Introduction
Materials and Methods
Plant Material and in Vitro Cultures
Rationing of Mineral Salts
Measurement of Shoot Growth and Plant Appearance
Quantification of Caffeine and Catechins
Quantitative Analysis of Amino Acids from Tea Extracts
Statistical Analysis
Results
Effect of Mineral Salts on Shoot Growth and Rooting of Tea Tree
Tea Plant and Leaf Morphology after Growth in Culture Media with Different Mineral Salts
Effect of Mineral Salts on Multiple Shoot Induction
Variations in Tea Extract Caffeine and Catechins Due to Minerals in Media
Variation of Tea Extract - Amino Acids Due to Different Minerals
Discussion
Introduction
Tea tree (Camellia sinensis L.) belongs to the family Theaceae. Tea extract is the oldest non- alcoholic beverage being consumed throughout the world. Chinese were among the first to use tea leaf extract as medicinal drink, later as beverage since 3,000 years (Eden, 1958). Tea tree is one of the most important plantation crops in the world. Tea leaves have more than 700 diverse chemical constituents including flavonoids, amino acids, vitamins (C, E, and K), caffeine, catechins and polysaccharides which are significant to human health.
Growth of plant and production of secondary metabolites are important aspects in tea tree cultivation. Nutrients affect plant growth and survival (Albert and Kinzel, 1973; Chae et al., 2006). Among nutrients, 17 of them influence tea plant growth and secondary metabolites production. Especially, the presence and variations in the amount of inorganic salts in the form of macro-elements and micro- elements are known to affect the productivity of plants and the content of useful substances (Orians et al., 2003; Park, 1995). Elevating the concentration of mineral salts considerably stimulated growth of the shoots, whereas the content of the sterol glycosides in their leaves decreased by about order of magnitude (Bondarev et al., 2003). The research so far has been conducted on determining the effect of total composition on plant growth and metabolite biosynthesis, however results directed towards establishing the contribution of each mineral salt on plant growth and metabolites biosynthesis have not been reported.
In vitro plant cultivation approach provides an advantage of controlling the environmental factors with ease. The information obtained with in vitro examination, one can grasp the conditions suitable for the in vivo cultivation of the plant. Therefore the so called “in vitro monitoring” of tea tree can aid in determination of conditions for optimal field cultivations. Nutrient media for plant tissue culture are designed to allow plant tissues to be maintained in a totally artificial environment. In vitro culture allows monitoring of the plant responses under varied biochemical and physiological conditions (Shibli et al., 2000). Examining the effects of plant growth and metabolites generation has not been carried out for tea under in vitro conditions. The purpose of this study was to assess the influence of different mineral salts on shoot growth and secondary metabolites production in cultured tea trees.
Materials and Methods
Plant Material and in Vitro Cultures
Three year old in vitro cultured tea trees (Camellia sinensis L.) from Institute of Hadong Green Tea, Hadong, South Korea were selected. Explant was a homogeneous clone selected from tea plants. The explants were cultured in Petri dishes (9 cm) containing 20 mL of 1/2 MS (Murashige and Skoog, 1962) supplemented with 1.44 mol of BAP (Duchefa, Haarlem, The Netherlands), sucrose (3% w/v), and gelite (0.38% w/v) (Duchefa, Haarlem, The Netherlands). Plantlets were then subcultured on 1/2 MS medium without plant growth regulators. All plates were incubated in a growth chamber fitted with a cool fluorescent light emitting 25 μmol・m-2・s-1 of photosynthetically active radiation (PAR) at 25 ± 1°C during 12 weeks. Each experiment involved a total of 5 plates per treatment and as triplicates.
Rationing of Mineral Salts
Individual shoots were cut from the multiple shoot clusters and allowed to proliferate on the basal 1/2 S solid medium without any plant growth regulators for 12 weeks. The effects of mineral salts on shoot growth and metabolites biosynthesis was determined by culture of the plantlets on 1/2 MS medium without each mineral salt. The medium for the in vitro culture consisted of macronutrients as in 1/2 MS (NH4NO3, 2.0625 g・L-1; KNO3, 2.375 g・L-1; KH2PO4, 0.2125 g・L-1; MgSO47H2O, 9.25 g・L-1; CaCl22H2O, 5.5 g・L-1; KI, 0.0415 g・L-1 and FeSO47H2O, 0.139 g・L-1). The experimental media were prepared as follows: M1, control (1/2 MS); M2, without NH4+; M3, without NO3-; M4, without H2PO4-; M5, without K+; M6, without Ca2+; M7, without Mg2+; and M8, without Fe3+. Each of the resulting media was supplemented with full-strength vitamins, sucrose (30 g・L-1), and gelrite (4.0 g・L-1). The pH of the medium was adjusted to 5.5 with 1N NaOH before autoclaving at 121°C for 15 min. All cultures were maintained under a 16 h light / 8 h dark photoperiod, in a growth chamber fitted with a cool fluorescent light emitting 25 μmol・m-2・s-1 of photosynthetically active radiation (PAR).
