Introduction
Material and Methods
Plant Materials
Analysis of Carotenoids
Analysis of Phenolic Compounds
Analysis of Volatile Organic Compounds
Analysis of Tomatines
Statistical Analysis
Results and Discussion
The Contents of Carotenoids
The Contents of Phenolic Compounds
The Contents of Volatile Organic Compounds
The Contents of Alkaloids
Introduction
Secondary metabolites are diverse compounds found in terrestrial and marine plant species. Plants produce secondary metabolites under biotic or abiotic stress because the compounds have antimicrobial, anti-herbivory, and allopathic effects (Dixon, 2001). In addition, various secondary meta-bolites show other beneficial properties such as color, scent, and flavor attractants (Frydman et al., 2004; Rohloff and Bones, 2005; Verdonk et al., 2003), UV-protectants, anti-oxidants, signaling factors, and nutraceutical and pharmacological efficacies (Aerts et al., 1999; Bagchi et al., 2000; Deavours and Dixon, 2005; D’Haeze and Holsters, 2002; Gidley, 2004; Manach et al., 2004; Merz-Demlow et al., 2000; Oldroyd, 2001; Relić et al., 1994; Setchell and Cassidy, 1999).
Tomato (Solanum lycopersicum L.) is known as a plant that contains many secondary metabolites. Profiling of secondary metabolites in tomato plants, particularly the fruits, has been performed by analysis of carotenoids (Moco et al., 2007), phenolic compounds (Slimestad and Verheul, 2009), volatile organic compounds (Buttery et al., 1987), and alkaloids (Friedman and Levin, 1998).
The carotenoids are important color compounds in flowers, food, and fruits (Verpoorte and Memelink, 2002). Lycopene is the most abundant secondary metabolites in fully red ripe tomato fruits. This carotenoids a natural pigment synthesized by plants to protect cells against oxidative damage (Rao and Agarwal, 1999) and to attract pollinators (Moco et al., 2007). Moreover, researchers have reported that lycopene induces communication between cells (Zhang et al., 1991) and controls immune systems and other metabolic pathways (Astorg et al., 1997; Fuhrman et al., 1997). Tomatoes also include β-carotene and lutein. β-Carotene is known for its provitamin A activity and lutein for its anticancer activity against lung cancer (Di Mascio et al., 1991).
Phenolic compounds are known to possess antimicrobial and antiviral properties (Dixon, 2001; French and Neil Towers, 1992). Many phenolic compounds in tomato fruits have also been found (Slimestad and Verheul, 2009). Walker (1962) reported that caffeic, chlorogenic, p-coumaric, and ferulic acids are found in tomato fruits, and contents of caffeic and chlorogenic acids decrease through the fruit development process. Wardale (1973) reported that chloro-genic acid is the major phenolic compound contained in fruits of various tomato cultivars, accounting for 75% of the total phenolics in mature green fruit and 35% in ripe fruit.
Volatile organic compounds in tomato plants affect the behaviors of pests and pollinators of tomato (Buttery et al., 1987). Andersson et al. (1980) who identified mono- and sesquiterpenes in tomato leaves had previously reported the volatile compounds of tomato. Urbasch (1981) additionally identified hexanal, (E)-2-hexenal, (Z)-and (E)-β-ocimeneter-pinolene, linalool, neral, geranial, methyl salicylate, nerol, geraniol, and 2-tridecanone. Buttery et al. (1987) improved upon the detection and isolation of mono- and sesquiterpenes and aliphatic and aromatic compounds in tomato leaves.
Tomatine from tomato was firstly isolated by Fontaine et al. in 1948. Friedman and Levin (1998) identified dehydro- and 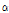 -tomatine in various parts of tomato plant. These tomato glycoalkaloids are of interest because of their implication in host-plant resistance. In particular,
-tomatine in various parts of tomato plant. These tomato glycoalkaloids are of interest because of their implication in host-plant resistance. In particular, 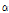 -tomatineis expected to protect tomato leaves against attack by micro-organisms (Morrissey and Osbourn, 1999). Sandrock and Van Etten (1998) reported that growth of eight saprophytic fungi and the tomato pathogens Stemphylium solani 11128 and Verticillium dahlia were greatly inhibited by
-tomatineis expected to protect tomato leaves against attack by micro-organisms (Morrissey and Osbourn, 1999). Sandrock and Van Etten (1998) reported that growth of eight saprophytic fungi and the tomato pathogens Stemphylium solani 11128 and Verticillium dahlia were greatly inhibited by 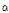 -tomatine.
