Introduction
Materials and Methods
Seed Collection
Alternating Temperature Regimes
Cold Moist Stratification
Anatomy of the Seed
Statistical Analysis
Results and Discussion
Seed Characteristics by Collection Phase
Germination Characteristics by Collection Phase
Germination Percentage at Alternating Temperatures
Germination by Cold Moist Stratification
Optimum Seed Collection Period
Introduction
The genus Deutzia (Hydrangeaceae) comprises about 60 species that are native to eastern and central Asia. The plants are deciduous shrubs with opposite leaves and serrated margins, white or white-pink tipped flowers and naturally peeling bark. They are widely used for ornamental purposes in city planters and botanical gardens. Deutzia paniculata Nakai is a native species of South Korea and is found only in Gyeongsang-do (Zaikonnikova, 1966; Kim 2003; Oh et al., 2005; Chang et al., 2007). Due to its restricted distribution, this species is designated as critically endangered (Chang et al., 2001), endangered (Korea National Arboretum, 2008) and vulnerable (National Institute of Biological Resources, 2012) in the International Union for Conservation of Nature and Natural Resources (IUCN) Red list. The plants grow up to two meters in height and produce 20 to 40 white hermaphroditic flowers in a panicle inflorescence during April or May (Lee, 1980; Kim, 2003). Fruit production is abundant; however, it is rare to find seedlings below 30 cm in height in its natural habitat. Due to its apparently limited natural reproduction and limited distribution, D. paniculata Nakai is considered threatened.
Germination and emergence of D. paniculata in natural habitats is influenced by environmental factors including temperature, light, soil fertility and moisture content (Karssen, 1982; Baskin and Baskin, 1998; Qu et al., 2008; Jha et al., 2015). Among them, temperature is the major factor promoting radicle protrusion and determining the rate of germination and seedling recruitment (Walck et al., 2011). In addition, alternating temperatures help to break dormancy and promote germination in D. paniculata as well as several weed species (Vincent and Roberts, 1977) including Brachiaria humidicola (Goedert and Roverts, 1986), Ranunculus sceleratus L. (Probert, 1988), Orobanche spp. (Kebreab and Mudoch, 1999), Carex spp. (Schutz, 2000), Solanum nigrum (Kamgari, 2009) and Hepatica asiatica (Chon et al., 2015). Besides temperature, the stage of seed maturity at harvest can also influence seed germination and seed viability in seedling propagation. Therefore, the seed collection period is important and can affect seed quality including proper embryo development, germination, seedling vigor, storability and viability (Hossain et al., 2005; Olasoji et al., 2012). For optimum propagation and storage, seed collection needs to be done in correct time (McCormark, 2004). With some plants, seed collection at an early stage of seed development can result in seeds with poor vigor and lower germination capacity (Coolbear et al., 1997) and can result in a rapid loss of seed viability due to evaporation of moisture from the endosperm (Weir, 1918; Scharpf, 1970).
A few studies have been conducted on the germination of Deutzia species. D. gracilis seeds germinate in cool temperatures in the spring and were reported to take more than a year to germinate under natural conditions. D. scabra seeds were reported to germinate within 1-3 months under controlled conditions maintained at 18°C (Gabr and Sayed, 2010). To date, there is limited information about the germination and growth of seedlings of D. paniculata. Due to its high conservation value, further study is required on the optimum seed collection time for promoting maximum seed quality. Therefore, we tested different seed collection times and alternating germination temperature regimes to determine the optimum seed maturity and germination conditions for seed propagation of D. paniculata. The results of this study provide insight into propagation procedures for the rare and endemic species D. paniculata.
