Introduction
Materials and Methods
Preparation of explants and surface sterilization of S. cumini leaves
Induction of embryogenic callus
Somatic embryo induction
Encapsulation of synthetic seeds
Physical properties of seeds
Seeds diameter
Roundness
Leaking
Regeneration ability
Statistical analysis
Results and Discussion
Induction, texture, and color of embryogenic callus
Time of appearance
Wet weight of embryogenic callus
Stages of embryogenic callus formation
Somatic embryo induction
Physical properties of seeds
Seeds regeneration ability
Conclusion
Introduction
Jamblang (S. cumini) is a medicinal plant from the Myrtaceae family, which is commonly found in tropical regions, including Asia and Australia, where it has been traditionally used in several forms of medicine. Various parts of the plant, including the fruit, bark, seeds, and leaves, possess therapeutic properties and have been used for centuries in traditional remedies. S. cumini seeds have been found to be useful for the treatment of conditions such as diabetes and digestive disorders (Qamar et al. 2021), while the leaves have been applied specifically to manage diabetes (Franco et al. 2020). The bioactivity of the plant is attributed to secondary metabolites, including flavonoids, polyphenols, saponins, and tannins (Qamar et al. 2022), which significantly contribute to its medicinal properties.
Due to the potential medicinal properties, preserving S. cumini is essential for further studies. The plant can be reproduced sexually using seeds, and vegetative propagation is achieved through grafting techniques (Qin et al. 2021; Singh et al. 2022). However, young, grafted plants are susceptible to collapse when exposed to strong winds due to their shallow roots. The application of grafting techniques on a large scale is also limited due to the relatively low number of suitable branches (Darwati et al. 2023). With regard to seed preservation, S. cumini seeds are considered recalcitrant and have a limited shelf life owing to their physical characteristics (Devi et al. 2018; Nair et al. 2020; Hardstaff et al. 2022). To address this challenge, one potential solution is to produce synthetic seeds using an encapsulation technique, where somatic embryos derived from in vitro culture methods are encapsulated.
Somatic embryos are obtained from the development of somatic cells without undergoing gamete fusion. Under stimulation with specific hormone compositions in the culture medium, somatic cells can form an embryogenic callus (Fang et al. 2022). An embryogenic callus is typically characterized by compact cellular structures, rounded and small-sized cells, large and uniform nuclei, and active cell division (Rajput et al. 2023).
According to previous studies, a combination of auxin and cytokinin hormones has been shown to stimulate embryogenic callus formation (Wu et al. 2020). The auxin 2,4-dichlorophenoxyacetic acid (2,4-D) promotes cell differentiation, organogenesis, and the induction of embryogenic calli, while 6-benzylaminopurine (BAP), a cytokinin, enhances cell division (de Sousa et al. 2022). A previous study showed that the synergistic interaction between 2,4-D and BAP can effectively induce embryogenic callus formation (Malik et al. 2020).
Embryogenic calli can subsequently develop into somatic embryos upon exposure to cytokinin supplementation. Thidiazuron (TDZ; N-phenyl-N´-1,2,3-thiazol-5-ylurea), a synthetic phenylurea compound with strong cytokinin-like activity, is known to promote induction, somatic embryogenesis, cell regeneration, and shoot organogenesis (Ahmad et al. 2021). TDZ induces somatic embryogenesis by modulating endogenous hormone levels and eliciting plant stress responses that trigger developmental reprogramming (Nautiyal et al. 2023).
Producing synthetic seeds using somatic embryos and applying an encapsulation technique is a valuable conservation strategy for the preservation of S. cumini plants. The encapsulation technique requires coating in vitro-grown plant parts with layers to produce seeds that can be replanted and grown into plantlets. The coating not only maintains the viability of somatic embryos but also offers protection against external materials that can reduce the quality (Saeed et al. 2020).
The use of compacting agents has been shown to significantly influence the success of encapsulation techniques. Alginate plays a significant role in preserving the active components of the compound used by preventing factors that can affect the quality of the enclosed seeds. Therefore, alginate is often used in the food industry because it is safe, biocompatible, and non-toxic (Thakur et al. 2018). The concentration of the compound as a capsule matrix compactor affects the quality and thickness of synthetic seeds as well as the penetrating ability of the embryo (Ouzhand et al. 2023).
One study reported that the encapsulation of Mentha spicata produced the best regeneration ability with a 3% alginate concentration, a storage temperature of 4°C, and a duration up to 12 weeks (Micheli et al. 2022). Another study on encapsulating synthetic shoot tips of Citrus sinensis L. found that treatments using 3% alginate with a storage temperature of 10°C up to 60 days led to successful regeneration and recovery (62.5%) (Mohamed et al. 2023). In addition, encapsulation of black zira with a 3% alginate concentration and 100 mM CaCl2 produced synthetic seeds with the highest germination rate (77.7%) (Ouzhand et al. 2023). Encapsulation of Viburnum dentatum L. has also been conducted at various temperatures and for different storage durations. The best results were obtained by the treatment of 4°C up to 80 days (Hatzilazarou et al. 2020).
