Introduction
Materials and Methods
Germination effect investigation
Seedling growth effect investigation
Air anion treatments
Plant growth characteristics
Endogenous hormones in seeds
Chlorophyll content
Cell division
Statistical analysis
Results
Germination rate of spinach seeds
Growth characteristics of spinach seedlings
Endogenous hormone content in spinach seeds
Chlorophyll content
Cell division
Discussion
Conclusion
Introduction
Environmentally friendly and resource-efficient crop production to ensure a sustainable food supply has become a major global focus amid growing concerns over environmental pollution and climate uncertainty (Ferrarezi et al. 2024). Vertical farming is a controlled environmental cultivation system that enables crop production in limited spaces by vertically stacking multiple layers. It maintains optimal environmental conditions, such as light, temperature, humidity, carbon dioxide concentrations, nutrient supplies, and airflow, to accelerate plant growth and ensure high-quality products (Kozai et al. 2019; Mempel et al. 2021). Because vertical farms can produce food regardless of the season, climate, or location, while utilizing space, nutrient, and water resources more efficiently than field cultivation, they have emerged as a next-generation urban food supply solution and future agricultural strategy.
Leafy vegetables are particularly suitable for vertical farming owing to their short growth cycles and sustained market demand. Compared to traditional field or greenhouse cultivation, leafy vegetables grown on a vertical farm exhibit remarkably higher yields (more than eleven times) and much better light use efficiency (0.55 vs. 0.23 g·mol-1 in the field), while requiring over 92% less water, as reported for lettuce (Barbosa et al. 2015; Jin et al. 2023). Along with lettuce, spinach (Spinacia oleracea) is widely grown in vertical farming systems and is considered one of the major leafy vegetables. Spinach, a cold-season vegetable native to Southwest Asia, is valued for its rich nutritional content—including dietary fiber, β-carotene, vitamin C, folic acid, iron, and calcium (Lim et al. 1990; Roberts and Moreau 2016). However, spinach seeds have irregular shapes and hard coats, making pretreatments for uniform germination difficult, with a germination rate below 60% (Katzman et al. 2001; Magnée et al. 2020; Thakur et al. 2025). Such issues hinder uniform seedling establishment and pose major challenges for commercial spinach production. According to Kozai et al. (2019), leafy vegetable cultivation typically consists of three stages: germination, seedling growth, and vegetative growth. The germination and seedling stages jointly account for up to 70% of the total cultivation period. Therefore, accelerating such stages could shorten the overall growth cycle significantly. To meet the demand for stable and efficient spinach production in vertical farming systems, it is essential to develop cultivation techniques that promote rapid and uniform seedling establishment. Accordingly, this study investigates the effectiveness of electrical stimulation via air ionization, applied to seeds and seedlings, on improving germination and the initial growth of spinach.
The earth is surrounded by an electromagnetic field and plants are exposed to it continuously (Ellis and Turner 1978). Under fair weather conditions at ground level, the electric field strength typically ranges from 100 to 150 V·m-1 (Harrison 2013). In the atmosphere, both positive and negative ions coexist, maintaining overall electrical neutrality (Krueger and Reed 1976). However, the electric field can vary locally, depending on meteorological conditions and topographical factors. For example, during thunderstorms, the electric field between clouds and the ground intensifies rapidly, leading to discharge phenomena such as lightning, which in turn generate large amounts of negative ions that contribute to the formation of localized electric fields (Standler and Winn 1979). Similarly, at the base of waterfalls, falling water generates a large number of negatively charged fine particles, which alter the electrical properties of the air markedly and cause local variations in the electric field (Standler and Winn 1979; Borra et al. 1997; Jiang et al. 2018).
Several studies have demonstrated that numerous types of plant growth and physiological responses are positively affected by the strength and distribution of the electric field under electrical stimulation (Bratton and Henry 1977; Li et al. 2019, 2022; Morales et al. 2021a, 2021b). For example, increased fresh weight in carrot plants and enhanced radicle elongation in buckwheat seeds have been reported in response to electric field treatments (Lemström 1904). Additionally, low strength of the electric field improves the availability of soil nutrients, thereby promoting plant mineral absorption and enhancing photosynthetic activity, resulting in broad physiological effects on plants. Based on such positive effects, electroculture is an emerging agricultural technique that applies electrical stimulation to improve crop productivity and overall agricultural efficiency (Lemström 1904; Pohl and Todd 1981; Christianto and Smarandache 2021; Lee and Oh 2023).
In our previous work, we demonstrated that the electrostatic potential increased linearly with the concentration of negative air ions produced by negative air-ion generators (Jeon 2025). This suggests that continuously released negative charges may accumulate in localized areas of the cultivation space, resulting in an increase in potential and generating electrical stimulation that could lead to changes or localized enhancement in the electric field distribution.
Therefore, the present study investigated the growth, physiological, and hormonal responses of spinach to air-based electrical stimulation at different growth stages, focusing on germination and seedling growth in a vertical farming system. Furthermore, the potential of this approach to enhance the cultivation efficiency and address current challenges in spinach production was evaluated.
Materials and Methods
Germination effect investigation
Two sheets of filter paper (TO5B–110; ADVANTEC, Tokyo, Japan) were placed in a Petri dish (150 × 20 mm), diameter × height and distilled water was supplied until the filter paper was sufficiently wet. One hundred spinach seeds were sown on the fully wet filter paper and placed in a controlled environment cultivation module. The experiment was conducted for 14 days immediately after sowing.
The air temperature in the module was maintained at an average of 20 ± 3°C, the relative humidity at 60 ± 3%, and the photosynthetic photon flux density at 130 µmol·m-2·s-1. The photoperiod was set to 12 h. During the air ionization treatment, the Petri dishes were opened from 9 a.m. to 9 p.m., and distilled water was supplied at 4 h intervals via a sprayer to prevent desiccation of the seeds. For each air ionization treatment, there were four Petri dish replicates, which were rotated clockwise daily to minimize experimental errors due to varying environmental conditions.