Materials and Methods
Seed Collection
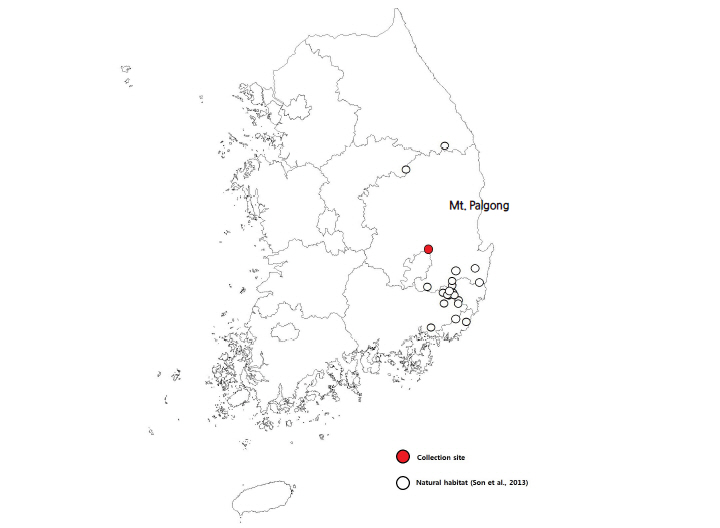
D. paniculata seeds were obtained from naturally growing populations of Mt. Palgong, Gyeonsangbuk-do, South Korea (Fig. 1). Young to mature fruits were collected every two weeks over a three month period from August 11th through October 24th, 2014, totaling five collections (collection phase I – V, Table 1). The first collection occurred when the fruits were light green in color with a high moisture content (collection phase I). The last collection was made when the fruits were dried and deep brown in color (collection phase V). The fruits were collected from randomly selected fruiting branches and fallen fruits. The seeds were surface sterilized with 0.5% NaOCl for 5 min, rinsed with running water for 20 min and air-dried at room temperature just before the experiment.
Table 1. Seed characteristics according to collection date.
| |
Seeds were collected five times in three months (collection phase I-V) and the data represent the mean and the standard deviation (SD). | |
Alternating Temperature Regimes
The sterilized seeds were placed in a petri dish (9 cm in diameter) with 8% (w/v) distilled agar medium. Each petri dish with seeds was then incubated in four different temperature regimes: 30/20°C, 25/15°C, 20/10°C, and 15/6°C day/night temperature, with a 14 h photoperiod in a Bio Multi Incubator (LH-30-8CT, NK system Co., Japan). The germination test was carried out in three replicates, each having 40 seeds distributed in a completely randomized design.
The germination percentage (GP) was calculated every day for 30 days to obtain a cumulative GP for each seed collection phase at the different temperature regimes. Seeds were considered to have germinated when at least 1.0 mm of the radicle had emerged. The mean germination time (MGT) was calculated by multiplying the number of germinated seeds on each day by the number of days from the beginning of the test and dividing by the total number of germinated seeds at the end of the experiment (Ellis and Roberts, 1981). The germination index (GI) was calculated using the formula by Kendrick and Frankland (1969) which proposed a germination index for comparisons of germination times, expressed as GI = Σ(1/t) where t is the time for each seed to germinate.
Damaged seeds were excluded from the germination assays. The germinated seeds were not removed from the petri dishes, in order to observe the vitality and vigor of the seedlings and the rate of survival was calculated.
Cold Moist Stratification
Forty seeds from each collection phase were placed on a petri dish with 8% (w/v) distilled agar medium and stratification was carried out at 4°C for 30 or 60 days in the dark. Three replicates were used for each treatment. After the desired period of cold moist stratification, the petri dishes were arranged in a growth chamber at 25/15°C or 30/20°C day/night temperature with a 14h photoperiod. The germination counts were performed for 30 days and only normal seedlings, excluding seedlings that died or showed low vigor, were counted.
