Introduction
Materials and Methods
Plant materials
Morphological analysis
Scanning electron microscopy (SEM)
Flow cytometric analysis
HPLC analysis of carotenoids
DNA extraction and SSR (simple sequence repeats), cpSSR (chloroplast SSR) analysis
Statistical Analysis
Results and Discussion
Morphology characters of leaf and fruit
Morphology of mature pollen using SEM
Ploidy determination
Identification of Carotenoids
Characterization of nuclear genome and chloroplast genome
Introduction
Citrus is the most important fruit crop based on the annual production and consumption. The fresh citrus market favors easy-peeling, seedless, and strong flavor in some areas. During the past 30 years, more than 100 new cultivars of citrus have been released through bud sport selection, crossing and other breeding pipelines in the world (Deng, 2005).
Plants chimera refers to plants whose meristems in stem tip are composed of two or more kinds of cells of different genetic types, out of which the plant develop into a complete one. In other words, two genetically different cells can coordinate to develop an entire plant, even flower and set fruits. Using the grafting technology, Winkler (1907) for the first time synthesized the interspecific chimera of tomato and nightshade. Since then, the synthesis, identification and analysis of plant chimera has been deployed in the research and application in various fields of botany (Marcotrigiano and Bernatyzky, 1995; Szymkowiak and Irish, 1999; Szymkowiak and Sussex, 1992), cytology (Chen et al., 2006), developmental biology (Wang et al., 2011), molecular biology (Bae et al., 2000), the study of the origin and development of tissues and organs (Zhu et al., 2007), and the interactions among cells and the breeding of crops (Zhou et al., 2002).
With the deepening of chimera research, more and more knowledge has been understood, as has promoted the utilization of chimera in plant breeding (Burge et al., 2002). In fact, as early as the 1920s, researchers have tried to achieve the synthesis of chimera with the purpose of helping plants to resist diseases (Jorgensen, 1927). There were also reports on the application of chimera in anti-insects breeding (Clayberg, 1975; Goffieda et al., 1990). Also, Occurrence of graft chimeras spontaneously or artificially in citrus have been reported previously (Kuhara, 1989; Ohtsu and Kuhara, 1994; Shen et al., 1998; Sugawara et al., 2002; Tanaka, 1980; Wu et al., 2004; Zhang et al., 2007).
Recently, a naturally occurring citrus graft chimera named ‘Hongrou Taoye’ (HT) with deeper red flesh color, has been discovered during screening of citrus bud mutants in Hubei province of China. It arose from the grafting junction of the scion ‘Taoye’ sweet orange (TSO) and Satsuma mandarin. TSO is a native variety in China with total soluble solids (TSS) content over 12% when ripening, and selected from the normal sweet orange and denominated in 1965. In this paper, the morphological, cytological, biochemical and molecular characters of this chimera are described.
Materials and Methods
Plant materials
The chimera HT was discovered in an orchard in Zigui county of Hubei province during the bud mutation investi-gation (Fig. 1A). It arose at the junction where a TSO bud was top-worked onto Satsuma mandarin in 1980s. It has been observed later that one branch produced fruits with their pericarp like that of TSO and flesh like that of Satsuma mandarin. Later, the buds from the branch were budded onto trifoliate orange to evaluate the stability of the traits. All the samples including the fruits and leaves were from the budded tress (designated as V1). The V1 individuals of HT and its donor individuals viz. TSO and Satsuma mandarin were maintained at the same orchard.
Morphological analysis
The length and width of 20 leaves, and the fruit width and height of both the chimera and donor plants were measured. Fruit samples consisted of ten fruits in a tree from three trees.
Scanning electron microscopy (SEM)
The FAA-fixed anthers were dehydrated using a gradient ethanol series (50, 70, and 95%) and then degreased by passing them through xylene twice for 20 min each, followed by critical point drying, mounting on copper stubs, and sputter coating with gold. The samples were examined under a scanning electron microscope (JSM-6390/LV, NTC, Japan) and representative images were obtained.
Flow cytometric analysis
Ploidy level of HT was determined by flow cytometry according to Zhang et al. (2007). To determine the standard peak of diploid cells (2C DNA), at least 10 leaves were collected respectively from TSO and Satsuma mandarin trees grown in the greenhouse. The standard peak was managed to appear at about channel 50 of relative fluorescent intensity.