After 14 days of the air ionization treatment, seed germination in each Petri dish was assessed, and the germination rate was calculated and expressed as a percentage.
Seedling growth effect investigation
The spinach seeds (‘Mustang’, FarmHannong, Seoul, Republic of Korea) were sown in plastic trays filled with horticultural soil (approximately 18 g per cell). Germination and cultivation were conducted under the aforementioned environmental conditions. Five days after sowing, only one seedling was left per cell, and the plants were cultivated for three additional weeks under the same conditions while being subjected to the air ionization treatment. The Hoagland nutrient solution (500 mL, pH 5.8, and EC 1.0 dS·m-1) was supplied daily. In each treatment, 100 seedlings were cultivated, with 50 seeds being sown in each of two trays.
Air anion treatments
Negative air ion generators (EGT1A2–2; EGtech, Incheon, Republic of Korea) were used to create negative air ions over the spinach seeds and seedlings. Nine air ionization generators were installed per unit cultivation area (60 × 60 cm), following the method described by An et al. (2021), including the specified installation height. The target negative air ion concentrations were achieved by adjusting the distance between the plant canopy and the generators, as shown in Fig. 1 and Supplementary Fig. 1. The ion concentrations selected for the treatments were within ranges that have shown positive effects on leafy vegetables in our previous studies (Song et al. 2014; Lee et al. 2015; An et al. 2021). The concentration of negative air ions was controlled at three levels: low air ions (LA, 3.6 × 105 ion·cm-3), moderate air ions (MA, 5.1 × 105 ion·cm-3), and high air ions (HA, 8.3 × 105 ion·cm-3). For the HA treatment, a voltage regulator (SD–500–0.5 kVA; Hanil Transformer, Namyangju, Republic of Korea) was used to ensure that the negative ion concentration was uniform. In the control treatment (Con), no air ionization generator was installed, and the average negative air ion concentration was 5.9 × 102 ion·cm-3. To minimize interference among the treatments, the cultivation areas were spatially separated, and the treatments were arranged at intervals of 50 cm. Atmospheric negative air ions were measured using a negative air ion meter (COM–3600F; COM SYSTEM Inc., Tokyo, Japan). The cultivation area was divided into four zones, and measurements were recorded for 5 min at each point. The mean concentration was then calculated.
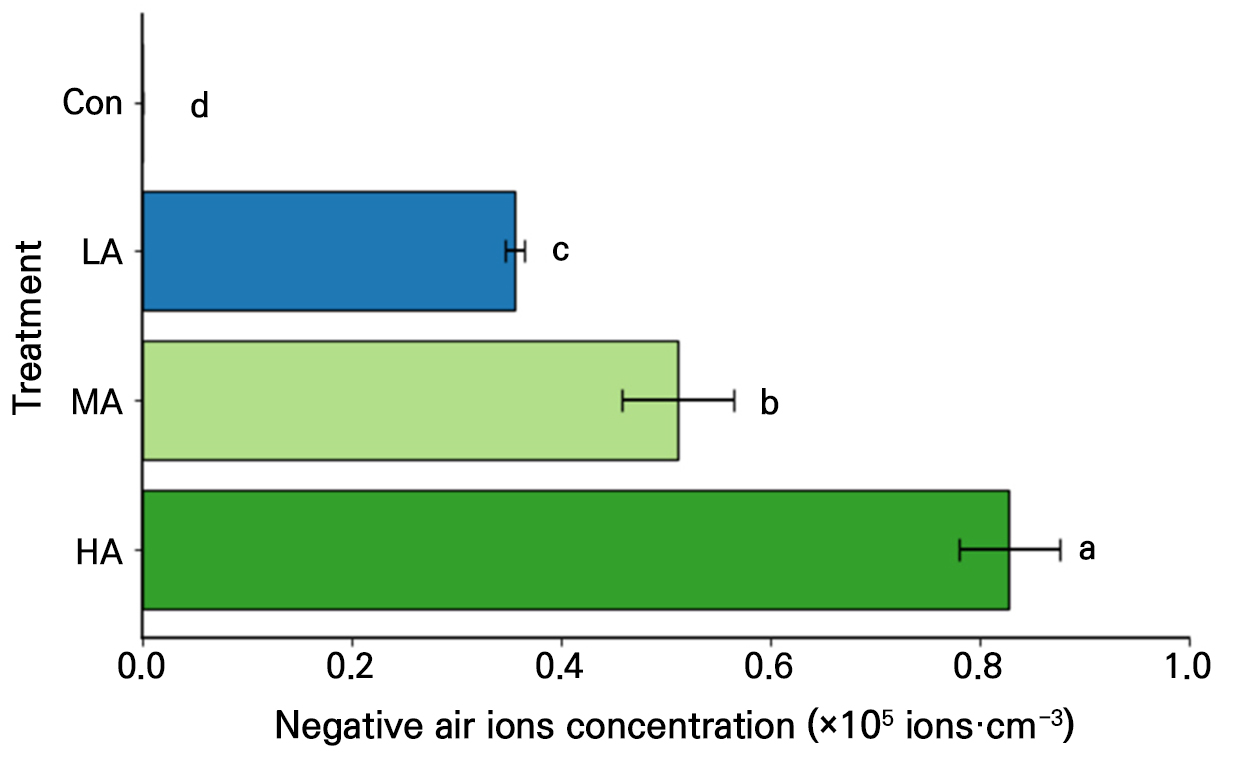
Fig. 1.