Despite the growing interest in somatic embryogenesis as a promising technique for plant propagation and conservation, there remains a significant gap in the literature regarding the encapsulation of S. cumini somatic embryos. At present, no studies have systematically investigated the effects of varying alginate concentrations, temperatures, and storage durations on the encapsulation efficiency and subsequent development of S. cumini somatic embryos. Understanding how different alginate concentrations (2%, 3%, 4%), temperatures (15°C and 25°C), and storage durations (15, 30, and 45 days) influence the viability, the physical properties of the synthetic seeds, and long-term storage ability is essential for optimizing the encapsulation process. It is also important for enhancing the success rate of micropropagation protocols for economically and ecologically important species. Therefore, this study aims to address this gap by providing valuable insights into the applicability of encapsulation and its potential to enhance the conservation and propagation efforts of S. cumini. The focus is on determining the optimal concentration of alginate solidifying agent for encapsulating S. cumini somatic embryos based on certain physical property parameters, in this case the seed diameter, roundness, and leakage parameters. In addition, the study evaluates the ideal alginate concentration and storage duration leading to the best regeneration ability at temperatures of 15°C and 25°C.
Materials and Methods
This study was conducted at the Plant Biotechnology Laboratory, Faculty of Biotechnology, Universitas Atma Jaya Yogyakarta, Indonesia, from November of 2021 to April of 2022. Embryogenic callus induction was carried out using six combinations of hormone treatments. Alginate concentrations of 2%, 3%, and 4% were tested as parameters with which to assess the resulting physical properties. Meanwhile, the regeneration ability was evaluated using the same alginate concentrations along with storage durations of 15, 30, and 45 days at temperatures of 15°C and 25°C, and each test was conducted in five repetitions (Fig. 1).
Preparation of explants and surface sterilization of S. cumini leaves
A total of 13 isolates of the explant were used here; these were S. cumini leaves that were neither too young nor too old, typically the second or third leaf from the plant's branch. The selected leaves were clean and free from aphids, fungi, or signs of insect damage. In the laboratory, the base and tip were removed and the leaves were cut in half.
The leaves were washed under running water three times and with a detergent for three minutes, rinsed until the foam disappeared, then placed in a beaker and covered with aluminum foil. These explants were transferred to a laminar air flow (LAF) cabinet, rinsed twice with sterile water, and sterilized with 70% alcohol for one minute (Reshma et al. 2020). The plants were rinsed for an additional three times with sterile water and were sterilized using 16.67% chlorine from 35% NaClO and two to three drops of Tween 20 for five minutes. In addition, the samples were rinsed once with sterile water, with this followed by sterilization with 11.9% chlorine from 25% NaClO for ten minutes. Finally, the explants were rinsed three times with sterile water and dried on a petri dish lined with sterile filter paper (Sitthichoptham et al. 2023).
Induction of embryogenic callus
The tips of the tweezers and scalpel were sterilized by burning over a spirit lamp, after which the explant was cut into 1 cm × 1 cm pieces. In this study, the rim of the petri dish was also sterilized using the flame of the spirit lamp before being opened. The explants were then planted in the treatment medium containing varying concentrations of 2,4-D and BAP hormones (Ahn and Lee 2021). In addition, the induction date was recorded on the label, and observations were made with regard to the parameters, in this case the texture, color, appearance time, wet weight, and cell histology of the embryogenic callus.
Somatic embryo induction
Somatic embryo induction was conducted by transferring the embryogenic callus to Murashige and Skoog (MS) medium supplemented with 5 mg of L-1 TDZ hormone. Observations at this stage included color, embryogenic traits, and somatic embryogenic development. Embryogenic properties were assessed through morphological characteristics and observations of the somatic embryo preparations. This stage was essential to identify somatic embryos suitable for encapsulation.
Encapsulation of synthetic seeds
Encapsulation was conducted in the LAF sewing chamber using the necessary tools and materials. Somatic embryos were cut into 4–5 mm pieces with sterile scalpels and blades and then handled with sterile tweezers and placed into MS media containing Na-alginate. In addition, embryos were collected using a sterile plastic pipette, slowly dripped into a 100 mM calcium chloride solution, and left to stand for 15 minutes. The resulting capsules were then removed and immersed in a sterile aqueous solution for an additional 15 minutes.
Physical properties of seeds
The alginate concentrations used were 2%, 3%, and 4%, and the physical property parameters observed were the seed diameter, roundness, and leakage. In addition, the tests were conducted using 30 synthetic seeds, 15 for size and roundness measurements, and 15 for the leakage analysis.
