Introduction
Materials and Methods
Plant Materials and Treatments
Morphological Injury Index
Electrolyte Leakages
MDA Content
Chlorophyll Content
Electrolyte Leakages
Protein Concentration
Proline Content
Statistical Analysis
Results
Effect of Temperature on Electrolyte Leakage of Gerbera Cultivars during Heat Treatment
Changes in Electrolyte Leakage of Gerbera Cultivars by Heat Treatment Period
Phenotypes after Heat Treatment
Changes in Proline, Soluble Protein, Chlorophyll, and MDA Contents of Gerbera Cultivars during Heat Treatment
10 Korean Journal of Horticultural Science & Technology Differences in Growth of Gerbera Cultivars during Recovery Phase after Heat Treatment
Discussion
Introduction
Heat stress, namely exposure to temperatures that exceed the tolerance range of the corresponding species, is an agricultural problem for a wide range of plants across the world (Hall, 2001; Wahid et al., 2007). Heat stress may cause a series of negative effects on the growth and productivity of plants, including reduced germination percentage, abnormal seedlings, poor seedling vigour, and increased sterility (Hasanuzzaman et al., 2013; Kumar et al., 2011; Piramila et al., 2012; Siddique et al., 1999; Toh et al., 2008). It has also been one of the most important factors underlying the reduction in yield and dry matter production in crops such as maize, wheat and rice (Giaveno and Ferrero, 2003; Mohammed and Tarpley, 2010; Zhang et al., 2013).
Either transitory or sustained high temperature can cause morpho-anatomical, phenological, physiological and molecular responses in plants (Ahuja et al., 2010; Mcclung et al., 2010; Mittler, 2006; Mittler et al., 2010; Mittler et al., 2012; Tubiello et al., 2007; Wheeler et al., 2000). The sensitivity and response of a plant to heat stress is complex and varied because it depends on many factors, such as the degree and duration of heat stress, the season, day versus night, the plant species, and the developmental stage of the plant (Guilioni et al., 1997; Young et al., 2004). Generally, the morphological damage from heat stress in plants includes scorching of leaves and twigs, sunburn on leaves, branches and stems, acceleration of leaf senescence and abscission, inhibition of shoot and root growth, flower abortion, and fruit discoloration (Guilioni et al., 1997; Ismail and Hall, 1999; Vollenweider and Gunthardt-Goerg, 2005; Wahid et al., 2007). Physiological responses of plant to heat stress are often associated with cell membrane thermostability, photosynthesis, accumulation of compatible osmolytes, water relations, hormonal changes, assimilate partitioning, and secondary metabolites (Hasanuzzaman et al., 2013; Wahid et al., 2007). Among them, cell membrane thermostability is one of the physiological characteristics most sensitive to heat stress in plants. Heat stress can alter the tertiary and quaternary structure of membrane proteins, make the lipid bilayer of biological membranes more fluid, and thereby enhance the permeability of membranes, leading to increased loss of electrolytes (Wahid et al., 2007). Heat stress also induces oxidative stress, which causes membrane lipid peroxidation and protein denaturation and accelerates the disruption of cell membrane stability (Camejo et al., 2006; Hasanuzzaman et al., 2013; Rodríguez et al., 2005). Photosynthesis is another physiological process sensitive to heat stress in plants. Any suppression of photosynthesis caused by high temperature can limit plant growth. Thus, changes in various photosynthetic traits under heat stress are good indicators of plant thermotolerance or thermosensitivity (Crafts-Brandner and Salvucci, 2002; Wahid et al., 2007). In addition, heat stress responses involve changes in the contents of a variety of compatible osmolytes such as sugars, sugar alcohols, glycinebetaine, proline, soluble protein, quaternary ammonium, and tertiary sulphonium compounds (Sairam and Tyagi, 2004; Wahid et al., 2007). The accumulation of osmolytes may be involved in osmotic adjustment, and it is a key adaptive mechanism in many plants against abiotic stress conditions (Hasanuzzaman et al., 2013; Wahid et al., 2007).
Gerbera jamesonii is one of the most popular cut flowers and has broad markets worldwide. During the production of gerbera flowers, when the temperature is high in summer, especially when the highest temperature is around 40°C during July and August in southern China, gerbera plants develop deformed flowers and shortened stems, both of which significantly reduce ornamental value. Moreover, the yield of cut flowers is also decreased by heat stress, leading to economic losses (Peng et al., 2010). However, little is known about the effect of heat stress on the physiological and biochemical characteristics of gerbera plants. Screening of heat-resistant and heat-sensitive cultivars is an important step to understand the mechanism by which heat stress limits growth and development in gerbera and to breed stress-hardy gerbera cultivars.
In this study, we analyzed the morphological, physiological and biochemical responses of six varieties of Gerbera jamesonii to heat stress. The results showed that high temperature led to loss of green color, yellowing, wilting, drying and death of the leaves of Gerbera plants. It also caused an increase of electrical conductivity, decreases in soluble protein and chlorophyll, and accumulation of MDA and proline in leaves. Moreover, our results also demonstrated that heat-stress tolerance is variable among gerbera cultivars.
Materials and Methods
Plant Materials and Treatments
Six commercial gerbera cultivars (Gerbera jamesonii ‘Meihongheixin’, ‘Beijixing’, ‘Dadifen’, ‘Shijihong’, ‘Linglong’, and ‘Taiyangfengbao’) were grown in a greenhouse under natural illumination. Temperature and relative humidity were monitored with thermometers and hygrometers and maintained between 18-26°C and 60-75%, respectively, through shade, water spraying or ventilation. Plantlets of the six cultivars were transplanted into 6-cm-diameter square plastic pots filled with a mixture of peat moss, perlite and vermiculite (2:1:1 v/v/v) and fertilized with N 128 mgㆍL-1, P 46 mgㆍL-1, and K 204 mgㆍL-1. Plants were watered as needed to ensure adequate soil moisture for growth. Three-month-old plants with 5-7 expanded leaves and 12-15 cm height were used for the study.
