Introduction
Materials and Methods
Plant Material and Treatment Strategy
Quality Indices and Physiological Disorder Assessment
Phenolic Compound Analysis and Enzyme Assay
Results and Discussion
Pre-Drying and SBI
Delayed Cooling and SBI
CO2 and SBI
CO2 Effects on Total Phenols, PPO, and POD Activity
Introduction
Skin blackening (SB) is a major physiological disorder that occurs in commercially cultivated Asian pear (Pyrus pyrifolia Nakai) cultivars, such as ‘Niitaka’ and ‘Shinsetsu’, and it lowers fruit marketability (Choi et al., 1995; Choi et al., 2014; Kuo and Yiu, 2006; Seo et al., 2001). SB mainly develops during cold storage and it has been reported that its occurrence can be reduced by post-harvest measures, such as heat treatment before cold storage and curing through field stacking (Choi et al., 1995; Park and Kwon, 1999). However, detailed studies on the causes and mitigations of SB disorder in newly bred
Asian pear fruits, like the late-season cultivar ‘Chuhwangbae’, are still lacking.
Cold storage is essential for long-term maintenance of fruit quality. However, it can cause a variety of chilling injuries in peach fruit; woolliness and leatheriness symptoms occur at low temperatures, resulting in diminished quality (Neves et al., 2013). Cold acclimation and delayed storage retarded those symptoms effectively; then, low temperature storage preserved quality and prolonged shelf life (Li et al., 2012; Neves et al., 2013). The superficial scald that occurs after low temperature storage in apple fruits is a result of chilling injury (Watkins et al., 1995). In litchi fruits, external visual quality quickly declined at room temperature after fruits were removed from storage at 2°C, due to browning. To prevent this, application of low temperature acclimation was reported to suppress browning by delaying polyphenol oxidase (PPO) activity (Jiang and Li, 2003). Pretreatment before cold storage is effective for increasing shelf life and suppressing physiological disorders in Western pear cultivars ‘Bartlett’ and ‘Spadona’. Short-term low-O2 treatments before storage reduce chilling injury, such as superficial scald, by reducing α-Farnesene production and lowering ethylene and CO2 levels (Pesis et al., 2014).
Generally, the occurrence of browning in plant tissues has been known to be induced by PPO (EC1.10.3.1), which catalyzes the oxidation process of o-diphenols in the pericarp, and o-quinone compounds or their derivatives thereof, which form melanin pigments by non-enzymatic polymerization (Kollias et al., 1991; Nicolas et al., 1994; Seo et al., 2001). The polyphenols in pear (Pyrus spp.) fruit are mainly distributed in the skin and major compounds were identified as arbutin and chlorogenic acid (Lin and Harnly, 2008); polyphenols gradually decrease with fruit maturation (Cui et al., 2005). Seo et al. (2001) noted a quantitative difference in chlorogenic acid between the SB-sensitive Asian pear ‘Niitaka’ and non-sensitive ‘Kamchunbae’.
The purpose of this study was to investigate the effects of pre-drying and delayed cooling before cold storage (1°C) to mitigate SB incidence (SBI), and to reveal the influence of carbon dioxide (CO2) on the disorder. These results will suggest a viable alternative to alleviate the SB disorder in specific Asian pear fruits that are stored and exported in a sealed state at a low temperature for an extended period.
Materials and Methods
Plant Material and Treatment Strategy
‘Chuhwangbae’ pear (Pyrus pyrifolia Nakai) fruits were harvested 177 days after full bloom (October 9, 2016) from a private orchard in Nonsan City, Chungnam Province, Korea. The harvested fruits were immediately transported to the laboratory at Chungnam National University and were kept in fruit bags at 25°C for three days until they were used for the experiments.
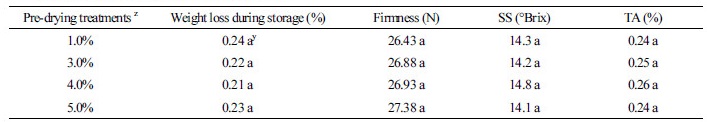
In the pre-drying experiments, we applied the drying treatment on the harvested fruits after removing the double-layer paper bags, using a blowing fan for six days at 25°C. Individual fruits were weighed during this period and classified into four groups: 1, 3, 4, and 5% weight loss, on average. Thereafter, the fruits were stored in a low temperature incubator (HB-103LP, Hanbaek Scientific, Korea) at 1°C for four weeks; we compared SBI and fruit quality. Each treatment was replicated three times, and the number of fruits in each treatment ranged from 36 to 40. SBI was measured according to the percentage of the whole fruit surface that was spotted (Fig. 1).
