Introduction
Materials and Methods
Plant materials and growth conditions
Quantitative analysis of IAA
Sample embedding
Immunostaining
Detection of lignin deposition
Results
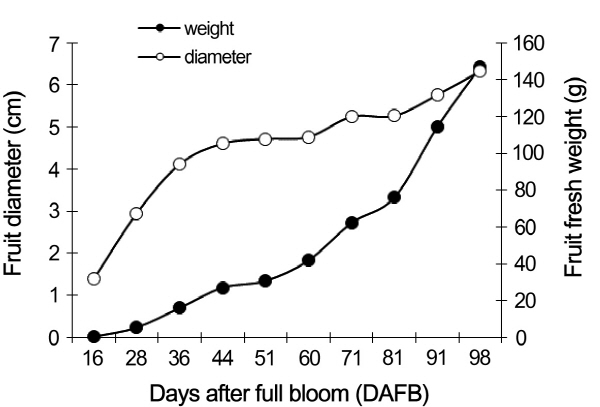
Growth curve of peach fruit
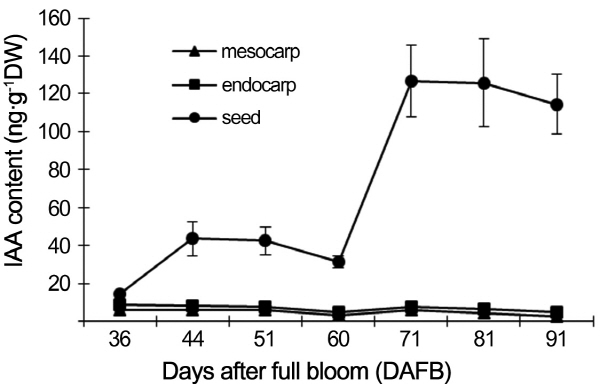
IAA levels in different tissues of peach fruit
Immunohistochemical location of IAA in various fruit tissues at different development stages
IAA distribution in seeds
IAA distribution in endocarp and mesocarp
Effect of NPA or TIBA treatment on IAA distribution in mesocarp of peach fruit
Verification of immunohistochemical localization technique for IAA distribution
Discussion
Seed coat as the potential source of auxin biosynthesis
The IAA distribution in the endocarp was consistent with the lignification
IAA in the mesocarp may be derived from external transport
Conclusion
Introduction
The phytohormone auxin plays an important role in the regulation of many physiological processes of plant growth and development (Kalluri et al., 2011; Petrášek et al., 2011). In fruit development, great progress has been made toward understanding the regulatory mechanism of auxin on climac-teric fruit, although the majority of research has focused on tomato (Solanum lycopersicum) (Molesini et al., 2009; Wang et al., 2005; Goetz et al., 2007; Mounet et al., 2009). Strawberry (Fragaria ananassa), which is widely used as a research model to study nonclimacteric fruits, has also been associated with auxin-induced ripening, and previous studies focused on the genes related to auxin signal transduction and in situ distribution of auxin (Liu et al., 2011; Hou and Huang, 2005).
Over the past few decades, peach (Prunus persica L.) has become one of the model species in the Rosaceae family (Verde et al., 2013). Ethylene and auxin have been regarded as the main phytohormone regulators of peach ripening (Trainotti et al., 2007). This study demonstrates that auxin plays a role in ripening peaches and interacts with ethylene during this process. The transcriptomics approach used in this work confirms that many genes related to the auxin signal transduction pathway are expressed in the pulp. Another study demonstrated that the auxin transport carriers of the PIN1 gene were expressed in the ripening process of peach fruit (Paponov et al., 2005). Torrigiani showed that jasmonic acid and spermine could affect fruit ripening and were related with ethylene and auxin (Torrigiani et al., 2012). Auxin is an important molecule both in early and late development (Bonghi et al., 2011), but the signal generating sites and transport pathway of auxin are poorly understood at present.
