Introduction
Materials and Methods
Seed Material
Temperature Treatment
Cold Stratification Period
GA3 Treatment
Data Collection and Analysis
Results and Discussion
Effects of Temperature on Seed Germination
Effects of Cold Stratification Period on Seed Germination
Effects of GA3 Treatment on Seed Germination
Introduction
Spiraea L. belongs to the Rosaceae family and comprises 50-80 species distributed throughout northern temperate regions (Potter et al., 2007). These ornamental plants produce colorful flowers and are highly ecologically adaptable, making them ideal for horticulture in many countries. For example, the North American native species S. tomentosa and S. alba are used primarily for landscaping and habitat restoration (Mickelbart et al., 2012). S. fritschiana (fritsch spirea), valued for landscaping, is native to the mountains and valleys of South Korea. This species has potential as a hedge shrub and a cut flower with its short height and abundant small flowers (compound corymb) (Kim and Sun, 1996), however little research has been conducted to examine the cultivation of S. fritschiana.
The success of mass propagation in woody plants is highly dependent on the propagation method chosen. Propagation by cuttings of woody plants can often be more complex and more restricted by harvest time than parallel applications in herbaceous plants. When plants are required for a rapid coverage of slopes or habitat restoration, seed propagation can be much less labor intensive than cutting propagation, however each Spiraea species has different temperature range requirements for optimum seed germination. In the eight cultivars of Japanese spirea (S. japonica) over 94% of seeds germinated in field conditions, whereas germination of S. chinensis did not occur when grown in night/day temperatures of 15/25, 20/30 or 25/35°C (10/14 h), nor under field conditions (Kim et al., 2002; Wilson and Hoch, 2009).
Seeds of Rosaceae species often require cold stratification to break seed dormancy and increase germination, and their growth can be aberrant and slow without it (Baskin and Baskin, 1998). In S. prunifolia for. simpliciflora, stratification at 3°C for 7 d increased the seed germination (Cho et al., 2012). During cold stratification, the balance of growth factors in seeds changes considerably. The abundance of growth promoters such as gibberellic acid (GA) is elevated in seeds subjected to low temperatures of 4-5°C (Yamauchi et al., 2004), and cold stratification also increases tissue sensitivity to GA (Derkx and Karssen, 1993).
GA functions to overcome the mechanical restriction conferred by tissues that surround the embryo, such as the aleurone and the testa, and increases the growth potential of the embryo (Ogawa et al., 2003). Exogenous GAs including GA3, GA4, and GA7 increase the success of germination in seeds of Rosaceae species (De Mello et al., 2009); for example, around 62% of Prunus serotina seeds soaked in 500 ppm GA3 germinated, whereas germination was only half as successful in seeds soaked in distilled water after 120 d of cold stratification (Phartyal et al., 2009). The inhibitory effect of a GA biosynthesis inhibitor on germination was partially reversed by treatment with GA4, and an increase in GA4 content in embryos led to germination (Chen et al., 2007).
To find efficient ways to increase and enhance the germination of S. fritschiana for mass propagation, revegetation, or habitat restoration, we investigated the optimal germination temperature, duration of cold stratification, and GA treatment, with the aim of improving the germination of S. fritschiana seeds.
Materials and Methods
Seed Material
S. fritschiana seeds were harvested in Geumwonsan Ecological Arboretum, Guchang, Korea (latitude 35°43′19.9″N, longitude 127°47′00.6″E) in October 2013. The seeds were air-dried at room temperature (22-25°C) for about one month then stored in sealed plastic jars at 5°C. Seeds were sorted by hand to ensure size uniformity. The mean seed weight was 0.013 g (n = five replicates, 300 seeds per replicate). The mean seed size was 1.44 × 0.33 mm (n = 50).
Temperature Treatment
Seeds were germinated at constant temperatures of 5, 10, 15, 20, 25, 30, or 35°C in a multi-room incubator (HB-302S-2, Hanbaek Scientific Co., Bucheon, South Korea) and illuminated by fluorescent lamps (FCL32SD/30, Kumho Electric, INC., Seoul, South Korea) with a 12 h photoperiod (20 μmol·m-2·s-1). All seeds were incubated in 9 cm Petri dishes containing two layers of filter paper (Whatman No. 2) moistened with distilled water that was warmed to each experimental temperature to eliminate water temperature effects. A seed was considered to be germinated when the radicle protruded through the seed coat (> 1 mm). Germinated seeds were recorded and then removed throughout the 30 d experiment. Each experimental treatment used 50 seeds, with three replications per treatment.
Cold Stratification Period
Seeds were sufficiently moistened on 9 cm Petri dishes containing two layers of filter paper (Whatman No. 2) and incubated at 5°C for 0, 6, 8, 10, 12, or 14 weeks in the dark. After cold stratification, the seeds were germinated at constant temperatures of 25 and 30°C. Seed germination tests were conducted using the method described above.