Anatomy of the Seed
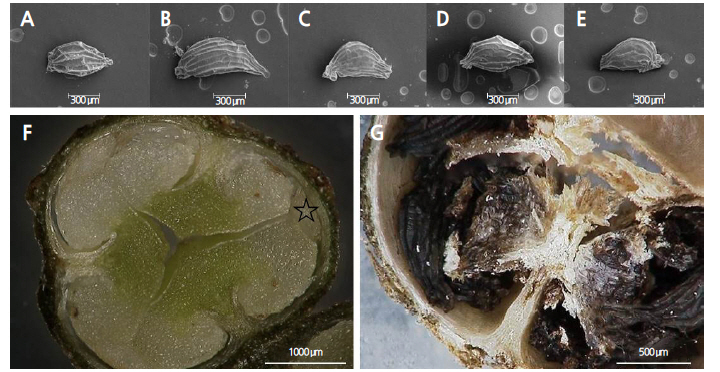
Morphological characteristics such as the length, width and weight of the seeds were measured. The size (length and width) of 30 seeds from each collection phase were measured with light microscopy (Leica MZ16FA, Leica Microsystem Co. Ltd., Germany). Seed length was measured using the longest axis from end to end and width was measured using the longest axis at a 90° angle of the length. The fresh weight of 100 seeds from each phase with five replicates was also determined.
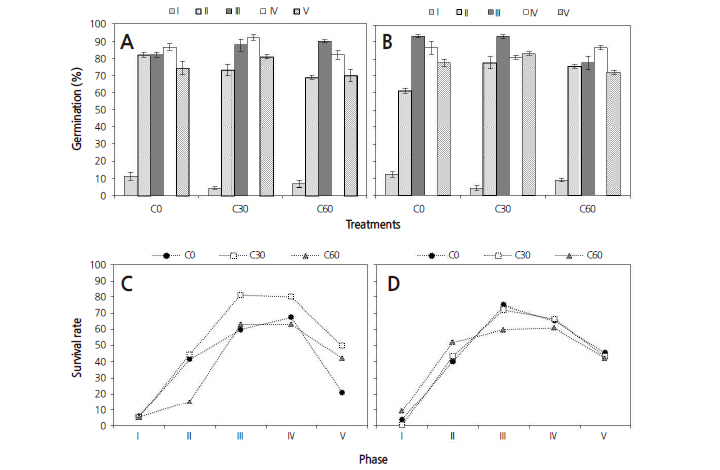
For scanning electron microscopy (SEM), the seeds were sputtered with gold coating in a KIC-IA COXEM Ion-Coater (COXEM. Co., Ltd, Korea). SEM imaging was carried out with a COXEM CX-100S scanning electron microscope (COXEM) at 20 kv. For anatomical studies, the seeds were dehydrated in an ethanol series (50, 70, 80, 90, 95, and 100%). After complete dehydration, the seeds were passed through combinations of alcohol:Technovit (3:1, 1:1, 1:3, and 100% v/v) and then embedded in Technovit 7100 resin. The embedded seed materials were then cut into serial sections of 4-6 μm thickness using a Leica RM2255 rotary microtome (Leica Microsystems GmbH, Germany) with disposable blades. The sections were stained with 0.1% Toludine blue O and mounted with Entellan (Merck, Co., Germany). The samples were observed under a light microscope (Hirox, Japan). Embryo shape and growth was shown on the longitudinal seed sections.
Statistical Analysis
Data was subjected to statistical analysis using SPSS software. Two-way analysis of variance (ANOVA) was conducted and means were compared using Tukey’s HSD mean separation at a 0.05 level of probability.
Results and Discussion
Seed Characteristics by Collection Phase
Each fruit of D. paniculata contained an average of 22-57 seeds. The average seed length ranged from 0.6-1.2 mm and average width ranged from 0.30-0.47 mm. The largest and heaviest seeds [0.95 ± 0.17 mm (length), 0.45 ± 0.09 mm (width), and 4.43 ± 0.23 mg/100 seeds (weight)] were obtained from the fruits collected during the end of August (collection phase II, Fig. 2) and had high moisture contents. Although the fruits from collection phases I and II were similar in appearance and moisture content, most of the seeds from phase I were non-viable. The fruits and seeds collected during the middle of September (collection phase III) had already begun to desiccate and appeared dry. The fruits collected on October 2nd (collection phase IV) were almost completely dry and had lost their endocarp. As the water content of the seeds diminished, the seeds decreased in length, width and weight (Table 1). As a result, the seeds from collection phase V were completely dry and weighed only 2.33 ± 0.19 mg/100 seeds (Table 1). This gradual loss of moisture may relate to dryness and the viability of the seed. Bing (2004) reported that the viability of Deutzia seeds was reduced as the seeds desiccated, thus limiting storability. Norman (1996) found similar results as the germination percentage of D. staminea dropped from 100% to 20% after three years of dry storage. Seed development at harvest are important to ensure quality in terms of germination ability and vigor (Khatun et al., 2009), as well as viability after storage.
