Introduction
Materials and Methods
Plant materials and growth conditions
Monitoring the flower lifespan
Flower dry weights
Observations of floral anatomy
Microscopic observations of the scape anatomy
Analysis of scape metabolome
Data analysis
Results
Floral longevity and functional traits at different positions on the scape
Anatomical differences in the scape
Changes in primary metabolites within the scape
Discussion
Flower functional traits within racemose inflorescences
Scape storage within racemose inflorescences
Nutrient recycling in flowers
Conclusions
Introduction
Orchids belonging to the genus Dendrobium have economic significance as potted plants and cut flowers. Most species of Dendrobium are characterized by the production of racemose inflorescences, and the inflorescence size is an essential characteristic that influences plant reproduction (Zhang et al. 2022). In addition, larger inflorescences are preferred by consumers as an important ornamental feature (Shibuya 2018). Investigating the morphological and anatomical foundations of racemose inflorescences in Dendrobium orchids and generating insights into their physiological maintenance mechanisms will contribute to a more comprehensive understanding of the reproductive ecological adaptation mechanisms in orchids.
Studying the functional traits of plant organs is an important approach used to understand plant adaptations (Wright et al. 2004). However, current research on the functional traits of floral organs is somewhat limited compared with that on other organs, and existing research has tended to focus on traits such as longevity, thickness, vein density, and biomass (Zhang et al. 2017; Zhang et al. 2021; Li et al. 2023). Among these, flower longevity reflects the conditions and functions of plant growth, and reproduction and is positively correlated with traits related to resource investment in plants (Roddy et al. 2021). Petal thickness is associated with the acquisition and retention of resources and water in flowers, with thick petals being able to store more substances and water, thereby reducing the internal water loss in plants (Gonzalez-Paleo and Ravetta 2018). Flower veins play a key role in the transport of water and nutrients within the petals, thereby determining the physiological responses of flowers to water and nutrient availability (Jin et al. 2019). Flower biomass directly reflects the resource investment in flowers and is an important trait of floral displays that influences plant reproduction through its effect on floral size. Flower biomass is also closely associated with the ability of flowers to withstand external forms of stress (Jin et al. 2019). Studies of the functional traits of orchid flowers have also determined varying degrees of differences among different species or varieties, with these findings indicating that orchids that are characterized by prolonged flower longevity often possess a large floral biomass, a high dry weight per unit area, and greater floral thicknesses (Zhang et al. 2021; Li et al. 2023). Exploring plants with long lifespans that possess racemose inflorescences and understanding their floral adaptation strategies through the integration of flower functional traits and scape traits will hold significant importance.
Significant variations in the longevity of flowers produced have been observed at different positions within an individual inflorescence (Zhang and Li 2009), and some researchers have proposed that such spatial variation serves as an adaptive mechanism that enables plants to maximize the benefits of floral display while reducing detrimental effects (Zhang and Li 2009). For example, in the case of Narthecium asiaticum (Liliaceae), flowers in the distal portion of the inflorescence have significantly shorter longevity than those in more central and proximal positions (Ishii and Sakai 2001). In relation to this, Ishii and Sakai proposed that continuing to maintain the flower display within the distal portion of the inflorescence is a wasteful investment of resources (Ishii and Sakai 2001). Stpiczyńska also suggested that limited resources could be a primary factor contributing to the shortened longevity of flowers in the distal portion of the inflorescen ces in this plant (Stpiczyńska 2003). To date, however, comparatively few studies have focused on determining whether orchids with long individual flower longevity follow the patterns observed in previous studies. Similarly, few studies have examined the structural and physiological mechanisms underlying the maintenance of this trait.
Integrating metabolomics provides an effective way to reflect the physiological response processes of plants. Previous primary metabolite studies have demonstrated that the flower lifespan is associated with the metabolism and storage of nutrients within the flower itself or in the plant. For example, significant changes in saccharide substances also occur in the pseudobulbs of Oncidium ‘Gower Ramsey’ during different flower development stages (Hsiao et al. 2011), whereas organic acids in Solanum nigrum increase significantly after flowering (Samardjieva et al. 2015). In addition, lipids play important roles in cell membranes, the regulation of cellular processes, and defense mechanisms during flower growth, development, and responses to stress, thereby influencing flower growth and development (Li et al. 2015; Sánchez-Martín et al. 2018), whereas amino acids and their derivatives are prominently associated with flower resistance (Chen et al. 2022). Previous research on the metabolomics of flowers has focused primarily on examining the abundance of different metabolites within the flower or plant and the corresponding correlations with flower growth and development. However, relatively few studies have investigated the metabolic changes within the flower scape.
This study aims to investigate the variation trends in the anatomical structure and metabolomics in the Dendrobium genus at the inflorescence level. Dendrobium ‘Garnet Beauty’ with a racemose inflorescence was used as the research material. Anatomical features were compared between the distal and proximal portions of the scape. Metabolomics was employed to examine the changes in stored substances across different locations of the scape at both the mature and senescent stages.
Materials and Methods
Plant materials and growth conditions
In this study, we specifically focused on the orchid Dendrobium “Garnet Beauty.” Seedlings propagated by tissue culture were used as the research material, and all specimens were cultivated in a greenhouse within the Nanning Botanical Gardens. The growth year and cultivation conditions of all materials were identical. Growth conditions were carefully maintained; the air temperature ranged from 25°C to 30°C, the relative humidity was held between 70% and 90%, and sufficient sunlight (40%) was provided through shading nets. The experimental plants were planted in 1.5-L plastic pots filled with a bark mixture, and all samples were grown within the same designated area. To ensure optimal growth, plants were watered daily at 08:00 to maintain a substrate moisture content of 65–75%.
Our aim was to investigate the role of stored substances within the scapes in maintaining inflorescence longevity throughout the entire inflorescence architecture of Dendrobium. To this end, we conducted a comprehensive study involving the metabolic profiling of the scapes, an investigation of the physiological responses of the floral organs, and observations of the morphological and anatomical features of the scape and floral organs. Specifically, we examined the differences in the flower lifespan between the distal and proximal portions of the scape. We assessed different functional and physiological traits of the perianth and scape as well as the anatomical characteristics of the perianth and scape in the distal and proximal sections of the inflorescence. To ensure an accurate analysis, we obtained data from six to ten biological replicates. The anatomical and physiological response samples were collected in August from flowers in full bloom (15–20 days after first bloom). Additionally, to reveal how storage in the scapes of Dendrobium “Garnet Beauty” contributes to maintaining the racemes, we measured the levels of primary metabolites (including amino acids and their derivatives, nucleotides and their derivatives, organic acids, lipids, and saccharides) in young and senescent blooms at distal and proximal portions of the scape and conducted pairwise comparisons.
