Introduction
Materials and Methods
Plant Materials
Cultivation Method
Investigation of Above- and Below-ground Characteristics
Investigation of Physiological Disorder, Disease, and Insect Pest Damage
Determination of Salt Resistance
Extraction and Analysis of Ginsenoside
Statistical Analysis
Results
Qualitative Characteristics of the Above-and Below-ground Parts
Emergence, Flowering, and Berry Maturation Periods
Growth Characteristics of the Above- and Below-ground Parts
Growth Characteristics of Kowon Seeds
Resistance to Diseases and Insect Pests
Comparative Physiology of Salinity and Heat Stress
Root Yield and Contents of Ginsenosides
Discussion
Introduction
Ginseng (Panax ginseng Meyer) is a perennial plant that is grown exclusively in the shade. It is difficult to be cultivated and its growth rate differs significantly with environmental conditions (Choi et al., 1980). Upon transplantation of superior seedlings to the field, ginseng can flower in 2 years (Chung et al., 1989), bearing fruits with only 2 to 5 seeds. Generally, flowering begins in year 3, although the plants still do not display mature phenotypes. Mature phenotypes appear in year 4, if growth progresses normally, implying that one generation of ginseng is 4 years old (Kwon et al., 1998). Thus, breeding of a new cultivar requires a considerable time.
Each 4-year-old plant yields approximately 40 to 60 seeds. Because the proliferation ratio is extremely low at approximately 10:1, the distribution rate of a new cultivar is only 13.0% (Kim et al., 2013). In addition, at low temperatures, ginseng seeds become dormant for approximately 90 to 100 days (Kim et al., 2015). In summary, breeding a new ginseng cultivar requires a long time due to the limited number of seeds produced and their dormancy. In 2002, ‘Chunpoong’ and ‘Yunpoong’ were registered as the first and second ginseng cultivars, respectively (The plant variety protection database, 2001a and 2001b).
At a value of USD 1.55 million in 2015, ginseng was the number one agricultural export of Korea (MAFRA, 2016). Owing to higher incomes and increased interest in health and wellbeing, the production and consumption of ginseng continues to increase each year. However, stable ginseng production began to decline because of global warming, which causes heat and salt stress along with various diseases (Jo et al., 2016). To overcome this problem, it is important to develop superior cultivation methods and cultivars. To date, the development of ginseng cultivars predominantly focused on the quantity and ease of processing of red ginseng. It is critical to develop additional cultivars that display enhanced tolerance to biotic and abiotic stress to stabilize ginseng production in a changing environment.
In the present study, we described the breeding process and main agronomic characteristics of a new cultivar of ginseng, ‘Kowon’.
Materials and Methods
Plant Materials
Seeds were obtained from four-year-old Korean ginseng plants grown in the breeding field of the Ginseng Division, Department of Herbal Crop Research, National Institute of Horticultural and Herbal Science, Rural Development Administration (RDA), Eumseong, Chungbuk. From 2009 to 2013, the characteristics of the ‘Kowon’ cultivar were compared to those of the control cultivars, ‘Chunpoong’ and ‘Yunpoong’, grown in Chungbuk province (Eumseong), Gyeongbuk province (Youngju), and Gangwon province (Cheorwon), Republic of Korea.
Cultivation Method
Seeds harvested on October 30, 2009, were cultivated to the seedling stage for one year and transplanted to the main fields at a planting density of 7 rows × 10 columns/90 × 180 cm in 2010. Shading nets comprised of 3 layers of blue and 1 layer of black nets were installed before the emergence of seedlings. In June 2010, two black layers were added over the nets to prevent heat injury. The cultivation process, including pre-planting treatment and pest management, complied with the Ginseng Good Agricultural Practices (GAP)(NIHHS, 2009). The plants were cultivated in sandy clay loam and analysis of the physicochemical properties of the soil was conducted according to the soil analysis method prescribed by the National Institute of Agricultural Sciences, Korea (NIAST, 2000).
Investigation of Above- and Below-ground Characteristics
Above- and below-ground properties, such as quantity, physiological disorder, pest resistance, cultivation management methods, and cultivar characteristics of 4-year-old plants were evaluated according to the Ginseng GAP (NIHHS, 2009) and the International Union for the Protection of New Varieties of Plants (UPOV) inspection standards (UPOV, 2005). The aboveground properties consisted of six qualitative characters (stem color, leaf color, leaf shape, shape of vertically cut stem, fruit color, and leaf color at senescence) and seven quantitative characters (stem length, stem diameter, leaf length, leaf width, leaf area, number of stems, and peduncle length). The below-ground properties included root length, root diameter, root weight, main root length, and number of lateral roots.
