Introduction
Materials and Methods
Plant Materials
Measurement of Light Parameters
Analytical Measurements
Instrumentation
Statistical Analyses
Results and Discussion
Light Parameters
Yield
Ascorbic Acid
Pungency
Chlorophyll Fluorescence
Conclusion
Introduction
Chili pepper species (Capsicum spp.) originate from humid environments in Central and South America (Votava et al., 2005). Peppers with moderate pungency may be consumed as a fresh vegetable in addition to their typical use as a seasoning, and therefore can be a valuable source of both capsaicinoids and ascorbic acid in the human diet (DeWitt and Bosland, 2009). Capsaicinoids primarily activate the peripheral terminals of specific sensory neurons (i.e. pain receptors), induce a heat sensation (Caterina et al., 1997), and are also potentially effective against age–related memory loss (Yang et al., 2015). Several studies have described the effects that agricultural practices such as fertilizer use (Johnson and Decoteau, 1996; Guinoza et al., 2015) and irrigation (Estrada et al., 1999; Mahendra and Bandara, 2000; Sung et al., 2005) have on the pungency of chili peppers. In addition, biological factors such as plant genotype (Dubey et al., 2015; Mo et al., 2015), node position of the pod (Zewdie and Bosland, 2000), and ripening stage (Nagy et al., 2015) affect final chili pepper pungency.
Ascorbic acid is essential nutrient that supports the human immune system through its antioxidant properties (Lutsenko et al., 2002). The final ascorbic acid content in pepper is sensitive primarily to the environmental factors light intensity and temperature (Lee and Kader, 2000). Maximum ascorbic acid contents are found in fully ripened fruits (Bae et al., 2014); however, the absolute level of ascorbic acid varies among different pepper species and cultivars (Kumar and Tata, 2009).
Although numerous studies have described capsaicinoid and ascorbic acid contents in various pungent peppers and the beneficial health effects of these phytochemicals, there is a poor understanding of how certain agricultural Revise lines and spacing: practices - in particular net shading - influence pepper traits such as pungency and ascorbic acid content. Net shading is used to modify the light environment, thus affecting plant light-regulated processes (Shahak, 2008). Furthermore, the limitation of high light exposure via net shading is a well-established horticultural practice in sweet pepper cultivation (Ilić et al., 2011; Kong et al., 2013; Selahle et al., 2015). The report of Son and Oh (2015) indicates that blue light qualities (400-500 nm) have a large impact on secondary metabolite accumulation in lettuce. Abiotic stresses can have differential effects on the photosynthetic activity of pepper plants, and therefore chlorophyll fluorescence measurements can give an insight into plant health. The optimal photochemical quantum yield of PSII, meaning the Fv/Fm measured using a chlorophyll fluorescence meter, is expected to be 0.72-0.85 in plants that are not under stress (Bolhar-Nordenkampf et al., 1989). In agreement, a decrease in fluorescence was observed by Guang-cheng et al. (2011) in pungent pepper during water stress.
The objective of this study was to investigate the combined effect of various light treatments and variation in harvest time on the pungency, ascorbic acid content, and market-quality yield of peppers from two Capsicum annuum hybrids, namely the yellow ‘Star Flame’ and ‘Fire Flame’ chili peppers. Furthermore, we also investigated the physiological condition of pepper plants under the various experimental conditions by measuring chlorophyll fluorescence as a reflection of plant health.
Materials and Methods
Plant Materials
The study was conducted in 2014 at the experimental field of the Horticulture Institute, Szent István University, Gödöllő, Hungary (lat. 47°61′ N, long. 19°32′ E). The soil of the experimental field was sandy loam classified as Cambisol with 1.8-2.0% humus content and an approximate pH of 7. Three DL-121TH data loggers (Voltcraft®, Germany) were placed in different positions within the greenhouse to collect ambient temperature data. The daily average temperature was calculated as the mean of 24 temperature measurements made throughout the day (Fig. 1).
Fig. 1.
The average daily greenhouse temperature from July 1 to October 26, 2014. Pepper harvest times were on July 27, September 16, and October 26 indicated by vertical lines.