Negative air ion concentration under different air ionization treatments. Con, LA, MA, and HA indicate control, low, moderate, and high negative air ion concentration treatments, respectively. The vertical bars indicate the standard error. The ion concentration in Con was 5.9 × 102 ion·cm-3. Different letters indicate significant differences at p < 0.001 (n = 6).
Plant growth characteristics
The fresh weight of the spinach seedlings was measured using an electronic scale (SI–234; Denver Instruments, Denver, CO, USA). The leaf areas of the seedlings were measured using a leaf area meter (Li–3100C; LI–COR, Lincoln, NE, USA).
Endogenous hormones in seeds
For the hormone analysis, seeds were placed in Petri dishes, treated with LA for 12 h and then collected. Spinach seeds were dried in a freeze dryer (Alpha 2–4 LSCplus; CHRIST, Osterode am Harz, Germany) for 72 h and pulverized using liquid nitrogen for hormone extraction. The powdered seeds (50 mg) were extracted with 500 µL of extraction solvent [2–propanol/H2O/concentrated HCl (2:1:0.002, v/v/v)] and homogenized at 4°C and 100 rpm for 30 min. Afterward, 1 mL of dichloromethane was added to each sample, which was then mixed and shaken at 4°C at 100 rpm for 30 min. The mixture was centrifuged (Smart R17 Plus; Hanil, Daejeon, Republic of Korea) at 13,000 × g for 5 min and the supernatant was transferred to a microtube. Subsequently, the eluate was evaporated by a rotary evaporator (HyperVAC VC2124; Hanil, Daejeon, Republic of Korea) and the sample was finally dissolved in 100 µL of methanol and used as a separation sample. Samples were stored at ‒20°C until the analysis.
For the hormone analysis, an HPLC system (Agilent 1260; Agilent Technologies, Santa Clara, CA, USA) equipped with a degasser, pump, auto sampler, and column oven was used, with an Eclipse plus C18 column (4.6 × 50 mm, 3.5 µM, Agilent, CA, USA) used for material separation. For the mobile phase, 0.1% formic acid in water (A) and 0.1% formic acid in methanol (B) were used, starting with 5% B, changing to 95% B for 1 min, and 95% B for 4 min, which was then changed quickly to 5% B for 6 s and maintained for 54 s. The flow rate of the solvent and the column oven temperature were maintained at 0.5 µL·min-1 and 30°C, respectively, and the injection amount of each sample was 10 µL. 2–cis, 4–trans–abscisic acid (ABA), gibberellic acid (GA3), and indole–3–acetic acid (IAA) were used as standards. The stock solution was dissolved in 1 mL methanol and prepared at a concentration of 1 mg·mL-1.
An API 4000 mass spectrometer (Applied Biosystems Sciex, Ontario, Canada) equipped with an electrospray ionization source was used. The analysis was conducted in negative and MRM modes, and BioAnalystTM v1.6.1 and Analyst v1.6.1 were used for equipment operation and data analysis, respectively. During ionization, high-purity nitrogen gas was set to 60 psi and used as a spray and dry gas. Q1 and Q3 were analyzed by means of a LC–MS/MS multicomponent analysis at the unit resolution.
Chlorophyll content
To investigate seedling growth, the chlorophyll contents of seedlings 7, 14, and 21 days after the negative air ionization treatment were analyzed. Spinach leaves (0.2 g) were ground in a mortar using liquid nitrogen to obtain a fine powder. Acetone (80% v/v) was added to the powdered samples and mixing was subsequently conducted. The mixed solution was then centrifuged at 13,000 × g at 4°C for 2 min. The supernatant was diluted four times with acetone. Diluted samples were used for chlorophyll content measurements. The chlorophyll content was expressed as the ratio of chlorophyll a to chlorophyll b. Chlorophyll a was calculated as (12.25 × A663) – (2.79 × A645), while chlorophyll b was calculated as (21.5 × A645) – (5.1 × A663), where A663 and A645 represent the absorbance values measured at 663 and 645 nm, respectively, using a spectrophotometer (UV–1800; Shimadzu, Kyoto, Japan), following the method described by Sumanta et al. (2014).
Cell division
Flow cytometry was used to analyze the DNA content and cell cycle of spinach seedling leaves following the negative air ionization treatment. A spinach leaf disc (0.5 cm2) was placed in a Petri dish, and 200 µL of extraction buffer (Cystain PI Precise P; Sysmex Partec, Görlitz, Germany) was added to extract the nuclei of the cells. The extracted samples were filtered by a 50-µM nylon mesh filter (Partec CellTrics; Sysmex Partec, Görlitz, Germany). Subsequently, 800 µL of a staining solution (staining buffer: PI solution: RNase solution was mixed at a ratio of 1:6:3) was added, and the mixtures was left in the dark for 20 min. Each sample was analyzed using CytoFLEX (Beckman Coulter, Brea, CA, USA), and 50,000 cells were analyzed.
Statistical analysis
Statistical analyses were performed by means of an analysis of variance in SAS (Statistical Analysis System, Version 9.4; SAS Institute, Cary, NC, USA). Significant differences in the measured values were confirmed using Tukey’s studentized range (HSD) test. All graphs were generated using Python v3.13.7 (Python Software Foundation, Fredericksburg, VA, USA) with the Matplotlib, NumPy, Pandas, and Seaborn libraries.
Results
Germination rate of spinach seeds
The air anion treatment tended to increase the germination rate of spinach seeds after 3 days of treatment (DAT), and the rate in LA was 36% higher than those in Con at 13 and 14 DAT (Fig. 2A). The fresh weight of spinach seedlings at 14 DAT in the treatment was significantly higher than that in the Con. In particular, seedling fresh weights in the MA and HA treatments were 29% and 25% higher, respectively, than those in the Con (Fig. 2B).