Investigation of Physiological Disorder, Disease, and Insect Pest Damage
Physiological disorders and diseases were investigated by visual inspection of the above- and below-ground sections of 100 individuals, according to the inspection standards prescribed by the RDA (RDA, 2012). The above-ground physiological disorder, manifested as leaf discoloration, and the below-ground physiological disorders, categorized into rusty roots and roots with rough skin, were distinguished into six grades based on the percentage of plants displaying physiological disorders per 100 individuals. Grade 0 indicated absence of the disorder, and grades 1, 3, 5, 7, and 9 indicated disorders below 1%, between 1-10%, between 10-25%, between 25-40%, and over 40%, respectively.
To check the above-ground diseases, such as Alternaria blight, anthracnose, gray mold, and Phytophthora blight, and the below-ground diseases, root rot was inspected in the fields. Ginseng pathogens can be distinguished easily in the field because the time of occurrence and symptoms are different. The disease symptoms were divided into six grades, based on the percentage of diseases plants per 100 individuals. Grade 0 indicated the absence of disease, and grades 1, 3, 5, 7, and 9, respectively, indicated disease below 1.0%, between 1-5.0%, between 5.0-10.0%, between 10.0-20.0%, and over 20.0%.
Determination of Salt Resistance
To test the salt resistance of ginseng, one leafstalk was cut from 20 plants, placed in a solution that had an electrical conductivity (EC) of 5 dS·m-1 and a salt composition of 21 mM KNO3, 6.1 mM KH2PO4, and 6.2 mM MgSO4·7H2O for 15 days, and subsequently examined following the method of Kim et al. (2008).
Determination of Heat Resistance
To measure heat resistance, three seedlings were transplanted to a pot in three replicates. Two months later, the pots were placed in a temperature-controlled chamber at 46°C for 1 h, a treatment that stops respiration in ginseng. After 1 d, the plants were examined for survival rate and chlorophyll fluorescence reaction. The fluorescence reaction was tested according to Nedbal et al. (2000) and Park et al. (2010) using an image fluorometer (Handy FluorCam, Photo System Instruments, Drasov, Czech Republic). After testing the chlorophyll fluorescence reaction, Fo, Fm, Fp, and Ft were measured and used for determining Fm/Fo, Fv, Fv/Fm, Fp-Fs (Fd), Rfd, and Fd/Fp.
Chlorophyll fluorescence parameters were defined as follows (Gorbe and Calatayud, 2012): Fo, minimal chlorophyll fluorescence intensity measured in the dark-adapted state when all PS II RCs are open; Fm, maximal chlorophyll fluorescence intensity measured in the dark-adapted state during the application of a saturating pulse of light; Fp, maximum fluorescence
value under saturating illumination when all reaction centers are closed or in reduced state; Ft, fluorescence at time t after onset of actinic illumination; Fv, variable chlorophyll fluorescence (Fm-Fo) measured in the dark-adapted state when nonphotochemical processes are at a minimum; Ft, fluorescence at time t after onset of actinic illumination; Fv/Fm, maximum quantum yield of PSII photochemistry measured in the dark-adapted state; Rfd, fluorescence decrease ratio. In this case, Fs is obtained after illumination with continuous saturating irradiance. Rfd is an indicator of the photosynthetic quantum conversion and is correlated with CO2 fixation rates.