Measurement of Shoot Growth and Plant Appearance
The shoot growth of cultured plant was measured after 4 weeks of culture. The growth rate tea shoot was represented as growth index (GI) as calculated using the following equation;
GI = (Final shoot length – Inoculated shoot length) / Inoculated shoot length
The leaf browning and necrosis were measured visually for 12 weeks with two weeks make observations of the tea tree. The appearance of plant, browning and necrosis of leaves were scored visually after 12 weeks of growth. The measurements were undertaken for three times.
Quantification of Caffeine and Catechins
The quantitative analysis of catechins was validated by HPLC analysis. The samples of tea extracts were subjected to HPLC as described previously (Kim et al., 2010). Quantitative analysis of catechins was achieved by co-chromatogram of the standards and samples and by comparison of the retention times. The samples for HPLC were selected on the basis of the primary screening of tea trees through colorimetric method. Quantification was repeated for a minimum of three times.
Quantitative Analysis of Amino Acids from Tea Extracts
To estimate free amino acids found in tea extracts, the leaves (FW, 100 mg) were homogenized and extracted with 10 mL of trichroloacetic on a hot water bath (ANALAB KSB-201) at 30°C for 2 h. Extracts were clarified by centrifugation at 4,500 rpm for 15 min and filtered through 0.45 μm CA membrane (MFS-25, Advantec, MFS, Inc., CA, USA). Amino acid analysis of samples was conducted as described pre-viously (Kato et al., 2003). The results were expressed as in Millenium 32 program.
Statistical Analysis
The experiments were conducted for a minimum of three to five times with repetitive results. Duncan’s multiple range test (DMRT) was used to analyze the variations. Values were represented as mean ± standard deviation (SD).
Results
Effect of Mineral Salts on Shoot Growth and Rooting of Tea Tree
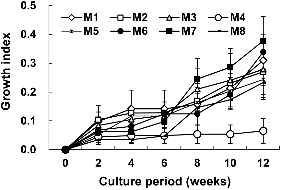
Mineral salts had dramatic effects on tea plant growth (Fig. 1). The growth rate was high when plant was cultured on M7 (Mg2+, 0.38 ± 0.17 cm) less medium while the plantlets cultured on M4 (H2PO4-, 0.07 ± 0.08 cm) free medium showed poor shoot growth. This observation indicated that H2PO4-has a positive effect on tea cultures while Mg2+ does not influence the same. Growth pattern of tea plant was similar in response to every treatment over 12 weeks.
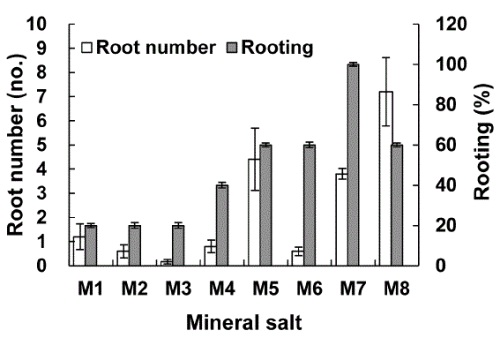
Root induction from tea plantlets was dependent on type of mineral salts (Fig. 2). Removal of M7 (Mg2+, 100%)from medium is beneficial for root formation in tea tree plants. However, M2 (NH4+, 20%) and M3 (NO3-, 20%) less media had varying influence on rooting. In addition, removal of M4 (H2PO4-, 40%), M5 (K+, 60%), M6 (Ca2+, 60%) and M8 (Fe3+, 60%) from culture media also enhanced rooting of tea trees.
The root formation was highly dependent on the presence of particular minerals (Fig. 2). K+, Ca2+, and Fe3+ removed media showed enhanced rooting to the extent of 60%. However, the removal of NH4+ and NO3- from medium lead to decreased rooting.
