Introduction
Materials and Methods
Experimental Conditions
Management of Nutrient Solutions
Irrigation Based on Solar Radiation
Analyses of Plant Growth and Ion Composition in Plants
Estimation of Water and Nutrient Uptake
Experimental Management and Statistical Analysis
Results and Discussion
Changes in Ion Concentrations in the Recycled Nutrient Solution and Root Media
Changes in Nutrient Uptake Concentrations
Changes in Ion Ratios in the Recycled Nutrient Solution and Root Media
Growth and Yield of Paprika Plants
Mineral Contents in Leaf and Fruit
Introduction
Due to limited water resource, increased fertilizer price, and environmental pollution, the importance of closed soilless cultures systems is steadily increasing (Holmer et al., 2013). Nevertheless, applications of the system to commercial soilless cultures are still scarce with technical difficulties (Massa et al., 2011) including ion imbalance in the recycled nutrient solution (Hao and Papadopoulos, 2002; Zekki et al., 1996). To maintain ion balances in both recycled nutrient solutions and root media, an adequate management of nutrient solutions is required (Ahn and Son, 2011; Hao and Papadopoulos, 2002; Zekki et al., 1996).
Although a real time measurement by ion sensors is an ideal approach for accurate nutrient control, stable operations are not still guaranteed on commercial basis (Bratov et al., 2010). Instead, electrical conductivity (EC)- based nutrient control is widely used in commercial closed soilless cultures (Savvas, 2002). To minimize the change in ion balance in the EC-based system, consumed nutrient solution was normally replenished by tap water and stock solutions manually or by an automated system (Savvas and Manos, 1999). Subsequently the periodical adjustment of recycled nutrient solutions was introduced for more efficient management of nutrient solutions, for instance, every two weeks. (Savvas, 2002). However frequent analysis acts as a drawback in commercial closed soilless cultures and therefore adequate intervals should be reviewed.
For the adjusting interval of recycled nutrient solutions, Ko et al. (2013a, 2013b) investigated the growth of paprika under different renewal periods with the constant interval. However, for more practical operation of closed soilless culture systems, extended adjusting intervals are more favorable. As nutrient uptake rate increases with growth stage (Pardossi et al., 2005; Silberbush et al., 2005), adjusting intervals can be flexibly designed with variable renewal patterns considering the stage. The aim of this study was to investigate the ion balances in recycled nutrient solutions and root media at different renewal patterns, and their subsequent effects on uptakes of water and nutrients by paprika for seeking possible ways to extend the adjusting periods.
Materials and Methods
Experimental Conditions
This experiment was conducted in a Venlo-type greenhouse at the experimental farm of Seoul National University (Suwon, Korea, Latitude 37.3°N, Longitude 127.0°E). Paprika (Capsicum annuum L. cv. Fiesta) seedlings were planted on the rockwool cube (10 cm × 10 cm × 6.5 cm) at the beginning of April, 2012. One month after planting on rockwool cubes, plants were transplanted in rockwool slabs (100 cm × 15 cm × 7 cm) (Grotop expert, Grodan, The Netherlands) at a density of 3 plants/m2. The experiment started 30 days after transplanting (DAT) when average plant height was 90 cm at fruit initiation. Average radiation ranged from 2.6 to 5.5 MJ・m-2・d-1 and average temperature was controlled at 24.6 to 31.6°C by hot-water heating systems inside the greenhouse. All the plants were supplied with the nutrient solution of EC 2.5 dS・m-1 and pH 5.5 to 6.5. Nine slabs were used for the experiment. Each slab, in which three plants were grown, was placed on each gutter (100 cm × 20 cm × 10 cm). Additional plant management of pruning and training was carried out every week. Different renewal patterns of the recycled nutrient solutions were applied: 4-4-4 weeks (constant renewal at week 4, 8, and 12) as a control, 6-4-2 and 8-2-2 weeks (different renewals at week 6, 10, and 12 and at week 8, 10, and 12, respectively) as treatments were compared.