Extraction and Analysis of Ginsenoside
We performed a comparative analysis of the ginsenoside content in the roots of 4-year-old plants of ‘Kowon’, ‘Chunpoong’, and ‘Yunpoong’ cultivars. Each root was washed, cut into pieces of the same length, snap frozen in liquid nitrogen, and dried for five days in a freeze-dryer. The dried ginseng roots were ground into a fine powder using a ball mill. Root powder (100 mg) was mixed with 1.5 mL of 70% methanol in a 2 mL tube, sonicated for 30 min, and incubated in a water bath at 60°C for 30 min. The samples were vortexed for 7 days at room temperature, and then centrifuged at 15,000 ×g for 10 min. The supernatants were filtered through 0.45 μm polytetrafluoroethylene syringe filters (Advantec DISMIC-13HP, Toyo Roshi Kaisha, Ltd., Tokyo, Japan). Ginsenosides were analyzed using a 1200 series HPLC system (Agilent Technologies, CA, USA) equipped with a Kinetex XB (C18) column, 100 × 4.6 mm with a 2.6 μm particle size (Phenomenex, Torrance, CA, USA). The HPLC analysis was carried out at a flow rate of 1.0 mL·min with a column oven temperature of 40°C and a wavelength of 203 nm. The solvent system employed was (A) ultra-pure water (+0.0005% formic acid) and (B) 100% acetonitrile. The gradient program used was as follows: 0 min, 18% B; 0.1-8 min, 20% B; 8.1-10 min, 22% B; 10.1-18 min, 33% B; 18.1-23 min, 33% B; 23.1-33 min, 50% B; 33.1- 42 min, 80% B; 42.1-47 min, 100% B; 47.1-52 min, 18% B. For each sample, ginsenoside (1 mg ·mL-1) was used as an external standard. The quantification of different ginsenosides was based on peak areas and was calculated as equivalents of representative standard compounds. The contents were expressed as milligram per gram dry weight (DW).
Statistical Analysis
Significance tests were performed by applying Duncan’s multiple range test (DMRT) using SAS (version 9.2, SAS Institute Inc., Cary, NC, USA). A p-value < 0.05 implied that the case was statistically significant. The data are presented as mean ± SD of three sample replicates.
Results
To cultivate a high-quality ginseng cultivar with stable production characteristics, ‘Kowon’ seeds collected from a farming house of Suwon in Korea in 1999 were sown. In 2003, elite samples were selected from 4-year old plants. Evaluation of the cultivation and propagation characteristics were conducted from 2003 to 2007. The line name ‘Korea No. 1’ was assigned based on the observed yield trials in the G03136-3 line from 2008 to 2010. Local adaptability tests were performed from 2011 to 2013. ‘Korea No. 1’ was acclaimed for retaining a high yield and resistance to Alternaria blight. It was named ‘Kowon’ and was registered as a new cultivar in November 2013. The breeding history of ‘Kowon’ is illustrated in Fig. 1.
Fig. 1.
Pedigree diagram of the new ginseng cultivar, ‘Kowon’. The Korean ginseng selection, breeding, and development project began in 1999 with a focus on collecting the phenotypic variance of the aerial part, Panax ginseng Meyer. The evaluation of the cultivation characteristics and propagation were conducted from 2003 to 2007 and then a yield test (observed yield trials) was conducted for three years (2008-2010). ‘Korea No.1’ consistently performed well for three years (2011-2013) at the three locations of the local adaptability test (LAT) in Korea, and it was released as ‘Kowon’.
Qualitative Characteristics of the Above-and Below-ground Parts
The color of ‘Kowon’ leaves is green during the growth period and red when the plant is old, unlike the yellow color of leaves in ‘Chunpoong’, the control cultivar. ‘Kowon’ has broad elliptical leaves with plain leaflets, unlike the narrow elliptical leaves and concave leaflets in ‘Chunpoong’. Although ‘Kowon’ has yellowish-green stems, it displays a light purple color around the petioles and the base. It produces fruits that have a red-colored pericarp, which is clearly distinguishable from the orange-colored pericarp of ‘Chunpoong’. ‘Kowon’ has a semi-recurved umbel whereas that in ‘Chungpoong’ is horizontal. Leaf color, leaf color at senescence, leaf shape, stem color, fruit color, and inflorescences of ‘Kowon’ are similar to ‘Yunpoong’; however, ‘Yunpoong’ has more stipules and a stronger degree of serration of leaf margins than ‘Kowon’ (Fig. 2). The roots of ‘Kowon’ are cream in color whereas those of ‘Chunpoong’ are bright cream. ‘Kowon’ and the control cultivars possess a stolon near the rhizome. The main roots of ‘Kowon’ are cylindrical and intermediate in size between those of ‘Chunpoong’ and ‘Yunpoong’ (Fig. 3).
Fig. 2.
Qualitative characteristics of the above-ground parts of the three ginseng cultivars. Qualitative traits were examined for leaf shape, distribution of anthocyanin coloration of the stem, presence or absence of pigment in the petiole, fruit color, and pigmentation of autumn leaf. ‘Kowon’ and ‘Yunpoong’ have broad elliptical leaves with plain leaflets whereas ‘Chunpoong’ has narrow elliptical leaves and concave leaflets. Anthocyanin coloration on the stem in ‘Chunpoong’ is only present in the lower section. The color of berries and autumn leaves are orange. Leaf color, leaf color at senescence, leaf shape, stem color, fruit color, and inflorescence of ‘Kowon’ are similar to that of ‘Yunpoong’.