Seeds diameter
A total of 15 synthetic seeds from each treatment were measured for their diameter using a digital caliper, and the size obtained from the average of five seeds represented the diameter. The larger the diameter, the better the quality of the synthetic seeds, as it showed that the somatic embryo was well covered.
Roundness
Roundness was determined by dividing the diameter of the largest seed by that of the smallest. Results closer to 1 showed a higher level of roundness, while values farther from 1 showed a lower level of roundness (oval shape).
Leaking
A total of 15 synthetic seeds were weighed to determine the initial weight; these were then stored at room temperature in a sterile aqueous solution for 30 days in culture bottles and covered with aluminum foil in a dark room. On the 30th day, the seeds were weighed again to determine the final weight. The smaller the percentage of leakage, the better the quality of the synthetic seeds. The leakage percentage was calculated using the following formula.
Regeneration ability
Regeneration ability was tested by planting synthetic seeds in a medium containing 2 mg L-1 BAP hormone in LAF. All materials to be used were placed in the LAF, and each culture bottle contained six planted synthetic seeds. Planting was carried out on the 15th, 30th, and 45th days. The parameters used in this test were morphology and the percentage of regeneration, calculated by dividing the number of seeds that successfully formed buds by the total number of seeds planted (six seeds per bottle) and then multiplying by 100%.
Statistical analysis
The data analysis in this study was conducted both qualitatively and quantitatively. The qualitative data, which were the embryogenic callus texture, color, cell histology, as well as the somatic embryogenic cell histology and morphology, were analyzed descriptively. Quantitative data were analyzed using a factorial block design for the embryogenic callus emergence time and wet weight, and a completely randomized design for the seed diameter, roundness, leakage, and regeneration ability. All data were subjected to analysis of variance (ANOVA) at a 95% confidence level. When significant differences were found, the analysis was followed by Duncan's multiple range test (DMRT). All tests were performed using SPSS version 23.
Results and Discussion
Induction, texture, and color of embryogenic callus
The results obtained in the induction embryogenic callus stage pertained to the texture and color of the embryogenic callus, the time of emergence, the wet weight, and the stages of formation from the S. cumini leaf explant. The texture and color of the embryogenic callus were visually observed, and the results showed that an intermediate texture was produced at each treatment (Table 1).
Table 1.
Texture and color of embryogenic calli from S. cumini leaf explants at 60 days after planting (DAP)
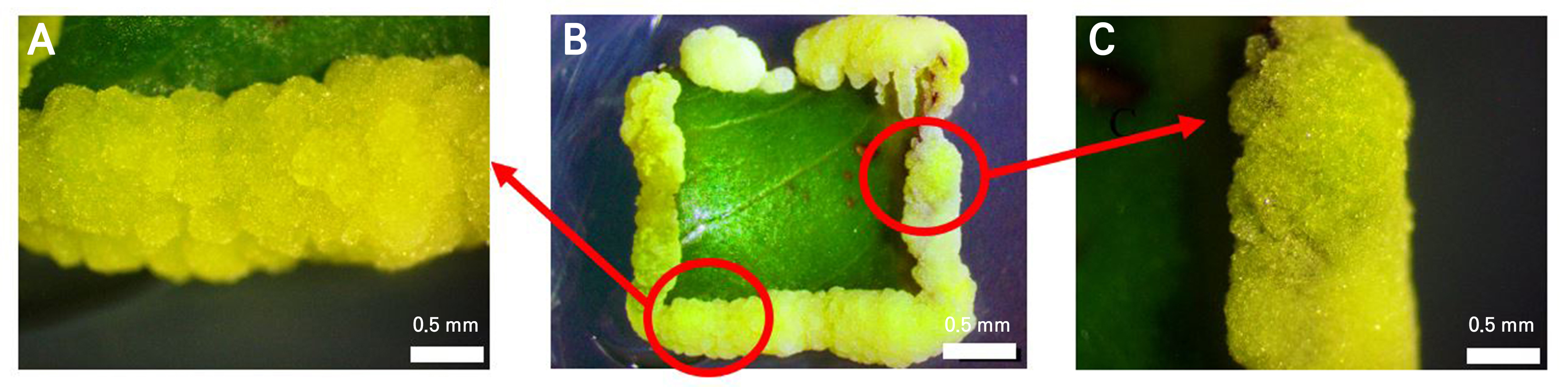
The embryogenic callus induced by the 2,4-D 1 mg L-1 + BAP 0.5 mg L-1 treatment had an intermediate texture that was yellowish-green in color, as shown in Fig. 2. The embryogenic callus (Fig. 2B) that formed had an intermediate texture with crumbly sides (Fig. 2A) and compact sides (Fig. 2C).