Management of Nutrient Solutions
Compositions of the nutrient solution was based on the standard of Research Station for Floriculture and Glasshouse Vegetables (PBG, the Netherlands): 14.17 NO3-, 1.14 H2PO4-, 5.92 K+, 8.85 Ca2+, 3.17 Mg2+, and 3.20 SO42- (in meq・L-1) as macro elements; and 1.05 Fe2+, 0.64 Zn2+, 0.09 Cu2+, 0.57 Mn2+, and 0.08 MoO42- (in ppm) as micro elements. After irrigation event, the drainage was returned to the reservoir tank (52 cm × 26 cm × 26 cm). EC and pH in the reservoir tanks were monitored every three days by using a multi meter (Multi 3420 SET C, WTW, Germany). EC and water content in the root media were measured by using a TDR sensor (Grodan, WCM-control, Denmark). To maintain the set EC of 2.5 dS・m-1, fresh water (EC 0.17 dS m-1 and pH 7.11) containing 0.21 Na+, 0.29 Cl-, 0.04 K+, 0.71 Ca2+, 0.21 Mg2+, 0.19 SO42-, 0.39 NO3-, and 0.04 PO43- (in meq・L-1) was added and then fresh nutrient solution added to reach the fixed volume of nutrient solution (20 L) in the tank. Total nine tanks were used for the experiment. To adjust the pH, 1N nitric acid was used.
Irrigation Based on Solar Radiation
The nutrient solution was supplied by drip irrigation with one emitter (average flow rate of 3 L・h-1) per plant based on solar radiation integral (SRI). Solar radiation (W・m-2) inside the greenhouse was measured every 10 s by a pyranometer (SQ-110-L10, Apogee, USA) at a height of 2 m from the ground and recorded at was connected to a data-logger (CR-1000, Campbell Scientific, USA). Whenever inside greenhouse SRI reached the set value, irrigation was started. Drainage was controlled within 30 to 50%. SRI was set 50 J・cm-2 and the irrigation lasted 3 to 4 min per event (150-200 mL/dripper) depending on the percentage of drainage amount. The irrigation frequency was checked every day online and was 8 to 10 times per day in average. During rainy day, the nutrient solution was manually supplied.
Analyses of Plant Growth and Ion Composition in Plants
Plant height and leaf area were measured from the start (30 DAT) and to the end (115 DAT) of the experiment. Leaf area was calculated by using leaf length and width according to Tai et al. (2009). Fully-matured leaves at the 4th and 5th nodes from the top of the stem were collected at 115 DAT to measure the nutrient uptake by the plants. 9 leaves were sampled in each treatment. Leaves were washed with tap water to remove dust or other contamination and dried for 48 h at 70°C. Fruits were harvested two times at 87 DAT (8 weeks after treatment) and 115 DAT (at the end of experiment). At each harvest, fruit number, fruit fresh and dry weights were measured. For the analysis of fruit mineral content, the matured fruit (three fruits per treatment) at the end of experiment were dried in a thermo-ventilated oven at 70°C until they reached a constant weight. The dried leaf and fruit samples were grinded by boll-mil. The 0.5 g of grinded sample was placed into a teflon tube and 10 mL of concentrated nitric acid was added to each tube. For rapid processing, the tubes were placed into blacks and heated to 90°C on a hotplate. The samples were heated at 90°C for about 1 h or until copious fume evolution subsides. The 1 mL of concentrated perchloric acid was added and the solution temperature was set to maintain at 180°C. The samples were digested until the denseness of the white fumes and continually heated to a clear solution. After the digest, the tube was cooled and 25 mL of deionized water was filled up. The nutrient contents of K, Ca, Mg, P, and S were determined by an inductively-coupled plasmaoptical emission spectrometer (ICP-730ES, Varian Australia Pty Ltd., Australia). Total-N was measured by Kjeldahl (Kjeltec 8400, Foss, Sweden).
