Introduction
Silicon (Si) is the second most abundant element in the soil and a mineral element for most of the world’s plant life. Most Si is present in the soil as insoluble oxides or silicates, although soluble silicic acid occurs in the range of 0.1-0.6 mM (Epstein, 1994). The availability of Si in the soil is governed not only by Si polymorphs but also by a number of factors which include soil moisture, temperature, soil pH, organic matter complexes, redox potential, particle size distribution, sesqui-oxide colloids, the presence of aluminium, iron and phosphate ions, as well as various exchangeable/dissolution reactions (Drees et al., 1989). Silicon has been reported to be beneficial for growth and development of graminaceous plants such as rice, sugarcane and wheat (Liang et al., 2007). However, recent studies have shown that the Si nutrition enhanced growth traits in carnation (Bae et al., 2010), chrysanthemum ‘Backwang’ (Moon et al., 2008), gerbera (Kamenidou et al., 2010; Savvas et al., 2002), kalanchoe (Bae et al., 2010), sunflower (Kamenidou et al., 2008), rose (Ehret et al., 2005; Hwang et al., 2005), and zinnia (Kamenidou et al., 2009). Furthermore, application of Si has been shown to be able to control diseases (Rodrigues et al., 2003; Voogt and Sonneveld, 2001) and pests (Jeong et al., 2012; Ranganathan et al., 2006), alleviate toxicity of some heavy metals (Shi et al., 2010), and enhance resistance to drought (Chen et al., 2011), salinity (Mateos- Naranjo et al., 2013), and temperature stresses (Epstein, 1999; Ma and Yamaji, 2006).
Ferns have been cultivated for their ornamental foliage and their demand as indoor plants has been increasing. The most frequently used indoor ferns are the Boston ferns (Nephrolepis exaltata and cultivars), which come in a variety of textures. Nephrolepis exaltata ‘Corditas’ is a fast and uniform grower belonging to the family Nephrolepidaceae. Its modest size and maximum frond length of 30 cm make this dwarf fern ideal for smaller pot sizes and arrangements. Plants frequently encounter adverse growth conditions (Krasensky and Jonak, 2012). Temperature is one of the primary abiotic factors which influence the growth and development of many plant species. Ferns vary greatly in their temperature requirements. Most grow best at daytime temperatures of 18 to 27°C and the nighttime temperature is about 5°C cooler than the daytime temperature (Hoshizaki and Moran, 2001). Boston ferns exposed to temperatures below 15.5°C and above 35°C may display stunted growth and yellow fronds (Dawson et al., 1991; Erwin et al., 1993). The optimal supply of Si increases the plant tolerance to many abiotic stresses including low and high temperature (Ma and Yamaji, 2006). Scientists have observed silica bodies in fern tissues (Hodson et al., 2005; Piperno, 1988), but no reports are yet available concerning the effect of Si on growth of ferns. In this study, we investigated the effect of Si on growth and temperature stress tolerance in N. exaltata ‘Corditas’.
Materials and Methods
In vitro-grown plantlets were acclimatized in the green-house. After a month, uniform plantlets were transplanted into 10 cm (370 mL) plastic pots containing a coir-based substrate (Tosilee medium, Shinan Grow Co., Korea). A nutrient solution containing 0, 50, or 100 mg・L-1 Si was supplied through a drip-irrigation system. Silicon was added to the nutrient solution in the form of potassium silicate (K2SiO3), and additional K introduced by K2SiO3 was subtracted from KNO3 and the resulted nitrate loss was supplemented with nitric acid. The composition of nutrient solution was (mg・L-1): 354 Ca(NO3)2・4H2O, 123 MgSO4・7H2O, 252.5 KNO3, 115 NH4H2PO4, 0.62 H3BO3, 0.062 CuSO4・5H2O, 2 Fe-EDTA, 1.1 MnSO4・4H2O, 0.04 H2MoO4, and 0.575 ZnSO4・7H2O. All treatment solutions were adjusted to pH 5.8 with nitric acid or potassium hydroxide at the beginning of the experiment and replacement. Each treatment consisted of three replicates and each replicate contained 45 plants. The plants were randomly arranged among nine beds in the greenhouse and were grown under a normal day-light condition with night/day set temperatures of 19/27°C and 60-70% relative humidity (RH). Plant height, width, number of fronds, root length, and fresh and dry weights of frond and root were recorded after 5 months of cultivation. Dry weight was measured after 72 h of drying at 70°C in a forced air dry oven.