GA3 Treatment
Seeds were soaked in 0 (distilled water), 500, or 1000 mg·L-1 GA3 at a constant temperature of 25°C for 24 h. After soaking, the seeds were rinsed several times with tap water. All seeds were germinated at constant temperatures of 25 or 30°C. Seed germination tests were conducted using the method described above.
Data Collection and Analysis
The germination characteristics were examined the final germination percentage (FGP), mean germination time (MGT), and the mean number of days for 30% germination (T30). MGT was calculated as MGT = Σ (ti × ni )/Σ ni , where ti is the time (d) from the beginning of the test and ni is the number of germinated seeds at the recorded time (Ellis and Roberts, 1981). The entire germination test was conducted in a completely randomized block design. Data were subjected to ANOVA and Duncan’s multiple range test. Statistical analysis was performed using SAS version 9.2 (SAS Institute Inc., Cary, NC, USA). A p value < 0.05 was considered to indicate a statistically significant result. Regression and graph module analyses were performed using Sigma Plot software version 10.0 (SPSS Inc., Chicago, IL, USA).
Results and Discussion
Effects of Temperature on Seed Germination
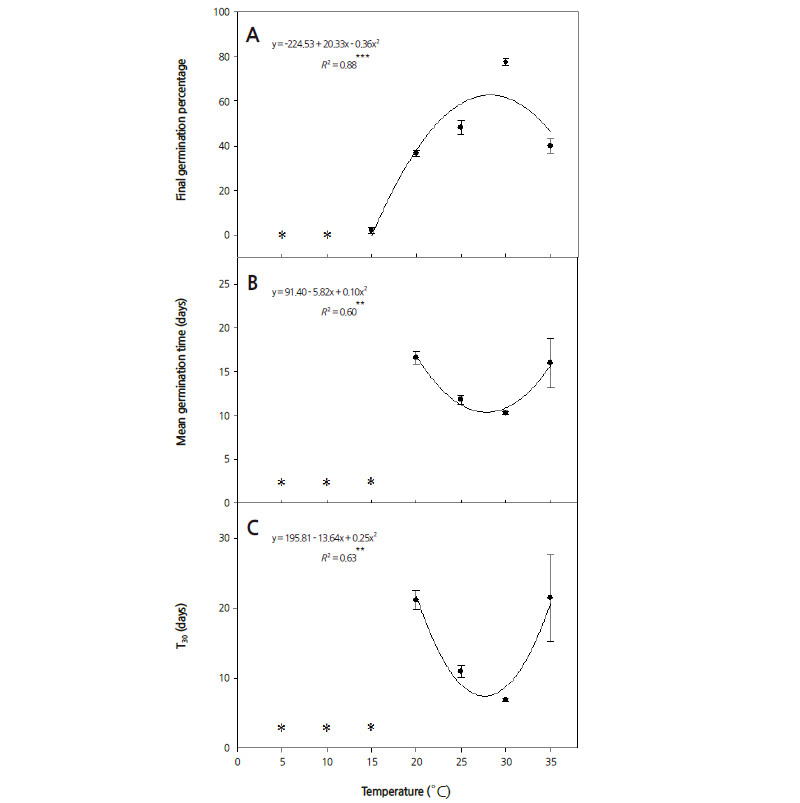
The close relationships between temperature and FGP, MGT, and T30 are illustrated in Fig. 1, with R 2 ranging from 0.60 to 0.88 (p < 0.01 or 0.001). Seeds of S. fritschiana did not germinate at 5 and 10°C, and MGT and T30 were not calculated at 5, 10, and 15°C (Fig. 1). FGP increased as the temperature for seed germination increased up to 30°C. The lowest MGT was obtained at 25-30°C (Fig. 1B). The fastest T30 occurred when seeds were germinated at 30°C (Fig. 1C). The optimal temperature for seed germination is considered as the temperature yielding the highest germination percentage in the shortest time. These results indicate that the optimal temperature for germination of S. fritschiana seeds was about 30°C, although the maximum germination percentage was below 80%. High germination temperatures may be preferred because S. fritschiana is adapted to occupy forest edges and rocky areas with greater exposure to sunlight and higher temperatures (Korea NIBR., 2014; Matlack, 1993).
Some species have a specific temperature range for seed germination because temperature acts to regulate germination under natural conditions and prevents germination under unfavorable thermal conditions for seedling growth. Seeds of a few shrubs native to deciduous forests, including Rosa rugosa and Rubus idaeus, germinate well at 20-30°C (Baskin and Baskin, 1998).
