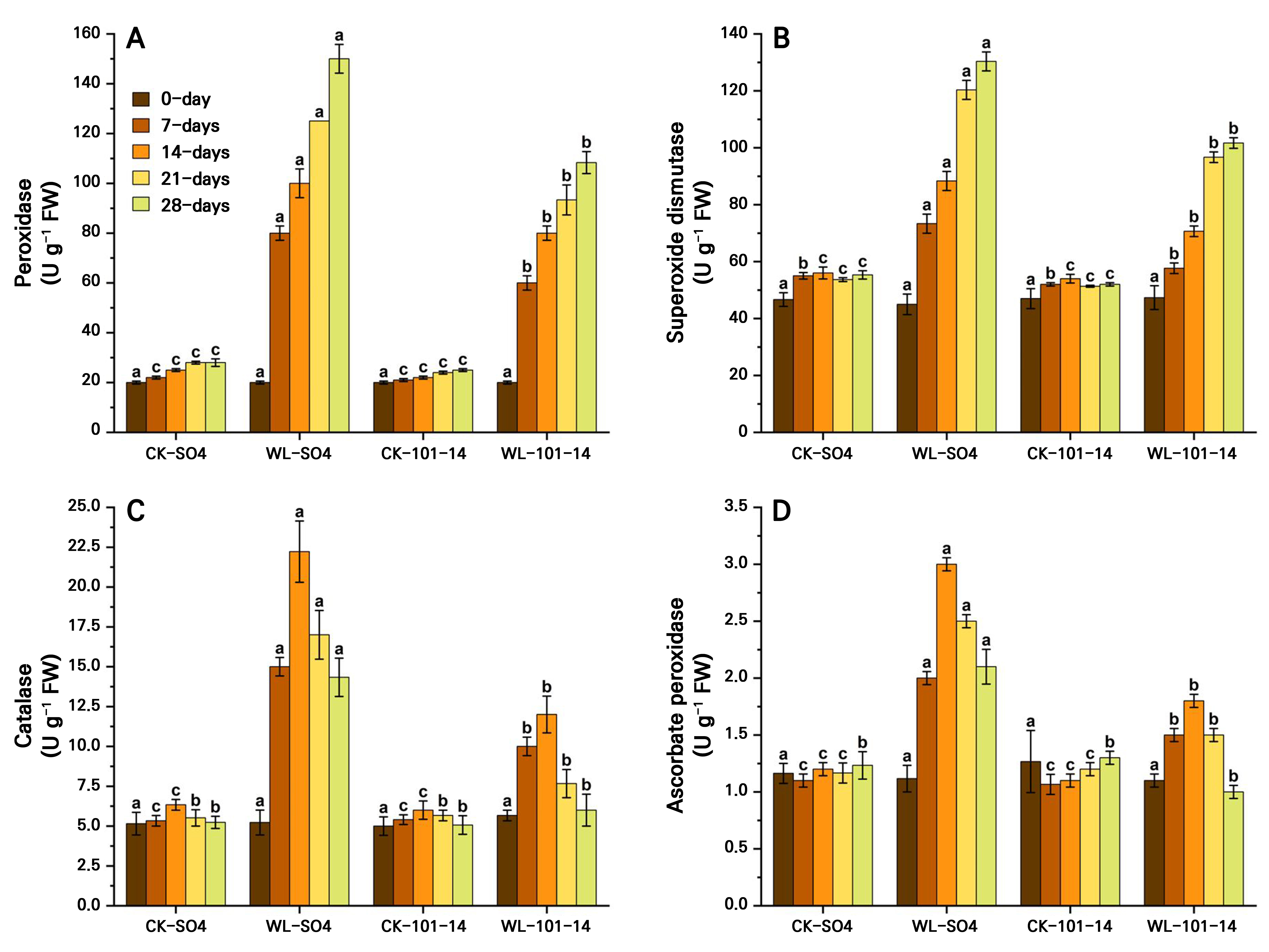
Fig. 8.
Effects of waterlogging on antioxidant activities, in this case peroxidase (A), superoxide dismutase (B), catalase (C), and ascorbate peroxidase (D) in the two grapevine rootstocks (SO4 and 101–14) during 28 days of stress. The data were analyzed by means of a two-way analysis of variance (ANOVA) with three replicates, using rootstocks and waterlogging as the two components. The data were computed as the means ± standard error (SE), with significance denoted by distinct letters for p < 0.05 (Tukey’s HSD test). WL: waterlogging, CK: control.