Monitoring the flower lifespan
To investigate the disparity in the flower lifespan between the distal and proximal segments of the Dendrobium “Garnet Beauty” inflorescence, we specifically selected scapes that had all flowers in the bud stage and supported more than ten buds. All buds on the scape were labeled and observed, and the flower cluster was divided into three equal parts, referred to as the upper, middle, and lower parts. The upper third of the scape was designated as the “distal” portion, and the lower third was designated as the “proximal” portion. The lifespan of the flowers was recorded by calculating the average lifespan of all flowers in the distal and proximal parts separately. To ensure an accurate analysis, we obtained data from ten biological replicates and documented the lifespan of individual flowers in terms of days. The lifespan of an individual flower was determined from the moment it fully opened until the closure of the labellum or the onset of senescence and wilting of the labellum, petals, and sepals.
Flower dry weights
To determine the dry weights of individual flower components, in this case the labellum, petals, and sepals, flowers from both the distal and proximal sections of the scape were carefully selected from six inflorescences. The isolated parts were then oven-dried at a temperature of 60°C for 48 h until a constant weight was attained. Subsequently, the dry weight of each flower part was measured using an analytical scale with a sensitivity level of one in ten thousand.
Observations of floral anatomy
To assess the density of the veins in the labellae, sepals, and petals, fully opened flowers from six inflorescences were carefully selected and immersed in a formalin: acetic acid: alcohol: distilled water fixative solution (FAA) at a ratio of 10:5:50:35 for a period exceeding 30 days to ensure correct fixation and decolorization. Upon obtaining complete decolorization and transparency of the floral organs, entire petals were placed directly onto glass slides and flattened using a cover slip. Images of the veins were captured at ×5 magnification, carefully avoiding the main veins of the flower, using a Leica DM2500 microscope (Leica Microsystems Vertrieb GmbH, Germany) equipped with a digital camera. For each section, five views were examined. The length of the floral veins was subsequently measured from the digital images using Image J software. The flower vein density was quantified as the length of the veins per unit area.
To measure the thickness of the floral perianth, perianth cross-sections were meticulously prepared utilizing a hand-sectioning technique, as follows: a slit was cut in a carrot, and the petals were then inserted into the slit. Ensuring that the blade was perpendicular to the incision, the entire carrot along with the petals was horizontally sliced to obtain slices of flower petals with a cross-sectional thickness of approximately 30 µm. Subsequently, these sections were photographed using a Leica DM2500 microscope to capture three distinct views of each section at ×10 magnification. For an accurate analysis, we obtained datasets from six biological replicates. The resulting images were then processed, and the thickness of the floral perianth was accurately measured using Image J software.
Microscopic observations of the scape anatomy
To conduct a comparative analysis of the anatomical variances of the distal and proximal segments of the scape, fully developed scapes bearing fully opened flowers were carefully selected from six inflorescences. The scapes were then divided into their respective distal and proximal portions and subjected to fixation and decolorization utilizing an FAA solution. After satisfactory decolorization, the scapes were manually sectioned, removed from the FAA solution and then cut into 30 µm slices using a blade perpendicular to the scape. Subsequent observations and photographic documentation were conducted using a Leica DM2500 microscope operating under magnification of ×10. For each segment, three distinct views were captured, and Image J software was used to measure the areas occupied by the xylem, phloem, and fibers precisely within each vascular bundle.
Manually sectioned slices were generated by removing the scape from the FAA solution and cutting it into 30 µm slices using a blade perpendicular to the scape. The slices were stained by placing them in a diluted 5% safranin solution and were then photographed using a Leica M205A stereoscopic microscope (Leica Microsystems Vertrieb GmbH, Germany) under visual magnification of ×20.0 and with a zoom factor of 2.00. For each section, three views were obtained, and Image J software was used to measure a range of parameters, including the total cross-sectional area of the scape, the number of vascular bundles, and the areas occupied by the xylem, phloem, and fibers within the vascular bundles. To ensure an accurate analysis, we obtained data from six biological replicates. Other parameters were calculated as follows:
Analysis of scape metabolome
The most significant differences in metabolite levels post-anthesis likely occur during the transition from full bloom to complete senescence (Li et al. 2023). Therefore, to conduct a widely targeted metabolomics analysis, we collected both young and old scapes from Dendrobium ‘Garnet Beauty’ and divided both sets into distal and proximal parts, after which all samples were frozen in liquid nitrogen immediately. Prior to sample extraction, the freeze-dried materials were ground into a powder using a grinding mill (MM 400; Retsch, Haan, Germany) at 30 Hz for 1.5 min. Thereafter, 100 mg of the powdered sample in each case was dissolved in 1.0 mL of a 70% methanol extraction solution and stored at 4°C for 24 h, after which the homogenate was centrifuged at 10,000 × g for 10 min to remove undissolved residue. Prior to performing the LC-MS analysis, the extracts were absorbed into CNWBOND carbon-GCB SPE cartridges (ANPEL, Shanghai, China) and filtered through a SCAA-104 membrane (0.22 µm; ANPEL, Shanghai, China). The metabolites were analyzed using a liquid chromatography-electrospray ionization-tandem mass spectrometry (LC-ESI-MS) system (LC: Shim-Pack UFLC Shimadzu CBM30A System; ESI, MS: Applied Biosystems 6500 QTRAP System). Chromatographic separation was conducted using an ACQUITY UPLC HSS T3 C18 column (1.8 mm, 2.1 mm × 100 mm; Waters). Water and acetonitrile containing 0.04% acetic acid (v/v) were used as mobile phases A and B, respectively. The A:B (v/v) elution profile was as follows: 95:5 v/v (water/acetonitrile) at 0 min, 5:95 v/v at 11.0 min, and 95:5 v/v at 15 min. The flow rate was 0.4 mL/min, and the column temperature was 40℃. The effluent was alternatively connected to an ESI-triple quadrupole-linear ion trap (QTRAP)-MS.
Linear ion trap and triple quadrupole (QQQ) scans were acquired using an API 6500 QTRAP LC/MS/MS system equipped with an ESI turbo ion-spray interface operating in positive ion mode and controlled by Analyst 1.6 software (AB Sciex). The ESI source parameters were as follows: turbo spray, 5500 V, and 550°C. The ion source gas I (GSI), ion source gas II (GSII), and curtain gas (CUR) were set at 55, 60, and 25 psi, respectively, and the collision gas (CAD) pressure was set to high. QQQ scans were acquired when the collision gas (nitrogen) pressure for MRM experiments was at 5 psi.
Metabolite identification was based on parametric values (m/z data, retention time, and fragmentation partners) and was compared with a self-built database (MetaWare) for annotation results (http://www.metware.cn/).