-tomatine.
Some studies indicated that in edible parts of vegetables are available because they contain higher contents of secondary metabolites than edible parts. For instance, the leaves of strawberry and carrot include higher contents of phenolic compounds than edible parts (Kähkönenet al., 1999; Kim et al., 2013). The previous researches, however, have analyzed the contents of secondary metabolites in ripe fruits of tomato plants but few studies have reported the metabolites in inedible parts such as leaves, internodes, unripe fruits, and roots. The objective of this study was to identify the carotenoids, phenolic compounds, volatile organic compounds, and alkaloids present in leaves, internodes, fruits, and roots of tomato plants.
Materials and Methods
Plant Materials
‘Bacchus’ tomato plants (Monsanto Korea, Chochiwon, Korea) were transplanted in a greenhouse located in Gwangju (N 35.26, E 126.74; Korea) on August 19, 2011. The plants were fertigated with Yamasaki nutrition solution (N; 7.0 me・L-1, P; 2.0 me・L-1, K; 4.0 me・L-1, Ca; 3.0 me・L-1, Mg; 2.0 me・L-1, Fe; 15.38 mg・L-1, B; 1.14 mg・L-1, Mn; 0.81・mg L-1, Zn; 0.09 mg・L-1, Cu; 0.04 mg・L-1, Mo; 0.01 mg・L-1) using an automatic drip fertigation system (pH; 5.5-6.0, EC; 2.0-2.5 dS・m-1) and were pinched off at the two leaves above the 6th flower truss. Roots, 18th (above the 4th flower truss), 21th (above the 5th flower truss), and 24th (above the 6th flower truss) leaves and internodes, and green, pink, and red fruits were harvested on May 15, 2012.
Fruits were classified into three ripening stages by the percentage of red color: 0% (green); more than 30% but not more than 60% (pink); more than 90% (red). After classification, samples were stored at -20°C until phytochemical analysis.
Analysis of Carotenoids
The hexane extraction method was performed as in Sadler et al. (1990). Samples (4 g) were weighed into 125 mL flasks and the flasks were sealed with aluminum foil to block light. A hexane:acetone:ethanol (50:25:25) solvent (100 mL) was added to the flask and then agitated for 10 min. Finally, water (15 mL) was added, followed by another 5 min agitation. The solution separated into distinct polar (65 mL) and nonpolar (50 mL) layers. The carotenoids in the upper hexane layer were measured using a spectro-photometer (UV-2550, Shimadzu, Kyoto, Japan). The lycopene, β-carotene, and lutein contents of each sample were then estimated using absorbance at 503, 455, and 446 nm, respectively.
Analysis of Phenolic Compounds
Phenolic compounds were extracted and hydrolyzed according to the process described in Nuutila et al. (2002). Different parts (5 g) of the tomato plant were homogenized with 5 mL of 1.2 M HCl in 50% (v/v) aqueous methanol. Ascorbic acid (8 mg) was added to the mixture as an antioxidant. After agitating at 35°C for 16 h, the extract was cooled, made up to 10 mL, and sonicated for 3 min. The extract was filtered through a 0.45 μm syringe filter before injection to the HPLC apparatus (Ultimate 3000, Dionex, Sunnyvale, CA, USA). The mobile phase involved a 20-60% gradient of methanol in water with 300 mL・L-1 trifluoroacetic acid, eluted at a flow rate of 0.8 mL・min-1 for 25 min. After each analysis, the column was washed with 100% methanol for 2 min, returned to 20% methanol, and re-equilibrated for 10 min before the next analysis. The eluted components were monitored using a UV/Vis detector at 280 and 340 nm with a Zorbax SB C-18 column, 150 × 4.6 mm, i.d. 5 μm (Agilent Co., New York, NY, USA). Analytical standards were trans-cinnamic acid, p-coumaric acid, caffeic acid, chlorogenic acid, gallic acid, ellagic acid, p-hydrobenzoic acid, 2,5-hydrobenzoic acid, 3,4-hydrobenzoic acid, vanillic acid, kaempferol, quercetin, and morin.