For heat stress at different temperatures, three-month-old seedlings of six cultivars were transferred into controlled environment chambers (FPG-300C-20D, LF Apparatus, Ningbo, China) at 23 (control), 40, 45, or 50°C for 1 d. In the second round of treatment, the seedlings of six cultivars were treated with 43, 45, and 47°C for 1 d. For heat stress with different treatment times, plants were exposed to 45°C for 0.5, 1, or 1.5 d respectively, with 0 d as the control. To avoid drought stress caused by evaporation from the pots under heat treatment, additional water, which was equilibrated at 45°C in advance, was provided to maintain the humidity above 65% during the heat treatment. For the recovery phase after heat stress, plants were transferred to normal growth conditions in the same controlled environment chambers at 23°C for 1 month as a recovery after the 1-d or 0.5- to 1.5-d heat treatment of 45°C. All treatments were under continuous illumination from fluorescent lamps with a PPFD of approximately 285 µmol·m −2·s −1, and relative humidity was controlled between 60-75%.
Morphological Injury Index
The injury index was defined as described by Hong et al. (2009) with modifications as follows: grade 0, normal growth; grade 1, fewer than 1/4 of the leaves become lightly wilted or rosette-shaped; grade 2, 1/4 to 1/2 of the leaves become wilted or rosette-shaped and lightly yellowed; grade 3, 1/2 to 3/4 of the leaves become wilted, rosette-shaped and yellowed; grade 4, more than 3/4 of the leaves become rosette-shaped and severely yellowed; and grade 5, the whole plant dies. The injury index was determined at the end of 1-d heat treatment at 45°C as well as at 1, 2, 3 and 4 weeks after heat treatment.
Electrolyte Leakage
The measurement of leaf electrolyte leakage was as described by Jaglo-Ottosen et al. (1998) and Hong et al. (2009), with some modifications. Two leaves in the middle position were taken, and four discs of 7 mm diameter near the veins of each leaf were sampled. The eight discs were placed in a tube containing 10 mL deionized water. The tube was shaken gently for 4 h, and the initial conductivity (C1) was measured with a conductivity meter (DDS-307A, Shanghai Leici, China). Then the sample was boiled for 15 min, and the final conductivity (C2) was evaluated. The relative electrolyte leakage was calculated as (C1/C2) × 100%. Leaf electrolyte leakage of the six cultivars was measured at the end of heat treatment and 4 weeks after heat treatment.
MDA Content
The measurement of malondialdehyde (MDA) was as described by Heath and Packer (1968) and Deng et al. (2011) with some modifications. Fresh leaves of each sample (0.2 g) were taken at the end of the heat treatment and homogenized with 10% (w/v) trichloroacetic acid (TCA). The samples were centrifuged at 4,500 g for 10 min, and 2 mL supernatant was mixed with 2 mL 10% (w/v) TCA prepared in 0.5% (w/v) thiobarbituric acid (TBA). The mixture was boiled at 90°C for 30 min and then quickly cooled in an ice bath and centrifuged at 4,500 g for 10 min. The absorbance was read at 532 nm and 600 nm in a spectrophotometer (T6 New Century, Purkinje General, Beijing, China). The concentration of MDA was calculated using its extinction coefficient, 155 mM-1·cm-1.
Chlorophyll Content
Leaf chlorophyll content was measured using the method described by Hiscox and Israelstam (1979) and Sun et al. (2014). After the heat treatment, fresh leaves (0.2 g) were taken, sheared, and soaked in a tube with 10 mL dimethyl sulfoxide. The tube was kept in darkness for 72 h. A spectrophotometer (T6 New Century, Purkinje General) was used to measure the absorbance of the sample at 663 nm and 645 nm. The contents of chlorophyll a and b were calculated according to the equations of Doganlar (2010).
Chlorophyll a: (11.75 × A663-2.35 × A645) × 20/mg leaf weight
Chlorophyll b: (18.61 × A645-3.96 × A663) × 20/mg leaf weight
Protein Concentration
The soluble protein content was measured using bovine serum albumin (BSA) as a protein standard according to Bradford (1976) with some modifications. After the heat treatment, approximately 0.2 g leaf sample was homogenized in an ice bath with 5 mL distilled water and centrifuged at 4,000 g for 10 min. The supernatant was poured into a 10-mL volumetric flask, and 2 mL distilled water was added. The mixture was centrifuged for 10 min at 4,000 g. Distilled water was added into the supernatant to a total volume of 10 mL. A 100-μL aliquot of the diluted supernatant was mixed with 5 mL Coomassie Brilliant Blue G-250 reagent, and then a spectrophotometer (T6 New Century, Purkinje General) was used to measure the absorbance at 595 nm.
Proline Content
Proline content was measured using the acid ninhydrin method (Bates et al., 1973; Deng et al., 2011) with some modifications. After the heat treatment, leaf samples were taken and homogenized in 5 mL 3% (w/v) aqueous sulfosalicylic acid and boiled at 100°C for 10 min. The homogenate was cooled to room temperature and centrifuged at 4,000 g for 10 min. Then, 2 mL supernatant was sucked into a test tube and supplemented with 2 mL glacial acetic acid and 3 mL 2.5% (w/v) acid ninhydrin. The reaction mixture was boiled at 100°C for 40 min. Subsequently, the reaction was quenched on ice. The mixture was extracted with 5 mL toluene, and the absorbance was read at 520 nm by spectrophotometer (T6 New Century, Purkinje General).