Two Capsicum annum L. hybrids, each with a distinct morphology, were investigated in this study: ‘Star Flame’, which produces fruit approximately 12 cm in length that develop vivid yellow coloring upon maturation, and ‘Fire Flame’, which is characterized by fruit 14-15 cm in length that possess a deep red color. Seeds for these two hybrids were purchased from Seminis® (Kecskemét, Hungary) and were sown on February 29, 2014, into propagation trays with holes. Once seedlings reached the 2- 4 leaf growth stage, they were planted out into individual pots 9 cm in diameter. Experimental seedlings were maintained under identical conditions in a heated greenhouse that had an average daytime temperature of 20°C. Plants were then transplanted into a 30 m (length) × 4 m (width) × 2.5 m (height) greenhouse on April 27 in twin rows with a spacing of 0.4 m between plants and rows, and 0.8 m between twin rows, therefore creating a planting density of 4.167 plants/m2. All plants received equal watering via drip-irrigation, equal fertilizing via weekly application of Megasol (Yara Co., Hungary) that contained 16% nitrogen (NO3, NH4, NH2), 8% phosphorus (P2O5), 24% potassium (K2O), and 8% sulphur (SO4), and equal measures to protect against insects and other pathogens throughout the study. Different sections of the greenhouse were covered with different colored nets to alter the light environment: namely red (ChromatiNet, Ginegar, Israel), white, and green (Első Magyar Kenderfonó, Hungary) nets. A non-shaded control section was also maintained in the same greenhouse which was not covered with a net. Each net was specified by the manufacturer to have a 40% shading ratio. Nets were placed perpendicular to the twin rows over a space 8 m in width, thus creating a block under which four biological replicates were randomly appointed. Ten plants were selected from each replicate, which were used to measure chlorophyll fluorescence, market-quality pepper yield, and phytochemical contents during the growing season. The fruit qualities from each replicate were analyzed in three technical replicates. The greenhouse had a north-south orientation that avoided any potential overlapping and shadowing effects among the nets, and plants in the middle-northern parts of the net shaded areas were analyzed, whereas those located close the border of the shaded areas were excluded. Furthermore, the daily sun circulation was observed before plant selection to avoid bias. Only healthy and fully ripened fruits that displayed typical hybrid characteristics were included to determine market-quality pepper yield. The total weight of market-quality fruits from four plants in each replicate during all three harvests (July 27, September 16, and October 26) were combined and expressed as kilograms per square meter.
Measurement of Light Parameters
We used a PAM 2500 portable fluorometer (Walz-Mess und Regeltechnik, Germany) to measure chlorophyll fluorescence in experimental plants. One measurement was made per week during a four-week period on a sunny day at noon before each harvest time, whereby four fully developed top leaves of a single plant from each replicate were affixed with leaf clips for a 35 min dark adaption before fluorescence measurement. The plants used for chlorophyll fluorescence measurement were chosen from those selected for phytochemical analysis to minimize bias from inconsistent serial measurements and to investigate the correlation between phytochemicals and Fv/Fm value. Maximum photochemical quantum yield of PSII was quantified using the Fv/Fm ratio determined by the fast kinetics method in the PamWin 3.0 software (Van Goethem et al., 2013). The photosynthetic photon flux density (PPFD) was measured on September 16 with a micro quantum sensor of a Leaf-clip Holder 2030-B (Walz- Mess und Regeltechnik, Germany) attached to a Pam 2500 fluorometer whose sensor was factory calibrated. The essential optoelectronic element of this micro quantum sensor was a filter combination that selects the photosynthetic active wavelength range of 380-710 nm. The PPFD measurements were performed just above average plant height. We measured the extent of shading in each experimental glasshouse section in the spectral range between 325-1075 nm using a portable spectroradiometer (ASD FieldSpec® HandHeld 2) on September 16. The instrument had a spectral resolution of < 3.0 nm at 700 nm and a wavelength accuracy of ±1 nm. The extent of experimental shading was measured using a white reference panel (USP 119), and light quantity measurements were performed outside the glasshouse and under each shading treatment in the space of one minute. Relative transmittance (in %) was calculated as the proportion of the light quantity outside the glasshouse maintained under each shading treatment.