In order to understand the role and mechanism of auxin, we must primarily reveal its distribution in plant tissues. Immunohistochemical localization of endogenous indole- 3-acetic acid (IAA) has been applied to sunflower embryos (Thomas et al., 2002), Arabidopsis thaliana seedlings (Sieburth, 1999), tobacco zygote and embryo (Chen et al., 2010), and poplar rhizogenesis (Dong et al., 2012). Hou and Huang (2004) confirmed that an IAA signal was present in the vascular tissue of strawberry. In the earlier phase of fruit development, the IAA signal was mainly localized in the phloem, while at the late stage, the signal appeared in the xylem and gradually increased with strawberry fruit development. In the achene, the IAA signal was mainly distributed in the developing embryos and in the tissues around them. The distribution of endogenous IAA and its role in the regulatory mechanism of peach fruit development are unknown.
Embryo development was closely monitored with peach fruit growth. In the early stage of fruit development, a variety of hormones were involved in the cross-talk, and auxin may play a direct role in the signal transduction (Bonghi et al., 2011). In order to explore the source of IAA and observe its spatial distribution and dynamic changes, the immunohistochemical localization of IAA was carried out in different tissues of peach fruits. Additionally, the study aimed to understand the effect of exogenous auxin transport inhibitors on the distribution of IAA and to understand the possible sites where IAA might be produced.
Materials and Methods
Plant materials and growth conditions
Three neighboring peach trees (P. persica L. cv. Luwangxian) located in the experimental orchard of Beijing University of Agriculture (Changping District, Beijing, China) were the source of fruits. Three peaches, with no symptoms of pests and diseases, were harvested from each tree at 16, 28, 36, 44, 51, 60, 71, 81, 91, and 98 days after full bloom (DAFB) in 2012. At 60 DAFB, the carpopodium of 10 fruits were treated with 50 μmol・L-1 NPA (naphthylphthalamic acid, NPA) or 50 μmol・L-1 TIBA (2, 3, 5-Triiodobenzoic acid, TIBA) in vivo, the treatment with distilled water was used as control. The following steps were the treatment procedure. Firstly, carpogonium was rinsed with distilled water and its bark tissue was rubbed gently by forceps to remove them off. Then a piece of cotton soaked with NPA, TIBA reagent or distilled water was wrapped around the treated carpogonium, which was sealed with parafilm to avoid solution evaporation. Three days later, the treated fruits were sampled for a further study.
Quantitative analysis of IAA
The endogenous IAA content was analyzed according to the description of Liu (2013). The mesocarp, endocarp and seed of peach fruit were ground in liquid nitrogen separately, and one g of ground samples were extracted overnight at 4°C in 80% methanol with sodium diethyldithiocarbamate as an antioxidant. An stable isotope-labeled 13C6-IAA (13C6-IAA were donated by Professor Aloys Hüttermann, University of Göttingen, Germany) were added as internal standards. The sample was purified further with 3-cc Oasis anion MCX SPE (Waters, USA). The eluted solution was dried under vacuum, dissolved in 50 μL of HPLC initial mobile phase, and filtered through a 0.25-μm filter (4 mm in diameter). Ten microliters of sample solution was injected into the HPLC-MSn system (ESI). The separation of mixed compound was performed by Surveyor LC pump (Thermo Finnigan, Waltham, MA, USA) and C18 column (2.1 × 150 mm, 3.5 μm; Agilent, Santa Clara, CA, USA). An ion trap mass spectrometer (LCQ DECA XP MAX, Thermo-Finnigan, USA) coupled to an ESI source (Thermo Finnigan, USA) was used to identify the target compounds. The data were analyzed by Xcalibur 2.1 (Thermo Finnigan, USA). Each analysis was performed in triplicate.
Sample embedding
Harvested fruits were separated into seed, endocarp, and mesocarp quickly. Along the ventral suture, the mesocarp and endocarp were cut into approximately 0.05 cm3 blocks. Seeds were slit on one side of the raphe at the early development; after 60 DAFB, seeds were divided into three parts –proximal, medial and distal parts – to be used as embedding samples. All excised samples were pre-fixed immediately in 2% (w/v) aqueous solution of 1-ethyl-3 (3-dimethyl-aminopropyl)-carbodiimide hydrochloride (EDAC, Sigma, USA) for 1h, then vacuumed and washed three times with 0.1 mol・L-1 phosphate-buffered saline (PBS, pH 7.2). After washing, the samples were post-fixed overnight in a solution containing 4% paraformaldehyde and 2.5% glutaraldehyde at 4°C, dehydrated with n-butanol and ethanol, embedded in paraffin, and sectioned into 10 μm slices. The slides were spread with polylysine before the fixing of the sections. Dried sections were deparaffinized with xylene and hydrated in an ethanol-water series.