Data analysis
Statistical significance was analyzed using independent sample t-tests that were performed for distal and proximal scapes using SPSS 20.0 software (IBM, New York, USA), and differences for which p < 0.05 were considered to be statistically significant. Metabolite differences in pairwise comparisons were analyzed with a partial least squares-discriminant analysis (OPLS-DA). The relative importance of each metabolite in the OPLS-DA model was evaluated using the variable importance in projection (VIP) metric. Metabolites with a VIP value ≥ 1 and fold change ≥ 1 or ≤ ‒1 were identified as differentially accumulated metabolites. We conducted longitudinal and transverse comparisons of metabolites using samples from scapes at two stages of Dendrobium ‘Garnet Beauty’ development: young and senescent. Longitudinal comparisons involved comparing the levels of metabolites in the distal and proximal parts of young scapes (YD vs. YP) and senescent scapes (OD vs. OP). For transverse comparisons, we compared the levels of metabolites in the distal parts of young and senescent scapes (YD vs. OD) and in the proximal parts of young and senescent scapes (YP vs. OP). By performing these comparisons, we hoped to gain an understanding of the relationships between scape longevity and different metabolite components and of the accumulation of metabolites in scapes of different ages and in different positions within scapes of the same age.
Results
Floral longevity and functional traits at different positions on the scape
We monitored the entire lifespan of the inflorescences of Dendrobium “Garnet Beauty” and observed the sequential opening of flowers from the base to the top of the inflorescence. The individual flowers had lifespans ranging from 20 to 58 days, with an average of 42 and 44 days. With respect to floral longevity, there were no significant differences between the flowers on the proximal and distal portions of the scape (Supplementary Table S1). We also compared the biomass, vein density, and floral thickness of the petals, sepals, and labellae between the proximal and distal segments of the scape. The average biomass of the petals, sepals, and labellum was 20.0, 18.4, and 22.2 mg, respectively; the average vein density of the petals, sepals, and labellum was correspondingly 0.46, 0.52, and 0.44 mm mm-2; and the corresponding values for floral thickness were 0.61, 0.51, and 0.81 mm (Table 1). These values indicated that among the flower parts, the labellae and sepals had the highest and lowest biomass and thickness, respectively. However, the sepals had the highest density of veins. With respect to physiological responses, functional traits, and anatomical characteristics, we detected no significant differences between flowers produced on the distal and proximal portions of the scape (Table 1 and (Supplementary Table S1).
Table 1.
Functional flower traits
Anatomical differences in the scape
The vascular bundle size and quantity are anatomical features associated with the efficiency of vascular transport in the scape. We observed that vascular bundles in the scape of Dendrobium “Garnet Beauty” were segregated into two rings: an outer ring comprising small vascular bundles located at the edge of the scape and an inner ring comprising large vascular bundles surrounded by parenchymal tissue (Supplementary Fig. S1). The vascular bundles in the scape consisted of the xylem, phloem, and fibers (Fig. 1A). A comparison of the proportional areas of the xylem, phloem, and fiber in the vascular bundles of the distal and proximal scapes revealed no significant differences in the respective areas of the xylem. However, the area occupied by the phloem in the distal part (4.2 × 10-4 mm2) was significantly larger than that in the proximal part (2.8 × 10-4 mm2); similarly, the area occupied by fibers in the distal part was significantly larger than that in the proximal part (Fig. 1B).
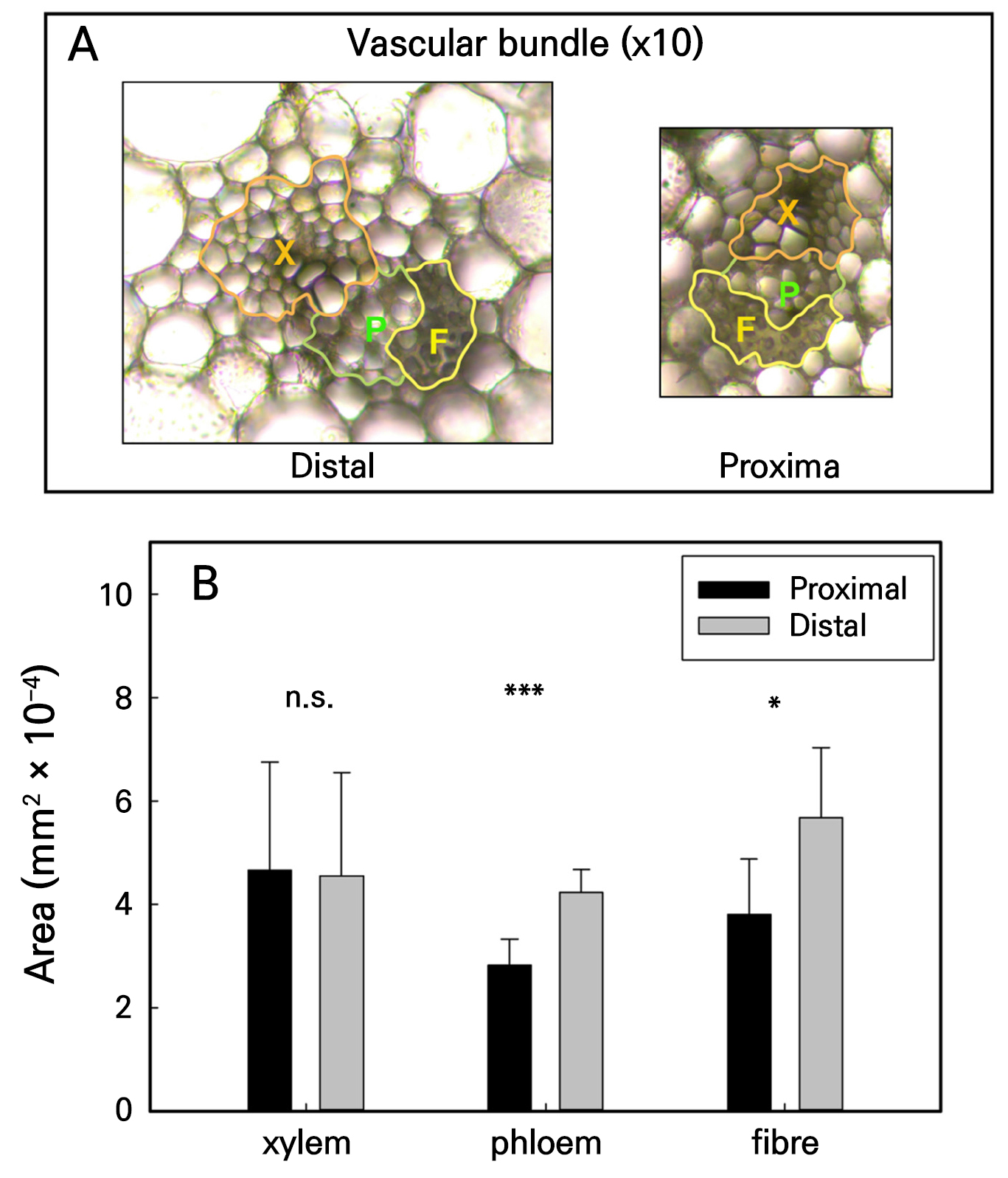
Fig. 1.
