Introduction
Materials and Methods
Plant materials
Pollen germination and morphology
Induction of hypo- and hypertetraploids
Observation of chromosome number in progenies obtained from open-, self-. or cross- pollination in hypo- and hypertetraploid grapes
Results and Discussion
Introduction
Seedlessness is one of the most important traits of table grapes, as consumer demand for seedless grapes has increased yearly in the global market. Thus, many grape breeders have strived to develop seedless varieties (Park, 2011; Heo and Park, 2015). Aneuploidy refers to the loss or gain of individual chromosomes from the normal chromosome set (Huettel et al., 2008). Aneuploid plants have been used in genetic studies to assign traits and molecular markers to specific chromosomes (Rick and Barton, 1954; Khush, 1973), but their economic usefulness is rarely evaluated because aneuploid plants often exhibit developmental abnormalities, sterility, or lethality (Henry et al., 2010). Male or female sterility in grapes, however, can provide the opportunity to induce seedless fruits via a one-time application of a growth regulator during the full blooming period (Park, 2010). In particular, near-polyploid plants are more tolerant than near-diploid plants because of their higher buffering capacity for chromosomal imbalance (Gao et al., 2016). Since polyploid plants generally have larger fruit, better disease resistance, and greater adaptability than diploid plants (Lewis, 1980; Gao et al., 1996), fostering hypo- or hypertetraploid (2n=4x±1 or 4x±2) hybrids is an effective way to develop new seedless grape varieties with highly desirable traits. In fact, ‘Takao’ (2n=4x-1=75) produces 100% seedless fruits with large berries and exhibits stable growth (Yamane et al., 1978). However, with the exception of 'Takao', no report has been published regarding the breeding of hypo- or hypertetraploid grape cultivars.
Hypo- or hypertetraploid seedlings have been produced from a few species. Bacchi (1940) obtained one hypotetraploid seedling from the open pollination of tetraploid citrus, and Esen and Soost (1972) produced one hypertetraploid seedling from the open pollination of tetraploid lemon. Gavrilenko et al. (2001) generated one hypertetraploid tomato from amphidiploid somatic hybrids. In Vitis, a small number of hypo- or hypertetraploid seedlings were also reported to result from para-fluorophenylalanine treatment and cross-pollination of tetraploid grape cultivars (Sarikhani and Wakana, 2006; Park et al., 2010). Therefore, the low occurrence of hypo- and hypertetraploid seedlings is the main barrier to developing new hypo- or hypertetraploid grape varieties. For this reason, efficient methods to generate more hypo- and hypertetraploid seedlings are needed.
It may be relatively easy to reduce chromosome numbers or to attain the original tetraploid state in the offspring of hypoor hypertetraploid plants during the course of sexual reproduction. In this study, we investigated the chromosome numbers in the progenies of hypo- and hypertetraploid grapes obtained from open-, self-, and cross-pollination to evaluate whether this technique could be applied to develop new seedless grape cultivars.
Materials and Methods
Plant materials
Hypotetraploid ‘Takao’, hypertetraploid ‘RB9127K’, and six tetraploid grape cultivars, including ‘Benefuji’, ‘Fujiminori’, ‘Honey Black’, ‘Shigyoku’, ‘RB9127’, and ‘Kyoho’, were used in this study (Table 1). Each hypo- and hypertetraploid grape was used as a female parent, and four tetraploids (‘Benefuji’, ‘Fujiminori’, ‘Honey Black’, and ‘Shigyoku’) were used as male parents to induce hypo- and hypertetraploid forms. In addition, ‘RB9127’ and ‘Kyoho’ were used to compare pollen germination rates and morphology with those of hypo- and hypertetraploid grapes. All grapes were grown in a vineyard at the Gangwondo Agricultural Research and Extension Services, located in Chuncheon, Republic of Korea. Five-year-old plants were used for this study and were trained to an overhead arbor at a planting density of 3.0 m × 4.0 m. The plants were irrigated at 15-day intervals, and the soil surface was managed by sod culture during the growing season.