Estimation of Water and Nutrient Uptake
The water uptake (L・m-2) by the plants was estimated using the difference between initial and final volumes in each reservoir tank (20 L) and the changes in the water contents of root media every two weeks. The evaporation or leakage from the tank was prevented and the evaporation from the rockwool cube was not considered. Water contents in the root media were 60%-70% when measured. Every two weeks, 50 mL of nutrient solutions from the reservoir tanks and root media were collected and analyzed. NO3-, H2PO4-, Cl-, and SO42- were measured by a spectrophotometer (PhotoLab, 6100 [VIS], Germany) with an anion test kit (Spectroquant, Merck KGaA, Darmstadt, Germany), and K+, Ca2+, Mg2+, and Na+ were measured by the ICP-730ES.
Ion in recycled nutrient solution (meq・L-1) = (CrVr + CwVw + CfVf) / (Vr + Vw + Vf) (1)
Water uptake (L, WU) = (Vw + Vf) + (Vr1 – Vr2) (2)
Nutrient uptake concentration (meq・L-1) = [Cf Vf – {(Vr1Cr1 – Vr2Cr2) + Vs (Cs1 – Cs2)}] / WU (3)
where, Cr, Cw, Cf, and Cs are ion concentrations (meq・L-1) in the recycled nutrient solution, added water, added fresh nutrient solution, and remained nutrient solution in the root media, respectively. Vr, Vw, Vf, and Vs are volumes of the recycled nutrient solution, added water, added fresh nutrient solution, and remained nutrient solution in the root media (L・m-2), respectively. 1 and 2 mean the beginning and the end of every two weeks.
Individual cation (or anion) ratio was expressed by dividing cation (or anion) concentration by sum of cation (or anion) concentrations. Water uptake (L/d/plant) was calculated as WU/14 d/3 plants.
Experimental Management and Statistical Analysis
There were three plants per slab and three slabs per treatment. A randomized block design with three replications was used. The levels of nutrient accumulation or depletion in the recycled nutrient solutions and root media, and the nutrient uptake by the plants were analyzed by using the software Sigma-Plot 10 (SPSS, Inc., Chicago, IL, USA). Ion ratios were analyzed by cumulative standard deviations with time. Data were subjected to analysis of variance (ANOVA) using SAS 9.2 (SAS Institute, Cary, NC, USA). Means and standard errors were analyzed for each nutrient. Values of p < 0.05 were considered statistically significant by using Duncan’s multiple range tests.
Results and Discussion
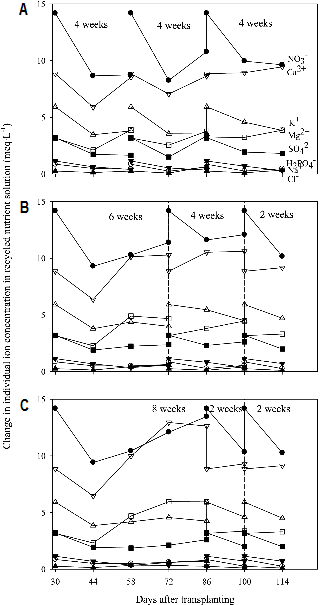
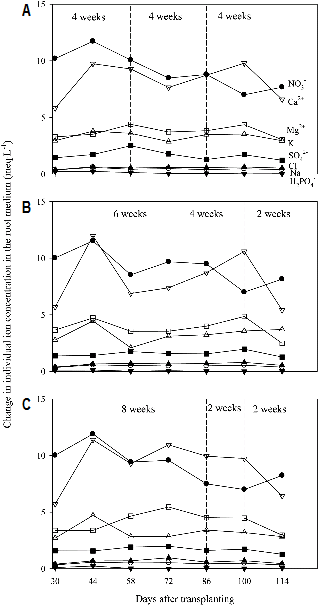
Changes in Ion Concentrations in the Recycled Nutrient Solution and Root Media