Tea Plant and Leaf Morphology after Growth in Culture Media with Different Mineral Salts
The number of leaves were less on tea plants cultured in medium without H2PO4. The leaves in tea plant were about 4.8 and 0.6 respectively after their culture in K+ and H2PO4- free media (Table 1). Leaf morphology also varied depending upon each mineral treatment. Generally browning of leaf was observed with passage of time and mineral salts removed treatments much affected. Leaves of plants cultured in Ca2+ and Fe3+ free media exhibited high browning. Also, leaf necrosis was high in H2PO4- and Ca2+ free media (Figs. 3D and 3F). However leaf browning in control plant was minimal.
Effect of Mineral Salts on Multiple Shoot Induction
The mineral salt composition of culture medium determined multiple shoot formation (Table 2). Multiple shoot formation was high in H2PO4,NH4+ and NO3- less media. NH4+ removal treatment resulted in about 2.4 multiple shoots. However, multiple shoots were not induced in Mg2+ and Fe3+ removed culture medium.
Variations in Tea Extract Caffeine and Catechins Due to Minerals in Media
The changes in caffeine and catechins were analyzed after 12 weeks of cultivation. There appeared many changes in the metabolites like caffeine and catechins (EC, EGCG, EGC, and ECG) depending on the presence or absence of minerals in media. There was a large difference in metabolites depending upon the existence of specific minerals.
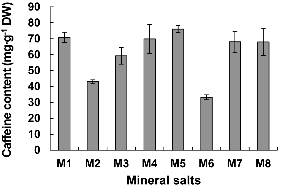
The concentration of caffeine was less in tea plants cultured in all other media except in medium deprived of K+ (Fig. 4). Especially, caffeine production was low in plants cultured on the NH4+ (43.08 mg・g-1 DW) and Ca2+ (33.23 mg・g-1 DW) less medium was lower by about 2.1 times than that of other treatments.
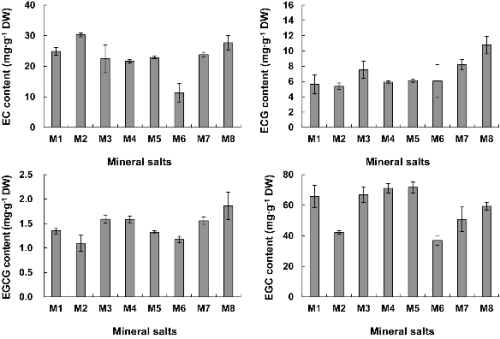
Mineral salts also affected the catechins production by in vitro cultured tea trees. Catechins production in Ca2+ and NH4+ deprived medium was lower than those of other medium conditions. However, Fe3+ absence in medium lead to enhanced catechin production.
All other medium conditions except NH4+ (30.28 mg・g-1 DW) and Fe3+ (27.69 mg・g-1 DW) absent treatments showed less EC content than the control (24.86 mg・g-1 DW). Especially, Ca2+ (11.36 mg・g-1 DW)removal from medium resulted in about 2.9 times reduced EC. Every other medium condition except NH4+ (5.37 mg・g-1 DW) devoid medium showed higher ECG. Among various mineral omissions, ECG content was high 10.78 mg・g-1 DW in Fe3+ less medium. EGCG and ECG levels were similar. Fe3+ deprivation resulted in 1.86 mg・g-1 DW. The EGC concentration of tea leaves was found to be 36.98 mg・g-1 DW after growth in Ca2+ less and on NH4+ removed medium they were lower than those of other treatments (Fig. 5).
Variation of Tea Extract - Amino Acids Due to Different Minerals
The changes in amino acids of tea extracts were analyzed after 12 weeks of culture (Table 3). A total of 22 amino acids could be detected. On the contrary to the result shown for amino acids analysis, the Ca2+ removed treatment (M6, 27.76 μg・g-1) showed 1.53 times higher total amino acid content than the control (M1, 18.2 μg・g-1). However, Fe3+ removed treatment (M8, 7.6 μg・g-1) showed a 3.65 times lower content than the Ca2+ treatment. Level of amino acid was variable based on treatments. Among the amino acids arginine was highest.
Discussion
Mineral salts composition of culture media results in enormous effects on shoot and root growth of tea tree. The growth rate of a tea tree was high when it was cultured on medium without Mg2+, while the plantlets cultured on medium without H2PO4- showed poor shoot growth. This observation indicated that both Mg2+ and H2PO4- are important factors governing tea plant growth. Magnesium (Mg2+) is one of the essential mineral nutrients for the growth and development of plants. Magnesium also acts as activator or regulator of many key enzymes in plant physiological processes (Marschner, 1995). However, both Mg2+ deficiency and oversupply have detrimental effects on plant photosynthesis (Shabala and Hariadi, 2005), consequently resulting in abnormal or restricted growth (Shaul, 2002). In our study the correlation of magnesium requirement for the shoot and root growth of tea could not be reached as previously reported.