HPLC analysis of carotenoids
Carotenoid pigments were analyzed by RP-HPLC using the modified binary gradient elution procedure originally developed by Lee et al. (2001). Chromatographic analysis was carried out with a Waters (Waters Corporation, Milford, Massachusetts) liquid chromatography system equipped with a model 600E solvent delivery system, a model 2996 photo diode array detection (DAD) system, a model 717 plus autosampler, and an Empower Chromatography Manager (Waters Corporation, Milford, Massachusetts). A C30 Carotenoid column (150 × 4.6 mm I.D., 3 μm) from YMC (Waters Corporation, Milford, Massachusetts) was used with MeOH–MTBE–H2O (81: 15: 4, v/v, eluent A) and MeOH–MTBE–H2O (10:90:4, v/v, eluent B) as mobile phases. Analysis was conducted under subdued light to avoid carotenoid degra-dation during analysis. The Violaxanthin and β-cryptoxanthin standards were obtained from CaroteNature (CaroteNature, Lupsingen, Switzerland).
DNA extraction and SSR (simple sequence repeats), cpSSR (chloroplast SSR) analysis
The total DNA of HT and its donor parents were extracted from the leaves by the method according to Cheng et al. (2003a).
Both SSR and cpSSR analyses were conducted according to the procedure of Cheng et al. (2003b, 2005). The PCR products were analyzed in the 6.0% (w/v) denatured polyacrylamide gels followed by silver staining according to the protocol of the technical manual on silver sequence DNA staining reagents (Promega, Madison, Wis.). Primers (TAA1, TAA15, TAA27, TAA33, TAA52) used for SSR analysis were based on those of Kijas et al. (1994). CpSSR analysis was performed with primers (SPCC1, SPCC3, SPCC9, SPCC11) synthesized according to the report of Cheng et al. (2005).
Statistical Analysis
Each treatment of all the experiments was replicated ten times. Statistical analyses were performed with SAS 9.1 (SAS Institute Inc., Cary, NC, USA). Means within a line followed by different letters are significantly different at p = 0.01 according to LSD test.
Results and Discussion
Morphology characters of leaf and fruit
The leaf shape of HT was similar to that of TSO. The shape of the spring leaves was similar to the leaves of peach and their petiole wings were not so obvious. But the leaf width of HT (4.60 cm) was broader than that of TSO (3.96 cm). So the leaf index of HT was between its donor parents (Table 1, Fig. 1B). Photographs of fruits of the chimeras and donor parents are shown in Figure 2A. TSO had short spheroid fruits which were 6.79 cm in diameter and 6.08 cm in height, and had yellow juice sacs (Fig. 2B). The aroma of its rind was strong. HT had short spheroid fruits with a diameter of 7.21 cm and a height of 6.16 cm, the aroma of which was similar to that of TSO. But the color of its juice sacs was deep orange, which was the same as that of Satsuma mandarin (Fig. 2B). HT had 11.5 seeds per fruit, which was similar to that of TSO (10.8 seeds per fruit) and different from the seedless Satsuma mandarin (Table 1, Fig. 2B). Interestingly, HT fruits are easy peeling; although not so easy as Satsuma mandarin, they are much easier to peel than TSO.
Morphology of mature pollen using SEM
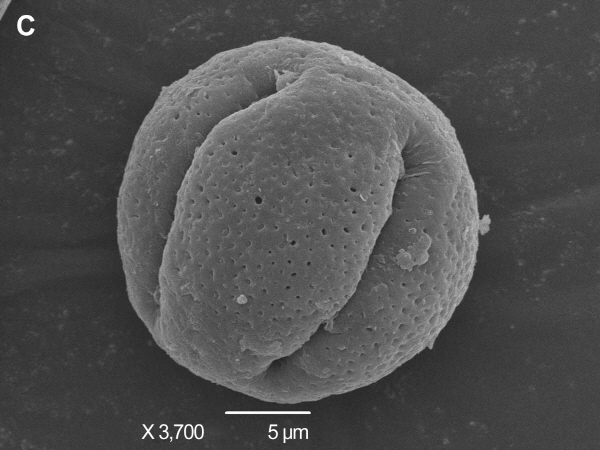
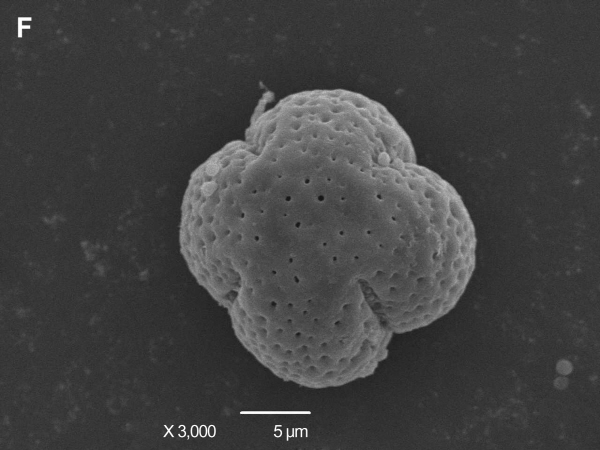
The pollens of HT and its donors were observed through scanning electron microscope. The morphology, the size and the ornamentation of exine of HT were similar to those of TSO but different from those of Satsuma mandarin. Mature pollen grains from TSO and HT were spherical and 23-25 μm in diameter. They were characterized by aperture ornamentation and 4-5 shallow colporates. There were some granules in the colporate (Fig. 3A, B, C, D). Compared with them, mature pollen grains from Satsuma mandarin were ellipse or spherical and 24-26 μm in diameter. They possess aperture ornamentation and 4 deep colporates (Fig. 3E, F).
