Introduction
Materials and Methods
Test materials
Acquisition of sterile explants of Hongyan and Shengdanhong strawberries
Stem tip stripping and primary culturing
Multiple shoot subculture multiplication culture
Adventitious roots induction
Detection of the detoxification effect of tissue culture seedlings
Results and Discussion
Effects of different concentrations of IBA and the 6-BA in strawberry microstem tip culture in vitro
Effects of different hormone combinations and concentrations on buds multiplication culturing
Effects of different combinations and concentrations of plant growth regulators on rooting culture
Virus-free detection of strawberry tissue culture seedlings
Conclusions
Introduction
Strawberry is a perennial herb with high nutritional and economic value, and its fruit is delicious and loved by people. The traditional production of strawberry seedlings mostly relies on separate propagation and creeping stem propagation, though the propagation speed is slow and virus infections readily occur, resulting in certain types of degradation, such as quality deterioration, which is not conducive to the promotion and planting of excellent varieties, such that the strawberry industry is encountering bottleneck problems (Hernández-Martínez et al. 2023). At present, the strawberry viruses that are seriously harmful in China mainly include strawberry crinkle virus (SCV), strawberry mottled virus (SMoV), strawberry mild yellow edge virus (SMYEV), and strawberry vein banding virus (SVBV) (Klerks et al. 2004; Wei et al. 2008; Takamura et al. 2020). Micropropagation is the most promising method of plant tissue culturing for the rapid multiplication and conservation of germplasm. Tissue culturing of the strawberry stem tip is the most effective method of maintaining the excellent traits of strawberries at present, having the advantages of a short breeding time, strong plant growth, high yields and excellent quality, all of which effectively improve the quality of seedlings (Yu et al. 2015; Żebrowska 2015; Farjana et al. 2023). The RT-PCR (reverse transcription-polymerase chain reaction) technique can be used for the detection of viruses (Chang et al. 2007; Veetil et al. 2016; Diaz-Lara et al. 2021; Zhao et al. 2022), with the additional advantages of being fast and sensitive.
At present, although there have been many studies of strawberry tissue culturing and rapid propagation systems, there are large differences in the combination and proportion of exogenous plant hormones (Debnath 2009; Sehrawat et al. 2016; Lee et al. 2017; Karmaker et al. 2023), which hinder the practical application of strawberry detoxification and rapid propagation technology and which also require further development. In the present investigation, creeping stem tips of the two genotypes of “Shengdanhong” and “Hongyan” were selected as explants and the most suitable hormone ratios of MS basal medium for the two strawberry varieties were explored by studying these process of shoot-induced culturing, adventitious bud proliferation culturing and rooting culturing, with RT-PCR method used to detect the virus-carrying conditions of strawberry tissue culture seedlings and to obtain virus-free seedlings, all in an effort to establish a strawberry stem-tip tissue cultivation and rapid propagation system. Doing this will provide an important theoretical basis for propagating virus-free strawberry seedlings and proper application methods.
Materials and Methods
Test materials
In this study, two annotinous strawberry varieties of “Hongyan” (originating in Japan) and “Shengdanhong” (originating in Korea) were selected as test materials, and the materials for trial research were cultivated in a greenhouse provided by the strawberry planting base of Agricultural Science and Technology Industrial Park, which is located in Nanyang, China.
Acquisition of sterile explants of Hongyan and Shengdanhong strawberries
Picked and cut shoot tips of the creeping stems of “Hongyan” and “Shengdanhong” about 2 cm in length had the outer bracts removed, the explants of which were then placed in a small beaker, rinsed under a water stream for an hour, soaked in a detergent solution for 20 min, and then rinsed again with running water for 30 min. The cleaned experimental material was placed in a sterile beaker of an appropriate size and sterilized on an ultra-clean bench. First, the specimens were soaked in a 70% alcohol solution for 30 s, rinsed twice with sterile water after removal, and then soaked and disinfected with a 0.1% mercury solution for 10 min. The solution was shaken continuously during this period and finally washed again with sterile water three to five times and set aside.