Anatomy of the scape. Transverse section of the distal and proximal parts of the scape, with bright red dots indicating vascular bundles stained with safranin dye (A). Microscopic schematic diagram of the vascular bundle at 10× magnification, where X, P, and F represent the xylem, phloem, and fiber, respectively (B). Analysis of the area differences in each part of the vascular bundle in the distal and proximal parts of the scape (C), with each vertical bar representing the mean ± SD of six measurements from different individuals. Statistical differences in the xylem, phloem, and fiber in the vascular bundles between the distal and proximal parts of the scape were determined by independent sample t-tests (*p < 0.05; **p < 0.01; ***p < 0.001; n.s., no significant difference).
The average cross-sectional areas of the proximal and distal parts of the scape were 50.0 mm2 and 27.4 mm2, respectively, and the distal portion had a significantly larger cross-sectional area than the proximal portion (Fig. 2A). The average vascular bundle size in the proximal and distal parts of the scape was 0.086 mm2 and 0.101 mm2, respectively, and the bundle size in the distal part was thus significantly larger (Fig. 2B). Similarly, the total area occupied by vascular bundles in the distal part (15.4 mm2) was significantly larger than that in the proximal part (13.3 mm2) (Fig. 2C), and the proportion of the total vascular bundle area to the cross-sectional area in the distal part (23%) was significantly larger than that in the proximal part (19%) (Fig. 2D). There were 29 and 116 vascular bundles in the distal and proximal parts of the scape, respectively, with the distal part thus having significantly fewer bundles (Fig. 2E). However, there were no significant differences between the number of vascular bundles per unit area in the distal and proximal segments (Fig. 2F).
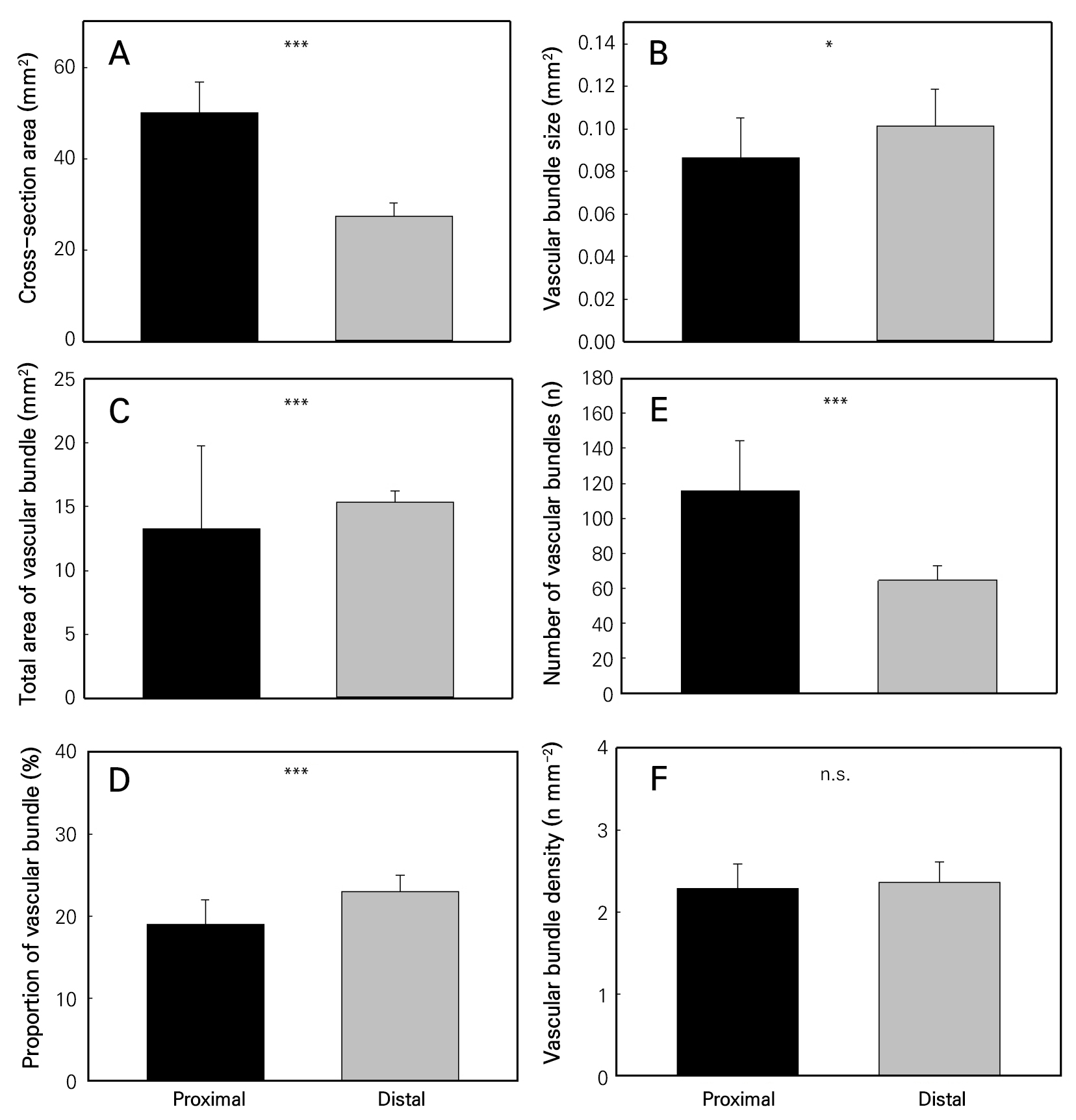
Fig. 2.
Anatomical characteristics of the scape. Proximal represents the average lifespan of flowers in the lower one-third of the inflorescence, and Distal represents the average lifespan of flowers in the upper one-third of the inflorescence. Cross-sectional area of the scape (A), size of the vascular bundle (B), total area of all vascular bundles in the scape (C), proportion of the total vascular bundle area to the cross-sectional area (D), number of vascular bundles in the scape cross-section (E), number of vascular bundles per unit area (F). Each vertical bar represents the mean ± SD of six measurements from different individuals. Independent samples t-tests were used to determine the statistical differences between the various anatomical characteristics of the scape at the distal and proximal parts (*p < 0.05; **p < 0.01; ***p < 0.001; n.s., no significant difference).