Statistical Analysis
In the electrolyte leakage and injury index data analysis, the statistical significance of differences between the treatments was analyzed using a one-way ANOVA test followed by Duncan's multiple range test (p < 5%) or/and the least significant difference (LSD) method by SPSS 16.0 (SPSS Inc., Chicago, IL, USA). In the proline, soluble protein, MDA and chlorophyll content data analyses, Student’s t-test (*p < 0.05, **p < 0.01) was applied to identify significantly different trait values by SPSS 16.0.
Results
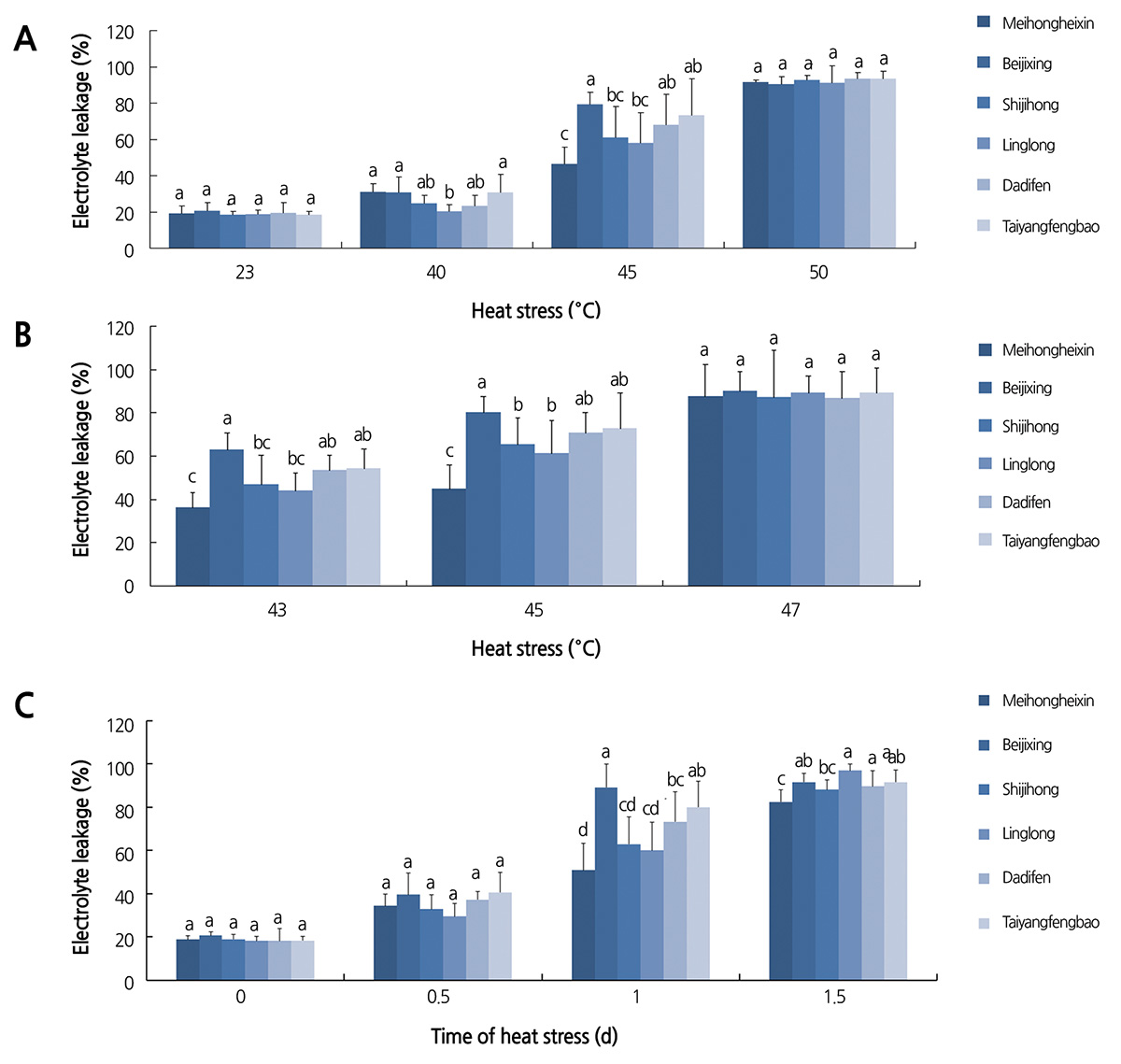
Effect of Temperature on Electrolyte Leakage of Gerbera Cultivars during Heat Treatment
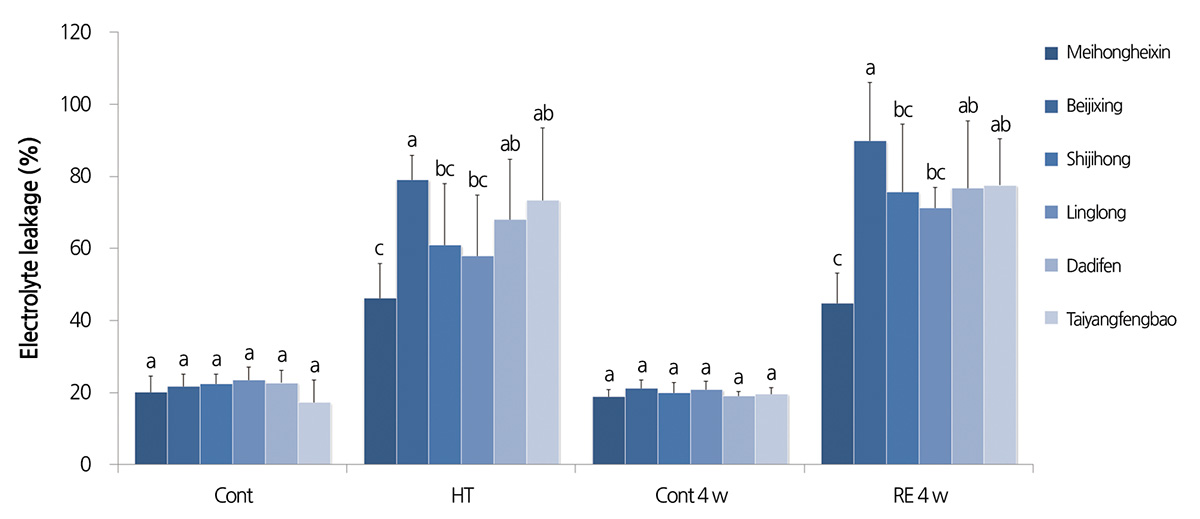
To determine the range of thermal tolerance of gerbera cultivars, three-month-old seedlings of six gerbera cultivars were subjected to different temperature treatments. As shown in Fig. 1A, when compared with normal growth temperature (23°C), treatment at 40°C only slightly increased the electrolyte leakage of all six cultivars, suggesting that 40°C induces mild heat stress in the gerbera seedlings. At 45°C, significant differences in electrolyte leakage were found between the six cultivars. The electrolyte leakage of ‘Meihongheixin’ was 46.2%, which was the lowest in all the tested cultivars. ‘Beijixing’ had the highest electrolyte leakage, of 79.1%, approximately 2.1 fold higher than that of ‘Meihongheixin’. Between them, there was ‘Taiyangfenbao’ (73.4%), ‘Dadifen’ (68.0%), ‘Shijihong’ (60.9%), and ‘Linglong’ (57.9%) in descending order. When the treatment temperature was raised to 50°C, the electrolyte leakage of all samples was sharply higher, and most of the treated plants died. We further examined the electrolyte leakage of the six cultivars after 1-d heat treatment at 43, 45, and 47°C. The electrolyte leakage of the six cultivars under 43°C showed a similar trend to that under 45°C, although the extent of electrolyte leakage of the former was smaller. At 47°C, the electrolyte leakage of all cultivars was higher than 85%, suggesting that membranes were seriously damaged under the heat treatment and that temperatures higher than 45°C exceed the range of heat tolerance of gerbera seedlings.