Immunostaining
The procedure for the immunolocalization of IAA was performed as described by Hou and Huang (2005) with some modifications. Slides were incubated in a blocking solution (3% H2O2) and washed briefly with a PBS solution. Antigen was restored with 0.01 mol・L-1citrate buffer. After washed with PBS, the slides were immersed in normal goat serum for blocking, and then washed briefly with the PBS solution. A drop of 100 μL primary IAA antibodies (Bioss, China) was added to each slide before the inner membrane was covered with plastic gloves and incubated overnight at 4°C. The slides were washed vigorously with the PBS solution. Subsequently, the slides were incubated overnight with phycoerythrin (PE)-Cy3-labeled goat anti- mouse IgG (Bioss, China) at room temperature. After washed with PBS, the slides were stained with the 4, 6-diamidino- 2-phenylindole (DAPI) solution, fixed by anti-quenching fluorescent mounting medium, then observed and photo-graphed by Olympus BX51-400X microscope (Olympus, Japan). Analysis was done using Motic Images Plus 2.0 software (Motic, China).
Two negative controls were used to verify the specificity of the antibody. One omitted the primary IAA antibody to test the non-specificity of IgG, while the other substituted non-immune goat serum for the IAA antibody to test the specificity of antibody. All other procedures were implemented as usual.
Detection of lignin deposition
A phloroglucinol-HCl reagent was used to stain tissues of different developmental peach fruit (Abeles and Biles, 1991). The fruit was cross-sectioned. After the reagent treatment, the stereomicroscope was used to observe the lignin deposition.
Results
Growth curve of peach fruit
The weight and diameter of growing fruits were monitored from 16 to 98 DAFB. Both parameters were characterized by three fruit growth stages: S1, S2, and S3 (Fig. 1). The first phase, rapid growth, was started from 16 DAFB to 44 DAFB, and the primary characteristic of this stage was the intensive equatorial growth. The stage from 44 DAFB to 71 DAFB was the hardening period, including a slow growth rate. The second fast-growth period was started from 71 DAFB to 98 DAFB, during which the diameter and fruit weight increased rapidly.
IAA levels in different tissues of peach fruit
The levels of IAA in mesocarp, endocarp and seed were assayed by HPLC-MS (Fig. 2). The level of IAA in seed was much higher than that in mesocarp and endocarp. There was no obvious difference between mesocarp and endocarp, in which tissues the level of IAA was little related with the development stage. After 60 DAFB, the level of IAA in seed was increased rapidly, reaching to a maximum at 71 DAFB.
|
Fig. 1. Growth curve of peach (‘Luwangxian’) fruit. |
|
Fig. 2. Changes in IAA content in different tissues of peach (‘Luwangxian’) fruit. Values are means ± SE (n = 3). |
Immunohistochemical location of IAA in various fruit tissues at different development stages
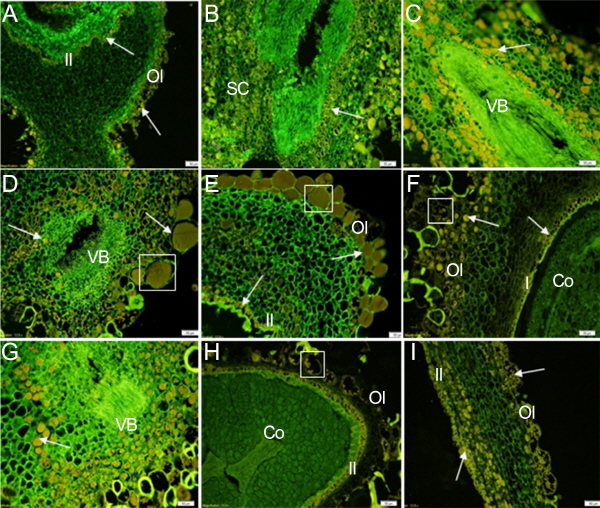
The immunolocalization assay was used to investigate the endogenous IAA distribution in various tissues of peach fruit during development. The results are shown in Figure 3-6 in which the green fluorescent color reflects the cell position stained by DAPI dye, and the yellow fluorescent color is the IAA immunolocalization signal reflecting the level and distribution of endogenous IAA.