Expression analysis of stress-responsive genes in grapevine rootstocks under waterlogging stress
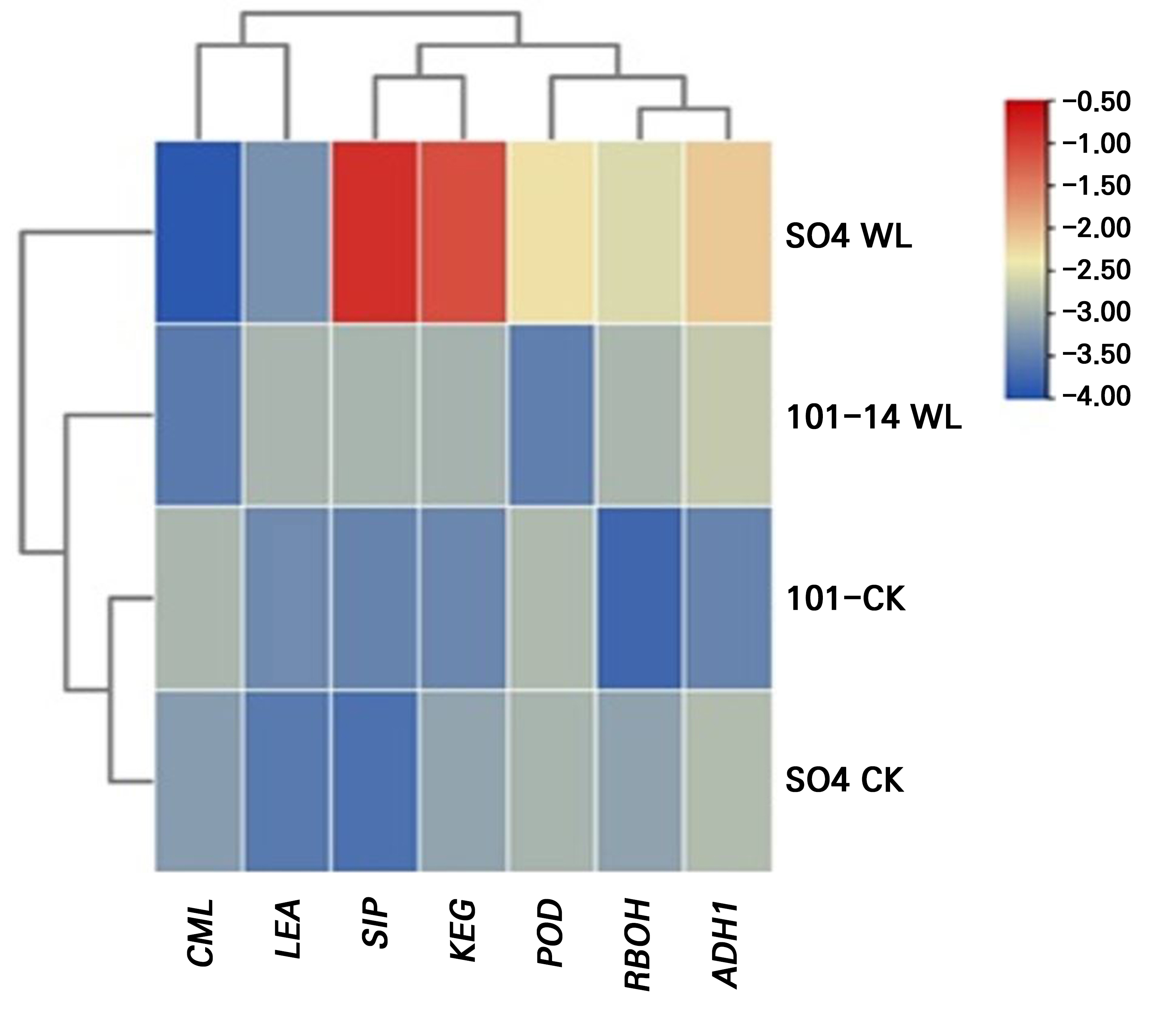
To investigate the molecular responses of the grapevine rootstocks to prolonged waterlogging stress, the expression patterns of selected stress-responsive genes were examined after 28 days of treatment (Fig. 9). A comparative gene expression analysis between the two rootstocks, SO4 and 101–14, revealed distinct transcriptional patterns in response to waterlogging. The 101–14 rootstock demonstrated relatively stable or moderately elevated expression levels across most genes, suggesting a constitutive or pre-adaptive expression pattern that may underpin its ability to maintain cellular homeostasis under stress. In contrast, SO4 exhibited more pronounced and stress-inducible upregulation of several defense-related genes, reflecting a reactive transcriptional response triggered by heightened stress perception.