Changes in Electrolyte Leakage of Gerbera Cultivars by Heat Treatment Period
When plants were treated at 45°C for 1.5 d, the electrolyte leakage of all tested cultivars was greater than 80%, suggesting that serious membrane injury was caused by the treatment in all tested cultivars. However, when the treatment time was decreased to 0.5 d, electrolyte leakage was only slightly higher than the control, and no difference was observed between the cultivars (Fig. 1C). By contrast, when the treatment lasted for 1 d, electrolyte leakage was significantly different among the cultivars as described above (Fig. 1A and B). Thus, these results indicated that a 1-d treatment at 45°C is suitable for detecting the effects of heat stress on the physiological and biochemical characteristic of gerbera seedlings and for distinguishing heat tolerance gradations between different gerbera cultivars.
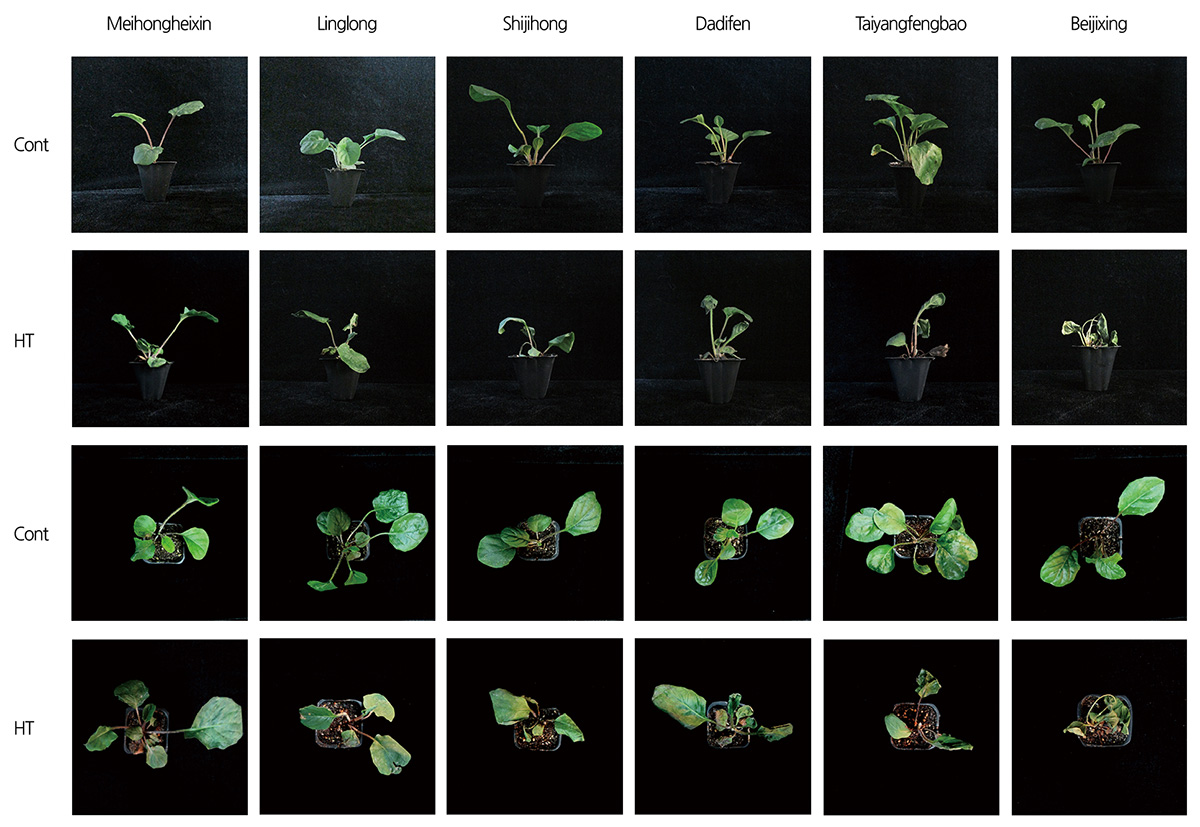
Phenotypes after Heat Treatment
The phenotypes of gerbera seedlings under heat treatment (45°C for 1 d hereafter) were observed. As shown in Fig. 2, when compared with untreated seedlings, the leaves of high-temperature-treated seedlings displayed different levels of injury symptoms such as yellowing, wilting, dried and dead. Among the six tested cultivars, ‘Meihongheixin’ showed the least illness after heat treatment, with leaves showing slight yellowing. ‘Beijixing’ showed the most serious injury, with leaves turning brown and wilted. The other two cultivars, ‘Linglong’ and ’Shijihong’, had similar phenotypes. Their leaves turned yellow, and the edges of some leaves were brown and dried. The phenotype of ‘Dadifen’ was similar to that of ‘Taiyangfengbao’, with half of the leaves becoming dried (Fig. 2).