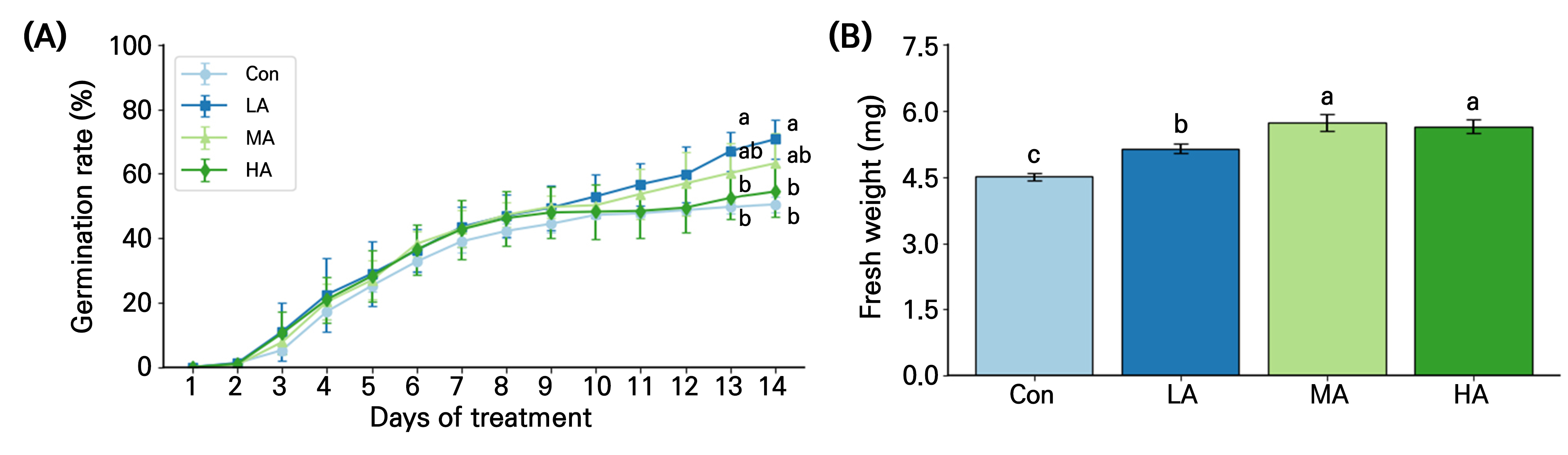
Fig. 2.
Germination rate (A) and seedling fresh weight (B) of spinach grown under negative air ionization treatments for 14 days. Con, LA, MA and HA indicate control, low, moderate, and high air ionization treatments, respectively. The vertical bars indicate the standard error. Different letters indicate significant differences at p < 0.05 (A; n = 100 and B; n = 20).
Growth characteristics of spinach seedlings
Shoot fresh weight in air anion-treated spinach seedlings was significantly higher than that of the Con seedlings at 21 DAT (Fig. 3A). At 7 DAT shoot fresh weights in the LA and HA treatments were significantly higher than that in the Con by 36% and 39%, respectively. At 14 DAT, the MA and HA treatments had higher shoot fresh weights than the Con and at 21 DAT, all air ionization treatments recorded higher values relative to those of the Con. In particular, the HA treatment consistently had the highest shoot fresh weight, with increases of approximately 40%, 31%, and 35% when compared with that in the Con at 7, 14, and 21 DAT, respectively.
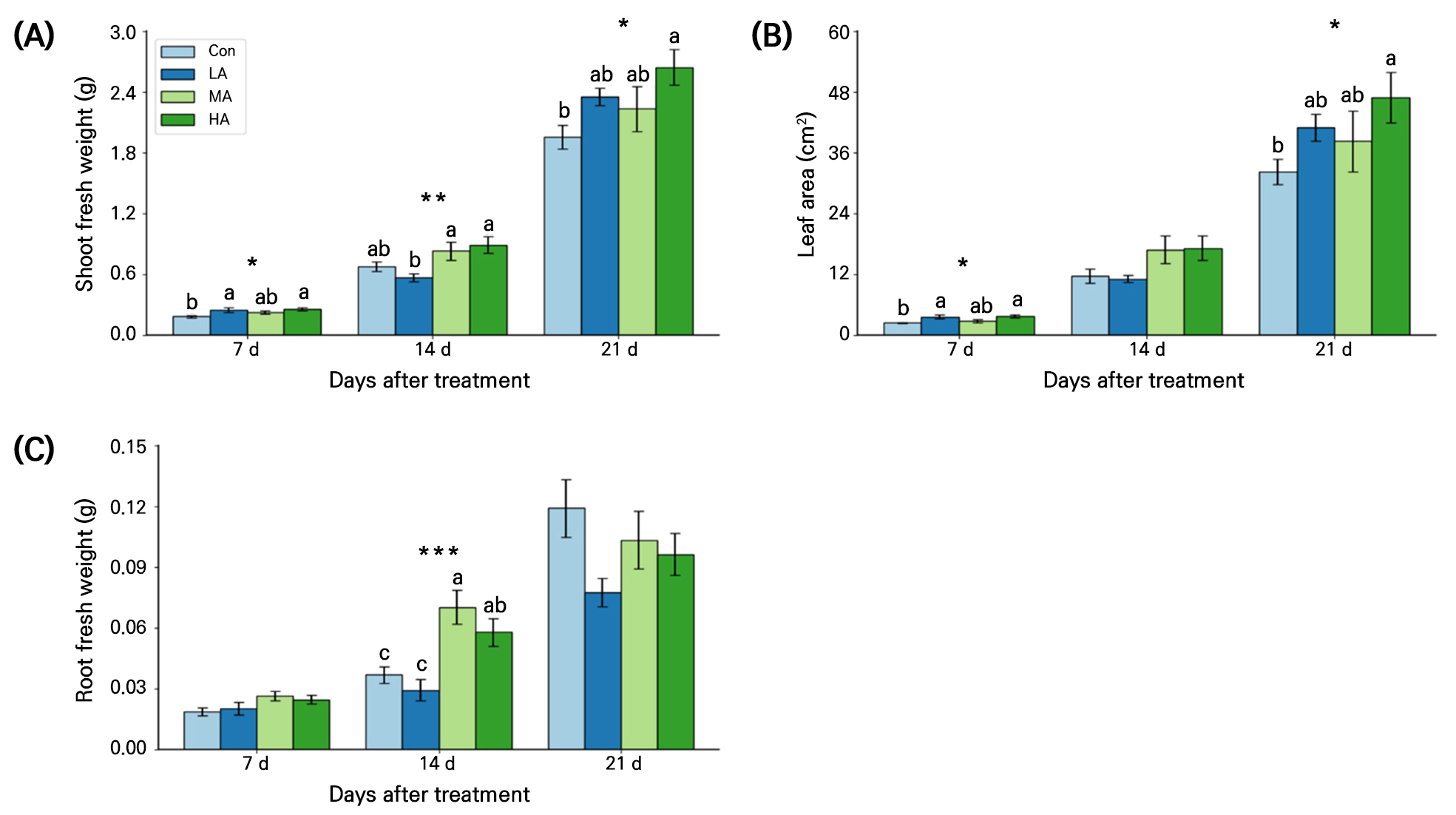
Fig. 3.