|
Fig. 2. Fruit morphology of ‘Hongrou Taoye’ and its donor plants. From left to right: ‘Guoqing No.1’ Satsuma mandarin; ‘Hongrou Taoye’; ‘Taoye’ sweet orange (bars = 3 cm) |
|
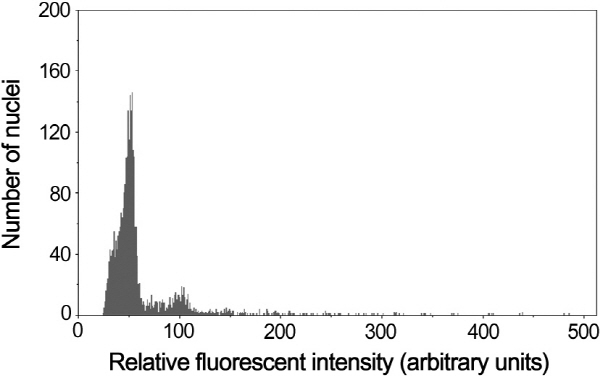
Fig. 4. Ploidy analysis of ‘Hongrou Taoye’ sweet orange and its donor plants (three samples were run together). |
Ploidy determination
Flow cytometry was used to analyze the ploidy levels of HT and its donor parents. When compared with the diploid control, TSO and Satsuma mandarin, whose fluorescence intensities were present to 50. HT demonstrated the fluorescence intensity similar to the control, indicating its diploid nature (Fig. 4).
Identification of Carotenoids
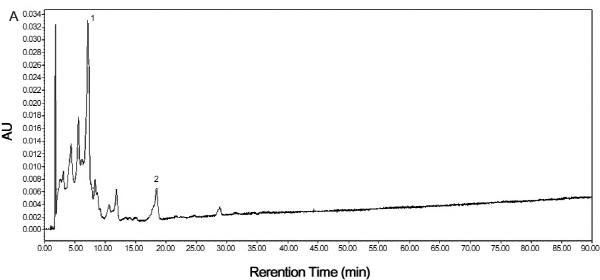
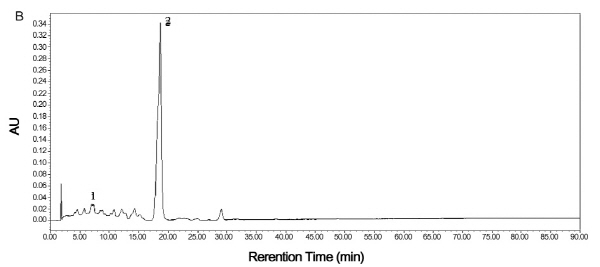
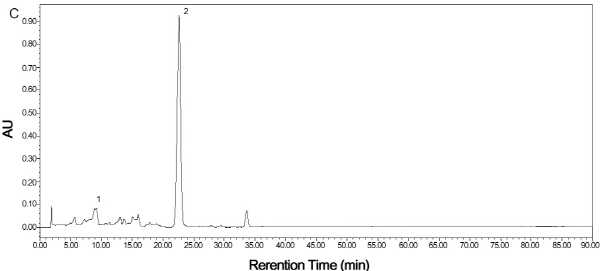
By comparing their absorption spectra and retention times with those of purchased authentic standards, mature TSO predominantly accumulated Violaxanthin isomers in the flesh (Fig. 5A). Violaxanthin is a yellowish pigment with the main cis-form in citrus having a visible absorption peaks of 413, 436 and 465 nm, while the pigment with the highest concentration in Satsuma mandarin was β-cryptoxanthin. It is an orange color carotenoid with visible absorption peaks of 479, 452 and 429 nm. Results showed that both HT and Satsuma mandarin accumulated β-cryptoxanthin predominantly in the juice sacs in the mature fruit (Fig. 5B, C).