Fig. 3.
Qualitative characteristics of the below-ground parts among three ginseng cultivars. Qualitative traits are skin color of the main root, presence or absence of stolons, and shape of the main root. The roots of ‘Chunpoong’ are bright cream in color whereas ‘Kowon’ and ‘Yunpoong’ are cream. All of the cultivars possess stolons near the rhizome. The main roots of ‘Kowon’ are cylindrical (medium cylinder) and intermediate in size between those of ‘Chunpoong’ (long cylinder) and ‘Yunpoong’ (short cylinder).
Emergence, Flowering, and Berry Maturation Periods
A comparative analysis of the emergence, flowering, and berry maturation periods of the three cultivars was performed. A three year study revealed that ‘Yunpoong’ was the earliest to emerge on April 18 or 19, whereas ‘Chunpoong’ emerged last on April 21 or 22. The emergence period of ‘Kowon’ was between the two. The order of flowering and berry maturation period was the same as that of the emergence period.
Growth Characteristics of the Above- and Below-ground Parts
Among the 4-year-old ginseng plant cultivars, stems of ‘Kowon’ were 31.3 cm long, 3.6 cm shorter than that of ‘Chunpoong’ (34.9 cm) but 3.1 cm longer than that of ‘Yunpoong’ (28.2 cm). Stem diameter positively correlated with root quantity (Chung et al., 1995); it was greater for ‘Kowon’ than for the other two cultivars. The number of stems per plant was highest in ‘Yunpoong’ (1.3) followed by ‘Kowon’ (1.1) and ‘Chunpoong’ (1.0). Leaf length (17.5 cm) and width (7.3 cm) in ‘Kowon’ were both higher compared to the two control cultivars. The number of leaves in ‘Kowon’ (25.0) and ‘Yunpoong’ (25.2) was greater than that in ‘Chunpoong’. This is probably because ‘Kowon’ and ‘Yunpoong’ have more stipules than ‘Chunpoong’. The total leaf area per petiole in ‘Kowon’ (202.4 cm2) was higher than the two control cultivars (Table 1). Many studies have reported that photosynthesis is higher in ginseng with a larger leaf area, which ultimately increases the size of the root (Parmenter and Littlejohn, 2000; Hughes and Proctor, 1981; Jang et al., 2015), and the same is expected in ‘Kowon’. The results of the below-ground characteristics of ‘Kowon’ are presented in Table 2. The root length in ‘Kowon’ was 27.7 cm, 0.6 cm shorter than that of ‘Chunpoong’, and 3.3 cm longer than that of ‘Yunpoong’ (24.4 cm). There was no significant difference in the length of the main root between ‘Kowon’ and the control cultivars. The diameter of the main roots in ‘Kowon’ and ‘Yunpoong’ were comparable, although they were 4.6 cm thicker compared to ‘Chunpoong’. ‘Yunpoong’ possessed the maximum number of lateral roots (3.4) followed by ‘Kowon’ and ‘Chunpoong’. The fresh weight of roots from ‘Kowon’ was 46.8 g, which was 9.5 g higher than that of ‘Chunpoong’ (37.3 g).
Growth Characteristics of Kowon Seeds
Characteristics of ‘Kowon’ seeds and control cultivars were compared. The number of fruits in ‘Kowon’ was 58.1, slightly higher than that of ‘Chunpoong’ and ‘Yunpoong’ (56.0 and 54.6, respectively), although the differences were not statistically significant. The weight of 100 seeds of ‘Kowon’, ‘Chunpoong’, and ‘Yunpoong’ was 5.6, 5.3, and 4.4 g, respectively. The seeds of ‘Kowon’ and ‘Yunpoong’ were 0.9-1.2 g heavier than those of ‘Chunpoong’. Seed length (6.0 mm) and width (5.1 mm) were highest in ‘Kowon’. ‘Chunpoong’ seeds were the hardest at 15.7 kgf·cm-2. The stratification rate was highest in ‘Yunpoong’ at 86.3%, followed ‘Kowon’ (83.5%) and ‘Chunpoong’ (80.1%). These findings are in agreement with those reported by Kim et al. (2014), and suggest that the stratification rate decreases as the hardness of the seed increases. According to Lee et al. (2008), the productivity of ginseng seeds increases with their weight. Because ‘Kowon’ has heavier seeds than the other two cultivars, it is expected to be higher yielding.