In the delayed cooling experiment, we applied a temperature acclimation (TA) treatment to the fruits to suppress the SB disorder. The harvested fruits were stored at 20°C for four days without refrigeration, and then three cooling treatments were applied: 1) 48-h cooling delay, 2) 24-h cooling delay, and 3) immediate cooling. The time to reach the final target storage temperature (1°C) was set to six days after cooling started, for each treatment. Fruits were stored in paper carton for export (5 kg, 9 to 10 fruits) for four weeks at 1°C using a low temperature incubator (HB-103LP, Hanbaek Scientific, Korea) and we compared SBI and other fruit quality indices. Each treatment was replicated eight times, and the number of fruits in each treatment ranged from 75 to 76.
To investigate the effect of CO2 treatment on SBI, fruits were placed in a specially made transparent plastic box (15L) equipped with inlet and outlet plugs. The boxes were stored (1°C) and subjected to CO2 or air (control) treatment for 20 days. The air mixed with a 2% concentration of CO2 was flushed at a rate of 1.0 L·min-1 through the front inlet to the rear outlet plug. The CO2 concentration was measured daily with a gas chromatograph (YL 6100-GC, Younglin, Korea) equipped with a thermal conductivity detector. After storage, we compared SBI, and collected samples for analysis of polyphenol components and related enzymes between sound and disordered tissues.
Quality Indices and Physiological Disorder Assessment
We measured the fresh weight of fruits using a digital scale (CB-3000, AND Korea, Cheongju, Korea), and flesh firmness, at two points on the equator after removing skin, with a rheometer (8 mm tip, TMS-Pro, Food Technology Corp., USA). The maximum pressure was measured under the condition of 5 mm sample move with 100 mm·min-1 speed. To determine soluble solids and titratable acidity, fruit juice was prepared by cutting the flesh of the fruit’s equatorial plane to a thickness of 10 mm. The soluble solids content was measured using a digital refractometer (PR-32a, ATAGO, Japan). For titratable acidity, 5 mL of juice was diluted with 35 mL of distilled water, and neutralized to pH 8.3 with 0.1 N NaOH, and then converted to malic acid concentration. Skin color difference (L*, a*, and b ) of the equatorial plane was measured using a chroma meter (CR-410, Minolta, Japan) to calculate the angle. The SBI score was based on the percent of damaged skin as shown in Fig. 1: 0, no occurrence; 1, < 1%; 2, < 10%; 3, < 30%; 4, 30-50%; and 5, > 50%). In the experiments, we used the percent disorder index (PDI) to calculate overall incidence (Iram et al., 2014). Briefly, we calculated total sum of numerical ratings after being multiplied by 100, and divided the value by the number of observations and the maximum number of ratings.
Phenolic Compound Analysis and Enzyme Assay
We prepared samples for protein extraction from tissue-cut sections of 2 mm thickness including skin, using a cork borer (12 mm diameter) for the unaffected tissues (SBI-0) and damaged ones (SBI-3 and -5). The quantification of total phenolics was performed per Costeng and Lee (1982), with slight modification. Briefly, 5 g of the tissue was placed in 20 mL 99% ethanol and homogenized at full speed for 1 min using a homogenizer (Ultra-Turrax T25, IKA-Labortechnik, Staufen, Germany). After homogenizing, we boiled the sample in a hot water bath for 10 min and then filtered it (Whatman #1, USA). The residues were further extracted three times with 80% ethanol and we obtained a total of 100 mL of the extracts. The reaction medium composition consisted of 1 mL sample extracts, 8 mL deionized water, and 1 mL phenol reagent (Folin & Ciocalteus). After 10 min, 1 mL 25% Na2CO3 solution was added and the absorbance was measured with a spectrophotometer at 725 nm after 2 h. Standard substances were quantified using (+) - catechin.
