Introduction
Materials and Methods
Plant Materials
Calcium Pre-treatment
Low Temperature Treatment
TEM Observation
Result
Loquat Leaves Damaged by Cold Treatment
Subcellular Calcium Localization
Changes in Cellular Ca2+ Level by CaCl2 Treatment
Changes in the Chloroplast Ultrastructure and Ca2+ Distribution by Cold Treatment
Discussion
Introduction
Loquat (Eriobotrya japonica Lindl.) is a subtropical evergreen fruit crop originated from southeastern China (Hueso and Cuevas, 2008). It has been introduced to many other countries, such as Japan, India, Australia, New Zealand, Madagascar, South Africa and Mediterranean countries in Europe (He et al., 2011). The fresh fruits of loquat can be consumed directly or processed into jam and jelly. Its leaves have been documented to possess medicinal values and have been used to treat skin diseases, diabetes, chronic bronchitis, coughs, phlegm and ulcers (Ferreres et al., 2009). China is the world’s largest producer of loquat with a cultivation area of 120,000 ha, producing more than 400,000 tons of fresh fruits per annum (Hueso and Cuevas, 2008). Putian city of Fujian province is one of the largest loquat producing regions in China with 80,000 tons of annual production (Jiang et al., 2010). Loquat plants bloom in autumn, and fruits develop during winter and mature in early spring (Reig et al., 2011). Hence fruit development is at high risk to the cold damage during winter. Generally, cold damage to loquat occurs when temperature drops to less than -6°C at flowering stage and to less than -3°C at the early fruiting stage (Yang et al., 2003). Jiang et al. (2010) reported that more than 80% loquat trees in Putian regions were cold damaged in 2004, causing 13,300 tons of yield reduction and 65 million Chinese Yuan of economic loss. Cold damage is therefore a major limiting factor for loquat production and area expansion (Wang et al., 2008).
Calcium plays an important regulatory role in plant’s response to multiple environmental stresses (Kader and Lindberg 2010; Shao et al., 2008; Yoon et al., 2010). It plays an important role in the signal reception, transduction and responses of plant to the external biotic and abiotic stresses (Guo et al., 2004; Snedden and Fromm, 2001). The role of calcium has been implicated in plant responses to cold tolerance (Braam and Davis, 1990), mechanical stimulation (Botella and Arteca, 1994; Braam and Davis, 1990), drought stress (Shao et al., 2008) and oxidative stress (Harding et al., 1997). Calcium also assists in maintaining stabilization and integrity of cell membrane and its function (Msadek et al., 1998), reducing lipid peroxidation (Zeng et al., 1987), and inducing gene expression and synthesis of specific proteins (Gao and Chen, 2002; Kim et al., 2009). The alleviatory effect of Ca2+ on the damage induced by different abiotic stresses has been reported in many fruit trees such as apple (Malus pumila Mill), pear (Pyrus spp.) and mango (Mangifera indica L.) (Chen and Liu, 2001). Pan et al. (2000) found that Ca+ regulated the activities of superoxide dismutase (SOD) by activating cellular calmodulin (CaM), thereby reducing the membrane injury of mango leaves by water stress. It has been reported that CaCl2 sprays or application of Ca(NO3)2 fertilizer increased Ca+ concentrations in leaves and consequently increased cold tolerance in pear trees, reducing the incidence of fruit disorders (Raese, 1996).
Cytosolic Ca2+ increases rapidly and its distribution in cell changes accordingly in response to environmental stresses, which is a critical step in the transduction of stress signal and the response of plant to stress (Guo et al., 2004; Liu et al., 2010). Biyaseheva et al. (1993) showed that heat shock resulted in increased intracellular Ca2+ in pea mesophyll protoplasts. However, little is reported about the changes of Ca2+ distribution in loquat during low temperature stress. The cytochemical method of transmission electron microscopy (TEM) has been widely used for subcellular calcium localization (Jian et al., 1999; Meng et al., 2000). The present study was to determine cold tolerance, changes in cell Ca2+ distribution and cell superstructure in the leaves of two distinct loquat accessions under cold stress. The possible role of Ca2+ in the alleviation of cold damage was also discussed.
Materials and Methods
Plant Materials
A commercial loquat cultivar Zaozhong 6 (Eriobotrya japonica Lindl.) and a wild accession oakleaf loquat (Eriobotrya prinoides Rehd) were obtained from the National Germplasm Nursery, Fujian Academy of Agriculture Sciences, China. The scions were grafted onto the rootstock of Jiefangzhong (E. japonica) and grown in pots (one plant per pot) in an orchard in Fujian Agricultural and Forestry University, Fujian, China. The grafted trees were placed randomly, irrigated and fertilized according to common district practices. Three years after the grafting, trees with uniform and vigorous growth were selected and used as experimental materials.