Shoot fresh weight (A), leaf area (B), and root fresh weight (C) of spinach plants grown under negative air ionization treatments for 21 days. Con, LA, MA and HA indicate control, low, moderate, and high air ionization treatments, respectively. The vertical bars indicate the standard errors. *, **, and *** indicate significant differences at p < 0.05, 0.01, and 0.001, respectively (n = 10).
The leaf areas at 7 and 21 DAT were significantly higher in the HA treatment than in the Con by 54% and 46%, respectively (Fig. 3B). In addition, the root fresh weight was 57% higher in the MA group than in the Con at 14 DAT (Fig. 3C). However, no significant differences in the root fresh weight were observed among the treatments at 21 DAT.
Endogenous hormone content in spinach seeds
The ABA content in the seeds treated with LA for 12 h was 31% lower than that in the Con (Fig. 4A). In contrast, the GA3 content in LA was 16% higher than that in the Con (Fig. 4B). In addition, IAA, a natural auxin, was 34% lower in the LA treatment than in the Con (Fig. 4C).
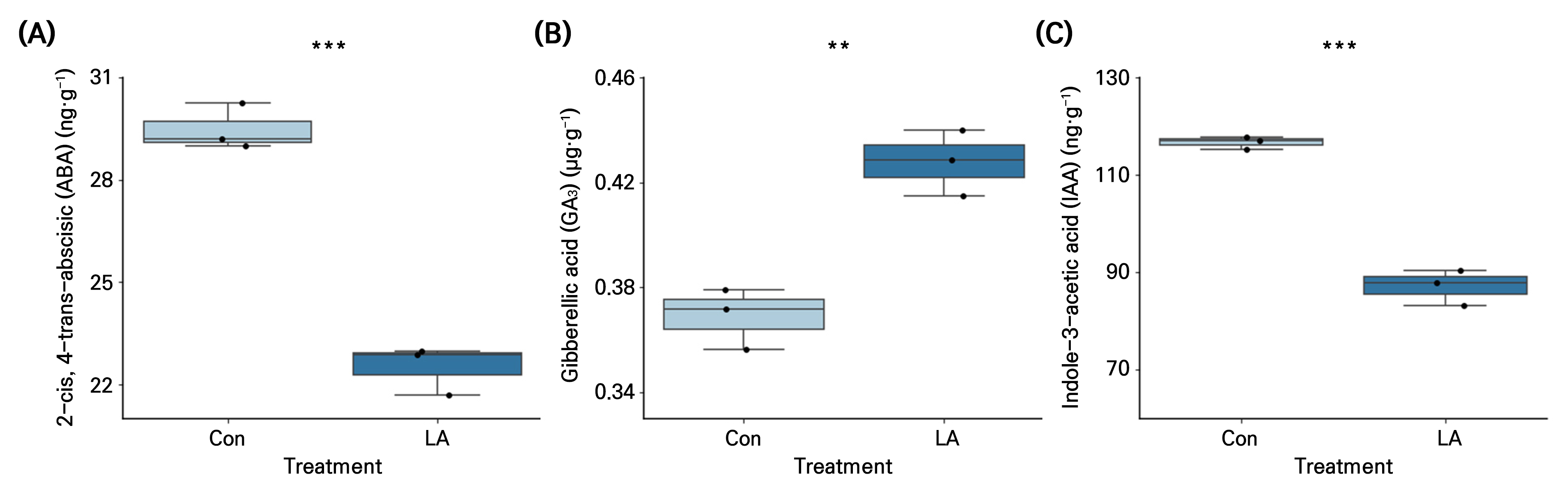
Fig. 4.
Abscisic acid (ABA) (A), gibberellic acid (GA3) (B), and indole acetic acid (IAA) (C) concentrations in spinach seeds under negative air ionization treatments. Con and LA indicate control and low air anion treatments. The vertical bars indicate the standard errors. ** and *** indicate significant differences at p < 0.01 and 0.001, respectively (n = 3).