Analytical Measurements
Capsaicinoid content, including the amounts of nordihydrocapsaicin (NDC), capsaicin (CAP), dihydrocapsaicin (DC), and other minor components (homocapsaicin derivatives and homodihydrocapsaicin derivatives), was determined following the method of Daood et al. (2015). Total capsaicinoid content was calculated as the sum of the individual compounds that appeared on the chromatogram. Three grams of homogenized pepper tissue (seed excluded) was crushed in a crucible mortar with quartz sand, after which 50 mL of analytical-grade methanol was gradually added before the mixture was carefully transferred to a 100 mL Erlenmeyer flask with a stopper. The mixture was subjected to ultrasonication in a bath ultrasonic device (Ultrasonic Cleaner, Raypa Trade, Turkey) for 3 min and then filtered through a filter paper (Munktell, Germany). The filtrate was further purified by passing it through a 0.22 mm PTFE (Chromfilter) syringe filter before it was injected into a HPLC column. For this, extracts were diluted appropriately and injected into a Nucleodur C18, Cross-Linked column (ISIS, from Macherey-Nagel, Duren, Germany). The separation was performed through isocratic elution using 50:50 water:acetonitrile and a flow rate of 0.8 mL/min. The fluorometric capsaicinoid detection was carried out at two wavelengths (EX: 280 nm and EM: 320 nm). Peaks corresponding to the different capsaicinoid compounds were identified by comparing retention times and mass data (Daood et al., 2015) created by analysing analytical standard material (purified from pungent red pepper, with 99% purity, by Plantakem Ltd., Sándorfalva, Hungary) with those resulting from experimental sample analysis.
Ascorbic acid contents were determined following the methods and HPLC protocols of Nagy et al. (2015). Five grams of homogenized pepper tissue (seed excluded) was crushed in a crucible mortar with quartz sand, after which 50 mL of 3% metaphosphoric acid solution was gradually added, before the mixture was transferred to a 100 mL Erlenmeyer flask with a stopper.
The mixture was filtered through a filter paper (Munktell, Germany) and then further purified by passing it through a 0.45 mm cellulose acetate (Wattman) syringe filter before it was injected into a HPLC column. For quantitative determination of ascorbic acid, sample data were compared to that generated using standard materials (Sigma-Aldrich, Budapest, Hungary).
Instrumentation
A Hitachi Chromaster HPLC instrument, which consists of a Model 5110 Pump, a Model 5210 Auto Sampler, a Model 5430 Diode Array detector, and a Model 5440 Fluorescence detector, was used to measure all compounds in the experimental samples.
Statistical Analyses
Statistical analyses were performed using IBM SPSS 22 software (IBM, USA). The effect of net shading on market-quality pepper yield was investigated using a linear model, in which pepper yield and shading were considered as the dependent variable and the explanatory variable, respectively. The effect of shading on chlorophyll fluorescence and contents of individual capsaicinoid compounds was investigated independently at each harvest time using a one-way linear model. We applied a linear model to investigate how various light treatments and harvest times affected the amount of ascorbic acid and total capsaicinoid content. Pairwise comparisons were made via a Tukey HSD post-hoc test. To check model assumptions, plot diagnostics were performed for each model. Pearson correlation tests were used to test the relationship between the above dependent variables (the most important explanatory variables in the recent study) such as ascorbic acid, total capsaicinoids, and chlorophyll fluorescence. The nutritional contents of hybrids were examined separately, because such properties are considerably different in red and yellow pepper cultivars as demonstrated by previous studies (Ghasemnezhad et al., 2011). Throughout the study, α was set to 0.05.