Changes in primary metabolites within the scape
We undertook pairwise comparisons of the accumulated levels of amino acids at different stages and in different locations in the scape. In the longitudinal comparisons (YD vs. YP and OD vs. OP), significant differences were observed in the contents of amino acids in the distal and proximal parts of the scape. Fifteen amino acids/amino acid derivatives differed significantly between YD and YP, with 14 showing significantly higher levels in the distal part. Thirty-eight amino acids/amino acid derivatives showed differences between OD and OP, with 35 characterized by significantly higher levels in the distal part (Supplementary Fig. S2). In the transverse comparisons (YD vs. OD and YP vs. OP), we also observed differences in the contents of amino acids among the different stages and locations of the scape. The YD vs. OD comparison revealed differences in 28 amino acids/amino acid derivatives, with 24 showing significantly increased levels in the distal part. Similarly, the YP vs. OP comparison revealed differences in 14 amino acids/amino acid derivatives, with nine showing significantly increased levels with scape aging (Supplementary Fig. S2).
With regard to saccharide changes at different stages and in different locations along the scape, we found that all saccharides were higher in the distal part of the scape in the longitudinal (YD vs YP) comparison. Among these, d-ribose and d-glucosamine 1-phosphate showed the most significant differences. The OD vs. OP comparison revealed differences in the levels of six saccharide substances, with higher levels observed in the distal part of the scape. Among these, pyridoxine-5'-O-(6'-feruloyl)glucoside showed the most significant difference (Fig. 3). The transverse (YD vs. OD) comparison showed differences in the levels of eight saccharide substances. Among these, five were lower in aging scapes, with d-glucosamine 1-phosphate showing the most significant reduction, while three showed higher levels, with d (+)-melezitose O-rhamnoside and maltotriose significantly increased. The YP vs. OP comparison revealed differences in seven saccharide substances, with five showing lower levels in aging scapes (Fig. 3).
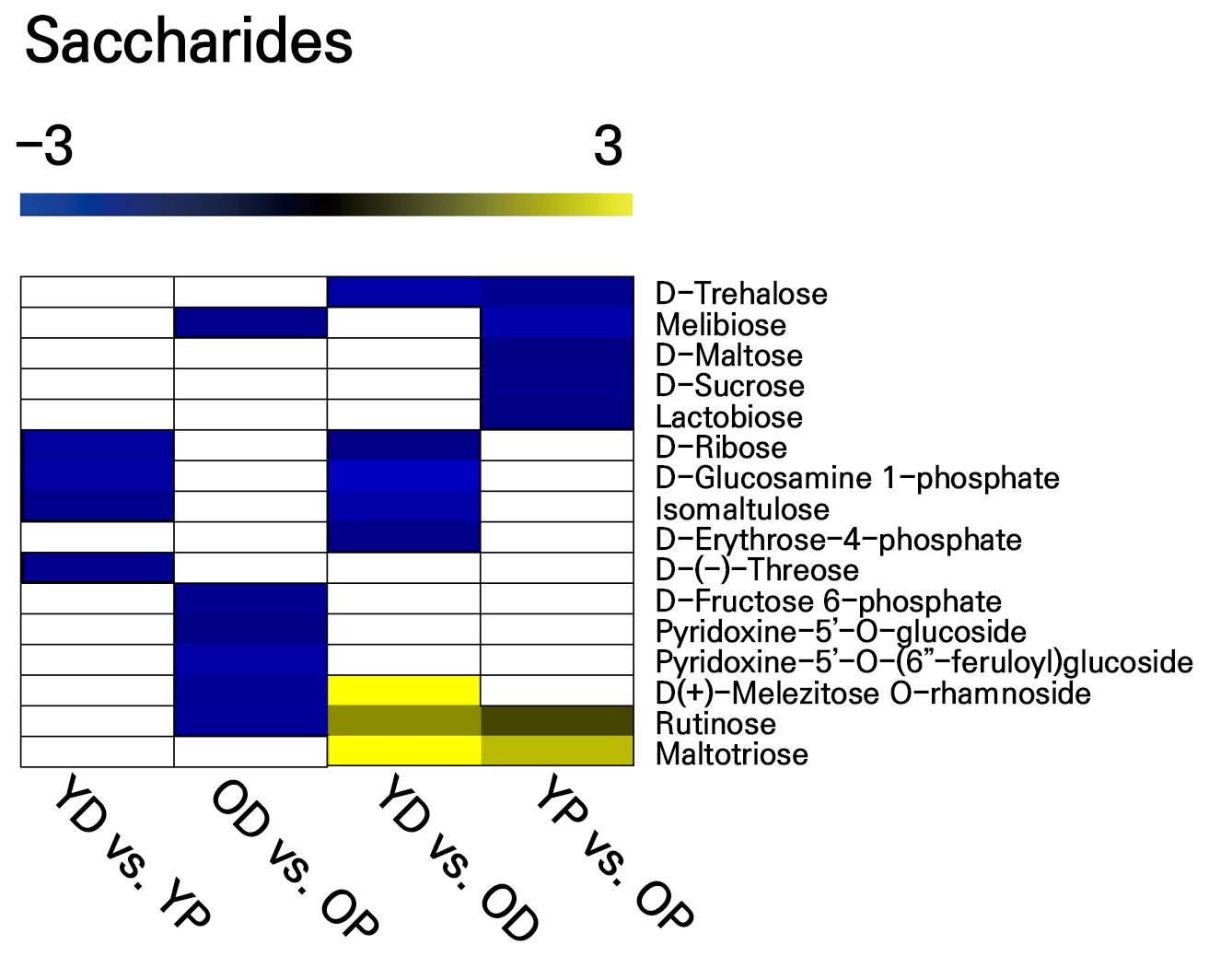
Fig. 3.
Saccharides were detected using widely targeted UPLC-MC to determine the contents of saccharides in the distal and proximal parts of young and old scapes, with the color in the heat map indicating the fold changes between the control groups. White in the heat map indicates no significant difference between the comparisons, and three independent replicates were performed for each part of the scape in each age group. YD: young distal; YP: young proximal; OD: old distal; OP: old proximal. YD vs. YP: YP/YD; OD vs. OP: OP/OD; YD vs. OD: OD/YD; YP vs. OP: OP/YP.
We conducted similar pairwise comparisons of the lipid level in different parts of the scape at different stages of development. The longitudinal comparison (YD vs. YP) revealed differences in the levels of 30 lipids, with only a single lipid (1,3-O-dicaffeoylglycerol) present at higher levels in the proximal part of the scape. The other 29 lipids were higher in the distal parts. The OD vs. OP comparison revealed differences in the levels of 44 lipids, with only two lipids (d-sphingosine and sphingosine 1-phosphate) higher in the proximal part. The other 42 lipids were higher in the distal part (Fig. 4). The transverse comparison (YD vs. OD) revealed differences in the levels of 28 lipids; of these, the levels of 24 were lower in aging scapes. Conversely, the levels of four lipids were higher in aging scapes. Furthermore, the YP vs. OP comparison revealed differences in the levels of 35 lipids, all of which were lower in aging scapes (Fig. 4).