We also analyzed individual polyphenols in the fruit tissues. 0.3g of ascorbic acid and 30 mL methanol were added to 5 g of the sample (2 mm thickness, including skin) and homogenized for 5 min in an ice bath. The homogenized sample was centrifuged at 15,000 rpm for 5 min at 5°C, and the supernatant was taken. After that, we added 20 mL methanol to the remaining residue, and stirred the mixture well. The centrifugation was repeated to make 50mL of the extract. The extract was concentrated using an evaporator at 40°C until we obtained a volume of 10 mL; then, 20 mL n-hexane was added and the solution was shaken to remove lipid components. We used the water layer for analysis of phenolic components by high performance liquid chromatography (HPLC) (LC 10-AD, Shimadzu, Japan) and Shim-pack CLC ODS (4.0×10 mm) with a guard column (4.0×10 mm). Pretreatment with C18 Sep-pak before injection into HPLC was followed by filtration through a 0.22 µm microfilter (Millipore, USA). HPLC analysis was performed under the following conditions. Standard phenolic compounds were prepared by dissolving arbutin, gallic acid, (+)-catechin, chlorogenic acid, benzoic acid, (‑)-epicatechin, homovanillic acid, caffeic acid, and p-coumaric acid in methanol. Column temperature was 37 °C and solvent was gradient elution with 0.01 M KH2PO4 (pH 2.0) and methanol. Detection was performed at 280 nm with a UV-visible variable detector (Shimadzu, Japan).
Polyphenol oxidase (PPO) activity was determined per Wong et al. (1971), with partial modification. We prepared samples for protein extraction by placing 1 g of the tissue in 10 mL 20 mM phosphate (pH 6.5, 0.4 g polyvinylpolypyrrolidone) and homogenizing at full speed for 1 min. The crushed material was used as crude enzymes after centrifugation (12,000 rpm, 20 min at 2°C). The assay system consisted of 3 mL 50 mM phosphate buffer (pH 6.5), 70 mM catechol, and 0.1 mL enzyme. In a control tube, the enzymes were replaced with water. PPO activity was determined by monitoring the increase of absorbance at 420 nm within 10 min at 37 °C. One unit of enzyme activity was regarded as a change in absorbance of 0.001 per minute of enzyme solution.
Peroxidase (POD) activity was determined per Doerge et al. (1997), with partial modification. The crude enzyme solution was the same as the PPO test. For the activity test, the reaction mixture was prepared immediately before use: 5 mM guaiacol and 0.3 mM H2O2 were added to phosphate buffer (50 mM, pH 6.5), and dark conditions were maintained. The reaction mixture was incubated at 37°C for 10 min with 1 mL enzyme in 3 mL substrate. POD activity was determined by monitoring the increase in absorbance at 470 nm within 10 min. One unit of enzyme activity was regarded as a change in absorbance of 0.001 per minute of enzyme solution. Protein concentrations were analyzed by bicinchoninic acid (BCA) method (Smith et al., 1985) using bovine serum (Sigma, USA) as a standard.
Statistical analysis was performed by Duncan's multiple range test (5% level) using the SPSS program (Version 20.0, SPSS, Inc., Chicago, Illinois, USA).
Results and Discussion
Pre-Drying and SBI
In this experiment, the early symptoms of SB were irregular spots on the skin (Fig. 1), similar to those of the Western pear ‘d’ Anjou’ when stored in low oxygen conditions (Lee et al., 1990). The PDI significantly decreased with increasing pre-drying time before low temperature storage (Fig. 2). That is, the highest PDI was 37.3 for the 1% weight loss group and the lowest was 5.1 for the 4% group. In addition, the 5% weight loss group did not show a statistically significant difference from the 4% group. As a result, pre-drying treatment to 4% weight loss was considered sufficient to prevent SB disorder in this cultivar.
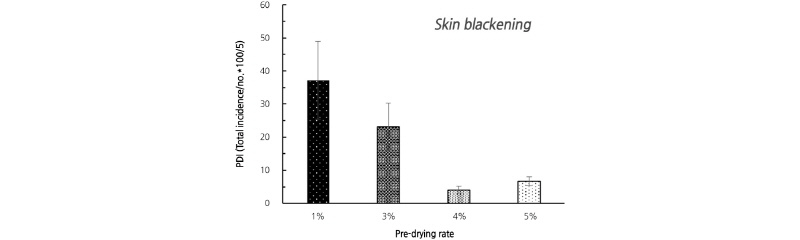
Fig. 2. The incidence of skin blackening disorder in ‘Chuhwangbae’ pear fruits treated with different rates of pre-drying before low temperature storage at 1°C for four weeks. During the pre-drying process, we treated the fruits with a blowing fan at 25°C for six days and sampled weight loss in the fruits. PDI, percent disorder index. Error bars represent the standard error of the mean (n = 36-40).