Stem tip stripping and primary culturing
After the sterilization treatment, asepsis stem tips were peeled off under a stereoscopic anatomical mirror. Young leaves and scale were peeled off layer by layer from the outside to the inside with a dissecting needle, fully exposing the growth points, with stem tip growth point tissue cut to 0.3 - 0.5 mm in size with two leaf primordiums as explants. These were then inoculated on microstem APEX tissue on a MS medium gel consisting of 0.8% (w/v) Phytagel with 30 g·L-1 sucrose added, supplemented with three 6-BA concentrations (0.5 mg·L-1, 1.0 mg·L-1 and 1.5 mg·L-1) and two IBA concentrations (0.1 mg·L-1 and 0.2 mg·L-1). In addition, a CK control was established (Table 1). A total of 560 explants were inoculated to obtain asepsis shoots. The inoculated cultures were grown and maintained at 25 ± 2°C with a 16 h photoperiod under white fluorescent light (3000 lux) and 8 h in the dark in a bioclean room.
Table 1.
Effects of Different Hormone Concentrations on the Callus and Adventitious Bud Induction from Stem Tip of Two Strawberry Genotype Varieties
Multiple shoot subculture multiplication culture
An efficient adventitious buds multiplication medium was investigated using MS medium containing cytokinins at various concentrations. Robust tissue culture buds were selected and induced by the primary cultivation of “Hongyan” and “Shengdanhong,” with callus and other browning parts carried by the explants disposed of, with inoculation following on MS medium for multiple shoot regeneration and gelling with 0.8% (w/v) Phytagel and 25 g·L-1 of sucrose added. In total, seven formulation treatments were used for the subculture: 6-BA (0.5 mg·L-1 and 1.0 mg·L-1) and IBA (0.1 mg·L-1 and 0.2 mg·L-1) were added to formulae 1-4, 6-BA (0.2 mg·L-1 and 0.5 mg·L-1) and NAA (0.1 mg·L-1and 0.2 mg·L-1) were added to formulae 5 and 6, and CK was set as a blank control (Table 2). A total of 336 explants were inoculated and maintained at 25 ± 2°C with a 16 h photoperiod under white fluorescent light (3000 lux) with 8 h in the dark in a bioclean room.
Table 2.
Effects of Different Hormones Combinations and Concentrations on the Multiplication of the Adventitious Buds of Two Strawberry Genotype Varieties
Adventitious roots induction
After the end of the subsequent proliferation culture experiment, robust tissue culture seedlings of “Hongyan” and “Shengdanhong” were selected and inoculated on a 1/2 MS medium gel with 0.8% (w/v) Phytagel and 30 g·L-1 of sucrose added to induced adventitious roots. Medium formulae 1-3 were supplemented with NAA 0.2 mg·L-1, 0.5 mg·L-1 and 1.0 mg·L-1, respectively, and medium formulae 4-6 had IBA 0.2 mg·L-1, 0.5 mg·L-1 and 1.0 mg·L-1 added, respectively, CK served as the blank control (Table 3). Half of each formula media had activated charcoal added (2 g·L-1), with the other parts not treated. A total of 424 explants were inoculated. First, these rootless seedlings cultured were placed in darkness for one week, after which the inoculated cultures were grown and maintained at 25 ± 2°C with a 16 h photoperiod under white fluorescent light (3000 lux), with 8 h in the dark in a bioclean room, as above.
Table 3.
Effects of Different Hormones Combinations and Concentrations on Root Formation of Two Different Strawberry Varieties
Detection of the detoxification effect of tissue culture seedlings
The RT-PCR procedure was utilized to detect the virus-carrying conditions of the strawberry tissue culture seedlings and to ensure that virus-free seedlings as pre-basic seeds were obtained. Here, 40 test tissue culture seedlings were selected from 14 treatments of “Hongyan” and “Shengdanhong;” three tissue culture seedlings were randomly selected in each treatment. In addition, three annotinous seedlings of “Hongyan” cultivated in an open field were selected as a control group. RNA extraction kits (Plant RNA kit r6827-01 kits) were used to extract total RNA from the leaves of the sample tissue culture seedlings. Detection was conducted with 1% agarose gel electrophoresis and imaging was undertaken with a Bio-Rad gel imaging system. In addition, cDNA was obtained based on total RNA reverse transcription, with the recommended test procedure of the cDNA strand synthesis kit followed.