Pollen germination and morphology
To measure the pollen germination rates in ‘Takao’, ‘RB9127K’, ‘Kyoho’, and ‘RB9127’, flowers were picked from clusters at the full bloom stage. The anthers were incubated in Petri dish under a fluorescent lamp at 25°C until the pollen grains were dispersed. Pollen grains were scattered gently onto artificial pollen germination medium containing 0.8 g·L-1 agar, 20 g·L-1 sucrose, and 10 mg·L-1 boric acid and incubated at 25°C for 4 hours under light. Pollen germination was defined as pollen tubes that elongated to three times the diameter of the pollen grain. The mean percentage of germinatedpollen was calculated from three samples, each containing approximately 500 grains. All pollen grains were examined under a microscope (Eclipse E400, Nikon, Japan). To observe pollen grain morphology, pollen grains taken from anthers were placed directly on stubs and coated with gold. The detailed surface ornamentation and aperture characteristics were examined under a scanning electron microscope (S-2460N, Hitachi, Japan).
Induction of hypo- and hypertetraploids
Progenies of ‘RB9127K’ and ‘Takao’ were derived from self-pollination, open-pollination, or crosses with tetraploid grapes. Fully developed seeds were extracted from mature berries approximately four months after pollination and crosses and separated into two categories, floaters and sinkers. Only sinker seeds were set in nursery boxes filled with wet sand and stored under natural environmental conditions. The following year, the nursery boxes were placed in a greenhouse to induce seed germination, and the number of germinated seeds was scored.
Observation of chromosome number in progenies obtained from open-, self-. or cross- pollination in hypo- and hypertetraploid grapes
Seedlings obtained from open-, self- or cross-pollination in hypo- and hypertetraploid grape were transferred to small pots, and the root tips of each seedling were collected for chromosome observation after one month of growth. The root tips were pre-treated with 2 mM 8-hydroxyquinoline at room temperature for 8 hours and fixed in acetic alcohol (1:3 v/v) at 4°C for 2 hours. The samples were washed and macerated in a solution of enzyme mixture consisting of 4% Cellulase RS, 1% Pectolyase Y23, 0.07 M KCl, and 0.075 M Na2EDTA for 30 minutes at 38°C. The macerated root tips were placed on a glass slide with a few drops of acetic alcohol (1:3 v/v) solution and dried at room temperature. The air-dried specimens were stained with 4% Giemsa diluted with 1/15 M phosphate buffer (pH 6.8) for 5 minutes, and chromosome numbers were counted under a microscope (Eclipse E400, Nikon, Japan).
Results and Discussion
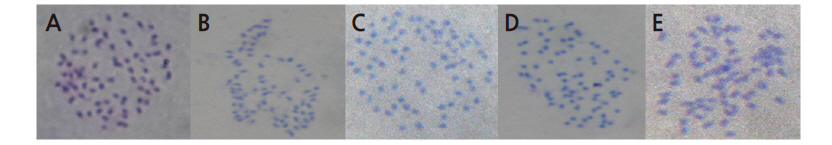
The germination rates of pollen grains from ‘Kyoho’, ‘RB9127’, ‘RB9127K’, and ‘Takao’ are shown in Table 1. The germination rates of tetraploid ‘Kyoho’ and ‘RB9127’ pollen were 36.5% and 39.1%, respectively. Hypo- and hypertetraploid grapes showed much lower pollen germination rates than the original tetraploid plants. The pollen germination rate in hypotetraploid ‘Takao’ was 0%, while the pollen germination rate in hypertetraploid ‘RB9127K’ was 20.3%. We examinedthe pollen morphology by SEM, finding that all grapes examined had pollen grains with furrows and germination pores (Fig. 1). In addition, the pollen grains appeared to be of relatively uniform shape. The grains were circular and commonly contained a protruding pore in the middle portion of each furrow, which concurs with descriptions of normal pollen grains from other grape cultivars (Abreu et al., 2006). Abreu et al. (2006) reported that grapes with pollen grains that include germination pores are usually fertile, but those with acolporated pollen grains without germination pores are sterile due to the lack germination structures. Hence, it appears that the sterility in hypotetraploid ‘Takao’ might not result from altered pollen morphology. Indeed, variations exist in pollen fertility among plants of different ploidy levels (Pringle and Murray, 1992; Hardion et al., 2015). In addition, pollen abortion might be associated with meiotic irregularity, gene dosage imbalance, reshuffling of chromosomes of different species into incompatible reaction systems, or the specific action of pollen-lethal genes (Souza et al., 2003). In hypotetraploid ‘Takao’, meiotic irregularity and gene dose imbalance caused by the lack of a chromosome are thought to lead to the abnormality of the vegetative and reproductive nucleus in pollen, resulting in pollen abortion. This assumption is supported by the abnormal behaviors of pollen grains observed in the current study. The pollen tubes in ‘Takao’ often displayed abnormal pollen behaviors, such as shriveling, branching, twisting, and swelling. Interestingly, the pollen germination rates differed between the tetraploid and hypertetraploid grapes used in this study. This result is in agreement with the finding of Fukushima and Tokumasu (1957) that pollen fertility in a hypertetraploid radish was much higher than that in a hypotetraploid radish. Thus, the abortion frequency of the vegetative and reproductive nucleus in hypertetraploid grapes might be lower than that of hypotetraploid grapes.