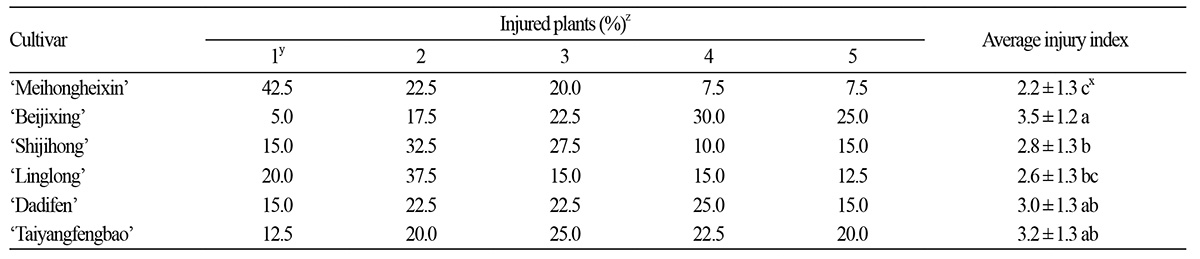
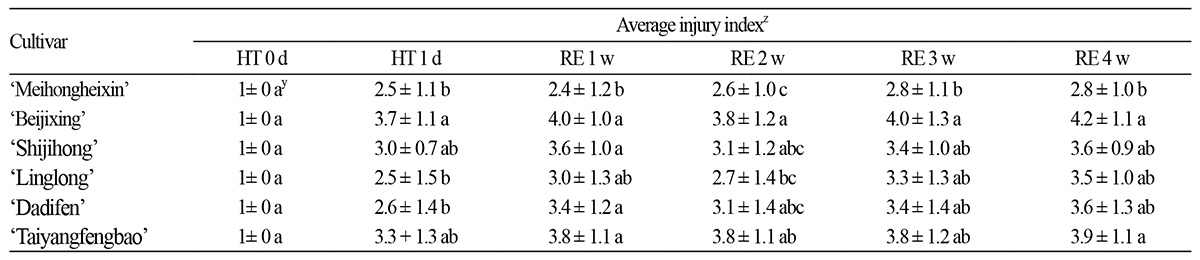
The injury severity was quantified using a 0–5 heat injury index (please see Materials and Methods for details). After the heat treatment, 42.5% of ‘Meihongheixin’ were in grade 1, which indicates the mildest symptoms, whereas only 5% of ‘Beijixing’ were in this category. In grade 5, indicating death, ‘Meihongheixin’ had the lowest percentage, i.e., 7.5%, among the six cultivars, whereas ‘Beijixing’ had the highest percentage, i.e., 25%. The average heat injury grade was calculated and listed in ascending order as follows: ‘Meihongheixin’, ‘Linglong’, ‘Shijihong’, ‘Dadifen’, ‘Taiyangfengbao’, and ‘Beijixing’ (Table 1).