Two pairs of specific primer sequences of the SMoV and SCV virus types were subjected to PCR amplification, with four strawberry viruses in total synthesized by the Wuhan Qingke Biological Company (Table 4). Using the 3G Taq Master Mix (Red Dye) (Vazyme Biotech Co., Ltd.) containing the Taq enzyme, 43 cDNA specimens obtained by reverse transcription were used as templates for PCR amplification and detection. The RT-PCR reaction system of 20 ul was as follows: 3G Taq Master Mix (Red Dye) at 10 ul, upstream and downstream primers at 1 ul, and cDNA template at 2 ul supplemented with ddH2O a 20 ul. The PCR amplification procedure based on specific primers of the SMoV virus was as follows: pre denaturation at 94°C for 2 min, followed by 35 cycles of denaturation at 94°C for 30 s, annealing at 55°C for 40 s, extension at 72°C for 30 s, and then extension at 72°C for 5 min, with storage at 4°C. The PCR amplification procedures based on specific primers of the SCV virus used the following steps: pre denaturation at 94ºC for 2 min,followed by 35 cycles of denaturation at 94°C for 30 s, annealing at 56.5°C for 40 s, extension at 72°C for 30 s, and then extension at 72°C for 5 min, with storage at 4°C. PCR amplification products were detected by means of 1.5% agarose gel electrophoresis and were photographed and analyzed using a Bio-Rad gel imaging system.
Results and Discussion
Effects of different concentrations of IBA and the 6-BA in strawberry microstem tip culture in vitro
Strawberry stem tip meristems were inoculated into the primary medium and clusters of buds could be distinctly differentiated in 60 days. The data in Table 1 show that when the IBA concentration was quantitative, the survival rate of stem tips and the induction rate of adventible buds showed a trend of initially increasing and then decreasing with an increase in the 6-BA concentration. When the concentration of 6-BA remained constant, the survival rate of stem tips and the induction rate of adventible buds decreased with an increase in the IBA concentration. For the MS medium supplemented with 0.5 mg·L-1 6-BA and 0.2 mg·L-1 IBA, the stem tip survival rate, callus induction rate and shoot induction rate of ‘Shengdanhong’ reached 84.21%, 81.58% and 84.21%, respectively (Fig. 1A and 1B). For the MS medium supplemented with 1.0 mg·L-1 6-BA and 0.1 mg·L-1 IBA, the stem tip survival rate, callus induction rate and adventitious bud induction rate of ‘Hongyan’ reached 100%, 85.71% and 88.10%, respectively (Fig. 1C and 1D). It can be seen that the hormone concentrations required for the ex vivo culturing of strawberries of different genotypes are different.

Fig. 1.
Shoot induction from the micro-stem tip of ‘Shengdanhong’ and ‘Hongyan’ strawberries on the medium component of the stem tip primary culture for 60 days: A, B) Initiation and regeneration of the maximum frequency of the ‘Shengdanhong’ strawberry multiple shoot culture in the 6-BA medium (0.5 mg/L) and the IBA (0.2 mg/L) medium. C, D) Initiation and regeneration of the maximum frequency of the ‘Hongyan’ strawberry multiple shoot culture in the 6-BA medium (1.0 mg/L) and the IBA (0.1 mg/L) medium. Scale bar = 1.0 cm.
Effects of different hormone combinations and concentrations on buds multiplication culturing
An analysis of the data in Table 2 shows that the adventitious bud proliferation coefficient of the ‘Shengdanhong’ and ‘Hongyan’ strawberry tissue culture seedlings in the MS induction culture medium component containing different hormone combinations and concentrations of subculture multiplication culture have significant differences. Figure 2 (A1, B1, C1, D1) shows different effects of the adventitious bud propagation of Strawberry ‘Shengdanhong’ and ‘Hongyan’ on the medium component of the different hormones combination and concentrations used for the subculture multiplication culture over a period of 40 days. When the concentration of 6-BA was 0.5 or 1.0 mg·L-1, the adventitious bud proliferation coefficients of both strawberry ‘Shengdanhong’ and ‘Hongyan’ types increased with an increase in the IBA concentration. When the IBA concentration was 0.1 or 0.2 mg·L-1, the adventitious bud proliferation coefficient of Poinsettia increased with an increase in the 6-BA concentration. A comprehensive analysis revealed that the maximum proliferation coefficient was achieved with the MS medium containing 0.2 mg·L-1 6-BA and 0.1 mg·L-1 NAA among these different medium treatments; the proliferation coefficient of “Shengdanhong” was 6.11, and the proliferation coefficient of “Hongyan” was 7.13, with strong seedlings with an abundant callus. This medium treatment was considered as the optimization medium for bud subculture multiplication.