The deviation of ion concentrations in the recycled nutrient solution from the initial supply was clearly observed in all the treatments and the ion imbalance was smaller in 4-4-4 weeks (control) than 6-4-2 and 8-2-2 weeks (treatments) (Fig. 1). Particularly, the imbalance of Ca2+ and Mg2+ in the recycled nutrient solution was affected by the renewal pattern. Ca2+ was more accumulated in 8-2-2 weeks. Savvas and Manos (1999) and Sonneveld (2000) reported that it is needed to estimate the accumulation or depletion of K+, Ca2+, Mg2+, and Na+ concentrations in the nutrient solutions managed by EC value. The depletion in NO3- concentration in the recycled nutrient solution was observed, while those in H2PO4-, SO42-, and Cl- were nearly the same within 8 weeks regardless of the renewal pattern. Ion concentrations in the root media changed within relatively narrow range in 4-4-4 weeks compared to those in 6-4-2 weeks (Fig. 2). Ca2+ and Mg2+ were more accumulated in the root media in 8-2-2 weeks around the middle of experiment period (72 DAT). Rouphael and Colla (2005) indicated that the accumulation of bivalent ion like Ca2+ and depletion of NO3- and K+ are well-known alterations occurring in the composition of nutrient solutions when recycled. Accumulation and depletion of ions were observed in all the treatments during the experiment period. However the changes in ion concentrations were different among all treatments.
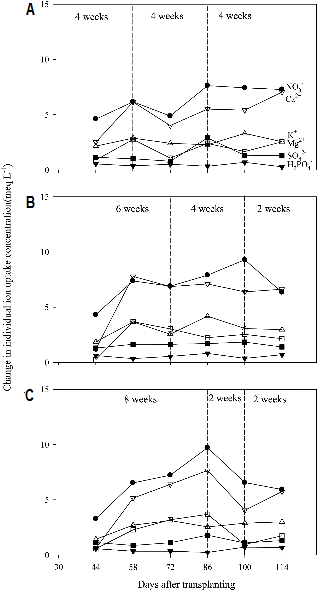
Changes in Nutrient Uptake Concentrations
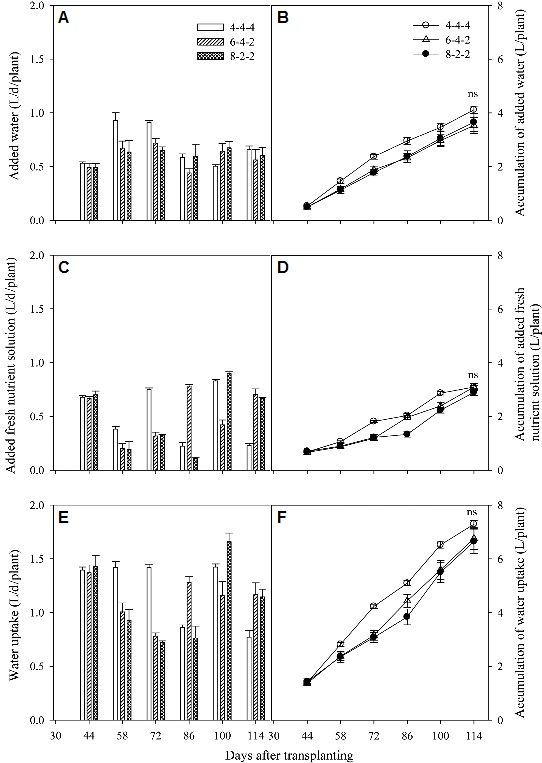
The added water, added fresh nutrient solutions, the total uptakes of nutrient solutions every two weeks and their accumulated values were described in Fig. 3. Although the added amounts and the water uptake showed different with time due to the different renewal patterns, there were no significant differences in the accumulated values at DAT 114 among all the treatments.
Changes in individual uptake concentrations for 2 weeks were smaller in 4-4-4 and 6-4-2 weeks than those in 8-2-2 weeks (Fig. 4). In general, the uptake of NO3- by the plants was the highest and followed by Ca2+, K+, Mg2+, SO42-, and H2PO4-. Marcussi et al. (2004) indicated that NO3- and K+ were the most extracted macronutrients by paprika and followed Ca2+, Mg2+, SO42- and H2PO4-. In this study, it was observed that NO3- > Ca2+ > K+ > Mg2+ > SO42- > H2PO4-. It might be due to the depletion of K+ when the nutrient solution was reused.