Characterization of nuclear genome and chloroplast genome
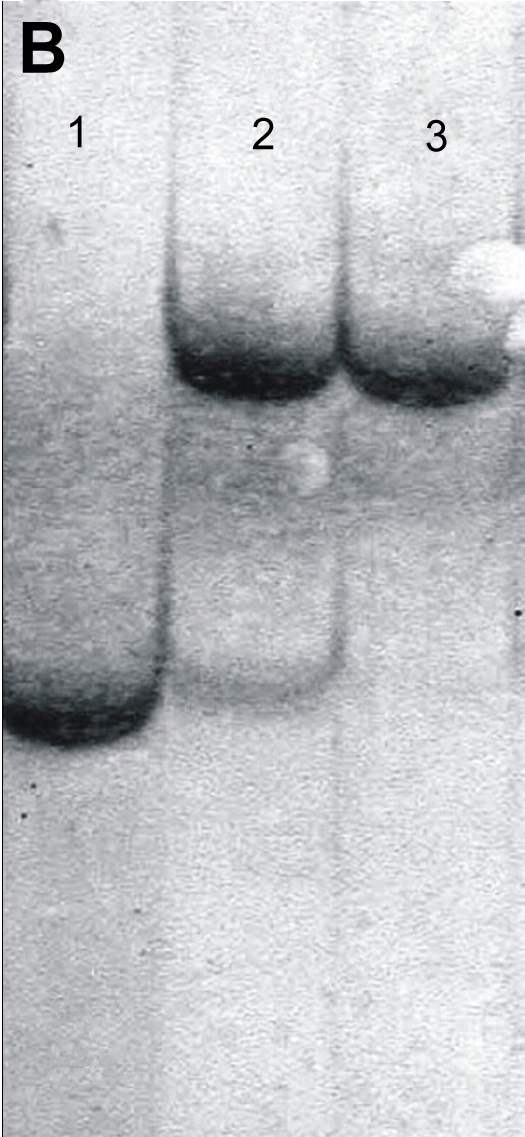
To determine the nuclear composition, SSR analysis was performed with HT and its two donor parents. One out of the five tested SSR primers, TAA15 revealed polymorphism between C. sinensis and C. unshiu. The amplified products of this primer showed that the band patterns of HT contained amplified bands from both TSO and Satsuma mandarin (Fig. 6A), which indicated that HT contained nuclear genomes from these two donors. For the chloroplast genome, HT was characterized by using four pairs of cpSSR primers (SPCC1, SPCC3, SPCC9 and SPCC11). The results revealed polymorphism between TSO and Satsuma mandarin. The graft chimera shared the same banding patterns with both TSO and Satsuma mandarin, which demonstrated that the graft chimera contained the chloroplast genomes from both donor parents (Fig. 6B). The above results show that HT is a graft generating periclinal chimera that consists of L1 from Satsuma mandarin and L2/L3 from TSO. Characters determined by L1 cell layer, such as juice sacs of the fruit were similar to those of Satsuma mandarin. While other characteristics, including petiole wing, fruit shape, the color and aroma of the rind, pollen, and seed determined by L2 cell layer, were the same as those of TSO. These were consistent with the results that juice sacs were originated from the L1 layer and gametes were usually produced by the L2 layer (Marcotrigiano and Bernatzky 1995; Zhou et al. 2002). In this study, the whole leaves were used to isolate DNA; because L1 cells of an apical meristem give rise to the epidermis of the leaf, and the cells of L2 and L3 give rise to the mesophyll (Frost and Krug 1942; Tilney-Bassett 1986), the SSR and cpSSR analyses showed that the graft chimera contained nucleic and chloroplast genomes from both donor parents.
Intercellular morphogenetic and physiological interactions and cell displacement have been observed in chimeric paints (Szymkowiak and Irish, 1999; Zhou et al. 2002). The observations reported here on leaf index (Table 1), both nuclear (Fig. 6A) and chloroplast (Fig. 6B) genome, and the aroma of flesh in this study indicated that chimeric cells likely were derived from both parental cultivars. Further studies are required to reveal the interaction between cells of Satsuma mandarin and ‘Taoye’ sweet orange in the chimera ‘Hongrou Taoye’.
The characteristics of HT have been maintained for several years and the phenotype is agronomic traits stable after vegetative propagation. Its fruit has a flavor and texture with mixture of Satsuma mandarin and TSO. The flesh is orange in color, fine and crisp in texture, and very juicy with strong aroma. The whole fruit has pleasant smell and excellent shelf life as TSO. So, HT combines the valuable traits of both donor parents. It is expected to be a commercially acceptable chimeric cultivar for the citrus industry.