Fig. 2.
Adventitious bud propagation of Strawberry ‘Shengdanhong’ and ‘Hongyan’ strawberries on the medium component of the subculture multiplication culture for 40 days. A1. Adventitious bud propagation of ‘Shengdanhong’ strawberries in the 6-BA medium (0.5 mg/L) and IBA (0.1 mg/L) medium; B1. Adventitious bud propagation of ‘Shengdanhong’ strawberries in the 6-BA medium (0.5 mg/L) and NAA (0.2 mg/L) medium; C1. Adventitious bud propagation of ‘Hongyan’ strawberries in the CK medium; D1. Adventitious bud propagation of ‘Hongyan’ strawberries in the 6-BA medium (0.2 mg/L) and NAA (0.1 mg/L) medium. Scale bar = 1.0 cm.
Effects of different combinations and concentrations of plant growth regulators on rooting culture
The optimal rooting induction culture medium formula was the 1/2 MS induction culture medium supplemented with 0.5 mg·L-1 IBA,30 g·L-1 sucrose and 7 g·L-1 agar. Using the 1/2 MS medium as the basic medium, healthy rootless strawberry seedlings were inoculated into the rooting medium with different hormone concentrations to observe the rooting condition. The datasets in Table 3 show that for the “Shengdanhong” variety with the 1/2 MS medium supplemented with 0.5 mg·L-1 IBA, the rooting rate was 72.41%, the average root number was 17.7 at most, and the plant was tall and robust. For the “Hongyan” variety, with 0.5 mg·L-1 IBA added to the 1/2 MS medium, the highest rooting rate was 91.67%, the average root number was 21, the root system was strong, the plants were strong and tall, and the callus appeared.