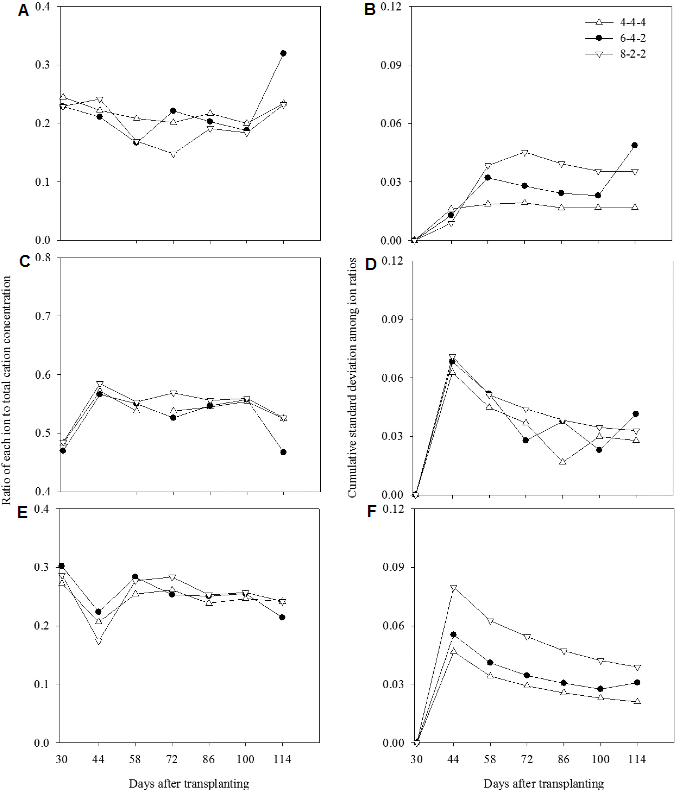
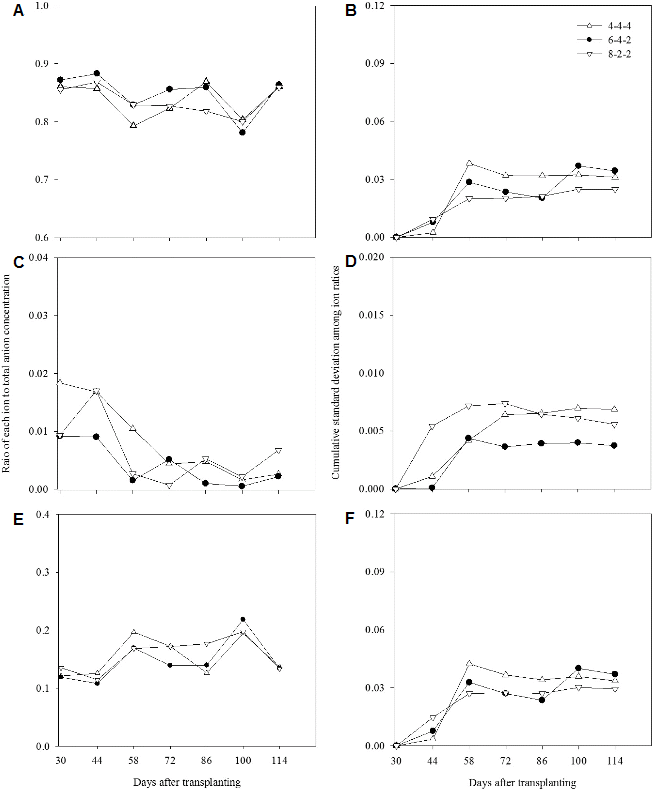
Changes in Ion Ratios in the Recycled Nutrient Solution and Root Media
The changes in cation ratios were similar among all the treatments before 100 DAT in the root media and became relatively higher in 6-4-2 weeks than 4-4-4 weeks (Fig. 5), while the change in anion ratio was stable in 4-4-4 weeks compared to other treatments (Fig. 6). Cumulative standard deviations in 4-4-4 and 8-2-2 weeks showed similar tendencies. After 58 DAT, the cumulative standard deviations reached plateau for both treatments in anions but decreased in cations, while abruptly changed after the second half of the experimental in 6-4-2 weeks. Stable changes in ion ratio in 8-2-2 weeks and a peak observed in 6-4-2 weeks mean that influence of the nutrient uptakes became greater on closed soilless cultures, and therefore the adjusting intervals could be extended in consideration of growth stage.