Germination Characteristics by Collection Phase
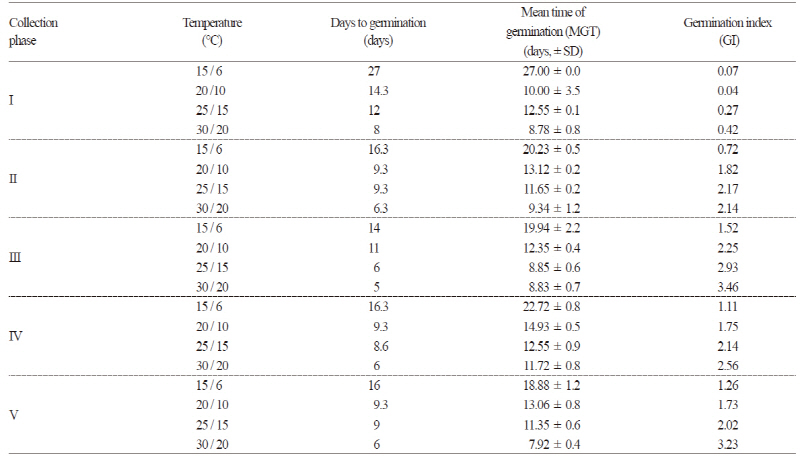
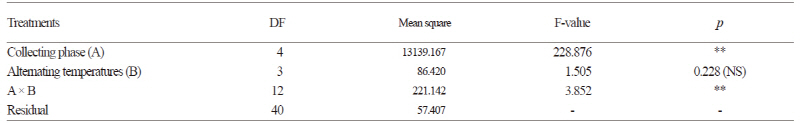
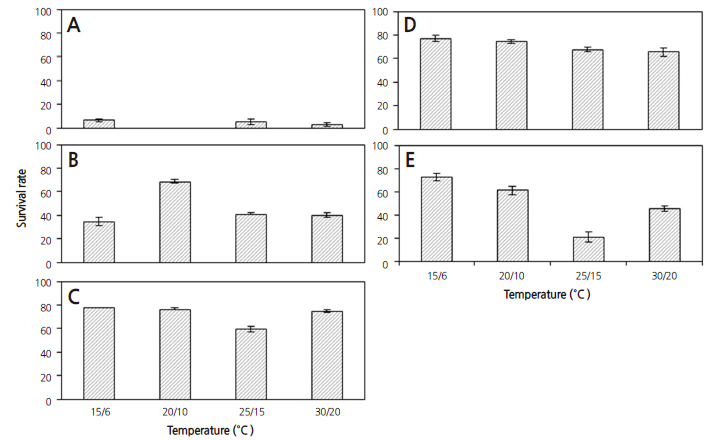
The germination characteristics of the seeds based on collection time and temperature regime were examined by determining the germination percentage (GP), germination index (GI) and mean time of germination (MGT) (Table 2 and Fig. 3). Seeds from collection phase III had an average GP of 90.5% at all four temperature regimes. The seeds from collection phase IV and V showed no significant differences among GPs; seeds from phase IV had a GP of 86.7% at the 30/20°C and 25/15°C temperature regimes and seeds from phase V had a GP of 82.2% at the 25/15°C temperature regime. Seeds from collection phase IV and V performed better than the seeds from collection phase I and II for GP and GI. The lowest germination was observed in the seeds from collection phase I with an average GP of 15.7%. The seeds from collection phase II showed a significant difference in GP, GI and MGT among the different temperature regimes. MGT was not significant by collection phase but increased with lower temperature. Overall, MGT was less than 14 days except for in the 15/6°C temperature regime. The results showed that the germination temperature and collection time play a major role in the germination of D. paniculata (p < 0.001, Table 3). The germination rates for the seeds collected in October were 70-80% regardless of temperature. Interestingly, the seeds collected in the middle of October (collection phase V) had lower germination percentages than the seeds collected in early October (collection phase IV).