Table 1. Origins and pollen germination rates of the hypo-, hyper-, and tetraploid grapes used in this study.
| |
| |
Fig. 1. SEM photographs of the pollen grains of four grapes, in polar view. A, ‘Takao’; B, ‘Kyoho’; C, ‘RB9127K’; D, ‘RB9127’; F, furrow; G, germ pore. | |
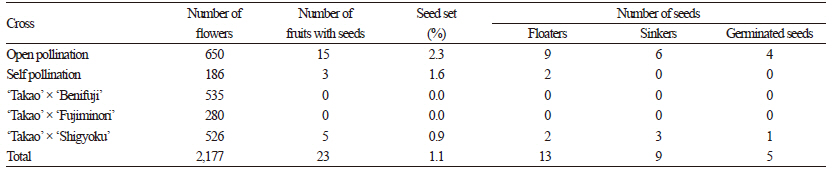
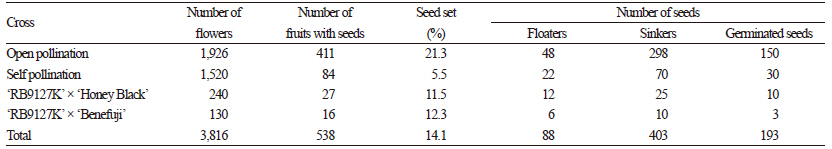
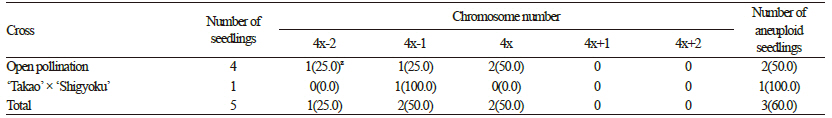
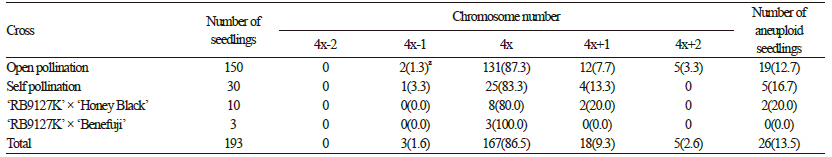
The percentage seed set values of ‘Takao’ and ‘RB9127K’ are summarized in Tables 2 and 3. The percentage seed set values varied from 0 to 21.3%, and we detected a difference between hypo- and hypertetraploid grapes. Overall, the percentage seed set values for hypertetraploid ‘RB9127K’ were higher than those of hypotetraploid ‘Takao’. The highest seed set percentage was observed after open pollination of ‘RB9127K’, whereas the lowest seed set percentage was found in ‘Takao’ crossed with ‘Benifuji’ and ‘Takao’ crossed with ‘Fujiminori’. Seed set is closely associated with pollen viability (Wang et al., 2004). Low pollen viability results in a low level of seed setting because the probability of pollination failure increases. Thus, it appears that the lower seed yield in hypotetraploid ‘Takao’ resulted from a low pollen level, as reported by Heo et al. (2007). As a result, the total number of progenies obtained from hypotetraploid ‘Takao’ was also much lower than that from hypertetraploid ‘RB9127K’. We observed the chromosomes of five seedlings derived from hypotetraploid ‘Takao’, finding that the chromosome number ranged from 2n=4x-2=74 to 2n=4x=76 (Table 3). Of the four seedlings obtained from open pollination of ‘Takao’, one showed 2n=4x-2=74, another showed 2n=4x-l=75, and the rest showed 2n=4x=76 (Fig. 2). The chromosome number of seedlings obtained from ‘Takao’ x ‘Shigyoku’ was 2n=4x-l=75. From hypertetraploid ‘RB9127K’, a total of 193 seedlings were obtained, and the chromosome numbers varied from 2n=4x-1=75 to 2n=4x+2=78 (Table 4). Among the observed seedlings, seedlings with 2n=4x=76 accounted for 86.5%, which was the highest percentage, whereas seedlings with 2n=4x-1=75 had the lowest frequency (1.6%). In addition, 11.9% of the seedlings gained one to two additional chromosomes. Hence, the occurrence frequency of hypo- or hypertetraploids from hypertetraploid ‘RB9127K’ was 13.5%, and a total of 26 aneuploid seedlings were generated. Hypo- and hypertetraploid plants among the progenies of tetraploid plants have been reported as being of spontaneous origin, asynaptic and desynaptic plants, and from crosses between different species (Khush, 1973; Edwards et al., 1980; Weber, 1991). In Vitis, hypo- and hypertetraploid seedlings were previously obtained from para-fluorophenylalanine treatment and from crosses of tetraploid grapes, but only two seedlings with aneuploidy were obtained, and the occurrence rate of hypo- or hypertetraploid seedlings was 0.85% (Sarikhani and Wakana, 2006). Although Park et al. (2010) generated a total of 23 seedlings with aneuploidy,these seedlings were obtained from approximately 4,700 seedlings. Thus, the occurrence rate and number of hypo- and hypertetraploid seedlings in the present study were much higher than in previous reports. This result suggests that the occurrence of hypo- or hypertetraploids might increase when hypo- or hypertetraploids are used as the female parent. Although we did not investigate