After five months of cultivation, Si-treated and the -untreated plants were placed in an environment controlled chamber. A nutrient solution containing 0, 50, or 100 mg・L-1Si was supplied uniformly twice a day. Plants were maintained at 10, 25, or 40 ± 1°C under a 12 h photoperiod with 530 µmol・m-2・s-1 photosynthetic photon flux density (PPFD), and 60% RH. Treatments were started 3 days after incubation at 25°C. Each treatment consisted of three replicates and each replicate contained 7 plants. After 7 days, chlorophyll (Chl) content and chlorophyll fluorescence parameters were measured. To estimate Chl content, frond tissue (0.01 g) was homogenized in 5 mL of ice-cold 80% (v/v) acetone, and then centrifuged at 2500 g for 15 min. The supernatant was decanted and the absorbance was measured at 645 and 663 nm. Chlorophyll concentration was determined according to the procedure developed by Arnon (1949). Chlorophyll fluorescence parameters were measured using a chlorophyll fluorometer (PAM- 2100, Walz, Germany). Fronds were dark adapted for at least 30 min before the chlorophyll fluorescence measurements.
Plants were harvested after 5 months of cultivation for chemical analysis. For each treatment five samples were taken. Dried samples (1.0 g) ground with a stainless mill (Cytclotec, Model 1093, Tector, Hoganas, Sweden) was ashed in a Nabertherm muffle furnace (Model LV 5/11/B180, Lilienthal, Bremen, Germany), for 4 h at 525°C. The ash was dissolved in 5 mL 25% HCl, followed by 20 mL of hot distilled water (DH2O) and brought to 100 mL with deionized water. Calcium (Ca), potassium (K), magnesium (Mg), phosphorus (P), sulfur (S), boron (B), copper (Cu), iron (Fe), manganese (Mn), and zinc (Zn) concentrations in digested frond and root samples were measured by using an inductively coupled plasma (ICP) spectrometer (Optima 4300DV/5300DV, Perkin Elmer, Germany).
Extraction of Si in the frond and root samples was con-ducted by a slight modification of the autoclave-induced digestion method (Elliott and Snyder, 1991). Dry, ground samples (0.1 g) were placed in 50 mL polyethylene tubes and wetted with 3 mL of a 30% hydrogen peroxide solution. Then five drops of octyl alcohol were added to prevent excessive foaming. To each tube 4.5 g of NaOH was added, followed by 6.0 mL of DH2O and each tube was gently vortexed and covered with a loose fitting plastic cap. The tubes were placed in an autoclave at 138 kPa for 1 h. After atmospheric pressure was reached, tubes were removed and the contents were brought to 50 mL with DH2O. The digested sample Si concentration was determined color-imetrically according to Sivanesan et al. (2010).
Statistical analyses of the data were carried out using SAS (Statistical Analysis System, V. 9.1, Cary, NC, USA) program. All data were subjected to analysis of variance (ANOVA) and Duncan’s multiple range tests was used to test significant difference between means.
Results and Discussion
Silicon nutrition either increased or decreased plant height in several plant species such as bracteantha, carnation, fuchsia, kalanchoe, lobelia, rice, and tomato (Bae et al., 2010; Kim et al., 2002; Mattson and Leatherwood, 2010; Sivanesan et al., 2011). In the present study, plant height was suppressed by 2.7 and 8.1% when the nutrient solution was supplemented with 50 and 100 mg・L-1 Si, respectively, compared with the control. Similar result was also reported in ornamental sunflower (Kamenidou et al., 2008), Tagetes patula ‘Boy Orange’ and ‘Yellow Boy’ (Sivanesan et al., 2010) and zinnia (Kamenidou et al., 2009). Plant width was also reduced by 6.9 and 13.8% when the plants were treated with 50 and 100 mg・L-1 Si, respectively, compared with the control (Table 1). The mean number of fronds decreased with increasing concentrations of Si in the nutrient solution. Compared with the control, addition of 50 and 100 mg・L-1 Si significantly reduced the mean number of fronds by 31.8 and 42.3%, respectively. Root length was not affected by 50 mg・L-1 Si treatment while, the longest root length was measured in 100 mg・L-1 Si treatment. However, Si treatment significantly reduced fresh and dry weights of roots as compared with the control (Table 1). Similarly, fresh and dry weights of fronds decreased with increasing concentrations of Si in the nutrient solution. When the nutrient solution was supplemented with 100 mg・L-1 Si, fresh and dry weights of fronds were reduced by 9.1 and 10.3%, respectively, compared with the control.