Chlorophyll content
The chlorophyll a content of spinach leaves was at least 22% higher in all treatments compared to the Con at 7 DAT, and that in HA was 35% higher than the Con at 14 DAT (Fig. 5). At 7 DAT, the chlorophyll b contents in the LA and MA treatments were 31% and 33% higher, respectively, than that in the Con, and the content in the HA treatment was 33% higher compared to the Con at 14 DAT. However, at 21 DAT, the chlorophyll a and b contents were not significantly different between the Con and any of the treatments.
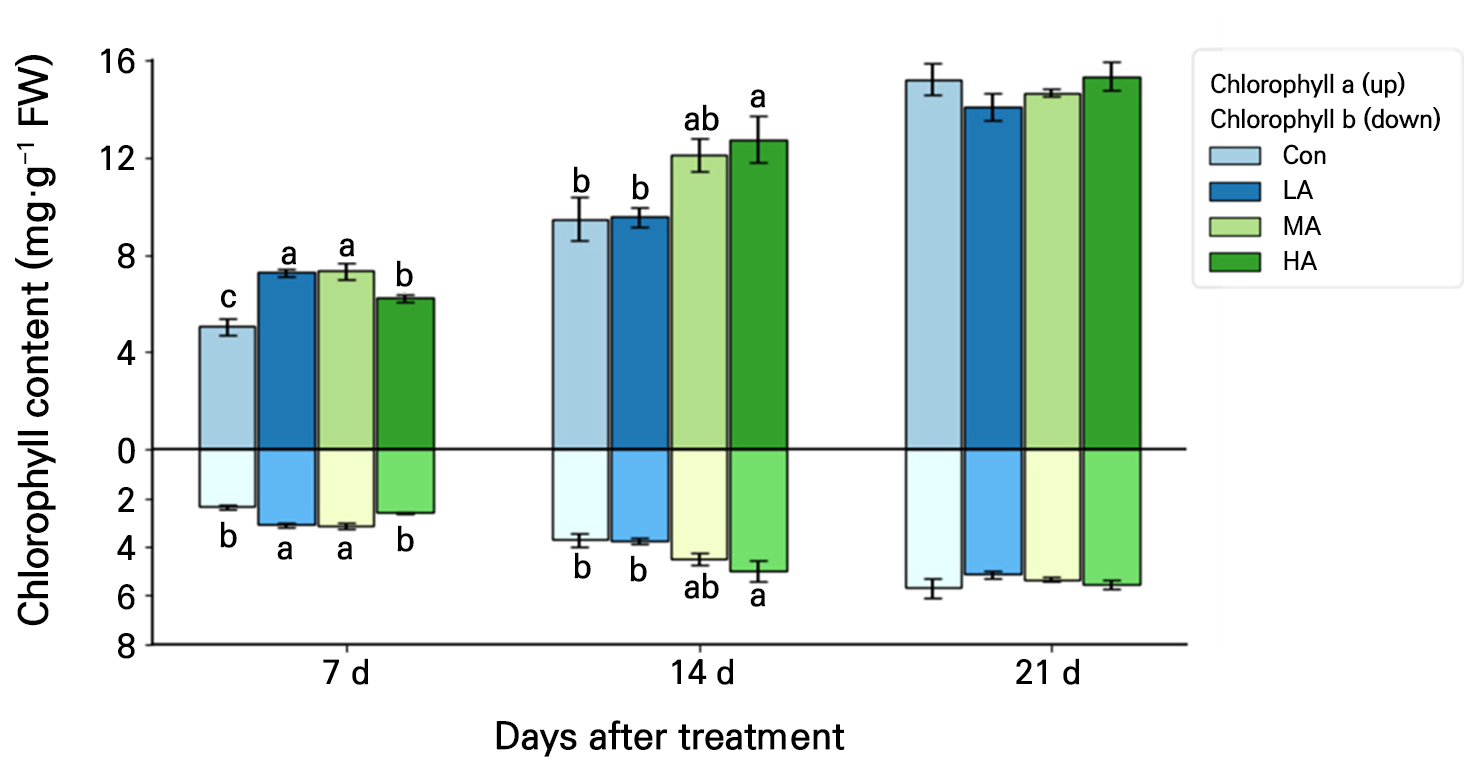
Fig. 5.
Chlorophyll a (A) and chlorophyll b (B) contents in spinach plants grown under negative air ionization treatments for 21 days. Con, LA, MA and HA indicate control, low, moderate, and high air ionization treatments, respectively. The vertical bars indicate the standard errors. Different letters indicate significant differences at p < 0.05 (n = 5).
Cell division
The DNA content and cell division activity in the spinach leaves were assessed at 16 DAT (Fig. 6). The 2C DNA content did not differ significantly between the air ionization treatments and the Con. However, the LA treatment had the highest 4C content among all treatments, at 33% higher compared to the Con. The proportion of G2M cells, indicating the division phase in the cell cycle, was at least 12% higher in all treatment groups than in the Con and the value in the HA group was 27% higher than that in the Con (Fig. 6B).
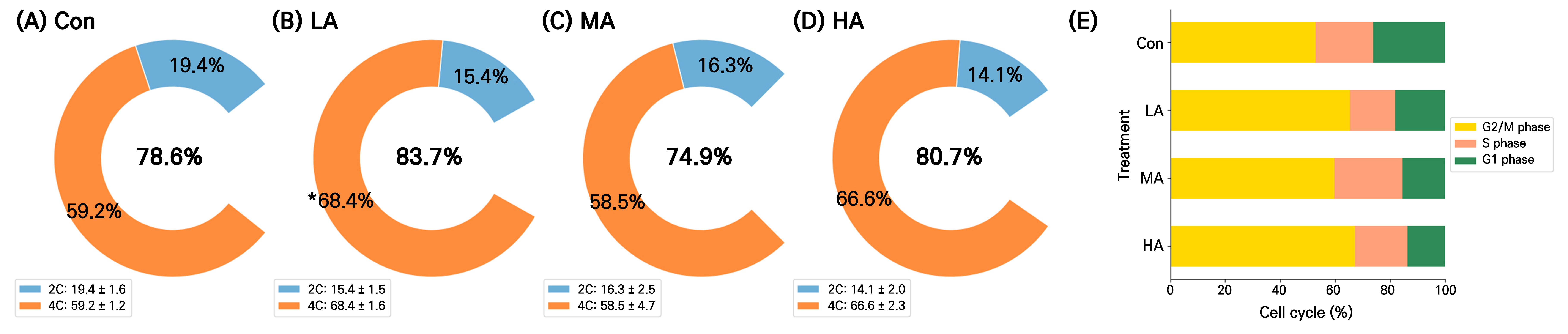
Fig. 6.