Effects of Cold Stratification Period on Seed Germination
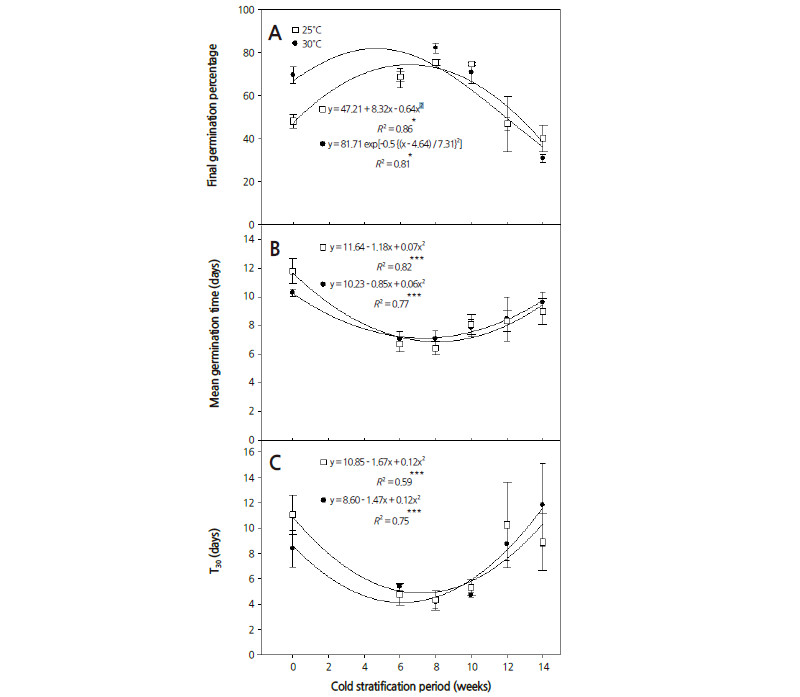
The germination of cold-stratified seeds was investigated at sub-optimal (25°C) and optimal (30°C) temperatures. FGP improved as the period of cold stratification increased up to a peak at 6 weeks at both germination temperatures (Fig. 2A). There were significant relationships between the cold stratification period and MGT and T30 (Fig. 2B, C; p < 0.001), as the cold stratification hastened germination of S. fritschiana seeds. These results suggest that seeds that fall in the autumn and overwinter under the snow will germinate rapidly at relatively high temperatures, which is a favorable condition for seedling development.
Woody species generally require extensive cold stratification treatments, which can be up to 180 d although 60-90 d is usually satisfactory (Bewley and Black, 1994). S. betulifolia seeds germinate at 0-2°C when kept under cold stratification conditions for more than 120 d (McLean, 1967), however in the present study, FGP of S. fritschiana seeds declined after 8 weeks of cold stratification. In some S. fritschiana seeds, dormancy had been induced by the prolonged cold stratification, preventing germination during winter (Milberg and Andersson, 1998).
Effects of GA3 Treatment on Seed Germination
Commercial seeds exhibit a high germination percentage, but their germination tests are conducted at optimal temperatures (Demir et al., 2011). Germination varies depending on the environmental conditions, which are not always ideal for seed germination in the field, however seeds for commercial use are required to achieve over 90% germination regardless of optimal temperature. To increase the germination percentage of S. fritschiana, exogenous GA3 was applied to the seeds.
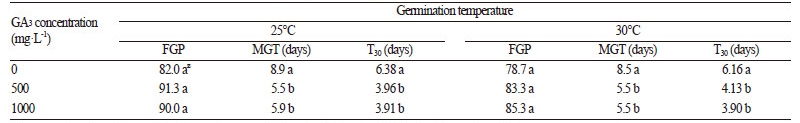
The application of GA3 often stimulates the germination of seeds belonging to Rosaceae, such as P. serotina (Phartyal et al., 2009). In the present study, the germination of S. fritschiana seeds increased from 48 to 82% by soaking them in distilled water at 25°C (Fig. 1A and Table 1), however soaking the seeds in GA3 solution did not improve FGP more than distilled water alone. The MGT of seeds treated with either 500 or 1000 mg·L-1 GA3 was 5.5 and 5.9 d respectively, which was lower than the 8.9 d MGT of seeds soaked in distilled water at 25°C (Table 1). The T30 of seeds treated with 500 mg·L-1 GA3 at 25°C was approximately 1.6 times faster than the 6.38 d taken by seeds soaked in distilled water. The similar results were obtained when seeds treated with 0, 500, and 1000 mg·L-1 GA3 germinated at 30°C.
Enhancing the germination and uniformity of seeds is required for mass production; however, wild-type seeds can have large variations in germination percentage, which results in non-uniform germinability (Qu et al., 2005). Variations in seed germination may have important ecological consequences, because a plant that produces seeds with different germination requirements has a higher chance of leaving descendants than one that does not (Fenner, 1991). In the present study, both cold stratification and GA3 treatment hastened the MGT of S. fritschiana. Exogenous application of GA3 has previously been substituted for cold stratification to germinate seeds in a short period of time (Kitchen and Meyer, 1991).
In conclusion, the optimal germination temperature is approximately 30°C for S. fritschiana seeds. Pretreatment with 6-8 weeks of cold stratification or by soaking seeds in distilled water or 500 mg·L-1 GA3 for 24 h accelerated and increased the germination of S. fritschiana seeds regardless of temperature. Further research might be useful to evaluate the effect of low concentrations of GA3 solution, such as 10, 20, 50, or 100 mg·L-1 on the promotion of germination in S. fritschiana seeds. Prolonged cold stratification can induce dormancy and result in a decline in germination. These results provide useful information for predicting germination characteristics of Spiraea species.