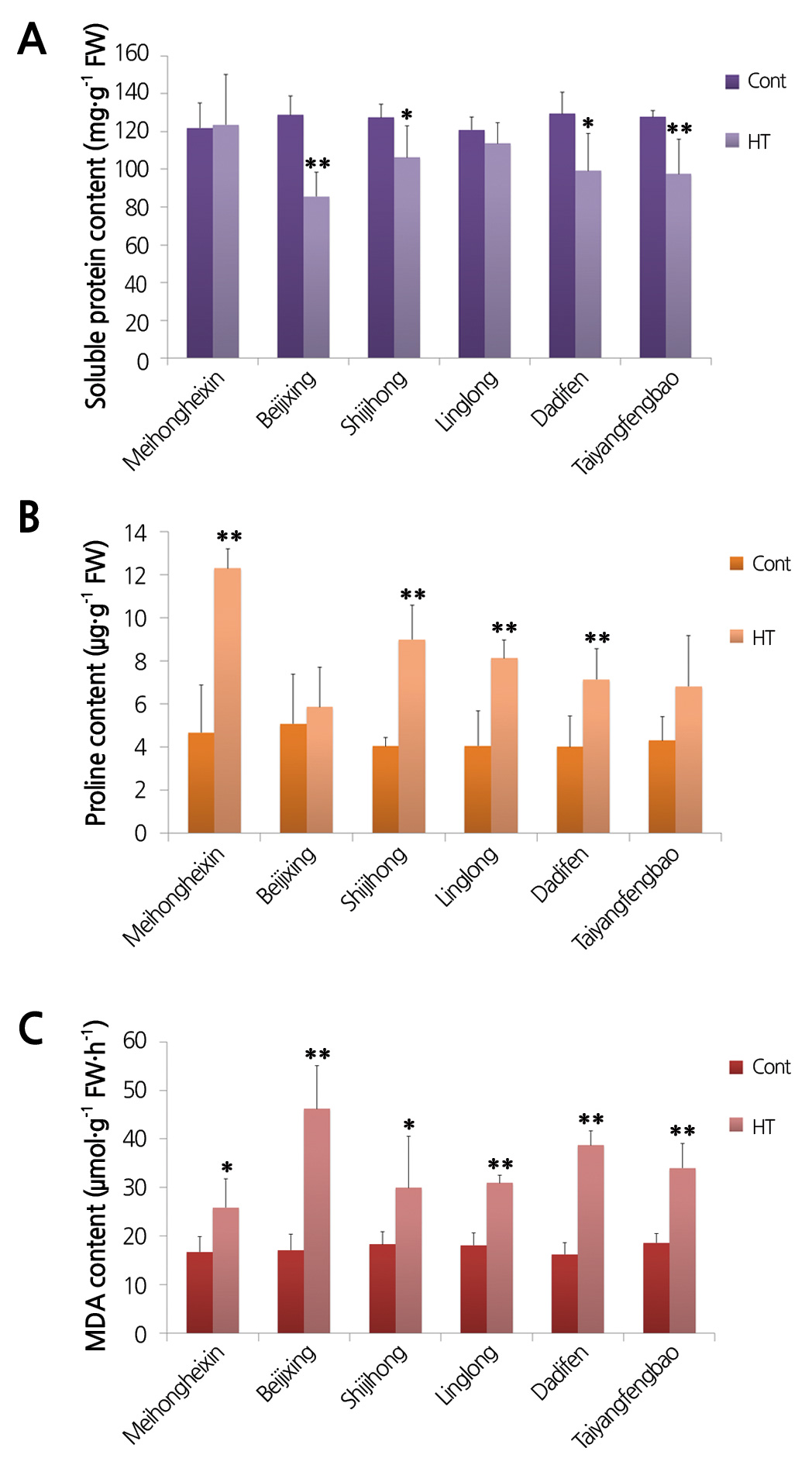
Changes in Proline, Soluble Protein, Chlorophyll, and MDA Contents of Gerbera Cultivars during Heat Treatment
The leaf soluble protein content of the gerbera seedlings was affected by heat stress, but the degree differed between the cultivars. As shown in Fig. 3A, the soluble protein content of ‘Meihongheixin’ and ‘Linglong’ remained stable as the temperature rose, and no significant difference was observed between heat treatment and control. By contrast, the soluble protein content of ‘Beijixing’, ‘Shijihong’, ‘Dadifen’ and ‘Taiyangfengbao’ decreased greatly under heat treatment. Among them, the extent of the change in ‘Beijixing’ was the most marked, nearly 33.5%, followed by ‘Taiyangfengbao’ (23.6%), ‘Dadifen’ (23.3%), and ‘Shijihong’ (16.6%). These results suggested that the heat tolerance of gerbera cultivars might be associated with the stability of soluble protein content under heat stress.
Leaf proline content increased under heat stress in most of the gerbera cultivars, indicating that as a general tendency, heat treatment had an inducing effect on proline content. As expected, the proline content of ‘Meihongheixin’ showed a drastic increase after the heat treatment, being approximately 2.6-fold higher than that of the control. ‘Shijihong’, ‘Linglong’ and ‘Dadifen’ also obviously increased their proline content by 2.2-, 2.0- and 1.8-fold, respectively, after the heat treatment. The other two cultivars, ‘Beijixing’ and ‘Taiyangfengbao’, did not show significant changes in proline content under heat stress (Fig. 3B).
Leaf MDA content in gerbera cultivars was increased by heat temperature in varying degrees. As shown in Fig. 3C, under heat treatment, the MDA content rose significantly in all the treated cultivars. The MDA content in heat-treated ‘Beijixing’ increased about 2.7-fold, whereas ‘Meihongheixin’ only increased 1.5-fold as compared with the untreated plants. The fold changes of the other cultivars were between these values. These results indicated that the heat stress suppressed cell viability in gerberas plants, and MDA content was associated with tolerance for heat stress in gerbera cultivars.