Texture is an important indicator of embryogenic callus regeneration (Wahyuni et al. 2020). A compact callus is typically dense and difficult to separate, whereas a crumbly callus is loosely arranged and easily disaggregated (Avila-Victor et al. 2023). An intermediate texture, representing a combination of both compact and crumbly characteristics, has been reported to support embryogenic competence (Setiawati et al. 2023). In this study, all treatments produced an embryogenic callus with an intermediate texture and nodular appearance (Table 1 and Fig. 2B). Although the callus surface appeared crumbly, its internal structure was firm and resistant to separation, reflecting solid and rigid characteristics. The consistent formation of an intermediate-textured callus was attributed to the synergistic effect of the 2,4-D and BAP application (Salihan and Yusuf 2020)
The 2,4-D hormone releases H+ ions, which lowers the cell wall's pH and causes it to stretch (Winnicki 2020). This loosening allows nutrients and water to enter the cell by diffusion, making it turgid. Adding BAP, on the other hand, speeds up cell wall formation, making the callus more compact (Ouyang et al. 2020). BAP quickens lignification, which strengthens the cell wall (Panggabean et al. 2023).
According to a previous study, callus color is linked to the degree of active cell division (Andaryani et al. 2019). The embryogenic callus appeared yellowish-green in only six treatments: BAP 1 mg L-1, 2,4-D 1 mg L-1, 2.4-D 1 mg L-1 + BAP 0.5 mg L-1, 2,4-D 2 mg L-1, 2,4-D 4 mg L-1, and 2,4-D 4 mg L-1 + BAP 1.5 mg L-1, while in the other treatments, it was yellow (Table 1). This was consistent with other work focusing on the biduri (Calotropis gigantea) plant. The results showed that explants induced with varying concentrations of 2,4-D and BAP hormones produced embryogenic calli that were yellowish-white, greenish-white, and green (Muthi’ah et al. 2023).
Yellow coloration in the S. cumini embryogenic callus was an early sign of somatic embryogenesis, showing high cell proliferation with little chlorophyll or tissue differentiation. The yellow-green color (Fig. 2), however, suggested that chlorophyll biosynthesis had begun (Handayani et al. 2019). This indicated a transitional phase of initial plastid development, suggesting enhanced embryogenic competence and structural differentiation that may lead to somatic embryo or organ formation. The hormones 2,4-D and BAP were essential for this chlorophyll biosynthesis process. The process started with glutamate, which was converted to 5-aminovulinic acid (ALA) and protochlorophyllide (DPP), with a final conversion into chlorophyll a and b (Liu et al. 2020).
Auxin and cytokinin hormones triggered one of the potential transcription factors, namely elongated hypocotyl 5 (HY5), for photomorphogenesis. Glut-RNA reductase was one of the target genes of HY5, and it converted glutamate into 5-aminovulinic acid (ALA). Its expression was regulated by auxin hormones and cytokinin (Liu et al. 2020).
Embryogenic callus colors are influenced by pigmentation, light, and by the hormone concentration (Mufadilah et al. 2024). Higher light intensity increases chlorophyll, making the callus greener (Handayani et al. 2019). Calli that appeared earlier also tended to develop a green color more rapidly.
Time of appearance
The embryogenic callus emergence time served as an indicator of the efficiency and effectiveness of the induction process. The shorter the time needed, the faster the embryogenic callus is obtained. In this study, the time of appearance was observed from day 0 up to when the embryogenic callus appeared on the explant. None of the treatments differed significantly, but significant differences were observed in the controls.
The treatment carried out using 2,4-D 2 mg L-1 without the addition of the BAP hormone had the fastest time of emergence and differed significantly from the control with a value of 10.8 DAP (Table 2). This result differed from the results of a study conducted on the Liberica coffee plant, which belongs to the same class as S. cumini. Liberica coffee leaf explants induced with 2 mg L-1 2,4-D and 1 mg L-1 BAP exhibited embryogenic callus emergence at 20 DAP (Lizawati et al. 2023).
Table 2.
Time to emergence of the embryogenic callus from S. cumini leaf explants (days)
The variation in the time of callus emergence was attributed to differences in callus growth rates, which could be influenced by several factors, including the types and concentrations of growth regulators (Ali et al. 2021). BAP, when combined with auxins such as 2,4-D, accelerated the initiation of callus formation. Concentrations of these two hormones played a critical role in callus induction, and specific concentration combinations could significantly reduce the time required for callus emergence (Dar et al. 2021). The application of 2,4-D and BAP at varying concentrations resulted in differences in the time of embryogenic callus initiation (Pathirana and Carimi 2023).
Auxins, specifically synthetic types such as 2,4-D, function as strong, singular signals that instruct specialized plant cells to dedifferentiate into an undifferentiated (meristematic) state and begin rapid, disorganized cell division. This direct signaling leads to more rapid initiation of callus development (Chandimali et al. 2024).