Results and Discussion
Light Parameters
The measuring of light parameters such as PPFD (Table 1) and relative transmittance in the photosynthetic active range (380- 710 nm, Fig. 2) gives an indication of the extent of shading resulting from the various colored nets combined with the greenhouse cladding material. The effect of different colored shadings on PPFD value was significant (F4,15 = 1752.60; p < 0.001). The highest PPFD value (712 ± 8.9 μmol·m−2·s−1) was measured in areas covered by greenhouse cladding material alone without net shading. The transmittance of light in the photosynthetic range (380-710 nm) was similar in areas under white and red shading net and also in the control treatment. The transmittance of light gradually increased with increasing wavelength. The light transmittance under white, red, and control (greenhouse cladding material alone) shading was approximately 60-72%, 42-60%, and 50-70%, respectively. The green net conferred a maximal light transmittance of 60% between 530-550 nm, and a low light transmittance rate (50-58%) compared to that of other shading nets for the longer wavelengths within the photosynthetic active range. We were also able to measure light transmittance at 325-380 nm, namely ultraviolet wavelengths that are shorter than the photosynthetic active range, which had approximately 35-40% transmittance under red and green shading nets, approximately 40-45% transmittance in the control treatment, and a higher transmittance of approximately 50% under the white shading net.
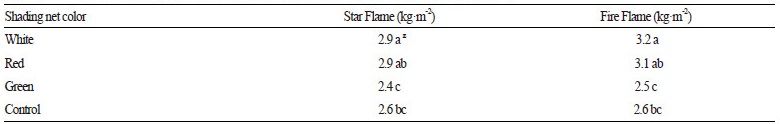
Table 1. The effect of shading net color on photosynthetic photon flux density (PPFD). zShared letters indicate no significant difference among values acording to Tukey HSD post-hoc tests atp = 0.05. |
Fig. 2.
The relative transmittance (%) of light through the experimental shading treatments. The measured wavelength range was 325–1075 nm and the photosynthetic active radiation (380-710 nm) is highlighted in grey.
Yield
We observed that both fruit quality components and pepper yield were influenced by net shading. In both of the examined hybrids, pepper yields were higher from plants grown under white and red net shading compared to that resulting from plants grown under either green net shading or control conditions (F3, 24 = 6.0, p = 0.003; Table 2). Furthermore, there was a similar effect regarding the average market-quality pepper yield in response to light treatments, with the highest yield observed from plants grown under white net shading. Similar yield effects were observed following red and pearl shading in sweet pepper, as reported by Ilić et al. (2011). In Hungary, green colored nets are the most commonly used shading material for pepper production (Ledó, 2011). However, our results with green net shading indicate that this light treatment caused a significant decrease in yield compared to that resulting from other light treatments. This yield decrease is probably due to the lower transmittance of light qualities in the upper photosynthetic range under green net shading compared to that under other net shadings. As stated by Son and Oh (2015), leaf cells effectively absorb red (600-700 nm) and blue (400-500 nm) wavelengths, and red light stimulates biomass production in plants such as lettuce. Thus, it is believed that green net shading considerably hinders red light exposure on plant leaves. In addition, Ombódi et al. (2015) measured PPFD in the photosynthetic active range under green net shading and found that it was reduced more than that under white and red net shading. As a result of less favorable environmental conditions for pod ripening, this reduction in PPFD could lead to lower pepper yields. According to Shahak (2008), net shading had a pronounced effect on pepper productivity during summer and early autumn months, which diminished at later stages in the season.
Ascorbic Acid
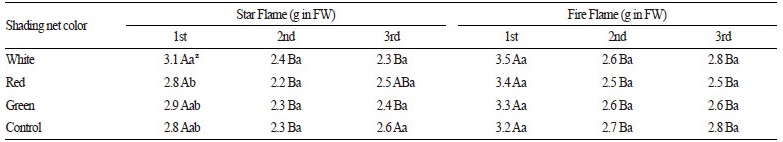
The ascorbic acid contents of ‘Star Flame’ peppers were significantly affected by the interaction of harvest time and shading (F6, 36 = 3.9, p = 0.004; Table 3). For all light treatments, fruits collected from the first harvest possessed higher ascorbic acid contents compared to that in fruits collected from the second harvest, which is in accordance with the findings of Kong et al. (2013), who emphasized that the effect of shading on ascorbic acid content may change with harvest times. The ascorbic acid contents of ‘Fire Flame’ peppers were significantly affected by harvest time (F6, 36 = 59.7, p < 0.001), as demonstrated by higher ascorbic acid contents in fruits collected from the first harvest compared to that in fruits collected from the second and third harvests. Before the first harvest, daily average temperature was 22-28°C (Fig. 1), which could have stimulated ascorbic acid biosynthesis. The increase in sunlight during the summer period may have had a positive effect on ascorbic acid contents, similar to that observed in previous studies whereby higher light exposure induced a higher amount of ascorbic acid in pepper (Mozafar, 1994). Although Ilić et al. (2011) found that the use of red net shading in greenhouse-cultivated sweet peppers could enhance ascorbic acid content compared to that in non-shaded peppers, such effects were not corroborated in the present study. The stimulating effect of white net shading on ascorbic acid contents was likely the result of the higher PPFD measured for this light treatment during summer. Our finding that white net shading stimulates ascorbic acid biosynthesis more than red net shading during the summer is supported by a prior report of the promotive effect of pearl net shading on ascorbic acid contents in stored red and yellow sweet peppers (Selahle et al., 2015).