There were no significant differences in weight loss during four weeks of cold storage in the pre-drying treated fruits regardless of drying rate, and the fruit quality parameters (flesh firmness, soluble solids and titratable acidity) did not differ between the treatments (Table 1). This suggests that even if the fruit is stored at room temperature for the pre-drying treatment, there is no significant deterioration in quality during the cold storage period to a 5% weight loss level. The external quality of Asian pear fruit may deteriorate due to excessive water loss after long-term storage, in the case of fruits whose weight was reduced by 5% or more (Lee et al., 2010). Therefore, it is necessary to examine whether there is a quality difference after long-term storage for several months due to excessive weight loss, apart from SBI.
Delayed Cooling and SBI
We investigated the effect of fruit-cooling delay as a process of low temperature conditioning (LTC) or temperature acclimation (TA) on SBI by comparing the drop rate to 1°C, which is the target temperature for long-distance export. Fruits were stored at 20°C for six days (TA-1), five days (TA-2), or four days (TA-3), followed by stepwise cooling to the target low temperature (1°C) for six days, and then stored at 1°C for four weeks. During the period without cooling, weight loss of fruits was 4.29, 4.24, and 3.72% for TA-1, TA-2, and TA-3, respectively, which showed that weight loss rate tended to be lower with faster cooling time (data not shown). SBI differed according to the LTC timing, the PDI after storage was 6.2, 9.2, and 19.1% in TA-1, TA-2, and TA-3, respectively (Fig. 3). These results suggest that if cooling is initiated soon (TA-3) after storing in a cold room, there is an increased risk of SBI because weight loss is less than 4%. Therefore, we found that SB-susceptible pear cultivars require TA treatment during cold storage, as well as sufficient pre-drying treatment after harvest.
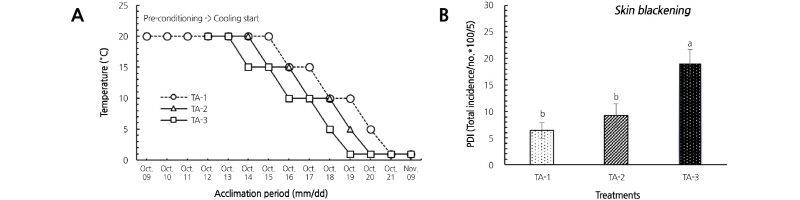
Fig. 3. Delayed cooling strategy (A) and skin blackening incidence (B) in ‘Chuhwangbae’ pear fruits stored for 20 days at 1°C. PDI, percent disorder index. Error bars represent the standard error of the mean (n = 75-76). Fruits were harvested on October 9, 2016 and pre-conditioned for four days at 20°C. TA-1: 48-h cooling delay at 20°C; TA-2: 24-h cooling delay at 20°C; TA-3: no delay.
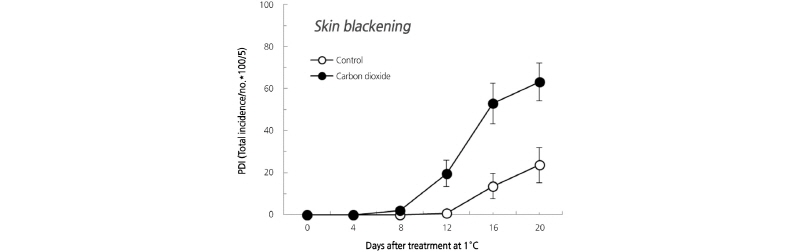
CO2 and SBI
We compared the results of the 2% CO2 treatment with the control treatment (0.03% CO2) on SBI during 20 days of cold storage (1°C) in ‘Chuwhangbae’ pear fruits. In the control fruits, SB occurred 12 days after cold storage and showed a PDI of 13.6 on day 16 and 23.6 on day 20. On the other hand, the 2% CO2 treatment group started to show SB from day 8, and the PDI increased from 19.6 on day 12 to 52.8 on day 16. After 20 days of storage, the PDI reached 63.2, which was three times greater than the control (Fig. 4).