Analysis of Volatile Organic Compounds
Volatile organic compounds were determined using the procedure described by Isleten and Karagül-Yüceer (2008). The sample gas was gathered by Tenax-TA (PerkinElmer Life and Analytical Sciences Inc., Waltham, MA, USA), a dynamic thermal extractor chamber system, which consists of a 26 mL glass tube and an air control system, containing an air supply unit and pumps. Purified nitrogen gas (39 mL・min-1) was consumed for ventilation. Volatile organic compounds in different parts of tomato plant were analyzed by thermal desorption gas chromatograph mass spectrometry (TDS-GC MSD and TDS2, Gerstel GmbH & Co. KG, Mülheim, Germany; 6890N and 5975, Agilent Technologies, Inc. Santa Clara, CA, USA).
Analysis of Tomatines
Tomatines were isolated and identified by the method of Friedman and Levin (1998). Samples (1 g in 20 mL of 1% acetic acid) were extracted by stirring for 2 h. The suspension was centrifuged at 13,300 RCF for 10 min. The supernatant was filtered through a Whatman GF/C filter and the pellet was resuspended, centrifuged, and filtered. The two extracts were combined. The extract was purified using SPE. C18 SPE tubes were conditioned with methanol (5 mL) followed by water (5 mL). The extract (about 30 mL) was allowed to gravity drip. When the sample was fully absorbed, the tube was washed with about water (10 mL), followed by 5 mL of 30:70 acetonitrile/1% NH4OH, and then water (5 mL). The dehydrotomatine and 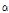 -tomatine were eluted with 10 mL of 70:30 acetonitrile/1 mM HCl. The sample was dried on a rotovapor and the residue was taken up in 1 mL of 50% methanol/0.1% acetic acid. The two samples were combined and filtered through a 0.45 μm HV membrane before HPLC injection.
-tomatine were eluted with 10 mL of 70:30 acetonitrile/1 mM HCl. The sample was dried on a rotovapor and the residue was taken up in 1 mL of 50% methanol/0.1% acetic acid. The two samples were combined and filtered through a 0.45 μm HV membrane before HPLC injection.
Commercial tomatine (Tokyo chemical industry Co., Tokyo, Japan) was separated into dehydrotomatine and 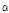 -tomatine by preparative HPLC using UV detection. Conditions were asfollows: 3 mL of eluent/min was passed through a 150 × 4.6 mm, i.d. 5 μm, Zorbax SB C-18 column; the eluent consisted of 25% acetonitrile and 100 mM ammonium phosphate brought to pH 3 with phosphoric acid. Tomatine (2 mg in 1 mL of 50% methanol and 0.1% acetic acid) was injected to the column; the two peaks were collected from the UV detector, which was observed at 200 nm. The structures of the dehydrotomatine and
-tomatine by preparative HPLC using UV detection. Conditions were asfollows: 3 mL of eluent/min was passed through a 150 × 4.6 mm, i.d. 5 μm, Zorbax SB C-18 column; the eluent consisted of 25% acetonitrile and 100 mM ammonium phosphate brought to pH 3 with phosphoric acid. Tomatine (2 mg in 1 mL of 50% methanol and 0.1% acetic acid) was injected to the column; the two peaks were collected from the UV detector, which was observed at 200 nm. The structures of the dehydrotomatine and 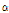 -tomatine were confirmed by mass spectrometry. The HPLC eluent for tomatine analysis was prepared by combining buffer (100 mL) with polished water (550 mL), acetonitrile (200 mL), and methanol (150 mL). The concentrated buffer was prepared by combining disodium phosphate (28.97 g) and citric acid (93.72 g) in water (1 L). This buffer was filtered through a 0.45 μm nylon membrane, passed through a 3 × 1 cm bed of Chelex 100, and then passed through a C18 SPE. The chromatography column was a 150 × 4.6 mm, i.d. 5 μm, Zorbax SB C-18. Flow rate was set to 3 mL・min-1. Wavelength was monitored at 200 nm.