Pungency
Some growers state that no-removing leaves improves the sugar content of fruit compared with CD. However, these growers typically combine NAD cultivation with other management methods, such as the addition of organic compost, proficient pruning, and maintenance of a suitable fruit load, thus introducing many other variables into cultivation. In the present study, only a slight increase in soluble solids content was observed in NAD-treated fruits, suggesting that to produce high-sugar fruits, growers must manage the leaves as well as manipulating other cultivation techniques.
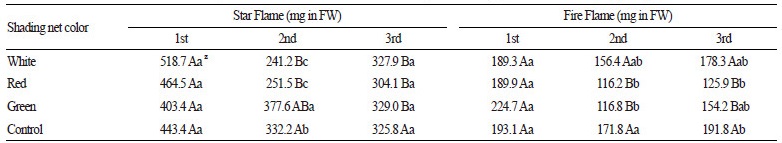
Similar to the effects described above on ascorbic acid content, net shading had a variable effect in different harvests on total capsaicinoid contents in both hybrids. In ‘Star Flame’ peppers, total capsaicinoid content was significantly affected by the interaction of shading treatment and harvest time (F6, 36 = 4.0, p = 0.004; Table 4). In fruits from plants grown under all shading treatments, total capsaicinoid contents were higher in those collected during the first harvest compared to that in fruits collected during the second and third harvests; however, no differences were observed between harvests for the control treatment. The higher average temperature of 22-28°C before the first harvest (July 27, Fig. 1) may have promoted capsaicinoid biosynthesis that has been shown to cause higher pungency in Capsicum fruits (Lindsay and Bosland, 1995). This hypothesis agrees with the findings of Otha (1960), who reported increased pepper pungency following higher night-time temperatures. In fruits collected during the second harvest, peppers from plants grown under green shading treatment had considerably higher total capsaicinoid contents compared to that in peppers originating from white and red shade-treated plants and control plants.
As described above, the different shading treatments were associated with notable differences in PPFD and the extent of light wavelength transmission. Green shading treatment resulted in variable light transmittance in the photosynthetic active range, with low transmittance of longer photosynthetic active wavelengths. Ombódi et al.(2015) found that PPFD in the photosynthetic active range was lower under green net shading than under white or red net shading, whereas this PPFD level was higher than that under control conditions. Our results combined with these previous findings could explain the variable effect of green net shading that leads to variation in pungency in the peppers resulting from this treatment.
In ‘Fire Flame’ peppers, total capsaicinoid content was significantly affected by both shading treatment (F3, 36=4.5, p = 0.009) and harvest time (F2, 36=15.3, p < 0.001), whereas the interactive effect of these two variables was marginally non-significant (F6, 36=2.4, p = 0.055). In ‘Fire Flame’ peppers, total capsaicinoid contents resulting from different shading treatments were similar in fruits collected during the two autumn harvests; however, differences were observed as a result of different shading treatments in fruits collected during the summer harvest. We found significantly higher total capsaicinoid contents in fruits from the control treatment compared that in fruits treated with red and green shading at the second harvest, and that in fruits treated with red shading at the third harvest. Gurung et al. (2011) demonstrated that a higher average temperature and increased solar radiation were associated with lower capsaicinoid content, yet we observed higher total capsaicinoid contents in fruits collected during the autumn harvests from plants under the non-shaded control treatment and white net shading. A further consequence of a lack of shading is the potential high-level water loss through evaporation from the plants leading to a water deficit. It was reported that soil moisture deficiency or limited water availability increased fruit pungency in other pepper varieties (Estrada et al., 1999; Mahendran and Bandara, 2000; Sung et al., 2005). According to available data in the literature, there is no evidence describing the relationship between distinct shading treatments and the extent of pepper pungency, and therefore it is difficult to explain the variable effects of shading treatments during different harvest times. It is possible that the amount of various secondary metabolites, such as capsaicinoids and ascorbic acid, is affected by the higher relative UV wavelengths transmitted under the white shading treatment and under non-shaded control conditions (Wink, 1988; Namdeo, 2007). According to Son and Oh (2015), blue light (400-500 nm) has a considerable impact on secondary metabolite accumulation in lettuce. We hypothesize that summer is the most suitable period to harvest peppers when considering secondary metabolite accumulation in pungent pepper hybrids.