IAA distribution in seeds
Histological observations of the seed are shown in Figure 3. A strong IAA signal could be detected during the develop-ment of seed. In the longitudinal sections (Figs. 3a, e, f, h, i), a layer of inner integument cells were relatively dense, and the distribution of the IAA signal was banded in these cells. It was interesting to see that outer integument cells were full of IAA signal during fruit development, with some of the mucilage cells having a strong IAA signal (Figs. 3d, e, f, h, i). The function of these cells has remained unclear, and although we did not observe direct evidence confirming their function whether they synthesize or store IAA, the presence of IAA in these cells was obvious. There was little IAA signal detected in tissues between the outer and inner integument. A strong IAA signal was detected in the seed coat in the transverse sections (Figs. 3b, c, d, g). In different development periods, there were no significant differences in IAA intensity. Vascular tissues contained no IAA signal, but strong IAA signals were detected in the layer of cells surrounding the vascular tissues (Figs. 3c, d, g). Little IAA signal was detected in cotyledon on the proximal side as the fruit was developing (Figs. 3f, h). The results of Figure 3 indicated that the IAA distribution in the seed was characterized by the spatial specificity.
IAA distribution in endocarp and mesocarp
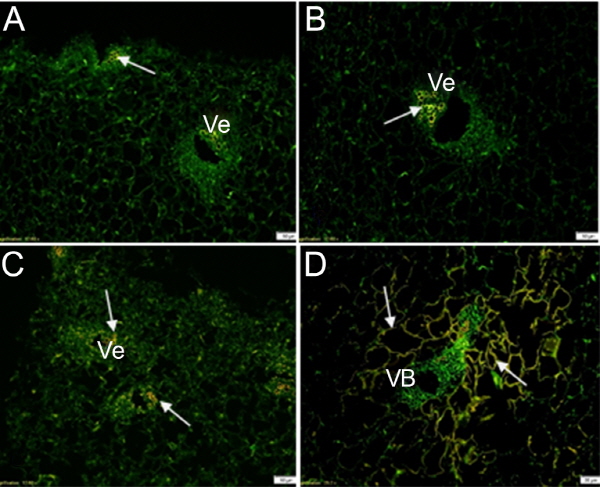
The distribution of IAA in the endocarp was analyzed before the tissue was lignified. At 28 DAFB, the endocarp cells were dense. A strong signal covering the entire cell was observed in the phloem surrounding the vessel tissue (Fig. 4a). Weak IAA signals were detected in endocarp cells at 51 DAFB (Fig. 4b).
The lignin deposition in the endocarp was observed via phloroglucinol-HCl staining (Fig. 5). At 71 DAFB, the endocarp was lignified entirely, except for the vascular tissue (Fig. 5b). The vessels within the vascular tissue were lignified, but the lignification process did not occur in the phloem tissue (Fig. 5c). Corresponding to the results of the IAA immunolocalization in the endocarp, there was no signal in the vessel. However there was a strong signal located in the phloem tissue within the vascular tissue (Fig. 4a). This result showed that auxin is not related to lignification of vessels, nor does it play the role in a negative regulation of lignification.
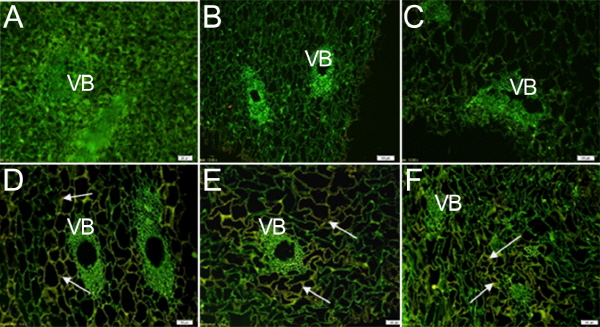
Compared with the seed, IAA signal in the mesocarp tissue was weakly detected throughout the whole growth period. In the earlier stages of development (before 51 DAFB), there was little IAA signal (Figs. 6 a-c); however, a little strong IAA signal was detected in the parenchyma cells around the vascular bundle at 60, 71, and 81 DAFB (Figs. 6 d-f);. The signal distribution was mainly concentrated in the outer layer of cells, near the cell wall, where it differed from the IAA signal in the seeds.