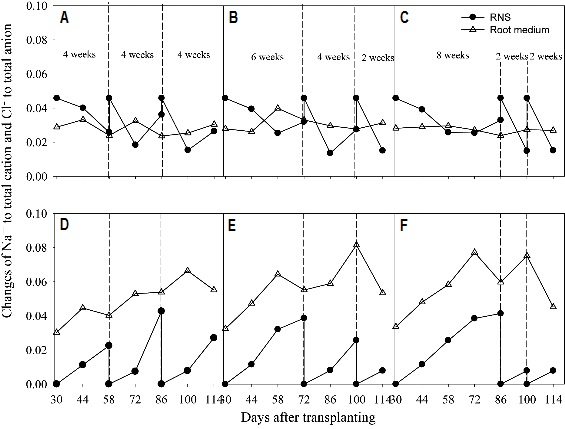
The accumulations of Cl- in the root media were higher than those in the recycled nutrient solution, however, Na+ concentration did not consistently change in the recycled nutrient solution and root media compared to the initial concentrations (Fig. 7). Increases in Cl- ratio were gradual in 4-4-4 weeks but fast after 72 DAT in 6-4-2 weeks in the root media. Unlike the essential nutrients, control of Na+ and Cl- concentrations are very difficult because the water containing both ions determines the levels of accumulations (Savvas et al., 2008).
Growth and Yield of Paprika Plants
There were no significant differences in growth and yield of paprika among all the renewal patterns of 4-4-4, 6-4-2, and 8-2-2 weeks (Table 1). According to Nukaya et al. (1991), yields are not affected by the accumulation of Cl- and SO42- in the recycled nutrient solution when the EC in the root media is maintained within the acceptable range. The specific ion-toxic effects of Na+ and/or Cl- were not related to the decrease in yield, while the accumulations of Na+ and Cl- were linearly related to the concentration of major elements in the nutrient solution (Baas et al., 1995).
Mineral Contents in Leaf and Fruit
There were no significant differences in mineral contents in leaf and fruit among all the renewal patterns (Table 2). K content in leaf was relatively higher in 4-4-4 and 6-4-2 weeks than that in 8-2-2 weeks, while the opposite trend was observed in fruit. Ca contents in fruit and leaf appeared to be higher in 4-4-4 weeks than those in 6-4-2 and 8-2-2 weeks. As shown in Fig. 4, Ca2+ uptake con-centrations in 4-4-4 and 6-4-2 weeks were higher than 8-2-2 weeks at DAT 114 (at the end of the experiment) and reflected to its contents in leaf and fruit. Marti and Mills (1991) reported that a decline in Ca2+ uptake occurred when fruits approach the mature green stage.
Renewal of the recycled nutrient solution using adequate renewal patterns can reduce the ion imbalance in the recycled nutrient solutions and root media without any detrimental effects on yield and plant growth. Relatively stable control of ion ratios in the root media was obtained in 4-4-4 weeks by adjusting the recycled nutrient solution. And the similar results were observed in 8-2-2 weeks compared to those in 4-4-4 weeks because the shorter renewal interval (2 weeks) was assigned to the latter growth stage. These results indicate that the renewal intervals can be extended rather than using constant intervals by considering the growth stage for more efficient and practical operations in closed soilless cultures.