Table 1. Growth characteristics of the above-ground parts of three ginseng cultivars.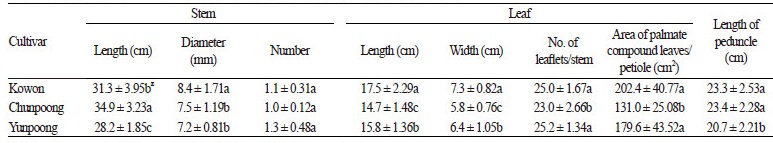 zDifferent letters (a, b) within a row indicate significant differences at p <0.05. |
Table 2. Growth characteristics of the below-ground parts of three ginseng cultivars. zDifferent letters (a, b) within a row indicate significant differences at p <0.05. |
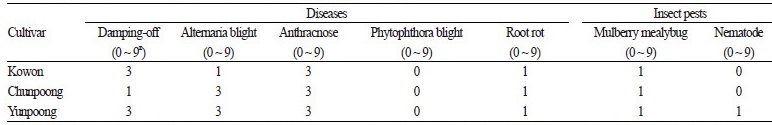
Resistance to Diseases and Insect Pests
The results of disease and pest infestation in ‘Kowon’ and control cultivars in the test fields are presented in Table 3. ‘Kowon’ displayed intermediate resistance against damping-off, anthracnose, and Alternaria blight, and these results were distinguishable from the control cultivars. Diseases, such as Phytophthora blight and root rot, and pests, such as mulberry mealybug and nematode infestation, were not observed, indicating a certain degree of resistance. Throughout the entire growth period, Alternaria blight is the most frequently occurring disease in all ginseng organs, including leaves, stems, and fruits (Lee et al., 2012).
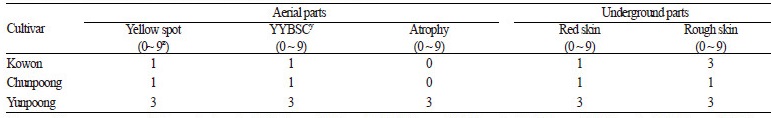
Comparative Physiology of Salinity and Heat Stress
Compared to ‘Yunpoong’, ‘Kowon’ had fewer incidences of leaf discoloration due to salt treatment, the frequency of which was approximately similar to ‘Chunpoong’ (Table 4). The below-ground part of ‘Kowon’ demonstrated frequent occurrences of rusty roots compared to ‘Yunpoong’. ‘Kowon’ and ‘Yunpoong’ had a similar number of occurrences of rough-skinned roots, which were higher than ‘Chunpoong’. The occurrence of physiological disorders in leaves impedes photosynthesis and assimilation. This restricts the transfer of nutrients to the below-ground portion and the roots cease to grow, thereby reducing their quantity. Rusty roots, roots with rough skin, and other physiological disorders in the below-ground parts result in a deterioration in quality, greatly reducing the potential profit (Lee et al., 2012). It is important to prevent physiological disorders, diseases, and pest problems as healthy growth of both above- and below-ground organs directly influences the quantity and quality of the ginseng produced.
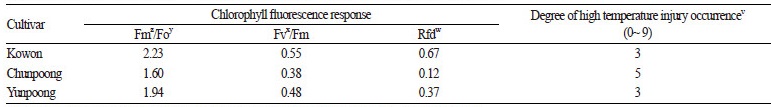
The results of fluorescence measurements made for assessing high temperature resistance in each ginseng cultivar are shown in Table 5. ‘Chunpoong’ was affected by high temperature; its leaves underwent withering and drooped down. On the other hand, ‘Yunpoong’ and ‘Kowon’ were relatively unaffected. Kim et al.(2013) used the values of fluorescence response parameters, Fm/Fo, Fv/Fm, and Rfd, as indicators for selecting heat resistant ginseng and reported the following values to be sufficient for each parameter: Fm/Fo ≥ 2.2, Fv/Fm ≥ 0.55, and Rfd ≥ 0.8. Comparison of the values of the fluorescence response parameters of the three cultivars showed that ‘Kowon’ had the highest values of 2.23, 0.55, and 0.67 for Fm/Fo, Fv/Fm, and Rfd, respectively, and satisfied two of the indicators (Fm/Fo ≥ 2.2, Fv/Fm ≥ 0.55) suggested by Kim et al. (2013). Considering the extent of high temperature damage and the values of the fluorescence response parameters, ‘Kowon’ is moderately resistant to high temperature.