The longitudinal (YD vs. YP) comparison of the levels of organic acids showed differences in the levels of seven organic acids, all of which were characterized by higher levels in the distal part of the scape. The OD vs. OP comparison revealed differences in nine organic acids, with eight showing higher levels in the distal part. Only a single organic acid, cis-citral, was present at lower levels in the distal part (Fig. 5). The transverse comparison (YD vs. OD) revealed differences in the levels of 16 organic acids; there were higher levels of 13 of these (including 3-ureidopropionic acid and 2,2-dimethylsuccinic acid) in the aging scape. Conversely, only three organic acids were characterized by lower levels in the aged scape. Similarly, the YP vs. OP comparison revealed differences in 15 organic acids; 11 of these were found at higher levels in the aging scape (Fig. 5).

Fig. 4.
Lipids were detected using widely targeted UPLC-MC to determine the contents of lipids in the distal and proximal parts of young and old scapes. The color of the heat map represents the fold change between the control groups, with white indicating no significant difference between the comparisons. Three independent replicates were performed for each different part of the scape in each age group. YD: young distal; YP: young proximal; OD: old distal; OP: old proximal. YD vs. YP: YP/YD; OD vs. OP: OP/OD; YD vs. OD: OD/YD; YP vs. OP: OP/YP.
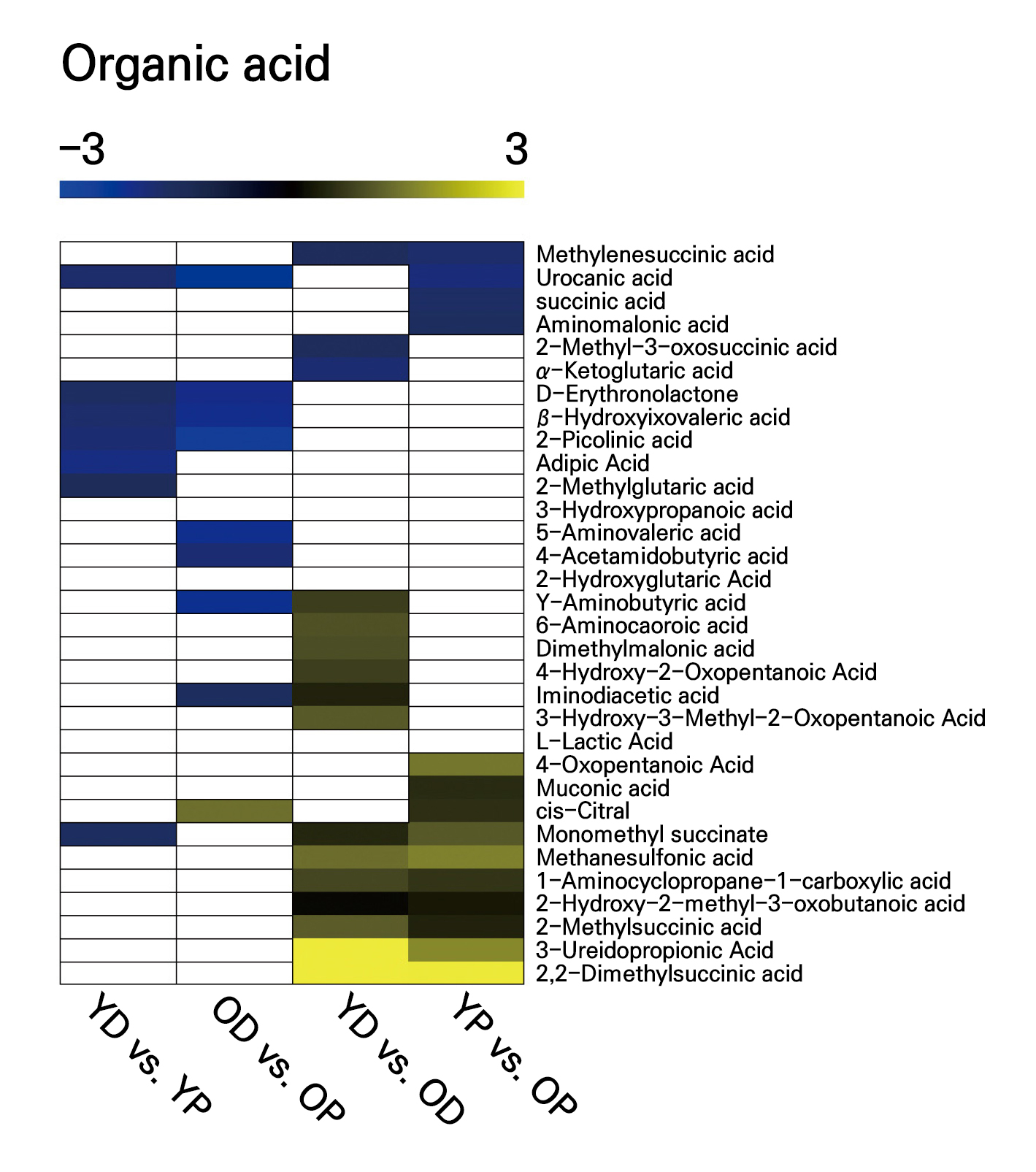
Fig. 5.
Organic acids were detected through a widely targeted UPLC-MC analysis of their contents in different parts of young and old scapes. The heat map colors indicate the fold change between the control groups, and white indicates no significant difference between the comparisons. Three independent replicates were conducted for each part of the scape in each age group. YD: young distal; YP: young proximal; OD: old distal; OP: old proximal. YD vs. YP: YP/YD; OD vs. OP: OP/OD; YD vs. OD: OD/YD; YP vs. OP: OP/YP.
Discussion
Flower functional traits within racemose inflorescences
Previous studies have reported significant differences between the lifespans of flowers produced at different positions within inflorescences (Zhang and Li 2009). For example, in their study of the floral lifespan of Narthecium asiaticum (Liliaceae), Ishii and Sakai (2001) found that flowers produced in the distal portion of the inflorescence were characterized by a significantly shorter lifespan than those produced in central and proximal locations. Similarly, Li et al. (2021) observed significant differences between the lifespans of flowers at different positions within the inflorescences of Stellera chamaejasme, a plant species found in alpine grasslands (Li et al. 2021). Flowers located at the edge of the inflorescence had the longest lifespan, followed by those in the central portion, whereas flowers at the distal end had the shortest lifespan (Li et al. 2021). In a study of the floral longevity of Platanthera chlorantha, the longevity of lower‐ or middle‐positioned flowers was found to exceed that of flowers in the upper position (Stpiczyńska 2003). In contrast, the present study detected no significant difference between the lifespan of flowers produced at the distal and proximal positions on the inflorescences of Dendrobium “Garnet Beauty” (Supplementary Table S1). This suggests that Dendrobium ‘Garnet Beauty’ employs a more conservative strategy for resource allocation of the inflorescence at different locations compared to several other plants.