Calcium Pre-treatment
Calcium pre-treatment was conducted at 9:00 am on 15 September, 2009. CaCl2 solution at 10 mM was sprayed onto each loquat plant until the solution began to run off from the leaves. The spraying treatments were made once every three days and repeated four times. This study used a randomised complete block design with two loquat varieties, two levels of CaCl2 (water and 10 mM) and three replicates. Each pot was properly labelled to identify the treatments and irrigated when necessary during the experi-mental period.
Low Temperature Treatment
The cold stress experiment was carried out in the Horti-cultural Laboratory of Fujian Agriculture and Forestry University from March to April, 2009. The cold treatment procedure was adopted from the methods of Xie and Li (2006) and Wang et al. (2008). The pre-treated loquat plants (whole plants in pots) with or without CaCl2 were placed in a temperature controllable freezer (ETC-100, Shanghai Jingchuang Electronics Manufacturing Co., Ltd. China). The temperature was initially set to 10°C and gradually decreased to 4°C at a rate of 10°C/h and then remained constant at 4°C for 10 h. The temperature was further decreased to -3°C at a rate of 2°C/h. The fourth and fifth leaves from the top were sampled after 0, 1.5, 3, 4.5, 6 and 7.5 h of the cold treatment at -3°C and used as the materials for ultrastructure observation. The treated plants were thawed under room temperature. Leaf browning was observed after 12 h. The leaf with more than 50% browning in total leaf area was considered as cold-damaged browning leaf.
TEM Observation
TEM observation was performed as previously described (Borgers et al., 1983) with minor modifications. Briefly, leaf tissue from one third of the top section of the harvested leaves was cut into pieces (1 mm × 2 mm ) and prefixed in 3% (v/v) glutaraldehyde in 2% (w/v) potassium antimonite solution containing 0.1 M phosphate-buffered saline (PBS, pH 7.6) at 4°C for 24 h. Then the prefixed leaf samples were washed four times, 30 min each, with 2% (w/v) potassium antimonite (0.1 M PBS, pH 7.6) and postfixed in 1% (w/v) osmium tetroxide containing 2% (w/v) antimonite and 0.1 M PBS (pH 7.6) at 4°C for 4 h. The postfixed samples were washed four times, 30 min each, with double distilled water (pH 6.0) and then two times, 30 min each, with double distilled water (pH 10.0). After this, the samples were dehydrated in a gradient series of ethanol and acetone, and finally embedded in Epon 618 resin. The sections (50 nm in thickness) were obtained by using an LKB-5 ultra-microtome (LKB, Bromma, Sweden), stained with uranyl acetate, and observed in a JEM 1010 TEM (JEOL,Tokyo, Japan) at 80 kV.
In order to confirm that the presence of Ca2+ deposits, chelation of calcium ions with ethylenediamine tetraacetic acid (EDTA) was performed. The mounted tissue sections with the presence of potassium antimonite previously confirmed by TEM were immersed in 100 mM EDTA (pH 8.0) and incubated at 37°C for 1.5 h. After incubation, the grids were rinsed briefly with distilled water and stained with uranyl acetate. EDTA chelates with Ca2+ resulted in the removal of Ca2+. The disappearance of deposited Ca2+ was examined under TEM.
Results
Loquat Leaves Damaged by Cold Treatment
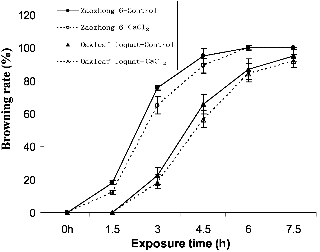
The wild accession oakleaf loquat was more tolerant than the commercial loquat cultivar Zaozhong 6 when exposed to the cold treatment at -3°C (Fig. 1). The leaves of the wild loquat accession remained intact after 1.5 h of the cold treatment, while those of the Zaozhong 6 were damaged by 12-18%, depending on the calcium treatment. Similarly, the nil-calcium control Zaozhong 6 plants had a frost injury index of 95.23% at 4.5 h after the cold treatment, as compared to 65.87% for oakleaf loquat.
The CaCl2 pre-treatment improved cold tolerance in both Zaozhong 6 and the wild oakleaf loquat. After 3 h, the CaCl2 treatment reduced the leaf browning rate by 9% in Zaozhong 6 and by 4% in the wild oakleaf loquat when compared with the untreated control. These results indicated that the CaCl2 treatment increased the cold tolerance of both accessions.