-tomatine were confirmed by mass spectrometry. The HPLC eluent for tomatine analysis was prepared by combining buffer (100 mL) with polished water (550 mL), acetonitrile (200 mL), and methanol (150 mL). The concentrated buffer was prepared by combining disodium phosphate (28.97 g) and citric acid (93.72 g) in water (1 L). This buffer was filtered through a 0.45 μm nylon membrane, passed through a 3 × 1 cm bed of Chelex 100, and then passed through a C18 SPE. The chromatography column was a 150 × 4.6 mm, i.d. 5 μm, Zorbax SB C-18. Flow rate was set to 3 mL・min-1. Wavelength was monitored at 200 nm.
Statistical Analysis
Statistical analyses were performed using SAS statistical software version 9.2 (SAS Institute Inc., Cary, NC, USA). Duncan’s multiple range test was used to assess differences in contents of carotenoids, phenolic compounds, and alkaloids. P ≤ 0.05 was considered statistically significant.
Results and Discussion
The Contents of Carotenoids
Lycopene was only detected in the red fruits, with a content of 196.15 μg・g-1 FW (Table 1). Kozukue and Friedman (2003) and Lenucci et al. (2006) reported that lycopene content ranged from 58 mg・kg-1 of fresh weight in ‘Momotaro’ cultivar to 253 mg・kg-1 of fresh weigh in ‘Kalvert’ cultivar. Accumulation of lycopene generally begins at the ‘breaker’ stage after tomato fruit has reached the ‘mature green stage’. Lycopene is mainly included in red-ripe tomato fruits (Approximately 80 to 90% of total pigment) (Lenucciet al., 2006). Ronen et al. (1999) reported that mRNA of CrtL-b which encodes lycopene β-cyclase and CrtL-e which encodes lycopene ε-cyclase decrease at the ‘breaker’ stage. The content of β-carotene was 23.23, 20.13, 14.60, 3.37, 1.54, 1.50, and 1.19 μg・g-1 FW in 24th leaves, 21th leaves, 18th leaves, red fruits, 21th internodes, 24th internodes, and 18th internodes, respectively (Table 1). β-carotene content in 24th leaves was 6.89 times greater than that in red fruits. The content of lutein was 25.61, 23.91, 17.91, 5.03, 1.91, 1.80, 1.47, and 1.06 μg・g-1 FW in 24th leaves, 21th leaves, 18th leaves, red fruits, 21th internodes, 24th internodes, 18th internodes, and pink fruits, respectively (Table 1). Lutein content in 24th leaves was 5.09 times greater than that in red fruits. Fraser et al. (1994) also showed that tomato leaf contains β-carotene and lutein but not lycopene. Lycopene plays an attractant role in seed dispersal that can affect the further propagation (Moco et al., 2007) and β-carotene and lutein, major carotenoids in green leafy vegetables, are potent antioxidants (Jiménez-Escrig et al., 2000). The results show that leaves and red fruits of tomato contain more antioxidants and attractants, respectively.