S. cumini leaf explants treated with BAP at all concentrations exhibited longer callus emergence times. When cytokinin (e.g., BAP) was applied simultaneously with auxin, the cells received dual signals; auxin promoted dedifferentiation, while cytokinin encouraged cell division and organization (often toward shoot formation). The integration of these signals required cellular rebalancing, which could delay the onset of undifferentiated growth compared to the singular auxin-induced response (Su et al. 2011).
Wet weight of embryogenic callus
The wet weight of the embryogenic callus indicated the presence of callus growth. This continuous growth of the embryogenic callus was characterized by larger clots and an increased mass (Wang et al. 2023). The highest value was produced by treatment 2,4-D 1 mg L-1 + BAP 0.5 mg L-1 with a weight of 158 mg (Table 3). The results presented in Table 3 differ from those reported by Wang et al. in their study of the pomegranate (Punica granatum) plant. They found that shoot tip explants cultured on a medium containing 1 mg L-1 NAA and 0.5 mg L-1 BAP produced an embryogenic callus with an average weight of 500 mg (Wang et al. 2023).
Table 3.
Wet weight variations (mg) of embryogenic calli from S. cumini leaf explants at 60 days after planting (DAP)
The wet weight produced was found to be dependent on the rate at which the embryogenic callus cells divided and multiplied (Wahyuni et al. 2020). Active cell division culminated in optimal callus growth and a greater callus wet weight. In addition, the wet weight of embryogenic calli can be affected by a high water content (Andaryani et al. 2019).
Hormones such as 2,4-D and BAP significantly affect the wet weight (Andaryani et al. 2019). The 2,4-D hormone increases the plasticity of the cell wall, facilitating the diffusion of water, minerals, and nutrients and thereby accelerating cell growth and enlargement (Malik et al. 2021). Meanwhile, the BAP hormone raises the volume of callus cells by inducing cell division and protein synthesis, which promote cell proliferation (Ali and Baloch 2020).
Stages of embryogenic callus formation
Callus formation was observed from the day of planting until the appearance of the callus, after which observation was continued once a week. The stages began with a slight swelling at the edge of the explant (Fig. 3B), followed by the appearance of a yellow embryogenic callus. In addition, growth occurred on the periphery of the explant, precisely on the incision scar. The swelling showed that the explant had responded to the hormone treatment. Nutrients in the medium were absorbed, and cell proliferation began, as evidenced by an increase in the mass of the embryogenic callus (Fig. 3C). Calli that showed embryogenic characteristics were then observed to assess their histological properties. Histological preparations showed the presence of cells in the embryogenic area, as presented in Fig. 4.

Fig. 4.
Histological preparation of embryogenic callus cells from S. cumini leaves treated with 2,4-D 2 mg L-1 at 28 DAP: (A) entire histological section of S. cumini leaf callus, ER = embryogenic region; magnification: 5×. (B) Enlarged view of a portion of (A); magnification: 10×45; stain: safranin.
The mechanisms of the 2,4-D and BAP hormones in the embryogenic callus induction required specific processes. The 2,4-D hormone released H+ ions that penetrated the cell wall and activated certain enzymes, leading to the release of hydrogen bonds within the cellulose molecular chain, which caused the cell wall to stretch. In addition, the proton pump (H+ ions) reduced the pH value, causing K+ ions to be transported from inside the cell. This uptake lowered the water potential in the cell, culminating in cell enlargement (Majda and Robert 2018).
Cytokines are compounds that can increase cell division, development, advancement, and the growth of leaves. In this case, the BAP hormone stimulated cell division by increasing the transition of G2 after DNA replication to mitosis. This protein synthesis is increased by the formation of a messenger RNA that remains stable and accelerates the translation process (Asghar et al. 2023). When lower sums of cytokinin (BAP) were presented to MS media enhanced with 2,4-D, the development rate and callus mass expanded (Qahtan et al. 2022).
Histological preparations were undertaken to observe the growth of embryogenic cells. These observations showed that embryogenic cells were located near the edges and had larger intercellular spaces. Meristematic cells lacked intercellular spaces, leading to a more densely packed appearance, while parenchymatic cells contained large vacuoles (Yang et al. 2021). The success of embryogenic cell formation depended on the initial level of cell differentiation, the environmental conditions, and on the interactions among the applied growth regulators (Avila-Victor et al. 2023).
The histological analysis also demonstrated the presence of cells in embryogenic areas, as shown in Fig. 4. These cells were small, with a large nucleus, a thick cell wall, and dense cytoplasm. When induced with the appropriate medium and growth regulators, such cells have the potential to develop into somatic embryos.