The distribution of vascular bundles can affect the efficiency of the long-distance transport of nutrients and water (Housley and Peterson 1982; Yang et al. 2021). The larger the total area of the vascular bundles is, the higher the transport efficiency becomes (Xie et al. 2012; Miyashima et al. 2013). The number of vascular bundles is positively correlated with the translocation of non-structural saccharides in stems and leaves, and the number, cross-sectional area, and phloem area of vascular bundles are positively correlated with the seed yield (Zhou et al. 2022). The area of the xylem and the density of the vascular bundles determine the water supply status of floral organs, and there is a significant positive correlation between the xylem area and hydraulic conductivity (Sun et al. 2017). A larger area of the phloem is considered more conducive to nutrient transport (Miyashima et al. 2013). In the present study, we found that the area of the vascular bundles and the proportion of the total vascular bundle area to the cross-sectional area were significantly larger in the distal part of the scape than in the proximal part (Fig. 2A–2D). Moreover, the area of phloem in the distal part was significantly larger than that in the proximal part (Fig. 1B), indicating that the distal part of the scape has greater nutrient transport efficiency.
Scape storage within racemose inflorescences
Varying degrees of resource allocation and competition have been observed among flowers within an individual inflorescence, leading to lifespan, quantity, size, development, morphology, and reproductive efficiency differences among the different parts of the inflorescence (Warringa et al. 1998; Ashman and Hitchens 2000; Liu et al. 2003). Given the limited resources available for the inflorescence and the fact that the proximal portion of the inflorescence is often closer to the nutrient source, the flowers produced proximally may gain access to a more abundant supply of resources than those produced more distally. Consequently, flowers produced in the distal portion of an inflorescence tend to have significantly shorter lifespans than those in the proximal portion (Nakamura 1986; Lee 1989; Guitian 1994; Medrano et al. 2000; Liu et al. 2003). For “Garnet Beauty,” no significant difference in the flower lifespan was observed between the distal and proximal parts of the inflorescence; however, different resource allocation strategies were observed.
Saccharides serve as energy sources for plants (Han et al. 2020), and the findings of previous studies have indicated a close correlation between the saccharide content and the flowering process. For example, sucrose can affect the flower longevity of rose (Rosa hybrida) flowers, and flower soluble sugars and total non-structural carbohydrates are positively correlated with flower longevity (Monteiro et al. 2002). Additionally, the flowering time of peonies is influenced by the saccharide concentration (Mornya et al. 2011). Saccharides also play an important role in orchid flowering (Hsiao et al. 2011). Oncidium pseudobulbs accumulate significant amounts of saccharide reserves during development, which are subsequently mobilized to support new shoot and inflorescence development (Carl and Choy 2000). For example, during the inflorescence initiation stage in Oncidium “Gower Ramsey,” the levels of mannose polysaccharides in the pseudobulbs decrease gradually as these polysaccharides are converted to starch. Subsequently, during the inflorescence development stage, synthesized starch is degraded to provide energy for flower development (Hsiao et al. 2011). Similarly, in the bud differentiation process of Eulophia flava, there is an initial reduction of the soluble sugar content followed by a subsequent rapid increase, thereby providing a sufficient supply of energy for bud differentiation (Wang et al. 2015). In the present study, we found that the scapes of Dendrobium “Garnet Beauty” inflorescences contained large amounts of saccharide substances, and the contents of some differed significantly between the distal and proximal portions of the scape and were consistently higher in the distal portion (Fig. 3). These differences indicate that larger amounts of saccharides are allocated to the distal portion of the scape to maintain a consistent lifespan among flowers, regardless of whether the flowers are located at the distal or proximal ends of the inflorescence.
Organic acids play essential roles in the growth, development, and metabolism of flowers. In tea flowers, certain organic acids have been shown to promote the tricarboxylic acid cycle (TCA cycle) (Jia et al. 2016), and previous studies have reported the need for the TCA cycle in the flowering process of Antirrhinum majus (snapdragon) (Muhlemann et al. 2017). In Solanum nigrum, the levels of citric and malic acids after flowering showed increases of 2.5- and 6.7-fold, respectively (Samardjieva et al. 2015). Gibberellic acid enhanced the post-harvest longevity of Zantedeschia elliottiana (W. Wats./Engl.) flowers (Janowska and Jerzy 2004). Naphthalene acetic acid (NAA) shortens Bougainvillea spectabilis “Killie Campbell” flower longevity and decreases the percentage of flowers reaching anthesis (Gago and Monteiro 2012). In the present study, we found that changes in organic acid contents in the distal and proximal portions of the scape tended to be consistent with the changes in saccharide contents (Fig. 5). These results further indicate that the distal portion of the scape stores greater energy reserves with which to maintain flower lifespans.
In the sepals of Dendrobium, the levels of free fatty acids remain stable throughout the flowering and senescence processes, contrasting with the gradual decline observed in the petals and labellae. This pattern underscores the dual functionality of free fatty acids, which serve as both an energy source and key regulatory molecules in the plant’s stress response (Li et al. 2023). In the present study, among those lipids showing differences in the contents between the distal and proximal portions of the scape, most were characterized by significantly higher levels in the distal portion (Fig. 4 and Supplementary Fig. S3), thereby indicating that the distal portion of the scape stores relatively abundant supply energy reserves and may possess stronger potential resistance. Previous studies have reported the key roles of amino acids and their derivatives in orchid flowering (Pangjai and Huehne 2015), and this study also showed that the differences between the distal and proximal portions of the scape with respect to the amino acid contents were consistent with those among other primary metabolites (Supplementary Fig. S2). This would likely be related to the consistency in the lifespan of flowers at different positions on the inflorescence.
Nutrient recycling in flowers
Flowers will transfer nutrients to other viable tissues for storage or direct utilization prior to senescence (Freschet et al. 2010; Mao et al. 2013). Nutrient recycling is common in plants, and research has shown that this process occurs during organ senescence, with recycled nitrogen and phosphorus accounting for 30–40% of a plant’s annual nutrient demand (Cleveland et al. 2013; Zhang et al. 2018). Transferring nutrients, primarily nitrogen and phosphorus, from aging plant tissues or organs to other viable tissues extends nutrient residence times in plants, improves nutrient-utilization efficiency, and reduces plant reliance on soil nutrients, which is an important adaptive strategy in plants (Aerts and Chapin 1999; Vergutz et al. 2012). Studies have shown that orchids generally have lower mineral nutrient uptake rates than those seen in most other plants (Zhang et al. 2022). Accordingly, the process of nutrient recycling is essential for orchids. Most studies of nutrient recycling in plants have tended to focus on the recycling of nutrients within the leaves. However, other plant organs, including fine stems, the woody cores of trees, and nutrient-storing roots can also undergo nutrient resorption (Brant and Chen 2015). Numerous studies have found that orchid flowers do not immediately wither as they age but continue to persist in the inflorescence for some time, during which the biomass of the flower steadily declines. These observations suggest that orchids have a strong ability to remobilize nutrients (Li et al. 2023).