The treatment duration also had significant impact on the browning rate. The leaf browning rate increased as the treatment time progressed. For example, the leaf browning rate of Zaozhong 6 was 18.23%, 75.67%, 95.25% and 100% at 1.5, 3, 4.5 and 6 h after cold treatment. All leaves of Zaozhong 6 were damaged after 6 h of cold treatment, irrespective to the CaCl2 pre-treatment.
Subcellular Calcium Localization
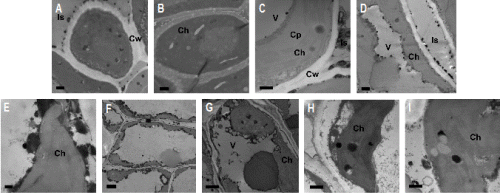
The free Ca2+ in plant cells reacted with antimonite and resulted in the formation of a calcium antimonate precipitate. The precipitate was present in black granules under TEM, indicating the presence of free Ca2+, which was clearly seen in the leaf cells of oakleaf loquat (Fig. 2A). After the EDTA treatment, the black granules disappeared, leaving a transient part on the same area (Fig. 2B). This was due to the chelating reaction of EDTA with Ca2+, resulting in the removal of Ca2+. This procedure verified the presence of free Ca2+ in loquat cells.
Changes in Cellular Ca2+ Level by CaCl2 Treatment
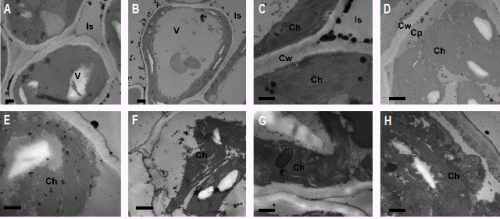
The calcium levels in the leaf cells of loquat were increased by foliar CaCl2 application, as clearly shown in the leaf cells of Zaozhong 6 loquat plants (Fig. 3B) compared to the nil-calcium control (Fig. 3A). As shown in the Fig. 3A, only a few black granules were observed in the intracellular space and the vacuole of the untreated control, while more black granules were observed in the CaCl2-treated cells, especially in the intracellular space (Fig. 3B).
|
Fig. 1. Browning rate (%) of the loquat leaves cold-stressed at -3°C, as determined 12 h after incubation at room temperature. Bars represent standard error of the mean. |
Changes in the Chloroplast Ultrastructure and Ca2+ Distribution by Cold Treatment
In the absence of cold stress, the stroma lamella and granal thylakoids of the chloroplast in Zaozhong 6 were clearly visible. The grana lamella was short while the starch granules were relatively large (Figs. 3B and 3C). The cells in oakleaf loquat were relatively larger with a compacted arrangement. They were rich in cell inclusion. The additional calcium precipitated in vacuoles and intracellular spaces (Fig. 3B). The cold treatment (-3°C) changed the chloroplast ultrastructure and Ca2+ distribution in Zaozhong 6, depending on the treatment durations. After 1.5 h of the cold treatment, the chloroplast ultrastructure of Zaozhong 6 was not affected and there was little change in calcium precipitates in the chloroplast (Fig. 3D). However, calcium precipitates in the cytoplasm around the chloroplast membrane increased, with about 10 black spots counted after 1.5 h cold treatment (Fig. 3D) as compared to virtually no calcium precipitates prior to the cold exposure (Fig. 3C). After 3 h of exposure, the chloroplast membrane was seriously damaged with abundant precipitates inside the chloroplast. There were about 40 black spots counted (Fig. 3E) as compared to only about 7 black spots after 1.5 h exposure (Fig. 3D), indicating the movement of calcium from the cytoplasm into the chloroplast. After 4.5 h of exposure, chloroplast membrane almost disappeared, causing leakage of stroma thylakoid and the aggregation of calcium precipitates (Fig. 3F). A clear void appeared between poorly structured grana lamella, and the calcium precipitates aggregated when the exposure time progressed to 6 h, which was indicated by the presence of hollow circles (Fig. 3G). Vesicles started to appear inside the chloroplast and calcium accumulation continued when the exposure time reached to 7.5 h (Fig. 3H).
As for the oakleaf loquat, there were only a few calcium precipitates which were evenly distributed in the chloroplast, with almost no precipitates found in the cytoplasm (Fig. 2C) prior to the cold treatment. No obvious changes occurred in chloroplast ultrastructure although there was slight increase in calcium precipitates at the 1.5 h exposure time (Fig. 2D). Meanwhile, calcium was found to have accumulated around cell wall (16 black spots) and plasma membrane (39 smaller black spots). At the 3 h exposure time, the chloroplast ultrastructure remained intact, while a large area with black granules and some calcium precipitates around the edge of the chloroplast and plasma membrane were observed, indicating the accumulation of calcium (Figs. 2E and 2F). After 4.5 h treatment, no obvious effect on chloroplast was observed and only limited calcium precipitates (about 3 small light-coloured black spots) was found inside the chloroplast, but the calcium precipitates aggregated in hollow circles around the chloroplast (Fig. 2G). After 6 h treatment, the thylakoid structure still remained intact and clear but chloroplast membrane became blurred. Large calcium precipitates (4 black spots) appeared (Fig. 2H). After 7.5 h exposure, the chloroplast membrane was hardly distinguished but the internal structure of chloroplast was still clearly defined. At this stage, the enlarged calcium precipitate granules (black spots) and starch grains (white spots) were observed (Fig. 2I).