Table 3. Correlation coefficients between collection phase and alternating temperature on germination of D. paniculata.
| |
***= p < 0.001; NS= non-significant (p < 0.01) | |
Germination Percentage at Alternating Temperatures
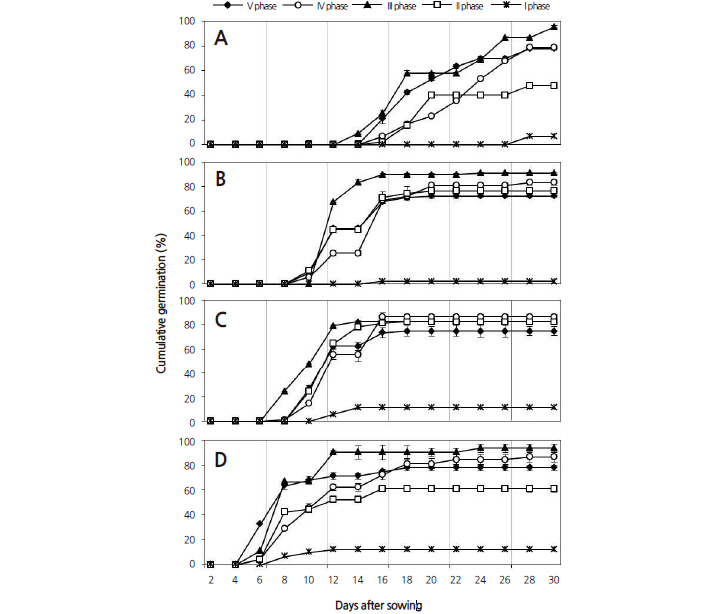
D. paniculata seeds showed a high cumulative germination percentage when incubated with alternating temperatures (Fig. 3). Radicle protrusion was faster at higher temperatures. The fastest radicle protrusion was observed on day five at the 30/20°C temperature regime in all collection times. Correspondingly, germination began on day eight at 25/15°C, day 10 at 20/10°C and day 14 at 15/6°C. The lowest mean germination time (MGT) in all collection phase was 9.3 ± 1.4 days at the 30/20°C temperature regime and MGT increased with a decrease in temperature: 11.4 ± 1.5, 12.7 ± 1.8, and 21.7 ± 3.3 days at 25/15°C, 20/10°C and 15/6°C, respectively (Table 2). The GP was similar with an average of 80.4% at the 25/15°C, 20/10°C and 30/20°C temperature regimes, except in phase I seeds. At the 30/20°C and 25/15°C temperature regimes, the rate of radicle protrusion became much slower when average GP reached 68% during 12-14 days. Similarly, this process appeared after 16 days at the 20/15°C temperature regime when GP reached 74%. However, the seeds at the 15/6°C temperature regime showed continued germination until 30 days, and as a result, average GP declined to 50% within 20 days. In all collection times, the GP of the D. paniculata seeds was high but the survival rate of the seedlings was inconsistent during the 30 day experiment. We observed that high temperatures facilitated faster germination at the beginning; however, the final germination percentage in the four temperature regimes was not significantly different. Fordham (1960) reported that seeds of Deutzia spp. had no inhibiting dormancy. Bing (2004) found that the temperature for optimum germination of D. parviflora was 25°C and the lowest germination was recorded at 15°C. The results of this study for D. paniculata are comparable to previous studies on the genus Deutzia. Nevertheless, some plants required low temperature for germination; for instance, the best germination of Deinanthe and Hydrangea were recorded at 18-21°C and 13-16°C respectively (Royal Horticultural Society, 2006).