In general, the SB disorders that appear only on the skin of pear fruits differ according to variety (Amiot et al., 1995), storage temperature (Kuo and Yiu, 2006; Choi et al., 1995), and harvest time (Choi et al., 2014). Studies have shown that the browning of fruit flesh increases with increasing CO2 concentration (de Castro et al., 2007), but the effect of CO2 on SBI in Asian pear has not been reported. Therefore, if controlled atmosphere storage is applied to Asian pear fruits, it is necessary to investigate the reactivity to CO2 to reduce the risk of physiological damage.
CO2 Effects on Total Phenols, PPO, and POD Activity
After 20 days of low temperature storage (1°C), we collected samples from unaffected and damaged fruits (Fig. 1) and compared total phenolic content. In the control fruits, we observed a significant decrease in total phenolic content in the severely damaged tissues. The total phenolic content in the severely blackened tissue (SBI-5) was 2.18 mg·g-1 fresh weight (FW), which was 36.6% lower than that of sound tissue (SBI-0, 3.44 mg·g-1 FW) and 27.9% lower than the slightly damaged tissue (SBI-3, 2.48 mg·g-1 FW). A decrease in total phenolic content appeared more pronounced in the fruits treated with 2% CO2. The total phenol in the tissues of SBI-5 was 0.65 mg·g-1 FW, which was 79.9% lower than that of SBI-0 (3.24 mg·g-1 FW). These results suggest that oxidation of total phenols was proceeded by the occurrence of the disorder (Fig. 5).
Increasing PPO activity in the control fruits coincided with increasing SBI, while there was no correlation between PPO activity and SBI in the CO2-treated fruits. PPO activity in the control fruit tissues of SBI-5 was 1.5 times higher than that of SBI-0 (Fig. 5). POD activity increased only in SBI-5 tissues in the control group, but no significant difference in POD activity was found in the CO2 treatment. POD activity was not significantly different between SBI-0 and SBI-3 tissues, but activity in SBI-5 tissues rapidly increased (Fig. 5).
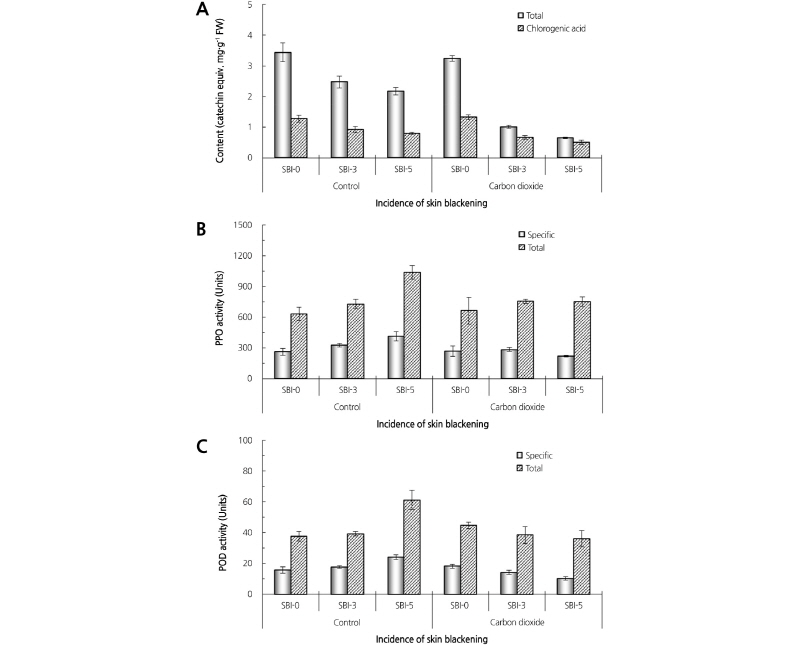
Fig. 5. Comparison of total phenolic contents (A), polyphenol oxidase (B), and peroxidase (C) activity between sound and skin blackened tissues in ‘Chuhwangbae’ pears treated with continuous air (control) or 2% carbon dioxide and stored at 1°C for 20 days. One unit of enzyme activity was defined as the change in absorbance of 0.001 per minute (total activity) or mg-1 protein (specific activity). Error bars represent the standard error of the mean (n = 3).