The Contents of Phenolic Compounds
Caffeic, chlorogenic, and vanillic acids were detected in all tested parts except root (Table 2), while trans-cinnamic acid, p-coumaric acid, gallic acid, ellagic acid, p-hydrobenzoic acid, 2,5-hydrobenzoic acid, 3,4-hydrobenzoic acid, kaempferol, quercetin, and morin were not founded. Chen et al. (2006) and Slimestad and Verheul (2009) reported that chlorogenic acid and its derivatives are the main simple phenolics in tomato leaves and fruits. Slimestad and Verheul (2009) reviewed that chlorogenic acid content ranged from 1.7 mg・kg-1 of fresh weight in ‘Izabella’ cultivar to 32.8 mg・kg-1 of fresh weigh in ‘Liso’ and ‘Senior’ cultivars. Results from the present study also show that the content of chlorogenic acid was higher than that of caffeic and vanillic acids. The content of chlorogenic acid was 40.11, 30.34, 25.91, 15.63, 9.95, 9.72, 8.69, and 7.69 μg・g-1 FW in 18th leaves, 21th leaves, 24th leaves, 24th internodes, 18th internodes, green fruits, 21th internodes, and red fruits, respectively (Table 2). Chlorogenic acid content in 18th leaves was 5.22 times greater than that in red fruits. The difference of the contents of the chlorogenic acid in leaves and internodes may be due to their senescence late. Chlorogenic acid that presents in tomato leaves related to insect resistance decreases growth and development of several tomato herbivores (Elliger et al., 1981; Stamp and Yang, 1996). Moreover, the compound is produced in response to green bug (Todd et al., 1971) and black rot infection in resistant sweet potato roots (Akazawa and Wada, 1961; Uritaniand Akazawa, 1955). Chlorogenic acid also has numerous beneficial properties related to potent antioxidant activities such as hepato-protective, hypoglycemic, and antiviral activities (Farah and Donangelo, 2006). Chlorogenic and caffeic acids are oxidized to more toxic quinones by polyphenol oxidase from the host or parasite, which then are polymerized to insoluble non-toxic melanins. Orthoquinones are very unstable, but the supplement of amino acids to the reactive center after orthophenol oxidation might prevent polymeri-zation and could explain how the quinones provide disease resistance (Clack et al., 1959; Johnson and Schaal, 1957). The contents of caffeic and vanillic acids in 24th leaves were 9.18 and 1.64 μg・g-1 FW, respectively, and was greater than that of the older leaves and other parts (Table 2). Brown et al. (2003) reported that young leaves of Arabidopsis have various secondary metabolites compared to old leaves, probably due to the re-allocation of secondary metabolites to decrease feeding by herbivores. Young leaves of strawberry plants also included greater content of phenolic compounds than old leaves (Kim et al., 2013). Antimicrobial activity of caffeic acid has been tested against various fungi and bacteria (Harrison et al., 2003; Kim et al., 2012; Rauha et al., 2000; Widmer and Laurent, 2006). The results show that leaves of tomato include more antimicrobial and antihervibore phenolic compounds than other parts.
The Contents of Volatile Organic Compounds
‘Bacchus’ tomato plants included 40 volatile organic compounds (Table 3). The contents of total volatile organic compounds were 444, 206, 152, 111, 108, 82, 37, 30, 26, and 18 mg・m-3 (fresh tissue) in red fruits, pink fruits, 24th leaves, 18th leaves, 21th leaves, green fruits, 18th internodes, 21th internodes, 24th internodes, and roots, respectively. The 2-butenal produced by the aldol condensation of ace-taldehyde was the predominant volatile organic compound in red fruits; however, it was not detected in roots, leaves, internodes, and pink fruits. Leaves included six monoterpenes such as alpha-terpinene, alpha-phellandrene, beta-phellandrene, di-limonene, alpha-pinene, and sabinene. Buttery et al. (1987) reported that the leaves of the red cherry tomato contain eight monoterpenes (2-carene, myrcene, terpinolene, alpha-pinene, limonene, alpha-phellandrene, beta-phellandrene, and alpha-terpinene) and three sesquiterpenes (δ-elemene, caryophyllene, and humulene). The content of beta- phellandrene in 18th leaves and sabinene in 24th leaves was higher than that in other parts. Tomato plants lay in volatile organic compounds in their glandular trichomes and the compounds are emitted by damage of the trichomes (van Schie et al., 2007). Herbivores increase the emission of the compounds from tomato trichomes because they have the ability to damage the trichomes (Gibson, 1971). In addition, sesquiterpene (alpha-humulene) was only detected in the 18th internodes. Degenhardt et al. (2003) reported that mono- and sesquiterpene are volatile compounds that attract herbivore enemies when plants were damaged by herbivores. The results show that the red fruits of tomato contained the highest contents of total volatile organic compounds, and the leaves of tomato contained the most diverse volatile organic compounds to attract herbivore enemies.