Somatic embryo induction
The S. cumini embryogenic callus cultured on MS medium supplemented with 2 mg L-1 2,4-D and 1 mg L-1 BAP was subsequently subcultured onto a medium containing 5 mg L-1 TDZ. The callus treated with 2,4-D and BAP exhibited an intermediate texture (Fig. 5A), while in the TDZ-treated sample, the texture remained in an intermediate stage, but with a more dominant compact region compared to the crumbly area (Fig. 5B). A compact texture is one of the morphological characteristics associated with somatic embryo formation. These results were consistent with those observed in Schisandra chinensis (Turcz.) Baill., a species from the same taxonomic class as S. cumini (Li et al. 2025). After the subculture step, the embryogenic callus entered the somatic embryogenesis process. Somatic embryogenesis typically progresses through distinct developmental stages, specifically the globular, heart, torpedo, and cotyledon phases (Botini et al. 2021). In this study, the globular phase appeared 21 days after subculture (Fig. 5G). Subsequently, somatic embryos developed from the late globular phase (Fig. 5LG) into the heart (Fig. 5H) and torpedo phases (Fig. 5T).

Fig. 5.
Comparison before and after the subculture step: (A) S. cumini embryogenic callus on MS medium supplemented with 2,4-D 2 mg L-1 + BAP 1 mg L-1 prior to subculture, (B) S. cumini somatic embryo on MS medium supplemented with TDZ 5 mg L-1 after subculture, and (C) somatic embryo developmental stages of S. cumini on MS medium supplemented with TDZ 5 mg L-1: G = globular phase, LG = late globular phase, H = heart phase, T = torpedo phase. (G), (LG), (H), and (T) represent magnified sections from (C). Scale bars: 0.5 cm and 0.1 mm.
Another characteristic of somatic embryos is the formation of the shoot apical meristem (SAM). Fig. 6 shows prospective shoots observed at the apex, characterized by a dense arrangement of nucleated cells that were evenly distributed and uniform in size. Somatic embryos that derived from S. cumini leaves exhibited a rounded morphology with a flat surface and bipolar characteristics.
Embryos can be regenerated from an embryogenic callus, as characterized by a smooth surface and compact texture, while non-embryogenic typically exhibit a crumbly and soft texture (Avila-Victor et al. 2023). Some plant species also produce somatic embryos with intermediate textures (Mazri et al. 2019). Previous studies have shown that S. cumini somatic embryos induced by the 2,4-D hormone exhibit an intermediate texture. Embryogenic calli produce globular-phase somatic embryos with an intermediate texture and a creamy yellow or brownish-yellow appearance when treated with 2.5 mg L-1 and 3 mg L-1 of 2,4-D, respectively (Ummah et al. 2019).
Plant cell growth during in vitro culturing is influenced by growth regulators. TDZ is an effective regulator, known for promoting callus formation, somatic embryogenesis, regeneration, and organogenesis (Karakas 2020). It has been shown to induce somatic embryos in S. cumini leaves and, in citrumelo plants, it outperformed the BAP hormone in producing quality embryos and buds (Al-Jalihawi et al. 2023). TDZ is a superior cytokinin, and its ability to replace the need for both auxin and cytokinin in somatic embryogenesis is a major advantage for some plants (Ahmad and Faisal 2018). Its pyridyl urea and thidiazolyl derivatives are more active than other adenine-type cytokinins. Moreover, TDZ reportedly exhibits de novo synthesis in conjunction with auxin hormones, specifically IAA (indole-3-acetic acid) (Wang et al. 2021).
One of the distinguishing characteristics of somatic embryos is the formation of SAM, which represents a prospective sprout that forms during plant embryogenesis. Plant cells divide and enter the globular stage, eventually developing into SAM, which forms buds (Godel-Jędrychowska et al. 2021). The characteristics (Fig. 6) in this study were consistent with the findings of earlier work, which emphasized that SAM exhibited a dark color with a denser cell arrangement, a relatively homogeneous appearance, and smaller cell sizes (Gaillochet and Lohmann 2015). The normal somatic embryogenic morphology includes a round shape, bipolar characteristics (prospective roots and shoots), and a flat surface (Godel-Jędrychowska et al. 2021). Somatic embryos are considered bipolar because SAM and the root apical meristem (RAM) form simultaneously (Zhu et al. 2022).