Germination by Cold Moist Stratification
After 30 and 60 days of cold moist stratification, the seeds were germinated at 25/15°C and 30/20°C temperature regimes (Fig. 5). After 30 days of cold moist stratification, the GP of the seeds from collection phase III and IV (> 80%) was only slightly higher than the non-stratified seeds at the 25/15°C temperature regime; however when collection phase III was excluded, the GP of the seeds treated for 60 days gradually decreased. The seeds treated with cold moist stratification at the 30/20°C temperature regime had a higher GP similar to the control at the 25/15°C temperature regime. Among Hydrangeaceae, Geer and Rinegart (2010) reported that the optimal germination conditions for Hydrangea seed was in 13-16°C temperatures and with GA3 treatment. In another report it was shown that Philadelphus lewisii seeds were germinated at temperatures of 22-26°C after two to five months of moist cold stratification at 1-5°C to overcome dormancy (USDA NRCS, 2010). Gabr and Sayed (2010) reported that D. scabra seeds had the highest survival of 96.67% at 24°C and the lowest survival of 41.67% at 4°C. In this study, we found that D. paniculata seeds did not germinate at or below 5°C, suggesting that a chilling period is not required for germination (Fig. 5). These results indicated that wild D. paniculata seeds require temperatures over 15°C for germination.
Optimum Seed Collection Period
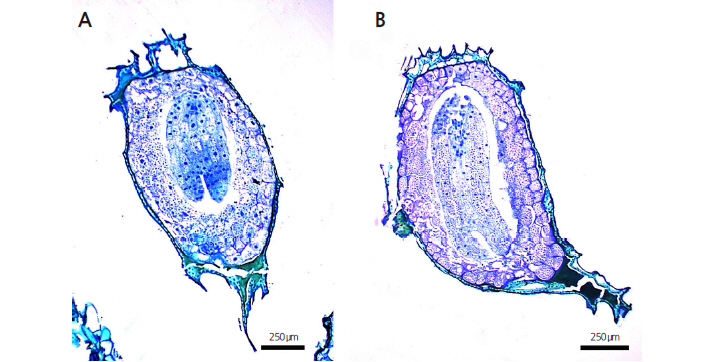
Each collection phase had a different germination percentage and the lowest germination percentage was observed in the seeds from phase I. The seeds collected in late August (phase II) were immature and the embryo was heart or torpedoshaped and approximately 269.9 μm in length. Thus, seeds of collection phase II had a low overall germination rate, though germination improved after two weeks (Fig. 6A). Seeds collected in early September (phase III) had embryos with a perfect torpedo shape, were approximately 391.2 μm in length and had a significantly higher germination rate and viability than those of earlier collections (Fig. 6B).
In some plants propagated by immature seed, for instance Geranium spp. (Geraniaceae), Trifolium spp. (Fabaceae) and Aethusa cynapium (Apiaceae), viability decreases significantly throughout the storage period (http://www.seedbiology.de/dormancy.asp). We found that immature seeds of D. paniculata collected during the early and middle of August germinated, but at a very low rate, and seeds collected during collection phase V had low viability probably due to desiccation. Kumar et al. (2002) reported that the stage of seed maturity had a large influence on the quality of the seed. On the other hand, Olasoji et al. (2012) suggested that collecting seeds of kenaf at a mature stage significantly improved the results of germination tests. We found that the optimum collection stage for D. paniculata was during the middle of September (Fig. 7). Although there were many fallen fruits (both intact and wounded) in the field, we did not find young seedlings throughout the field survey period. Germination is affected by temperature and moisture. The fruit in late October, that mostly fallen, had desiccated. Therefore, it is possible that seedlings were not found in the field due to factors related to temperature and moisture. We also collected fallen fruit, finding that seeds from wounded fruit showed little germination and that the seeds from intact fallen fruit did not germinate at all (data not shown). Based on these results, we suggest that D. paniculata seeds collected during late autumn (≤ 10°C) have a lower germination capacity than seeds collected during early autumn. This study provides indirect implications on the collecting strategy and life cycle for propagation of D. paniculata.