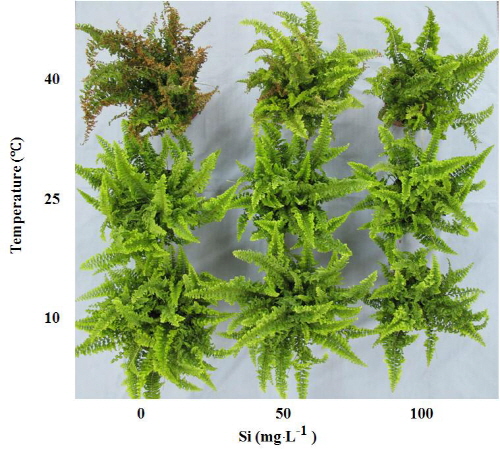
In the present study, Si nutrition had a negative effect on the growth characteristics of N. exaltata ‘Corditas’ by decreasing the concentrations of tissue nutrients content. However, many reports have shown that Si plays an important role in protecting plant from abiotic stresses (Liang et al., 2007; Ma, 2004; Ma and Yamaji, 2006). Temperature stress is one of the main abiotic stresses that limit plant growth and survival. The morphological symptoms of heat stress include scorching of leaves and twigs, leaf senescence and abscission, shoot and root growth inhibition (Wahid et al., 2007). In the present study, frond scorch was observed when plants were treated with extreme temperature. Severe scorch occurred in the Si-deprived plants when they were exposed to 40°C (Fig. 1). Heat stress may cause severe water loss when transpiration exceeds moisture absorption by roots. A portion or all of the leaf can be killed with long-term exposure to unfavorable temperatures. Studies indicate that Si is mainly transported passively in the transpiration stream, and is accumulated at sites of high transpiration (Wiese et al., 2007). Silicon application decreased transpiration rate in maize (Gao et al., 2006), and rice (Ma, 2004). In the present study, 100 mg・L-1 Si treatment significantly increased the Si concentration of frond. The result suggest that high amount of Si deposition may reduce the transpiration rate and thereby enhance high temperature tolerance in N. exaltata ‘Corditas’. The temperature treatment had a significant effect on Chl a, Chl b, and total Chl content of N. exaltata ‘Corditas’. The content of Chl a or Chl b reduced significantly when the Si-deprived plants were treated with 10 and 40°C, respectively. When the Si-treated plants (100 mg・L-1) were exposed to 10, 25 or 40°C, the total Chl content was increased by 52.7, 20.5 and 79.6%, respectively, compared with the control. Similar results were also observed in bermudagrass (Brecht et al., 2009). Silicon also increases the tolerance to heat stress in rice (Ma, 2004). Agarie et al. (1998) reported electrolyte leakage caused by high temperature was less pronounced in leaves grown with Si than in those grown without Si. These results suggest that Si may be involved in the thermal stability of lipids in cell membranes although the mechanism has not been elucidated (Ma, 2004). Chlorophyll fluorescence has been widely used as an early indication of many kinds of plant stress, including temperature (Chen et al., 2012; Kalaji et al., 2011; Maxwell and Johnson, 2000). Chlorophyll fluorescence parameter Fv/Fm was more sensitive to tem-perature in plants (Salvucci and Crafts-Brander, 2004). In the present study, Fv/Fm value was not significantly different when the plants were exposed to 25°C. However, significant difference in Fv/Fm was recorded when plants were exposed to 10 and 40°C. Thus, Fv/Fm could be used as an indicator of low and high temperature tolerance (Guo et al., 2006; Jiang and Howell, 2002). When Si-treated plants (100 mg・L-1) were exposed to 10 or 40°C, Fv/Fm value was increased by 34.6 and 110.7%, respectively, compared with the control. The results indicate that photosystem II function is not significantly affected by temperature stress, when Si was supplied to N. exaltata ‘Corditas’.