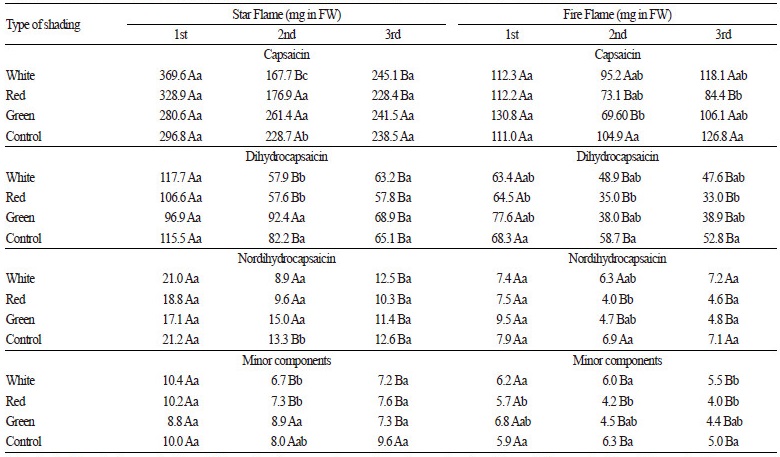
CAP and DC are the major compounds responsible for pungency in peppers and constitute up to 70% of total capsaicinoids in the Scotch Bonnet variety (Gahungu et al., 2011). Our analytical measurements revealed that, in addition to CAP and DC, NDC made up a notable proportion of total capsaicinoid content, and all three capsaicanoids were affected by shading treatments in both hybrids. In ‘Star Flame’ peppers, all capsaicanoids were influenced by the interaction of harvest time and net shading (F6, 36 2.5-4.3, p < 0.042 for each compound, Table 5), which was similar to the trend observed for total capsaicinoid content. In most cases, the peppers gathered during the first harvest returned higher pungency values, and the effect of shading treatments was observable only in peppers gathered during the second harvest. In ‘Fire Flame’ peppers, CAP and NDC contents were significantly influenced by the interaction of harvest time and net shading (F6, 36 = 7.9, p = 0.035 and F6, 36 = 2.6, p = 0.032, respectively). Under white net shading and control conditions, the amounts of CAP and NDC did not vary between fruits gathered from different harvests; however, red and green net shading was associated with significant differences in compound contents among harvests, with more CAP and NDC contents observed in fruits gathered during the summer harvest. The contents of DC and other minor components were affected by both harvest time (F2, 36=26.6, p > 0.001 and F2, 36 = 11.4, p > 0.001, respectively) and net shading treatment (F3, 36 = 4.2, p = 0.012 and F3, 36 = 5.4, p = 0.003, respectively). Comparable to in ‘Star Flame’ peppers, these compounds were significantly higher in fruits gathered during the first harvest than those collected during the autumn harvest. In the Serrano pepper variety, which produces fruit with a similar appearance and pungency to ‘Fire Flame’ peppers, Giuffrida et al. (2013) measured 707 ± 40 mg·kg-1 dry weight CAP contents. This level is approximately equivalent to 100 mg·kg-1 fresh weight CAP contents, which was measured in this study in ‘Fire Flame’ peppers grown under all shading treatments.