Physical properties of seeds
The average diameters of the synthetic seeds obtained with alginate concentrations of 3% (0.626 ± 0.032 cm) and 4% (0.656 ± 0.045 cm) showed no significant differences compared to that at 2% (0.556 ± 0.065 cm) (Table 4). Based on these results, the higher the concentration of alginate used during encapsulation, the larger the seed diameter. Encapsulation is a somatic embryo coating technique used to produce synthetic seeds that are planted and regrown later in vitro (Mohamed et al. 2023). The roundness parameter (Table 4) at an alginate concentration of 3% (1.30 ± 0.10) was not significantly different from those of the other two treatments, but the 2% concentration (1.11 ± 0.04) differed significantly from the 4% treatment (1.19 ± 0.04). In addition, the roundness of the synthetic seeds was determined by calculating the ratio of the largest diameter to the smallest diameter, as measured using a digital caliper. A roundness value close to 1.0 showed a more spherical and morphologically uniform seed (Cervantes et al. 2016). The values obtained at concentrations of 3% and 4% were closer to 1 (one) compared to that at 2%. The alginate concentration was found to affect the diameter of the encapsulated seeds, while the concentration of alginate affected the diameter of the synthetic seeds, and the greater the concentration, the larger the diameter (Volić et al. 2018). The physical shape of the synthetic seeds (Fig. 7) corresponds to the data shown in Table 4.
Table 4.
Physical characteristics of S. cumini somatic embryogenic synthetic seeds
The concentration of alginate also affected the shape of the encapsulated seeds that formed, and the recommended roundness values were those close to 1. In addition, the roundness values at concentrations of 3% and 4% were closer to 1 compared to that found when using the 2% alginate concentration (Table 4). This was consistent with earlier work in which the use of an alginate concentration above 2.5% produced synthetic seeds with a firm and uniform globular shape (Micheli et al. 2022).
Good synthetic seeds are characterized by a dense and rigid shape. Therefore, leakage is considered to be among the important parameters determining the quality of the physical properties. After 30 days of storage, the 3% alginate concentration resulted in the lowest leakage percentage (7.66 ± 0.29%). This was significantly lower (p < 0.05) compared to the corresponding results from the 2% (12.46 ± 0.63%) and 4% (15.26 ± 1.67%) treatments (Table 4). As a lower leakage percentage indicates better synthetic seed quality, the 3% alginate concentration was considered optimal.
The physical appearance of the synthetic seeds is presented in Fig. 7, corresponding to the data in Table 4. In this study, the largest seed diameter was observed in the 4% alginate treatment. In terms of shape, seeds with the most uniform roundness were produced by the 3% alginate concentration, followed by 4% and 2%. The highest leakage percentage was also recorded in the 4% treatment, which exhibited the most cracking with varying degrees of rupture after 30 days of storage, as shown in Fig. 8.
One study reported that the leakage rate is influenced by the concentration of alginate, and the lower the percentage of leakage, the better the quality of the seeds. Leakage also occurs due to incomplete solidification or damage to the seed capsule (Volić et al. 2018). In addition, this parameter determines the regeneration ability and viability of somatic embryos within the capsule, as essential nutrients could be released outside, limiting their absorption and utilization by seeds.
The use of a low alginate concentration produces soft capsules, explaining the high percentage of leakage in synthetic seeds derived from the 2% treatment. The hardness of the capsule is determined by the interaction between alginate and CaCl2. When alginate concentration is too low, the capsules formed were soft and brittle, making them prone to leakage (Micheli et al. 2022). The use of excessively high alginate concentrations increases the viscosity of seeds (Saeed et al. 2020). Elevated high viscosity reduces the moisture content in seeds, resulting in a change in the osmosis pressure. Here, the S. cumini synthetic seeds were stored in sterile distilled water. Based on the principle of osmosis (balance), water around the seeds could penetrate, promoting continued growth and eventually rupturing the seeds.
Seeds regeneration ability
The regeneration ability parameters were tested with variations in the alginate concentration and storage duration and at temperatures of 25°C and 15°C. The largest percentage of regeneration (66.67 ± 1.65%) at 25°C was produced by the 3% alginate concentration with storage durations of 15 and 30 days, as shown in Table 5. In general, the 3% alginate showed a higher percentage of regeneration ability compared to the other two concentrations. A good retention period ranged between 15 and 30 days after storage, while 45 days of storage resulted in the lowest percentage. The results of the analysis with SPSS showed that the results from the different alginate concentrations used here differed significantly, whereas the storage duration parameter outcomes did not show any significant differences.
Table 5.
Percentage of early globular-phase somatic embryo regeneration at a storage temperature of 25°C
A primary success metric for somatic embryos is their ability to regenerate from a capsule matrix (Arias-Pérez et al. 2021). Factors such as the media, compaction agents, temperature, and storage conditions all influence the regeneration of synthetic seeds (Hatzilazarou et al. 2020). Varying the alginate concentration changes the seed thickness, which impacts the embryo’s ability to emerge from the capsule (Ouzhand et al. 2023). Furthermore, embryo quality and viability are affected by the storage duration and temperature (Mohamed et al. 2023).