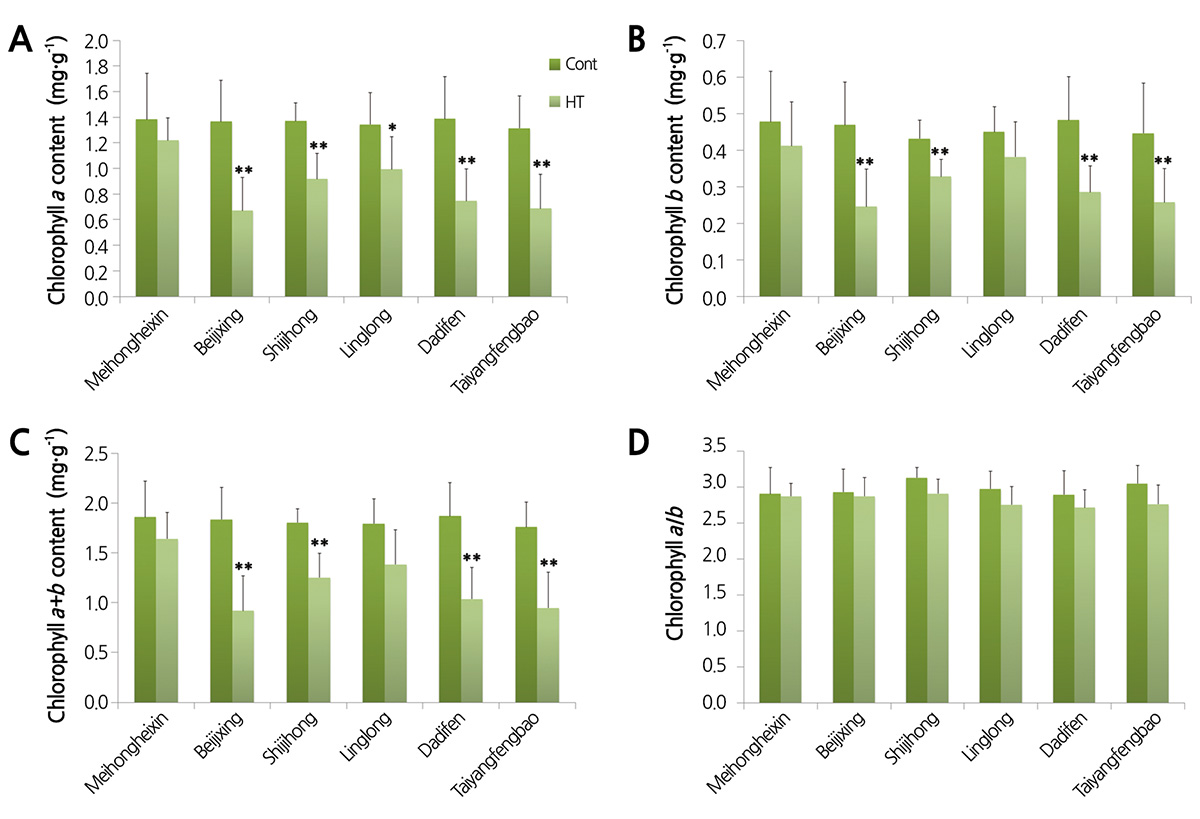
Heat stress reduced the content of both chlorophyll a and b in gerbera cultivars to varying degrees, but did not affect the chlorophyll a/b ratio. As shown in Fig. 4, chlorophyll a, chlorophyll b and total chlorophyll content showed pronounced differences between the heat treatment and control in ‘Beijixing’, ‘Shijihong’, ‘Dadifen’ and ‘Taiyangfengbao’. The total chlorophyll content of ‘Beijixing’ was reduced the most drastically, by about 50%, when the temperature was higher, followed by ‘Taiyangfengbao’ (46%) and ‘Dadifen’ (45%). In ‘Linglong’, the chlorophyll a content was significantly lower than the control, while the chlorophyll b content only changed slightly. Despite this difference in response of chlorophyll a and chlorophyll b, the chlorophyll a/b ratio was not significantly different. The chlorophyll content of ‘Meihongheixin’ remained stable under high temperature, suggesting that the growth of ‘Meihongheixin’ might be affected less by heat stress.
Differences in Growth of Gerbera Cultivars during Recovery Phase after Heat Treatment
During the recovery period, the injury index increased with time and varied among the cultivars. The injury indexes of ‘Linglong’, ‘Shijihong’, ‘Dadifen’, ‘Taiyangfengbao’, and ‘Beijixing’ increased gradually, indicating that the growth status of these cultivars got worse after the heat injury. At the end of the recovery period, the injury index of ‘Beijixing’ remained the highest among the cultivars; the growth rate of the injury index was highest in ‘Linglong’ and ‘Dadifen’ with an increase of 40.0% and 38.5%, respectively, over the recovery period. By contrast, ‘Meihongheixin’ maintained the lowest injury index, which was lower than 3.0 during the recovery period (Table 2), indicating that the growth of ‘Meihongheixin’ did not get worse, and only moderate heat injury was inflicted on this cultivar by the heat stress.
All treated plants were sampled and leaf electrolyte leakage was detected after one month of recovery. As shown in Fig. 5, the electrolyte leakage of ‘Meihongheixin’ was the lowest after the recovery period, whereas that of ‘Beijixing’ was the highest. Furthermore, ion leakage from ‘Beijixing’, ‘Shijihong’, ‘Linglong’, and ‘Dadifen’ increased by at least 15% during the recovery period, suggesting the continued deterioration of the membrane. The electrolyte leakage of ‘Meihongheixin’ and ‘Taiyangfengbao’ did not change significantly.
Based on the above studies, we concluded that among the six tested gerbera cultivars, ‘Meihongheixin’ is a heat-resistant cultivar, whereas ‘Beijixing’ is a heat-sensitive one. ‘Shijihong’ and ‘Linglong’ are relatively heat-resistant cultivars, whereas ‘Dadifen’ and ‘Taiyangfengbao’ are relatively heat-sensitive cultivars.
Discussion
Plants exposed to high temperatures exhibit a variety of responses, including symptomatic and quantitative changes in morphology and growth (Wahid et al., 2007). The changes in morphological characteristics can directly reflect the status of plant injury and are the most direct indicators for evaluating resistance to stresses (Tian et al., 2011). In this study, the results showed that a 1-d heat treatment at 45°C caused obvious damage to the aerial part of the gerbera plants. Under this heat treatment, leaves of the gerbera cultivars turned yellow and wilted to varying degrees (Fig. 2). This phenotype is similar to previous reports in chrysanthemum under heat stress (Hong et al., 2009).