Discussion
The present study clearly demonstrates that the superior tolerance of the SO4 grapevine rootstock to prolonged waterlogging, in comparison to the highly sensitive 101–14, is orchestrated by a multi-layered and coordinated adaptive response. While both genotypes exhibited signs of stress, SO4 effectively mitigated the adverse effects by maintaining key physiological functions, in this case moderate gas exchange and photosynthesis, and by preserving its photosynthetic machinery. This resilience was fundamentally linked to a more rapid and robust activation of its antioxidant defense system, which successfully limited the accumulation of damaging reactive oxygen species. Critically, these physiological advantages were underpinned at the molecular level by the significant upregulation of a suite of stress-responsive genes related to anaerobic metabolism, ROS homeostasis, and cellular stabilization, a response that was notably absent or delayed in 101–14, leading to its progressive physiological collapse. Our results align with those of Stevens and Harvey (1995), who revealed that waterlogging exacerbated the growth reduction caused by saline irrigation (61% decline under combined stress vs. 47% with salinity alone). Waterlogging had significant negative impacts on growth, nutrient (P, K, Ca, Mg) contents, and root metabolic activity (Kang et al. 2010).
Ruperti et al. (2019) indicated that a grafted scion can “delay the stimulus perception and rootstock reactivity to drought”, a mechanism that likely extends to waterlogging stress. This underscores that grafting is a dynamic partnership, and the selection of both the rootstock and scion is crucial for overall plant resilience. The tolerance and responses of rootstocks exhibit variations that depend on specific stress factors. Waterlogging decreases plant height, the leaf area, and the number of leaves and nodes, also inhibiting aboveground growth and the formation of the canopy. In grapevines, ongoing flooding often leads to a reduction in the stem diameter, stunted shoot growth, and early leaf senescence. Below the surface, the root length, diameter, and count are negatively affected. This type of stress is also known to cause fine root deterioration quickly, leading to a less dense and less effective root system, with certain genotypes displaying a diminished regenerative capacity following the alleviation of water stress (Alonso-Forn et al. 2025).
In our study, the results show notable reductions in chlorophyll a and b as well as carotenoid levels in grapevine leaves, thereby indicating a compromised photosynthetic apparatus. This loss of pigmentation was detected after 48 hours of water stress, reaching its lowest levels after extended exposure (28 days). The observed decline in the chlorophyll content is attributed to disrupted biosynthetic pathways and increased degradation of chlorophyll pigments, as shown by the downregulation of genes involved in chlorophyll metabolism. These alterations are associated with decreased Pn, reduced Tr, lower gs, and heightened Ci levels. Furthermore, the reduction in WUE indicates ineffective carbon assimilation per unit of water loss, indicative of compromised rootstock functionality (Zhu et al. 2018; Prinsi et al. 2021; Wang et al. 2021; Edwards et al. 2022). The accumulation of proline and soluble sugars represents key physiological adaptations to waterlogging, functioning as osmoprotectants and stabilizers of cellular integrity, respectively. The study shows both rootstocks, SO4 and 101–14 exhibit increased levels of proline and sugars in response to stress; however, the quantitative variations were dependent on the genotype, with SO4 displaying the highest soluble sugar content and the lowest proline levels compared to 101–14 under stress conditions. This aligns with findings by Zhu et al. (2018) and Ruperti et al. (2019), who reported that proline accumulation is a common adaptive response to waterlogging-induced hypoxia.