Effect of NPA or TIBA treatment on IAA distribution in mesocarp of peach fruit
TIBA and NPA, inhibitors of auxin transport by combining with a carrier protein that is responsible for IAA transport, could change the protein conformation or inhibit the specific binding to repress the polar transport of IAA. No changes in IAA distribution occurred in the endocarp or seed after the fruit treated with exogenous NPA or TIBA, but in the mesocarp, IAA distribution was dramatically altered. In the control (Fig. 7d), the IAA signal was located in the parenchyma cells surrounding the vascular tissues, as seen in Figure 6, but IAA signals were present in vessels after the fruit had been treated with TIBA (Figs. 7a, b) or NPA (Fig. 7c).
Verification of immunohistochemical localization technique for IAA distribution
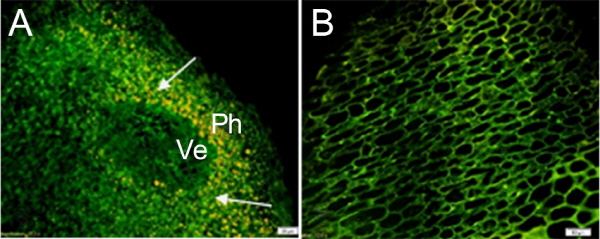
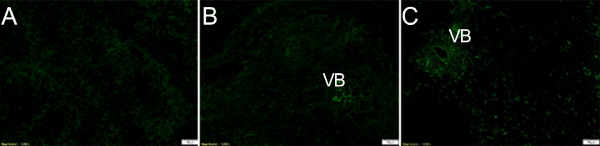
In order to verify the reliability of the immunolocalization technique, two controls of each tissue were carried out. The first control was the primary IAA antibody substituted by non-immune goat serum, and the second control was the primary IAA antibody omitted. The IAA distribution of the first negative controls in different tissues is shown in Figure 8. There was little IAA signal detected in these sections, indicating that the method was reliable and that the antibody was highly specific (Figs. 8 a-c).
Discussion
Auxin is one of the important phytohormones involved in regulating the development of peach fruit. IAA is a natural auxin in plants. We measured the level of IAA in peach fruit and found that the content of IAA was different in various tissues throughout the development. Miller (1987) showed that the level of IAA decreased with the growth of the embryo from 67 DAFB to 88 DAFB. Regardless of whether an enzyme immunoassay or mass spectrometry method was used, the distribution of auxin will be destroyed during the extraction process. Therefore, an immunohisto-chemical localization technique was used to explore in situ auxin distribution, which would be an important step towards clarifying the regulatory mechanism of auxin in peach fruit development.
Seed coat as the potential source of auxin biosynthesis
The study of IAA determination early in fruit development shows a specificity of the developmental stages in peach fruit (Miller et al., 1987). During fruit growth, the IAA content differed among different tissues. For the same growth periods, the IAA level was the highest in the seed, followed by endocarp and mesocarp. These results are consistent with our results determined by the immunohistochemical localization of IAA technique: The content of IAA was highest in seed, whereas it was low in mesocarp and endocarp tissues.
Bonghi et al. (2011) determined that peach embryogenesis and fruit development are tightly coupled. Moreover, phy-siological disturbances during early embryo development will lead to seed abortion and fruitlet abscission. Different hormones affect fruit and seeds differently, and auxin will induce specific gene expression in the early and late growth periods in peach fruit (Trainotti et al., 2007). Auxin produced by the seed may behave as a signal that is efficiently transported to and within the mesocarp. Normally in plants, the embryo is the vital source of auxin production (Yoshida et al., 2012). In our study, a strong signal was accumulated in the seed coat, indicating a specific distribution of endogenous IAA. In different parts of the seed, the outer and inner integument have one or more cell layers with IAA signals (Figs. 3a, e, f, h, i). In any developmental stage, stronger IAA signals were present in the mucilage cells. It can be inferred, therefore, that the seed coat of peach fruit may be the source of auxin. In dicotyledonous plants, such as peas, auxin is made in the endosperm (Bandurski et al., 1988). The auxin content can be assayed until the endosperm is reabsorbed, so it is speculated that the embryo is not the only source of auxin.