The highest percentage of regeneration (83.33 ± 1.65%) at 15°C was produced when using the alginate concentration of 3% with a storage duration of 30 days (Table 6). Overall, encapsulated seeds maintained at 15°C exhibited a higher regeneration rate than those stored at 25°C, indicating that 15°C represents a more suitable storage temperature for the regeneration of S. cumini somatic embryos.
For optimal seed quality and high regeneration rates, a 3% alginate concentration is best (Manokari et al. 2021a). Concentrations of 4–5% create seeds that are too hard for embryo growth, while low concentrations result in poor seed quality due to inadequate calcium alginate formation (Arias-Pérez et al. 2021). The low regeneration frequency with 2% and 4% alginate concentrations arises because synthetic seeds are either fragile or overly hard (Subrahmanyeswari et al. 2023).
Table 6.
Percentage of early globular-phase somatic embryo regeneration at a storage temperature of 15°C
A previous study found that sodium alginate at a concentration of 2.5% could maintain viability for up to 30 days (Hatzilazarou et al. 2020). Encapsulation was used to extend the shelf life of somatic embryos, but after storage for an extended period, this led to a decrease in oxygen levels of the capsule matrix and a reduction in seed nutrition caused by leakage (Maqsood et al. 2012).
After 45 days of storage at both 15°C and 25°C, the somatic embryos in the 2% and 3% alginate remained green, whereas those in 4% alginate turned brownish-yellow (Figs. 9 and 10). Seeds stored in sterile aqueous media also showed green growth. Room-temperature storage led to more rapid nutrient loss from evaporation as compared to low-temperature storage (Abbasi et al. 2020). Alginate and sugar protected the embryos, increasing their resistance to cold. Research shows that seeds stored at room temperature are more prone to leakage than those kept at a low temperature (Khilwani et al. 2016).

Fig. 9.
Morphology of S. cumini somatic embryos after encapsulation at 15°C: (A) 2% alginate, 15-day shelf life; (B) 3% alginate, 15-day shelf life; (C) 4% alginate, 15-day shelf life; (D) 2% alginate, 30-day shelf life; (E) 3% alginate, 30-day shelf life; (F) 4% alginate, 30-day shelf life; (G) 2% alginate, 45-day shelf life; (H) 3% alginate, 45-day shelf life; and (I) 4% alginate, 45-day shelf life. Scale bar: 0.5 cm.

Fig. 10.
Morphology of S. cumini somatic embryos after encapsulation at 25°C: (A) 2% alginate with a shelf life of 15 days, (B) 3% alginate with a shelf life of 15 days, (C) 4% alginate with a shelf life of 15 days, (D) 2% alginate with a shelf life of 30 days, (E) 3% alginate with a shelf life of 30 days, (F) 4% alginate with a shelf life of 30 days, (G) 2% alginate with a shelf life of 45 days, (H) 3% alginate with a shelf life of 45 days, and (I) 4% alginate with a shelf life of 45 days. Scale bar: 0.5 cm.
Seeds storage at 15°C is considered to be the best treatment because S. cumini is a subtropical plant prone to damage at storage temperatures below 10°C. Therefore, storing somatic embryos at 4°C can culminate in less regeneration ability due to the sensitivity of cells and tissues to low temperatures (Khilwani et al. 2016).
Higher concentrations of alginate, specifically 4%, hinder embryogenic growth by creating excessively hard and thick seeds. In contrast, a 2% concentration results in fragile seeds (Manokari et al. 2021a). The failure of encapsulated somatic embryos to grow into buds is linked to their incomplete globular phase and the use of a suboptimal regeneration medium. Optimal bud development is more likely when embryos are encapsulated during the cotyledon phase (Ouzhand et al. 2023). The regeneration medium can be enhanced with gibberellic acid (GA) to promote germination, which is particularly effective for the growth of synthetic seeds (Saeed et al. 2020). Another storage method for synthetic seeds involves a liquid medium (Manokari et al. 2021b).
Conclusion
In conclusion, the optimal concentrations of 2,4-D and BAP hormones to induce an embryogenic callus are 2 mg L-1 of 2,4-D and 0 mg L-1 of BAP, leading to an embryogenic callus with an intermediate texture and yellowish-green color. Variations in the alginate concentration (3%) and a storage period of 30 days at 15°C and 25°C produce S. cumini somatic synthetic embryo seeds with the best physical properties and regeneration ability. Optimization of the regeneration medium and the induction of embryos at the green cotyledonary stage are necessary to achieve successful somatic embryo encapsulation leading to shoot growth. This study represents an initial step toward increasing plant production, specifically for plants with medicinal properties such as S. cumini and other food plants. Somatic embryo encapsulation provides a solution for plant production, particularly for species difficult to propagate through conventional methods. In the future, somatic embryo encapsulation can be developed further for conservation, plant breeding, and to enhance plant resistance.