Proline functions not only during osmotic adjustments but also as a ROS scavenger, stabilizing proteins and cellular structures under stress. While proline and sugar accumulation are beneficial for a proper osmotic balance and for ROS detoxification, and as an energy supply, excessive induction as noted in 101–14 may divert resources away from growth, potentially reducing long-term tolerance (Ruperti et al. 2019). These findings are related to previous studies that report the upregulation of glycolytic and fermentative pathways under waterlogging conditions, serving to sustain ATP production in the absence of oxygen (Ruperti et al. 2019; Mudasir and Shahzad 2025). Moreover, sugars also serve as signaling molecules and precursors for protective metabolites, contributing to stress recovery and cellular repair mechanisms (Zhu et al. 2018). These findings reinforce the view that SO4 may be more inherently tolerant to prolonged waterlogging, while 101–14 exhibits a higher degree of stress sensitivity, relying on a stronger inducible defense mechanism.
Additionally, the levels of MDA and H2O2 increased, serving as markers of oxidative membrane damage and a heightened ROS burden, respectively. These results highlight the fundamental role of oxidative stress indicators in assessing plant responses to waterlogging and underpinning the genotype-specific variance in stress adaptation mechanisms (Sood 2025). Under the hypoxic condition caused by waterlogging stress in this study, SO4 exhibited the lowest concentrations of MDA and H2O2, whereas 101–14 showed the highest levels. The mechanisms of each rootstock that enable tolerance to oxidative stress are vital for their overall performance (Krishankumar et al. 2025).
On the other hand, our study shows an increase in the activities of antioxidant enzymes, with the levels of activity in SO4 increasing more than in 101–14 rootstock. These results are consistent with previous research demonstrating that waterlogging induces marked increases in antioxidant enzyme activities in grapevines, with tolerant rootstocks such as SO4 exhibiting stronger CAT and APX responses. The research on grapevine rootstocks under certain types of abiotic stress, such as drought or waterlogging, highlights the role of enhanced enzymatic defenses in mitigating oxidative damage (Wang et al. 2021; Iqbal et al. 2025a). These findings underscore the critical role of antioxidant enzyme systems in conferring resilience to waterlogging stress, supporting the genotype-specific differences observed in this study. The hypoxia condition caused by waterlogging triggers early and pronounced increases in the activity levels of antioxidant enzymes such as SOD, POD, CAT, and APX. Activity trends differ: SOD and POD peak quickly but decline after prolonged stress, whereas CAT and APX may follow a contrary pattern. This coordinated enzyme induction is essential for maintaining redox homeostasis and survival under hypoxic stress. The enhanced induction of antioxidant proteins and related gene expression levels, underlying better adaptation in tolerant genotypes, and the failure to sustain enzyme activity, are associated with greater cellular injury and lower survival mechanisms (Zhu et al. 2018; Wang et al. 2021).
Furthermore, the genes that are involved in signal transduction, redox regulation, and cellular defense exhibit precise and dynamic upregulation during waterlogging. Waterlogging-responsive transcriptomic networks converge on the antioxidant machinery, stress-responsive signaling capability (ABA, redox signaling), and cellular defense mechanisms (such as late embryogenesis abundant proteins, heat shock proteins, and hypoxia markers). The expression levels of ROS detoxification enzymes are tightly correlated with their physiological activity and with indicators of oxidative stress. Comparative transcriptomes show that tolerant rootstocks such as SO4 can induce more robust and rapid upregulation of these pathways than sensitive ones (Zhu et al. 2018; Wang et al. 2021; Sood 2025).
Although both rootstocks activate protective mechanisms under waterlogging, several differences in the intensity of stress were observed over the treatment period. SO4 generally shows more tolerance with superior abilities in osmolyte accumulation, antioxidant enzyme activation, root survival, and pigment preservation compared to 101–14 when exposed to 28 days of stress. These advantages are likely attributable to differences in gene expression and regulation, more effective ROS scavenging, and stronger root anatomical features (Zhu et al. 2018; Wang et al. 2021; Alonso-Forn et al. 2025). Our results show a significant increase in the expression of key defense and redox-related genes in SO4 during waterlogging. These include KEG kinase, CML, ADH1, POD, RBOH, and LEA. This strong transcriptional response suggests a highly adaptive regulatory mechanism that allows SO4 to quickly respond and adjust to stress through enhanced signaling, ROS detection and detoxification, and the activation of anaerobic metabolism to cope with low oxygen levels. The marked increases in CML and RBOH suggest enhanced calcium-dependent signaling and regulated ROS generation, while higher POD and LEA levels reflect strong antioxidant activity and cell protection (Zhu et al. 2018; Wang et al. 2021). In contrast, 101–14 shows relatively moderate increases in gene expression. This indicates that it may rely on more constant, pre-set levels of defense mechanisms, keeping key genes active even before stress occurs. While these changes could help maintain a proper long-term cellular balance, the less dynamic response may result in slower or weaker activation of critical stress responses, making 101–14 more susceptible to extended or intense waterlogging conditions (Wang et al. 2021).