In this study, we found that the levels of different primary metabolites were significantly higher in aging scapes than in young scapes. Among the substances showing differences in contents between young and aging scapes (specifically amino acids and their derivatives and organic acids), most were present at significantly higher levels in aging scapes, indicating that their contents increase with the wilting of the flowers (Fig. 5 and Supplementary Fig. S2). These increases in amino acids, their derivatives, and organic acids in aging scapes can be attributed to the recycling of certain nutrients within the flowers, which are subsequently transported through the scape to other parts of the plant.
Conclusions
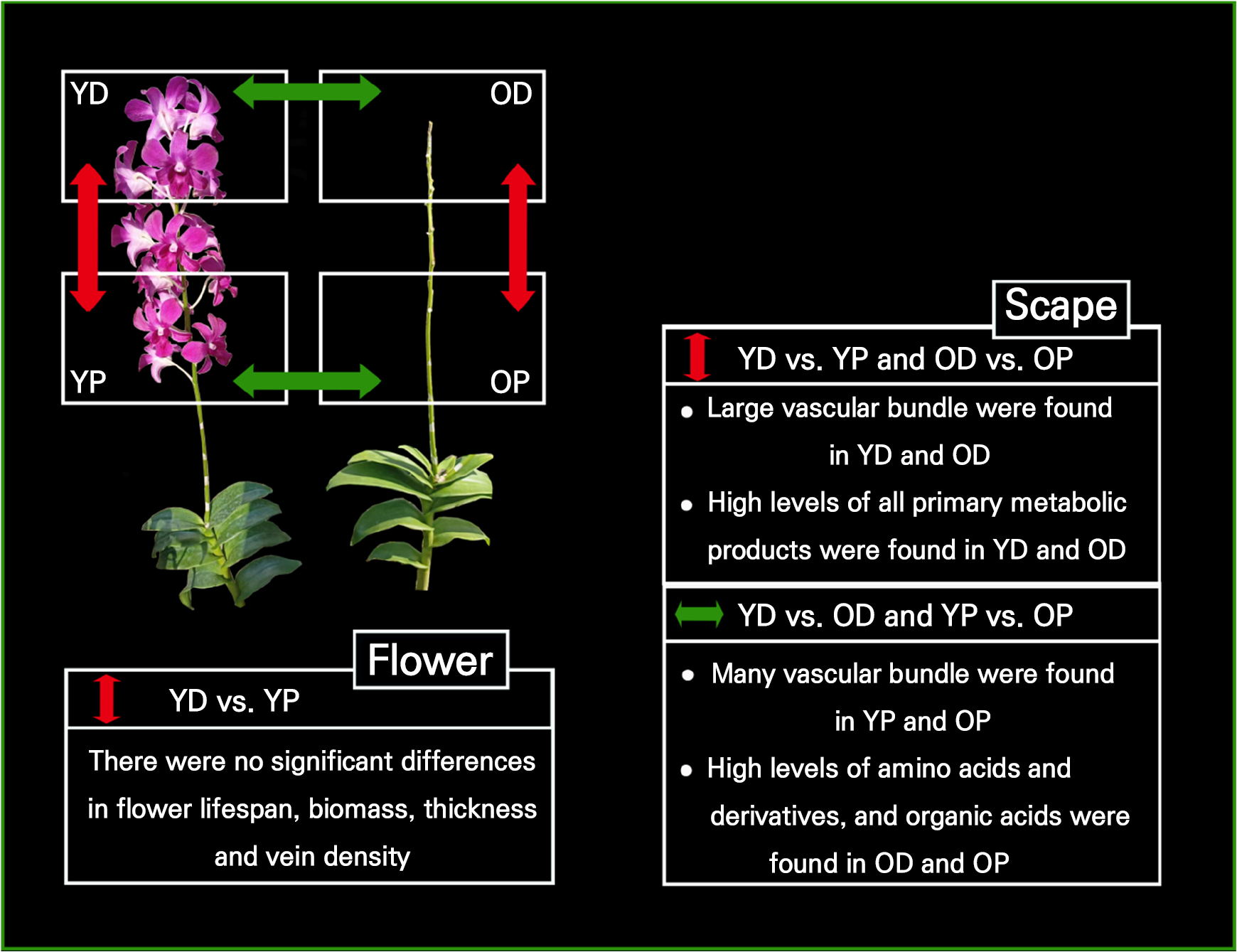
We compared the morpho-anatomical characteristics, primary metabolite contents, and corresponding physiological and morpho-anatomical responses of the distal and proximal scapes of Dendrobium ‘Garnet Beauty’ inflorescences. We observed significant differences in the levels of primary metabolites between the distal and proximal portions of the scape, with most showing higher levels in the distal portion. However, there were no significant differences in the lifespan, quantity, size, development, or morphology of the flowers among the different parts of the inflorescence (Fig. 6). Therefore, we propose that the scape of the orchid inflorescence has a certain storage capacity and that the orchid inflorescence may solve the problem of the shortened lifespan in the distal portion of the scape owing to resource limitations by increasing nutrient storage in the distal portion of the scape.
Supplementary Material
Supplementary materials are available at Horticultural Science and Technology website (https://www.hst-j.org).
- HORT_20250016_Table_S1.pdf
Supplementary Table S1. Floral longevity
- HORT_20250016_Fig_S1.pdf
Supplementary Fig. S1. Schematic representation of transverse section of scape. Transverse section of the distal and proximal parts of the scape, with bright red dots indicating the vascular bundles stained with safranin dye.
- HORT_20250016_Fig_S2.pdf
Supplementary Fig. S2. Amino acids and their derivatives were detected using widely targeted UPLC-MC to determine the content of amino acids and their derivatives in the distal and proximal parts of young and old scapes. The colors on the heat map indicate fold changes between the control groups, and white in the heat map indicates no significant differences between comparisons. Three independent replicates were performed for each different part of the scape in each age group. YD: young distal; YP: young proximal; OD: old distal; OP: old proximal. YD vs. YP: YP/YD; OD vs. OP: OP/OD; YD vs. OD: OD/YD; YP vs. OP: OP/YP.
- HORT_20250016_Fig_S3.pdf
Supplementary Fig. S3. Nucleotides and their derivatives were detected by a widely targeted UPLC-MC analysis of the levels in the distal and proximal parts of young and old scapes. The color in the heat map represents the fold change between the control groups, with white indicating no significant difference between the comparisons. Three independent replicates were performed for each different part of the scapes in each age group. YD: young distal; YP: young proximal; OD: old distal; OP: old proximal. YD vs. YP: YP/YD; OD vs. OP: OP/OD; YD vs. OD: OD/YD; YP vs. OP: OP/YP.