the frequencies of hypo- or hypertetraploids in the progenies when hypo- or hypertetraploids are used as the male parent, we assume that the same results could be obtained as with their use as the female parent, since aneuploid cells are continuously produced as a result of mitotic irregularities, such as multipolar division, lagging chromosomes, unequal distribution of chromatids, and chromosome fragments (Bayliss, 1973). In particular, we found that the occurrence frequency of hypertetraploids was higher than that of hypotetraploids. This difference might be due to either the lower functionality of hypotetraploid gametes (with chromosome loss resulting from meiotic irregularities) compared to hypertetraploid gametes or the greater vigor of seeds of hypertetraploid grapes versus hypotetraploid grapes. Pollen fertility usually depends upon the functionality of gametes (Silva et al., 2011). In this study, the pollen fertility rate in hypertetraploids was not much lower than that in true tetraploids, unlike the pollen fertility rate in hypotetraploids. Thus,gametes with one less chromosome are expected to be more functional than those with one more chromosome, and the combination of this abnormal gamete and a normal gamete could produce more hypertetraploids. In addition, hypertetraploid seeds are less shriveled and less disturbed in their development than hypotetraploids in red clover and rye (Muntzing, 1943; Ellerstrom and Sjodin, 1966). This greater tolerance of seeds and plants to an excess of chromosomes is also supported by the fact that plants that deviate most from the euploid chromosome number are more frequent among hypertetraploids (Ellerstrom and Sjodin, 1966).
Table 2. Seed set and germination in open-pollinated, self-pollinated, and cross-pollinated hypotetraploid ‘Takao’ grape.
| |
Table 3. Seed set and germination in open-pollinated, self-pollinated, and cross-pollinated hypertetraploid ‘RB9127K’ grape.
| |
Table 4. Chromosome numbers in seedlings obtained from hypotetraploid ‘Takao’ grape.
| |
zNumbers in parentheses indicate occurrence percentages. | |
Table 5. Chromosome numbers in seedlings obtained from hypertetraploid ‘RB9127K’ grape.
| |
zNumbers in parentheses indicate occurrence percentages. | |
In this study, we confirmed that it might be easier to establish hypo- and hypertetraploid seedlings using a hypo- or hypertetraploid as the female parent than with other reported methods. Although we noticed that it was still difficult to generate a large number of hypo- and hypertetraploid seedlings from a hypotetraploid grape, several seeds contained an embryo with normal endosperm at the initial stage of seed development. Hence, more hypotetraploids should be obtained using the embryo rescue technique after pollination. The hypo- and hypertetraploid hybrid grapes obtained from this study show stable vine growth characteristics (Park et al., 2015), indicating that they can be used for commercial purposes, unlike other aneuploid plants, which display abnormal leaf morphology and unstable growth patterns. In addition, most of the hybrid grapes displayed low pollen fertility and produced seedless fruits with large berries, as 100 ppm of gibberellic acid was applied during their full-bloom period. Thus, the production of hypo- and hypertetraploid grapes using this method will contribute to the development of new seedless grape varieties with large berries.