Conclusions
In conclusion, this study comprehensively elucidates the multi-layered mechanisms conferring superior waterlogging tolerance to the grapevine rootstock SO4 when compared to the highly sensitive 101–14. Our integrated analysis demonstrates that the resilience of SO4 is not reliant on a single trait but on a rapid, coordinated defense strategy encompassing morphological adaptations, physiological stability, and a robust molecular response. The development of adventitious roots, sustained gas exchange, an enhanced antioxidant capacity, and effective osmotic adjustments are all underpinned by the significant upregulation of key stress-responsive genes, including ADH1, RBOH, POD, and LEA, as well as stress signaling components. In contrast, the physiological and morphological collapse of 101-14 was directly correlated with its failure to mount a similar molecular defense, leading to uncontrolled oxidative damage and mortality.
From a theoretical standpoint, this research contributes a valuable comparative model for understanding plant adaptation to hypoxia in woody perennials. Our findings point out the vital role of a proactive and integrated defense network, where anaerobic metabolism (ADH1), ROS homeostasis (RBOH, POD), and cellular stabilization (LEA) are co-regulated. This work moves beyond identifying tolerance to explaining its molecular architecture, providing a clear link between specific gene expression patterns and whole-plant physiological resilience. The stark contrast between the two genotypes reinforces the concept that tolerance is an active process of genetic activation rather than a passive endurance of stress, offering a clear blueprint of the molecular machinery required for survival under waterlogged conditions.
The practical implications of these findings for the viticulture industry are significant. First and foremost, we provide robust, evidence-based support for recommending the SO4 rootstock for cultivation in regions prone to seasonal flooding or high rainfall levels or in vineyards with poorly drained soils. This investigation offers growers a tangible strategy for mitigating crop losses associated with climate change-induced extreme weather events. Second, the seven stress-responsive genes identified and validated in SO4 represent promising candidate biomarkers for future breeding programs. These genes could be used to develop molecular screening tools to assess waterlogging tolerance rapidly in new rootstock hybrids, accelerating the development of climate-resilient grapevine varieties without the need for lengthy and resource-intensive field trials.
While this study provides valuable insights under controlled conditions, future research should aim to validate these findings in a field environment, where waterlogging often co-occurs with other biotic and abiotic stresses. A logical next step would be to employ a systems-biology approach, such as transcriptomics (RNA-Seq) and proteomics, to capture the full spectrum of genes and proteins involved in the adaptive response of SO4. Investigating the long-term impacts of intermittent waterlogging on grape yield and quality when using the SO4 rootstock would also be crucial. Finally, functional genomic studies, such as using CRISPR/Cas9 to knock out key identified genes in SO4, would definitively confirm their roles in conferring tolerance and further solidify their utility as targets for genetic improvements. Ultimately, this study lays the groundwork for both immediate practical applications and future fundamental research aimed at enhancing the sustainability of viticulture worldwide.
Supplementary Material
Supplementary materials are available at Horticultural Science and Technology website (https://www.hst-j.org).
- HORT_20260007_Table_S1.doc
Supplementary Table S1. Primers used for qPCR
- HORT_20260007_Table_S2.docx
Supplementary Table S2. Two-way ANOVA summary table (F-values and significance levels) for the main effects of rootstock and treatment and their interaction.
- HORT_20260007_Table_S3.doc
Supplementary Table S3. Effect of waterlogging on oxidative stress markers, photosynthetic parameters, growth characteristics, antioxidant enzyme activities, and root architecture in two grapevine rootstocks (SO4 and 101–14) during 28 days of stress.
- HORT_20260007_Table_S4.doc
Supplementary Table S4. Effect of waterlogging treatment on oxidative stress markers, photosynthetic parameters, growth characteristics, antioxidant enzyme activities, and root architecture in grapevine rootstocks during 28 days.