Discussion
The leaf injury on loquat leaves increased with increasing exposure time under cold stress at -3°C. CaCl2 treatment reduced the leaf injury on both accessions. Similar results were observed in other plants (Chen and Liu, 2001; Raese, 1996; You et al., 2001). Raese (1996) reported that calcium foliar application increased calcium concentrations in leaves and fruits of apple (Malus demestica) and pear (Pyrus communis L.), resulting in improved cold hardiness and yield of the fruit trees.
The cold damage also varied with loquat accessions. Oakleaf loquat demonstrated higher cold-tolerance than Zaozhong 6. Qu and Li (2006) also found that different pear varieties differed in their tolerance to cold stress. The differences in cold injury between cultivars and species of fruit trees have been frequently reported (Rodrigo, 2000). Calcium application could alleviate the cold damage, which could be due to the improved osmotic stress through the signal transduction process (Liu et al., 2010). Chloroplasts are believed to be the most sensitive cell organelles to low temperatures (Kimball and Salisbury, 1973). The present study also showed that calcium application could reduce chloroplast damage during the initial phase of cold treatment. However, further increase in cold treatment duration would eventually result in the collapse of the chloroplasts even though loquat plants responded with the accumulation of calcium around chloroplasts.
TEM observations on two loquat accessions treated with CaCl2 indicated that free Ca2+ level in loquat leaves were very low under normal temperatures. However, under cold stress, Ca2+ level in cytoplasm was significantly increased, followed by the accumulation of Ca2+ around the chloroplast membrane. Similar changes in subcellular Ca2+ localization have also been observed in maize mesophyll cells under drought stress (Ma et al., 2011). The changes in cell Ca2+ distribution could be related to the signal transduction associated with Ca2+. It has been well documented that Ca2+ plays an important role in signal transduction in response to environmental changes (Chen and Liu, 2001; Kim et al., 2009; Shao et al., 2008). Liu et al. (2010) documented that Ca2+ participated in the signal transduction process of osmotic stress in maize. Generally, plant maintains a low level of Ca2+ in cytoplasm by regulating membrane Ca2+ transport and the intracellular Ca2+ pools (Song and Fu, 1997). Under abiotic stresses, Ca2+ was released into cytoplasm from cell walls and vacuoles through Ca2+ channel, the activation of Ca2+ pools, the reduction of Ca2+ pump functions and the Na+-Ca2+ exchange system (Liu et al., 2010; Saunders and Hepler, 1981; Williamson and Ashley, 1982). Our observation also indicated that Ca2+ level rose accordingly with increasing exposure time to the cold stress, resulting in the accumulation of Ca2+ in cytoplasm. Further increase in exposure time led to the collapse of chloroplast despite of the responsive accumulation of Ca2+ around the chloroplast membrane. The calcium ions were then transported into chloroplast through inward Ca2+ channels and Ca2+ transport protein (Wang and Zhang, 2002).
Low temperatures have significant impacts on the ultra-structures of chloroplasts (Kimball and Salisbury, 1973). As observed in our experiments, the cold damages on the chloroplast structure such as membrane, stroma lamella and granal thylakoids were more severe in Zaozhong 6 than in wild oakleaf loquat at each respective cold treatment duration. It was also found the Ca2+ level was lower in oakleaf loquat than in Zaozhong 6 and the calcium accumulation after cold treatment occurred much earlier in Zaozhong 6 than in wild oakleaf loquat. These results suggest that the cold sensitive cultivar Zaozhong 6 had a quicker response to the cold stress by the rapid accumulation of Ca2+, as compared to the cold-tolerant wild oakleaf loquat.
In conclusion, calcium foliar application improved cold tolerance in both loquat accessions. Loquat plants accumulated calcium around chloroplasts in response to cold stress and such accumulation of calcium occurred earlier in the cold sensitive cultivar Zaozhong 6 than in the cold-tolerant wild oakleaf loquat. In addition, cold damage symptoms to chloroplasts also appeared earlier in Zaozhong 6 than in oakleaf loquat. These results suggest that the two loquat species differed in cold tolerance and calcium accumulation dynamics.