The Contents of Alkaloids
The content of dehydrotomatine was 889.11, 852.33, 817.05, 358.29, 269.72, 242.71, 59.11, 11.32, 7.89, and 7.51 μg・g-1 FW in 24th leaves, 21th leaves, 18th leaves, 24th internodes, 21th internodes, 18th internodes, roots, green fruits, pink fruits, and red fruits, respectively (Table 4). The content of 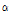 -tomatine was 1417.90, 1321.30, 1275.01, 307.36, 214.35, 167.52, 56.59, 43.02, 15.52, and 13.10 μg・g-1 FW in 24th leaves, 21th leaves, 18th leaves, 24th internodes, 21th internodes, 18th internodes, roots, green fruits, pink fruits, and red fruits, respectively (Table 4). The contents of dehydro- and
-tomatine was 1417.90, 1321.30, 1275.01, 307.36, 214.35, 167.52, 56.59, 43.02, 15.52, and 13.10 μg・g-1 FW in 24th leaves, 21th leaves, 18th leaves, 24th internodes, 21th internodes, 18th internodes, roots, green fruits, pink fruits, and red fruits, respectively (Table 4). The contents of dehydro- and 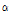 -tomatine were greatest in the leaves, followed by the internodes, roots and fruits. Younger leaves and internodes contained greater contents of dehydro- and
-tomatine were greatest in the leaves, followed by the internodes, roots and fruits. Younger leaves and internodes contained greater contents of dehydro- and 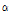 -tomatine than older leaves and internodes. Friedman and Levin (1998) reported that leaves of tomato contained greatest dehydro- and
-tomatine than older leaves and internodes. Friedman and Levin (1998) reported that leaves of tomato contained greatest dehydro- and 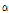 -tomatine and the compounds in immature green fruits are partly degraded as the fruit development. Dehydro- and
-tomatine and the compounds in immature green fruits are partly degraded as the fruit development. Dehydro- and 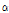 -tomatine in plants serve as natural defenses against fungi, bacteria, insects, and mammals. Especially,
-tomatine in plants serve as natural defenses against fungi, bacteria, insects, and mammals. Especially, 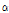 -tomatine that disrupts cell membrane by lysing of liposome has been implicated in plant defense (Morrissey and Osbourn, 1999). Boulogne et al. (2012) reviewed insecticidal and fungicidal activities of tomatine against Macrosiphum euphorbiae, Leptinotarsa decemlineata, Melanopus bivittatus, Heliothis zea, Hyposoterexiguae, Spodoptera exigua, Tribolium castaneum, Sitophilus oryzae, Fusarium solani, Nomuraea rileyi, and Fusarium oxysporum. The results showed that young leaves of tomato include more pesticidal alkaloids than other parts.
-tomatine that disrupts cell membrane by lysing of liposome has been implicated in plant defense (Morrissey and Osbourn, 1999). Boulogne et al. (2012) reviewed insecticidal and fungicidal activities of tomatine against Macrosiphum euphorbiae, Leptinotarsa decemlineata, Melanopus bivittatus, Heliothis zea, Hyposoterexiguae, Spodoptera exigua, Tribolium castaneum, Sitophilus oryzae, Fusarium solani, Nomuraea rileyi, and Fusarium oxysporum. The results showed that young leaves of tomato include more pesticidal alkaloids than other parts.
Pichersky and Gang (2000) reported that the ability to produce secondary metabolites has been selected throughout the process of evolution in different plant line ages when the compounds addressed specific needs. For instance, floral scent volatiles and pigments have evolved to attract pollinators and thus increase fertilization rates. The ability to synthesize toxic metabolites has evolved to ward off microorganisms and herbivores (from bacteria and fungi to insects and mammals) or to suppress the growth of other plants. The metabolites found in fruits prevent spoilage, and their color, aroma, and flavor signal the presence of potential rewards such as sugars, vitamins, and amino acids to animals who eat the fruit and thereby help seed dispersal. Other chemicals serve cellular functions that are unique to the particular plant in which they occur, such as resistance to salt or drought. We confirm that inedible parts (e.g., leaves, internodes, and unripe fruits) of tomato plants mainly included toxic compounds to microorganisms and insects (chlorogenic and caffeic acid, and tomatines) and edible parts contained pollinator attractants (lycopene and volatile compounds). Thus, after harvesting of fruits, the inedible parts of tomato plants can be used as a raw material for antioxidants, anti-inflammatory agents, fungistats, and pesticides.


 -tomatine
-tomatine