The IAA distribution in the endocarp was consistent with the lignification
There was a difference in IAA distribution between the mesocarp and endocarp in untreated fruit. The IAA signal was detected in the phloem of vascular tissues (Fig. 4). It has been previously reported that the vascular tissue in the endocarp of peach fruit belongs to collateral vascular bundles (Zhang et al., 2009). The IAA signal was located in the phloem tissue of vascular bundles in our research. The IAA signal in the endocarp is transported through the vascular tissue, but whether it comes from the seed coat or mesocarp was unknown. The results obtained by the phloroglucinol-stain technique showed that the vessels within the vascular tissue had lignified at the second stage of fruit development, but the lignification had not occurred in the parenchyma cells around the vascular tissue. At 28 DAFB, a strong IAA signal may have existed in parenchyma cells of vascular tissue to promote the vascular differentiation and formation, and the IAA signal did not appear in the vessels. Several studies confirmed that IAA was a negative regulator of lignin synthesis (Elobeid et al., 2012). Therefore, a weak IAA signal in the vessel is consistent with the physiological phenomenon of lignin synthesis.
IAA in the mesocarp may be derived from external transport
Several studies have addressed the role of auxin in plant and fruit development (Trainotti et al., 2007; Vanneste and Friml, 2009). These data indicate that an accumulation of IAA in the vascular bundles of the basal regions of the petioles induces the occurrence of rhizogenesis of poplar leaves (Dong et al., 2012). IAA plays a crucial role in fruit development and rhizogenesis and is transported by the vascular bundle to other tissues. Ruegger et al. (1997) proposed that continuous auxin transport is necessary for the formation of vascular bundles. Sieburth (1999) indicated that the auxin is important for the induction of vascular tissue development in Arabidopsis. Our results in peach did not correspond with the results of the previous studies. In our study, there was only a weak IAA signal detected in the vascular tissues, but in the cells around the vascular tissue there appeared strong IAA signals. Stronger IAA signals were located in the seed (Fig. 3). Bonghi (2011) determined that during peach fruit development, auxin is transported from the seed to other organs by the apoplastic pathway, which was coincided with our results that IAA was produced in the seed coat, but it was not transported symplastically, indicating that it was likely translocated through the apoplastic pathway.
Contradictory with our results, a previous study of the immunohistochemical localization of IAA was carried out in strawberry, which showed that the IAA signal spread all over the vascular tissues of the phloem (Hou and Huang, 2004). In the mescoap of peach (Fig. 6), a strong IAA signal was observed in the parenchyma cells surrounding the vascular tissue, but only a weak IAA signal was detected in the vascular tissue. This suggests that the mesocarp could produce IAA or depend on its transportation from other organs. At the later stages of fruit development, the IAA located in the parenchyma cells around vascular tissue might have been used to promote cell enlargement and mesocarp ripening. After the fruit had been treated with auxin transport inhibitors, however, the distribution of IAA within the mesocarp was switched: The IAA signal appeared in the vessels, a weak IAA signal was detected in the parenchyma cells, (Fig. 7). Treatment with TIBA or NPA would affect the normal transport of IAA, and an IAA signal could not be detected in the parenchyma cells surrounding vascular tissue after treatment. Consequently, the IAA within the parenchyma cells might be transported from other tissues in the mesocarp of normal fruit. It was interesting that the IAA signal was detected in the vessels in the mesocarp of treated fruit. As mentioned, the IAA within the seed might be transported out by apoplastic pathway, so IAA in the vessels might be transported from the endocarp while fruits were treated with TIBA or NPA.
Conclusion
The immunohistochemical analysis of IAA in peach fruit showed that the source of IAA in various fruit tissues was different. IAA was mainly derived from seed coat. In the endocarp, IAA was transported possibly through vascular tissue. In the mesocarp, IAA might come from seed coat, endocarp or other tissues, for instance, stem or leaves. Auxin differentially regulated peach development in various tissues, but the different modes of action might be consistent with their developmental characteristics. In future studies, we will assess different tissues of fruit to elucidate the role of IAA and its molecular mechanism in peach fruit development.