Nuclear 2C and 4C DNA contents (A–D) and the cell cycles (E) of spinach plants grown under negative air ionization treatments. Con, LA, MA and HA indicate control, low, moderate, and high air anion treatments, respectively. The legends represent the mean ± standard deviations. An asterisk (*) indicates a significant difference in 4C of the LA treatment compared with the Con (p < 0.05, n = 8).
Discussion
Air anions are produced when free electrons generated by energy excitation attach to gas molecules, thereby inducing localized microelectric field charges in the surrounding space (Standler and Winn 1979; Borra et al. 1997). Although in-depth studies that independently examine the direct effects of air ionization on plant physiological responses at the cellular level are limited, previous reports have confirmed its beneficial effects on growth promotion (Song et al. 2014; Lee et al. 2015; Won et al. 2018; Yeom et al. 2025). The present study provides evidence that air ionization exerts positive effects during the early developmental stages of spinach, including germination and seedling growth, as reflected by changes in hormone levels, the DNA content, and cell division activity. However, the direct effect of air anions on seed germination has not yet been reported. However, previous studies have confirmed interaction between electric fields and germination stimulation in wheat and barley seeds (Pittman and Ormrod 1970; Lynikiene and Pozeliene 2003).
Ji et al. (2016) reported that the degree of physical damage to the spinach seed coat increased when seeds were exposed to an electric field, as evidenced by more frequent cracking. In addition, Sudsiri et al. (2017) reported that exposure to electromagnetic fields (2.5–11.0 mT) increased the water content (%) in palm seeds relative to the Con and promoted germination. In their study, none of the Con seeds germinated. Therefore, electrical stimulation via air anions may break the spinach seed coat and improve water absorption. Subsequent water imbibition enhances the activity of GA3, a key germination hormone stored in seeds (Mahajan and Pandey 2015; Ji et al. 2016). ABA, a germination inhibitory substance in seeds, is observed at higher concentrations in dormant seeds, whereas the GA3 contents increase at the end of dormancy, and germination is promoted (Seo et al. 2006; Nambara et al. 2010; Liu et al. 2013). The antagonism between ABA and the GA3 content in seeds under negative air ionization treatment provide evidence of an improving germination rate (Fig. 4A and 4B). Bound auxin, an inactive storage form found in reserve organs or seeds, can be converted rapidly into free auxin (IAA) when needed (Taiz and Zeiger 2010). During this conversion, IAA levels may decrease temporarily as the bound forms are hydrolyzed, and free IAA is metabolized rapidly or oxidized.
In the present study, the IAA content in spinach seeds decreased following the air anion treatment, suggesting that the treatment promoted auxin metabolism (Fig. 4C). A similar trend was observed in sorghum seeds, in which IAA levels decreased after a wet pre-treatment for dormancy breakage (Dewar et al. 1998). Wu et al. (2020) reported that IAA acts synergistically with ABA, promoting ABA-mediated seed dormancy and suppressing germination via auxin signaling. In the present study, reduced IAA and ABA levels under the negative air ion treatment suggest a weakening of auxin-ABA associated inhibitory regulation, consistent with a shift from dormancy to germination. Although the role of auxins in spinach germination has not yet been clarified, our results demonstrate that air anions can significantly alter endogenous hormone levels, including IAA, ABA and GA3, implying that negative air ions may influence germination by reshaping the hormonal balance. In addition, the fresh weight of spinach sprouts at 14 DAT was significantly higher in all treatment groups by at least 14% compared with that in the Con (Fig. 2B). An increase in the germination rate of seeds had a positive effect on initial growth. Similarly, an electric field increased stem elongation in soybean and wheat sprouts by up to 35% compared with the Con (Kiatgamjorn et al. 2002; Leong et al. 2016).
Positive effects of electrical stimulation following air ionization were also observed at the spinach seedling stage before transplanting. After five days of germination, the seven-day air-anion-treated spinach seedlings already showed an increase in the shoot fresh weight and leaf area (Fig. 3A and 3B). Lee et al. (2015) and Song et al. (2014) reported that an air anion treatment enhanced the photosynthesis and growth of leafy vegetables (kale and lettuce) throughout the growth period after the seedling stage. In contrast, results here demonstrate that even a short exposure of seven days during the seedling stage can promote growth, possibly due to an increase in the chlorophyll content in the spinach leaves (Fig. 5), which may enhance the photosynthetic rate (Croft et al. 2017). No significant differences were observed between the treatments and the Con at 21 DAT, which is attributable to the dilution effect of the chlorophyll concentration caused by the expansion of the leaf area during the active growth phase (Figs. 3B and 5). Therefore, the significant increases in chlorophyll contents at 7 and 14 DAT likely contributed to the early growth of the spinach seedlings (Figs. 3 and 5).