The ability of plants to cope with or adapt to environmental stresses, including heat stress, varies across and within species as well as cultivars. Such variation of the tolerance or adaptability to heat stress has been subject to natural selection over hundreds of thousands of years, as well as being used for artificial selection by breeders to improve agronomic traits (Shavrukov et al., 2010; Sun et al., 2014). In this study, phenotype comparison and injury grade assessment revealed that the heat tolerance of the gerbera cultivars was significantly different (Fig. 1, Tables 1 and 2). Among the six tested cultivars, irrespective of the heat treatment or the recovery treatment, ‘Meihongheixin’ was less damaged by the heat stress, with the mildest symptoms on leaves and the lowest average heat injury grade. Conversely, ‘Beijixing’ was damaged heavily and had the highest average heat injury grade. The extent of injury of the other four cultivars was between these two extremes. Furthermore, we also determined the physiological and biochemical indicators of all the cultivars, and the results were consistent with the phenotypes, supporting the existence of a difference in heat tolerance among the gerbera cultivars.
High temperature can alter the membrane structure of a plant cell, leading to increased membrane permeability, so that the electrolytes and certain small molecules within the cell flow out, causing an increase in electrical conductivity (Wahid et al., 2007). Thus, electrical conductivity, which reflects the stability of the cell membrane, is widely used to assess the heat tolerance of a variety of crops. Our results showed that 45°C for 1 d was the optimal heat condition to distinguish the tolerance of different gerbera cultivars, as the electrical leakage among the six cultivars was significantly different under this condition. We found that the electrical leakage of ‘Meihongheixin’ was much lower than that of ‘Beijixing’ under high temperature and recovery treatments (Figs. 1 and 5), suggesting that the membrane structure of the heat-resistant cultivar ‘Meihongheixin’ remains relatively stable under heat stress.
Soluble protein metabolism is one of the important metabolic pathways for responding to environmental stresses in plants. Soluble proteins serve as osmotic adjustment substances, and might also improve antioxidant capacity through ROS elimination and stabilization of the structure of biological macromolecules (Fan et al., 2014). The soluble protein content is suppressed by high temperature in many plants, including mulberry (Chaitanya et al., 2001), strawberry (Gulen and Eris, 2004), grass (Xu and Zhou, 2006), and rice (Tang et al., 2008). Our study showed that soluble protein content was significantly decreased after heat stress in most gerbera cultivars except ‘Meihongheixin’ and ‘Linglong’, in which the protein 12 Korean Journal of Horticultural Science & Technology concentration was relatively stable (Fig. 3A). Soluble protein content under heat stress may be associated with protein biosynthesis and denaturation (Gulen and Eris, 2004). Thus, such phenomena suggest that the life cycle of proteins in ‘Meihongheixin’ and ‘Linglong’ remained relatively stable under heat stress, possibly leading to the higher heat tolerance of ‘Meihongheixin’ and ‘Linglong’.
Proline frequently accumulates in large quantities in stressed plants as an osmotic regulator, and it serves as an osmotic protectant for many cellular structures during abiotic stress (Kishor et al., 2005; Sairam and Tyagi, 2004; Ueda et al., 2008; Wahid et al., 2007). The accumulation of proline may buffer the cellular redox potential under heat and other environmental stresses and thereby enhance the stress tolerance of plants (Deng et al., 2011; Wahid and Close, 2007). In our results, the high proline content of heat-treated ‘Meihongheixin’ suggested that proline was biosynthesized as a means of regulating cellular osmosis and metabolism and conferring enhanced tolerance to high temperature, which minimized cellular damage under conditions of very high temperature. By contrast, the heat-stressed ‘Beijixing’ contained the least proline, consistent with its relative heat sensitivity (Fig. 3B).
The lipid peroxidation of cell membranes, which is usually caused by free radicals, occurs under stress conditions (Xu et al., 2006). The accumulation of MDA, a product of peroxidation of unsaturated fatty acids in phospholipids, is widely used as an indicator of the degree of cellular membrane lipid peroxidation (Li et al., 2000). Our study found that heat stress induced an increase in MDA content in all tested cultivars, suggesting that lipid peroxidation occurred in gerbera seedlings under heat stress. Moreover, the degree of MDA content increase in the cultivars was consistent with their level of heat hardiness (Fig. 3C).
Wang et al. (2009) reported that after heat treatment, the chlorophyll content decreased quickly, and the magnitude of the change in heat-resistant plants was smaller than that in non-resistant varieties. Similarly, through comparing the chlorophyll content of ‘Meihongheixin’ and ‘Beijixing’, we found that after heat stress, the content of chlorophyll a, chlorophyll b, and chlorophyll a+b had decreased less in ‘Meihongheixin’ than in ‘Beijixing’ (Fig. 4), indicating that the photosynthetic apparatus of the heat-resistant cultivar ‘Meihongheixin’ had better stability than that of the heat-sensitive ‘Beijixing’.
In conclusion, our study showed that heat stress not only damages the appearance of gerbera plants but also increases the electrical conductivity, MDA and proline content and decreases the soluble protein and chlorophyll content in leaves. The heat tolerance of the six tested gerbera cultivars was found to be significantly different. Among them, ‘Meihongheixin’ is a heat-resistant cultivar, whereas ‘Beijixing’ is a heat-sensitive one. ‘Shijihong’ and ‘Linglong’ are relatively heat-resistant cultivars, and ‘Dadifen’ and ‘Taiyangfengbao’ are relatively heat-sensitive ones.
The gerbera plants we used in this study were three-month-old seedlings. Heat tolerance will likely differ at different developmental stages, such as vegetative stage versus reproductive stage. In addition, the heat treatments were conducted under artificial controlled growth conditions. In natural conditions, high temperature stress is often accompanied by drought and other stresses. Therefore, field trials for cultivar evaluation and the mechanisms of the effects of heat stress on the growth of gerbera plants will be subjects of future studies.