Phosphorus is one of the three main nutrients that plants require to thrive. It functions as one of the major players in the process of photosynthesis, nutrient transport, and energy transfer (Fageria et al., 1997). A plant with the proper amount of phosphorus available to it will grow more vigorously however, while its deficiency causes stunted growth, lack of fruit or flowers, wilting and leaves may be greener or have a purple cast to them due to the photosynthetic process being affected. This study also showed that indeed phosphorous is required for good growth of tea plant.
The plant morphology in general and leaf appearance in particular varied depending upon presence of mineral salt in growth medium. The tea plants cultured in medium without H2PO4- showed less foliation. Chatuavedi (2006) reported that continuous supply of H2PO4- to the crop during crop-growth period, which is more beneficial and increased total number of tillers, dry matter accumulation and fertilizer-use efficiency and resulted in higher yields of the wheat. Mittal et al. (1978) had also reported similar findings. However, the relationship between leaf induction and H2PO4- deficiency has not been reported so far.
The observation that higher browning of tea leaf in Ca2+ and Fe3+ free medium indicates that both of these minerals are key players in tea plant metabolism. Also, H2PO4- and Ca2+ deficiency resulted in necrosis of tea plant again substantiating the important roles of these minerals for tea tree cultivation.
Multiple shoot induction was high on H2PO4,NH4+ and NO3- free media. Varying strength of nutrient components of the basal media have been shown to markedly influence the micropropagation process in many plant species (Klimaszewska and Keller, 1985; Seetharam et al., 2007). Decreased KNO3 and iron were required to improve shoot multiplication (Poothong and Reed, 2014). Mohamed et al. (1987) reported that FeSO4‐Fe had a great effect on the total amino acids content and distribution of tomato. Iron increased the translocation of proline from roots to tomato leaves.
Mineral salts composition influence metabolite production of in vitro cultured tea trees. In both Ca2+ and NH4+ free medium the catechin production was low, however, removal of Fe3+ from mediumwas beneficial in enhancement of the same. This fact indicated that these minerals; Ca2+, NH4+, and Fe3+ play critical role in catechin production in tea cultivations. Calcium can result in an enhancement of the secondary metabolite production (Sudha and Ravishankar, 2002). There is interplay of the signaling molecules also which regulates the entire pathway. Nitrogen on the soil NO3-N and NH4-N form is absorbed into plants is used in growth. The lack of nitrogen can weaken the growth of a plant and the growth of its roots (Mengel and Kirkby, 1987; Shear and Faust, 1980). Induction of secondary metabolite biosynthesis by lowering ammonium concentration was observed in ginseng saponin production by Panax quinquefolium (Zhong and Wang, 1998). Iron is also as important to health of plants. In plants, it acts as an oxygen carrier, helping form certain respirator enzyme systems in most crops. Hagendoorn et al. (1994) reported that addition of Fe3+ led to decreased lignin content and an increased cytoplasmic pH. Fe2+ stress enhances ROS production via the Fenton reaction and promotes h-thujaplicin production via ROS induced lipid peroxidation that may activate cyclic oxylipin and ethylene pathways (Zhao et al., 2005).
Mineral salts can influence on amino acid biosynthesis of in vitro cultured tea trees. About 5.58 times higher total amino acid strength was observed following removal of Ca2+ from growth medium. However, in Fe3+ removed medium the amino acid content was lower than Ca2+ removed treatment. Thus, Ca2+ and Fe3+ may also play key roles during amino acid biosynthesis by cultured tea plants. Calcium promote production of this essential amino acid (Sheldon et al., 1951). The tryptophane in the forage was increasingly higher with increasing concentration of calcium. The presence of Ca2+ suggests a time-dependent change in cell membrane properties that may require the synthesis of a protein involved in transport. The Ca2+ may be involved in the binding of such protein to the membrane or maintaining a membrane conformation that favors protein binding to maintain an intact transport system (Harrington et al., 1981). Also, iron deficiencies were associated with large increases in the free levels of the two amides asparagine and glutamine from tomato (Possingham, 1957).
This study investigated the effects of mineral salts on plant growth, its physiology and secondary metabolites generation in tea tree. Response of plant cultures to in vitro and in vivo induced stress was similar (Sawwan et al., 2000). These experimental results once again establish the important roles played by various mineral salts. Such results and observations can be used as a basic data for the proper technical development for various soil controls and design of fertilizers suitable for tea tree cultivation area in the future.