Virus-free detection of strawberry tissue culture seedlings
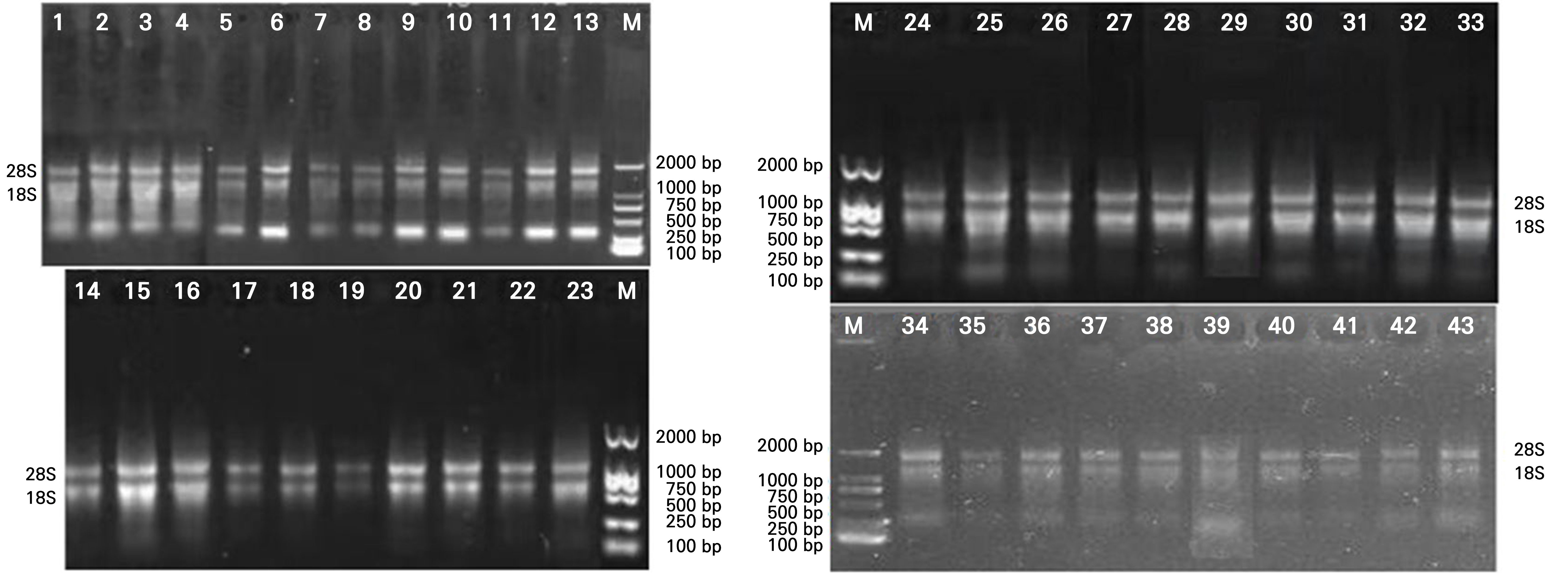
Total RNA from the leaves of 40 strawberry tissue culture seedlings was isolated and detected, with the 18S and 28S bands relatively complete and clear, possibly meeting the quality requirements for reverse transcription (Fig. 3). The virus-free rates of the stem tip cultured in-vitro seedling samples of the two strawberry varieties were ascertained and assessed by primer screening, PCR amplification, and 1.5% agarose gel electrophoresis (Figs. 4 and 5). Among the 40 stem tip cultured sample,the number of plants carrying the SMoV virus amounted to three, and the number of virus-free seedlings came to 37, indicating a virus-free rate of 92.5%. None of the strawberry stem tip tissue culture seedlings carried SCV, and the number of tissue culture seedlings without SCV was 40, for a virus-free rate of 100%. Comprehensive research presents that the virus-free rate examined by RT-PCR of the tissue culture seedlings in vitro cultivated was 92.5%, demonstrating that tissue culture seedlings micro-propagated from creeping stem tips were expeditious and reliable for the breeding of virus-free seedlings.
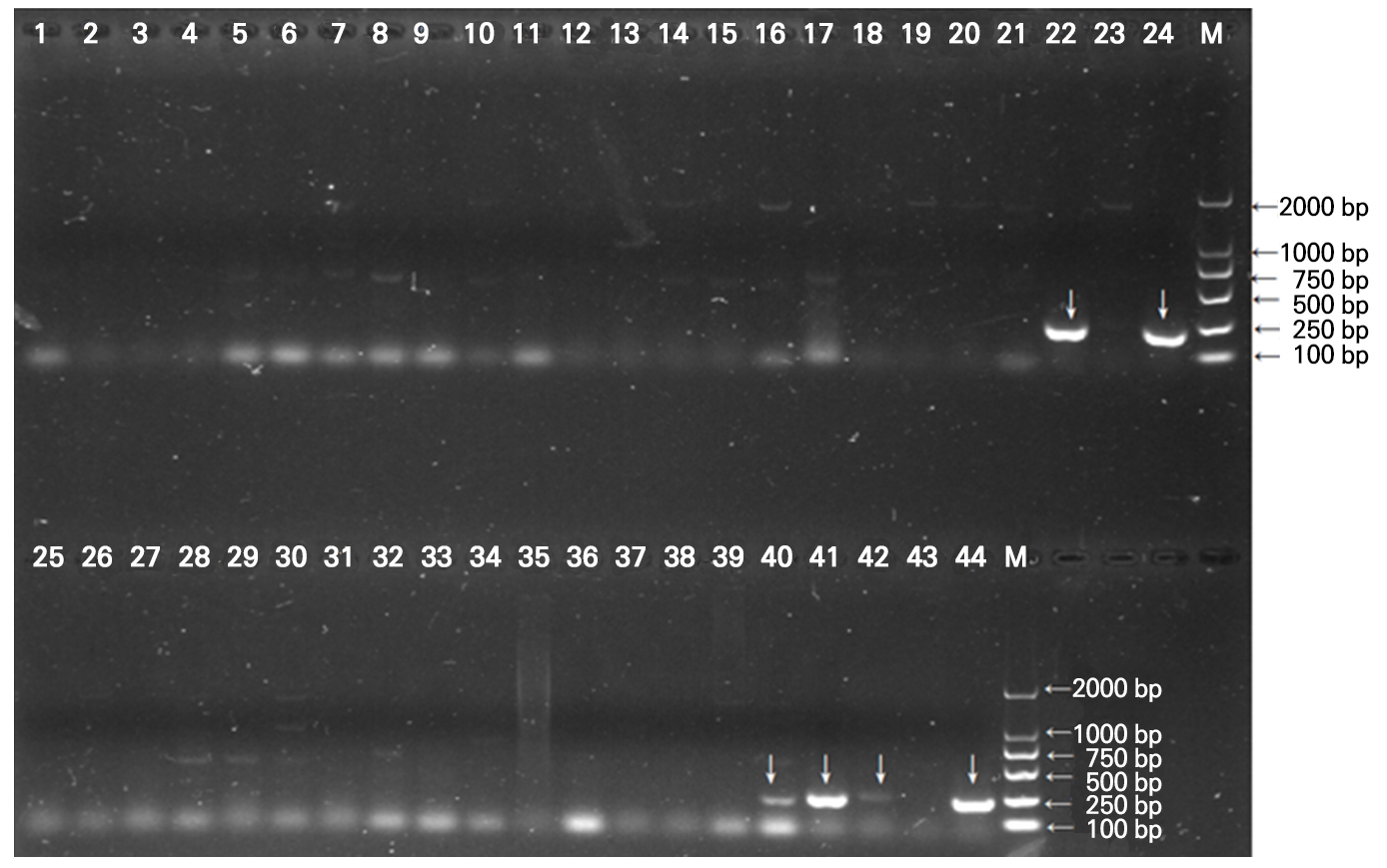
Fig. 4.