Dannehl et al. (2012) reported that electrical stimulation affects the transmembrane potential difference, increasing the permeability of voltage-gated ion channels, which, in turn, enhances ion uptake and leads to ion accumulation within the cells. In particular, an increase in the Mg ion concentration induces a higher chlorophyll content, photosynthetic rate, and greater biomass production (Dannehl 2018). Mg is a key atom in the chlorophyll molecule, and enhanced Mg uptake following an air anion treatment may promote chlorophyll biosynthesis (Shaahan et al. 1999). Previous studies have revealed a strong correlation between the chlorophyll content and Mg ions, indicating that electrical stimulation induces efficient nutrient uptake and chlorophyll biosynthesis, thereby promoting plant growth (Ward 1996; Rochalska 2005). Similarly, an air anion treatment has been reported to enhance the Mg content, in addition to growth and photosynthesis, in kale (Lee et al. 2015). Comparable effects have also been observed in photosynthetic bacteria (Rhodopseudomonas palustris), where electrical stimulation increased the formation of hydrogen bonds in chlorophyll a, thereby stabilizing molecules and facilitating electron transition. It also improved CO2 fixation and RuBisCO activity by enhancing the efficiency of light-harvesting proteins.
These findings suggest that the increase in chlorophyll observed in the present study may result not only from enhanced Mg ion uptake and chlorophyll biosynthesis but also electrochemical modulation of photosynthetic units, ultimately contributing to improved seedling growth. In our previous study, photosynthesis-related parameters, specifically the photosynthetic rate, transpiration rate, and stomatal conductance, were enhanced by an air ionization treatment in greenhouse-grown spinach during the spring and fall seasons (An et al. 2021). In the spring experiment, the HA group showed significantly higher transpiration and stomatal conductance than the Con at 24 DAT (1.7- and 1.3-fold higher, respectively). In the fall, the MA group exhibited the highest photosynthetic rate and stomatal conductance at 24 DAT, which were 8% and 56% higher than those of the Con, respectively. In addition, the transpiration rate was approximately 45% higher across all air ionization treatments. Correspondingly, in spring, the fresh weights of shoots and roots in the HA group were at least 26% greater than those of the Con, and in fall, all air ionization treatments showed increased shoot and root fresh and dry weights and leaf area by at least 50% compared to the Con.
The effects of air anions on spinach cultivation were stage-dependent and varied based on the concentration of negative ions. At the germination stage, the LA treatment exhibited the highest performance (Fig. 2A), whereas seedling growth was significantly enhanced by the MA and HA treatments at 14 DAT (Fig. 2B). HA maintained the highest fresh weight throughout the seedling stage (Fig. 3). These results indicate that the positive influence of air anions on crop growth differs depending on the growth stage and ion concentration. Therefore, appropriate negative air ion concentrations and corresponding levels of electrical stimulation should be applied according to the growth stage. An elevated DNA content can be regarded as an indicator of an accelerated cell division cycle (Darzynkiewicz and Juan 1997). The cell cycle consists of the G1 phase (cell growth), the S phase (DNA synthesis), the G2 phase (preparation for division), and the M phase (mitosis, in which one cell divides into two daughter cells) (Vázquez-Ramos and de la Paz Sánchez 2003). The results of the cell cycle analysis indicated that the ratio of the S phase representing DNA synthesis and the M phase representing the mitotic phase was higher in all air anion treatments than in the Con. The promotion of spinach shoot growth could be attributed to an increase in the cell division rate due to electrical stimulation by the air anion treatment (Fig. 6).
Gualtierotti et al. (1968) and Krueger et al. (1969) reported that negative air ions increased iron (Fe) absorption, promoted the biosynthesis of cytochromes and other Fe-containing enzymes, and enhanced oxygen consumption in plants. Increased oxygen consumption has also been consistently observed in animal (guinea pig) tissue experiments, where a negative ion treatment significantly enhanced the rate of conversion of succinate to fumarate (Krueger and Reed 1976). Overall, the results suggest that negative air ions stimulate a specific reaction in the Krebs cycle, thereby reinforcing the aerobic metabolism of carbohydrates. As the Krebs cycle is a fundamental metabolic pathway that operates universally at the cellular level, such effects are likely to occur in plants as well. Thus, it is hypothesized that electrical stimulation induced by air anions fully meets the energy demand of spinach, thereby promoting DNA synthesis and cell division. This hypothesized mechanism provides further supporting evidence of the growth-promoting effects of electrical stimulation induced by negative air ionization. Future studies should test this hypothesis by directly measuring electrical changes during negative air ionization exposure, conducting time-course analyses of hormone dynamics and related gene expression levels.
Conclusion
A specific range of air anion treatment could have a positive effect on crop growth not only at the harvest stage but also at the seed germination and seedling stages in spinach. Unlike conventional electrical stimulation methods, which are often hazardous and inefficient when applied practically, air-based electrical stimulation enables localized application at crop sites using a simpler and more manageable setup. When integrated into horticultural crop production, air ionization can be regarded as a key technology that can be integrated with fundamental environmental factors, such as temperature, humidity, light, and nutrients, to accelerate plant growth and enhance food production without requiring additional resources. To facilitate such applications, it is essential accurately to determine the strength of the electric field generated by air anions and quantitatively characterize the resulting physiological responses of plants.
Moreover, precise environmental control technology of vertical farms may enable the more efficient formation of air-induced electric fields than in field or greenhouse conditions, potentially eliciting stronger positive effects on crop growth through electrical stimulation by an air anion treatment, ultimately enhancing food productivity.
Supplementary Material
Supplementary materials are available at Horticultural Science and Technology website (https://www.hst-j.org).
- HORT_20250065_Fig_S1.pdf
Supplementary Fig. 1. Negative air ionization generators were installed between the LED lighting fixtures within the cultivation module. The negative air ion concentration was regulated by adjusting the distance between the generators and the plant canopy, thereby enabling precise adjustments of ion exposure throughout the growth period.