The application of Si has been reported to affect the tissue nutrient concentrations of many plant species such as alfalfa (Wang and Han, 2007), bean (Zuccarini, 2008), chrysanthemum (Sivanesan et al., 2013), gerbera (Kamenidou et al., 2010), and rice (Guo et al., 2005). In the present study, 100 mg・L-1 Si treatment reduced both frond and root tissue concentration of Ca by 14.9 and 14.0%, respectively, compared with the control. Similar result was also observed in sunflower (Kamenidou et al., 2008) and rice (Ma and Takahashi, 1993). Compared with the control, 50 mg・L-1 Si treatment significantly reduced (0.97%) or non-significantly increased (11.7%) the concent-ration of K in frond and root tissue, respectively. In contrast, 100 mg・L-1 Si treatment increased (10.9%) or reduced (3.6%) the concentration of K in frond and root tissue, respectively, compared with the control. Similarly, addition of Si decreased leaf tissue K concentration in bean (Zuccarini, 2008) and chrysanthemum (Sivanesan et al., 2013). Liang et al. (1996) reported the K uptake by solution-cultured barley plants increased following Si applications. Compared with the control, 50 or 100 mg・L-1 Si treatment reduced Mg concentration of frond and root tissue by 5.8 and 10.2%, respectively. Similarly, 100 mg・L-1 Si treatment reduced P concentration of frond and root tissue by 2.8 and 9.3%, respectively. However, 50 mg・L-1 Si treatment increased frond tissue concentration of P by 1.2%. The Si treatment tends to increase the P con-centration in the green tops of sugarcane (Roy et al., 1971), but decreased in rice (Guo et al., 2005; Ma and Takahashi, 1990). Compared with the control, 50 or 100 mg・L-1 Si treatment significantly increased (23.3%) or reduced (15.0%) the concentration of S in frond and root tissue, respectively.
The Si treatment increased (10.5%) or reduced (18.2%) the concentration of B in frond and root tissue, respectively, compared with the control. Both frond and root tissue concentration of Cu reduced with increasing Si concentration in the nutrient solution. The Fe concentration of frond and root tissue increased or reduced when plants were treated with 50 and 100 mg・L-1 Si, respectively. Similar results have also been reported in alfalfa (Wang and Han, 2007), chrysanthemum (Sivanesan et al., 2013), and gerbera (Kamenidou et al., 2010). The Mn concentration of frond and root tissue also reduced with increasing Si concentration in the nutrient solution. Compared with the control, frond tissue concentration of Zn decreased 9.9 and 13.8% when plants were treated with 50 and 100 mg・L-1 Si, respectively. Root tissue concentration of Zn increased (18.4%) or decreased (6.5%) when plants were treated with 50 and 100 mg・L-1 Si, respectively, compared with the control. Both frond and root tissue concentrations of Si significantly increased linearly with increasing Si concentration in the nutrient solution. The 100 mg・L-1 Si treatment increased the Si concentration of frond and root tissue by 268.1% and 348.6%, respectively, compared with the control.
In conclusion, Si nutrition significantly reduced the mean number of fronds, fresh and dry weights of fronds and roots. Similarly, Si application decreased significantly tissue concent-rations of Ca, Mg, P, Cu, Fe, Mn, and Zn. The reduction of biomass may have resulted from changes in the concentrations of inorganic nutrients in leaf or root tissue due to Si appli-cation. The Fv/Fm value decreased during low or high tem-perature stress suggesting that Fv/Fm could be used as an indicator of low and high temperature tolerance in ferns. The present study also suggests that application of Si may be used to enhance temperature tolerance of ferns.