Root Yield and Contents of Ginsenosides
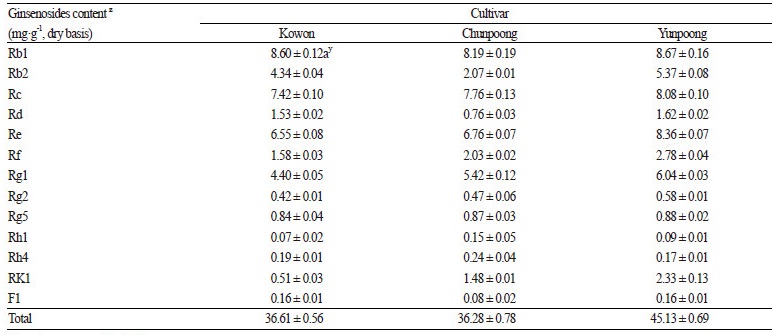
By comparing the productivities of control cultivars with ‘Kowon’ in three study regions, ‘Kowon’ was found to have the highest yield in all three regions, with an average of 666 kg·10a-1. This was 27 and 4% higher than that of ‘Chunpoong’ and ‘Yunpoong’, respectively. Of the key secondary metabolites in ginseng, the content of ginsenoside was highest (45.13 ± 0.69 mg·g-1) in ‘Yunpoong’, followed by ‘Kowon’ (36.61 ± 0.56 mg·g-1) and ‘Chunpoong’ (36.28 ± 0.78 mg·g-1). The Rb2 and Rd contents in ‘Kowon’ were 4.34 ± 0.04 and 1.53 ± 0.02 mg·g-1, respectively, which were higher than those of the control variety ‘Chunpoong’, but lower than those of ‘Yunpoong’ (Table 6). However, the concentration of F1 ginsenoside, known to have whitening effects, was confirmed to be 0.16 ± 0.01 mg·g-1, which was the same as that of ‘Yunpoong’. Overall, the levels of Rb1, Rb2, Rd, and F1 ginsenosides were confirmed to be higher in ‘Yunpoong’ than in ‘Chunpoong’, whereas the other ginsenosides were at comparable levels.
Discussion
Climate change is contributing to heat damage and disease outbreaks in the cultivated ginseng; it is therefore necessary to develop techniques and cultivars to reduce this damage. In the past, the yield and quality of ginseng were largely determined by the cultivation techniques and soil health. This has changed with advances in cultivation, with yield and quality now being determined by the use of genetically superior cultivars. Presently, 90% of the cultivated ginseng still comprises landrace cultivars. Because these landrace cultivars are not maintained as genetically pure lines, their yield and quality are lower than the genetically superior cultivars, even under the same cultivation conditions. This leads to difficulties in achieving stable production. Here we have demonstrated that ‘Kowon’ has a higher yield than the landrace cultivars, is more resistant to pests and physiological damage, and is superior in quality. Moreover, it is more reliable than the currently grown landrace cultivars, since it has passed tests for production rate and local adaptation during breeding, further implying that it will be better suited for achieving stable yield and quality.
Many individual ginseng breeding lines have been selected in Korea since 1968 to develop new cultivars with good quality and high yield. Sweveral promising lines were selected and ‘Chunpoong’ and ‘Yunpoong’ were registered as new ginseng cultivars by Kwon et al. (1998 and 2000). ‘Chunpoong’ displayed excellent physical characteristics (Kwon et al., 1998) and ‘Yunpoong’ was selected for its high-yielding properties (Kwon et al., 2000). To date, ginseng cultivars have been bred for good root shapes, high yield, and high saponin content (Kim et al., 2013). Recently, ginseng farmers are demanding cultivars with resistance to abiotic and biotic stresses exacerbated by climate change. Thus, it is essential to develop new cultivars which have resistance to heat stress and physiological disorders. The ‘Kowon’ cultivar has passed the local adaptation tests in five different locations. The results presented here confirm that it produces a higher yield and is more resistant to leaf spot disease compared to the landrace and the previously developed cultivars ‘Chunpoong’ and ‘Yunpoong’.
Prevention is the key for avoiding the occurrence of Alternaria blight; it is thus important to cultivate cultivars with resistance in order to reduce the frequency of its occurrence (Kim et al., 2010). ‘Kowon’ has shown resistance to Alternaria blight, and is thus predicted to stabilize production; this could be achieved if the ‘Kowon’ cultivar, with its excellent genetic traits, is distributed to farms for cultivation.