Chlorophyll Fluorescence
We used the maximum photochemical quantum yield of PSII measured as Fv/Fm to describe plant health prior to each of the various harvest times. For ‘Star Flame’, Fv/Fm differed between shading treatments at the first harvest (F3, 12 = 4.2, p = 0.031; Fig. 2), with lower Fv/Fm values measured in control plants compared to that in plants grown under any of the three shading treatments, although a significant difference was observed only between the control and green net shading. For ‘Fire Flame’, Fv/Fm values differed between shading treatments at the second harvest (F3, 12 = 5.1, p = 0.017; Fig. 3) as well as at the third harvest (F3, 12 = 5.6, p = 0.012), with differences observed in Fv/Fm values resulting from plants grown under control conditions and red net shading. The lowest Fv/Fm value was observed in plants prior to the summer harvest grown under non-shaded control conditions (0.720), which is considered to be a suboptimal value according to Bolhar-Nordenkampf et al. (1989). Previously, Hanying et al. (2001) showed that high temperature–induced stress inhibited photosynthetic activity in sweet pepper, which also supports the hypothesis that environmental stress caused suboptimal photosynthetic activity in experimental plants in this study.
Fig. 3.
The maximum photochemical quantum yield of PSII given as Fv/Fm of ‘Star Flame’ (left) and ‘Fire Flame’ (right). Shared letters indicates no significant difference in values between different shading treatments according to Tukey HSD post-hoc test at p = 0.05 (n=4).
Fig. 4.
Correlation between maximum photochemical quantum yield of PSII given as Fv/Fm and total capsaicinoid contents in ‘Star Flame’ (upper panel, r = 0.47, N = 48, p < 0.001) and ‘Fire Flame’ (lower panel, r = -0.45, N = 48, p <0.001).
Fig. 5.
Correlation between maximum photochemical quantum yield of PSII given as Fv/Fm and total capsaicinoid contents in ‘Star Flame’ (upper panel, r = -0.47, N = 48, p < 0.001) and ‘Fire Flame’ (lower panel, r = -0.45, N = 48, p < 0.001).
Fm values differed between shading treatments at the second harvest (F3, 12 = 5.1, p = 0.017; Fig. 3) as well as at the third harvest (F3, 12 = 5.6, p = 0.012), with differences observed in Fv/Fm values resulting from plants grown under control conditions and red net shading. The lowest Fv/Fm value was observed in plants prior to the summer harvest grown under non-shaded control conditions (0.720), which is considered to be a suboptimal value according to Bolhar-Nordenkampf et al. (1989). Previously, Hanying et al. (2001) showed that high temperature–induced stress inhibited photosynthetic activity in sweet pepper, which also supports the hypothesis that environmental stress caused suboptimal photosynthetic activity in experimental plants in this study.
Fv/Fm values and total capsaicinoid contents were significantly negatively correlated with each other in both ‘Star Flame’ (r = -0.47, N = 48, p < 0.001) and ‘Fire Flame’(r = -0.45, N = 48, p < 0.001), suggesting that a lower Fv/Fm value was associated with higher capsaicinoid content, and that peppers under environmental stress produced fruit with higher pungency. Ascorbic acid and total capsaicinoid contents were significantly positively correlated in ‘Fire Flame’ alone (r = 0.65, N = 48, p < 0.001). Topuz and Ozdemir (2007) found that higher capsaicinoid content was related to higher ascorbic acid content in hot red pepper, which is similar to the correlation observed here in the red-fruit pepper and absent in the yellow-fruit pepper. There was no significant correlation between ascorbic acid and Fv/Fm values in either ‘Star Flame’ or ‘Fire Flame’.
Conclusion
In conclusion, it may be a valid suggestion for growers to harvest during summer, particularly in combination with white net shading, in order to obtain high ascorbic acid contents in both ‘Star Flame’ and ‘Fire Flame’ peppers. However, it is important to consider that shading may contribute to a seasonal variation in pepper nutritional contents. Furthermore, for peppers harvested in autumn, either no or green net shading should be employed to improve the pungency of ‘Star Flame’ peppers. Finally, to improve pepper yield, both white and red shading may be integrated into the greenhouse cultivation of both ‘Fire Flame’ and ‘Star Flame’ hybrids.