It has been demonstrated that oxidation of phenolic compounds, including chlorogenic acid and catechins, occurs in model solutions by a combination of pear (P. communis cv. Williams) PPO and POD activities (Richard-Forget and Gauillard, 1997). They suggested that pear POD enhanced phenolic degradation, regardless of the presence of PPO, and could be explained by the additional consumption of H2O2 generated by PPO oxidation. In addition, the production of H2O2 in the oxidation of tea phenolic compounds, which are catalyzed by PPO, suggested that the effects of PPO and POD in the browning process may be synergistic (Subramanian et al., 1999). Therefore, the subsequent increase in POD activity, after the main oxidative increase in PPO activity in the damaged tissues of ‘Chuhwangbae’ pear, may be a major factor in the maximum SBI (SBI-5) observed in the control group.
While there was no increase in PPO activity with the 2% CO2 treatment, the total phenolic contents in the damaged tissue rapidly diminished; this appears to be a CO2 injury rather than enzymatic oxidation. These results suggest that if pear fruit are exposed to greater than 2% CO2 concentration during refrigeration, the risk of SBI is high.
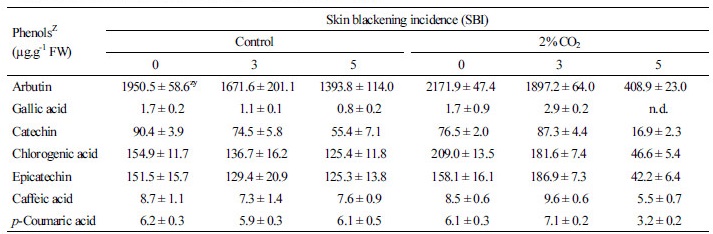
The phenolic content in the damaged tissues appeared to depend on CO2 treatment and SBI. We identified seven polyphenols in the sound or damaged tissues by HPLC. Arbutin accounted for more than 82% of total phenolic components, followed by chlorogenic acid, (‑)-epicatechin, and (+)-catechin, and a small amount of caffeic acid, p-coumaric acid, and gallic acid. The arbutin content found in this experiment was similar to that reported in Chinese pear (P. bretschnrideri) and Japanese pear (P. pyrifolia) cultivars (Cui et al., 2005; Lin and Harnly, 2008). Arbutin content decreased with increasing SBI in the control group: 14.3% lower in SBI-3 (1,671.6 µg·g-1 FW) and 28.5% lower in SBI-5 (1,398.8 µg·g-1 FW), when compared to SBI-0 (1,950.5 µg·g-1 FW). On the other hand, the change in arbutin content was more prominent in severely damaged tissues in the CO2 treatment: 81.2% lower in SBI-5 (408.9 µg·g-1 FW), when compared to SBI-0 (2,171.9 µg·g-1 FW). In addition, chlorogenic acid, epicatechin, major phenolics in Western pears (Amiot et al., 1995), and catechin contents significantly decreased in SBI-5 tissue, especially in the CO2 treatment (Table 2). These results concur with the report that total phenols were lower in Western pear ‘Williams’ when stored in high CO2 conditions (Amiot et al., 1995). Therefore, severe SB in the CO2 treatment group seems to be caused by the combination of increased enzyme activity, such as PPO and POD (Fig. 5), and direct damage due to high concentration of CO2 (Fernández-Trujillo et al., 2001).
This also suggests that CO2, accumulated by fruit respiration, may cause SB in fruits that are stored in closed carton boxes for a long period at low temperatures. Therefore, it would be desirable to recognize the importance of ventilation strategies for cold storage facilities and to use gas absorbers in each carton.
In conclusion, we examined the effects of pre-drying, delayed cooling, and CO2 treatment on inhibition or acceleration of SB disorder in ‘Chuwhangbae’ pears. The SBI can be reduced by pre-drying the fruits at 25°C, to 4% weight loss. Regarding a cooling strategy to alleviate SBI, we recommend starting the cooling after storing fruit for six days at 20°C. Total phenols in the fruit peel decreased significantly, and PPO and POD activity increased significantly, with increasing SBI in normal cold storage conditions. However, concerning CO2 treatment, severe SBI is likely a complex process involving related enzyme oxidation and direct CO2 injury.