Results of 1% agarose gel electrophoresis detection of ‘Shengdanhong’ and ‘Hongyan’ strawberries for the SMoV virus by RT-PCR. Lane M: Trans2K Plus Ⅱ DNA Marker. Lane 1-40: PCR amplification product detection of in-vitro regenerated seedlings; 41-43 show the PCR amplification product detection outcomes of cultivated plants. Downward indicating arrows denote the amplified positive DNA band of RT-PCR samples 22, 24, 40, 41, 42, and 44 in their corresponding lanes.

Fig. 5.
Results of 1% agarose gel electrophoresis detection of ‘Shengdanhong’ and ‘Hongyan’ strawberries for the SCV virus by RT-PCR.Lane M: Trans2K Plus Ⅱ DNA Marker. Lane1-40: PCR amplification product detection of in-vitro regenerated seedlings. 41-43 show the PCR amplification product detection outcomes of cultivated plants.
Conclusions
In the primary medium, an appropriate plant growth regulator such as 6-BA combined with IBA was beneficial for the induction of explants (Cappelletti et al. 2016; Rimy et al. 2024). The present study investigated the effects of different concentrations and combinations of plant growth regulators on the in vitro regeneration process of strawberries. The results showed that with the treatment of an induction culture MS medium containing the best combination of 0.5 mg·L-1 6-BA and 0.2 mg·L-1 IBA, the shoot tip survival rate, callus induction rate and adventitious shoot induction rate of “Shengdanhong” were found to be 84.21%, 81.58% and 84.21%, respectively. Under the induction culture of MS basal media containing the best combination of 1.0 mg·L-1 6-BA and 0.1 mg·L-1 IBA, the shoot tip survival rate, callus induction rate and adventitial shoot induction rate of “Hongyan” were 100%, 85.71% and 88.10%, respectively. It was therefore confirmed that explants of different varieties require different types and concentrations of optimal growth regulators (Wang et al. 2015; Naing et al. 2019). In the adventitious bud proliferation culture, the results found by a comparison of three hormones at different concentrations showed that adding 0.2 mg·L-1 6-BA and 0.1 mg·L-1 NAA led to the best proliferation coefficients of “Shengdanhong” and “Hongyan,” at 6.11 and 7.13, respectively. The increment coefficients of “Hongyan” were significantly different from those of other formulations (p < 0.5). Through rooting culture assessments, it was found that rooting induction was straightforward for strawberry seedlings, as a large number of roots could be induced in a medium without a plant growth regulator. The rooting effect of IBA was better than that of NAA. When the concentration of IBA was 0.5 mg·L-1, the rooting rates of “Shengdanhong” and “Hongyan” were phenomenal and efficient (72.41% and 91.67%, respectively), and the average root number was the highest (17.7 and 21, respectively). In addition, it was found that adding 2 g·L-1 AC could effectively prevent browning of the plants; in addition, the roots of the plants were stronger, the leaf color was dark green, and growth was robust. In this study, two strawberry viruses, SMoV and SCV, were successfully detected by the RT-PCR method, with virus-free rates up to 92.5%. In order to ensure the reliability and feasibility of a negative control, an internal standard can be introduced to further address this issue. Therefore, it is of great significance to use the virus-free technology related to in-vitro shoot tip culturing to establish a rapid propagation system for strawberries, to breed strawberry pre-basic seed seedlings, to improve the quality and yields of strawberries, and promote the industrialization of these fruit (Chen et al. 2019).