Introduction
Materials and Methods
Plant Materials and Growth Conditions
Light Quality Treatments
Growth Characteristics
Determination of Starch and Total Sugar Contents
Statistical Analysis
Results and Discussion
Growth Characteristics
Total Sugar and Starch Contents
Development of Tomato Plants After Transplanting
Conclusion
Introduction
Plug seedlings are usually grown in individual plug tray cells that contain a small amount of a growing medium. Some advantages of using plug seedlings are the ability to produce uniform seedlings, low labor requirement, high space utilization efficiency, flexible scheduling of transplants, and ease of transplanting (McAvoy, 1988; Peppler, 1990). For these reasons, plug seedling use has steadily increased since the 1990s (Jeong et al., 2016). Nowadays, most farms use plug seedlings, and
extensive research has been performed to produce high-quality seedlings.
The seedling age, cultivation environment, and starch and sugar accumulation that is used as a mid- to long-term energy source for plant growth at the seedling stage affect the growth and yields of plants after transplanting (Nesmith, 1993; Streb and Zeeman, 2012; White, 1980; Whitwell and Crofts, 1972). It has been reported that red (R) light-emitting diodes (LEDs) used in the seedling period led to higher initial yields and average weight of paprika fruit compared to blue (B) LEDs, fluorescent lamps (FL), and natural light (Lee et al., 2012). In addition, growth of strawberry plantlets and early fruit yields were dependent on the type of soil (Park et al., 2015). It was also observed that shorter seedling periods resulted in better quality of seedlings and better yields of pepper fruits (Kim et al., 2015). Therefore, it is very important to manage the seedling period by providing the appropriate environmental conditions. Seedlings with proper stem diameter and leaf size, highest possible leaf thickness with a high assimilation ability, and no excessive stretching are considered high-quality seedlings.
In recent years, LEDs have found their way into agriculture as supplementary light, in light culture, and in grafting and healing processes of plug seedlings. The biggest advantage of LEDs is that they can be used to select specific wavelengths, also known as the light quality (Yeh and Chung, 2009). R and B LEDs are mainly used to study plant growth responses in basic research because the light utilization efficiency is high for these LEDs (Hopkins and Hüner, 2004; Terashima et al., 2009). Many studies using these light qualities have been carried out for various crops to investigate their growth responses. For Cymbidium plantlets, R light improved and B light deteriorated the leaf growth (Tanaka et al., 1998). For strawberry plantlets, R light showed greater enhancement than B light of both the leaf growth and chlorophyll content (Nhut et al., 2003). Paprika grown under B LEDs was significantly taller than those grown under R LEDs (Lee et al., 2012). These results demonstrate that the light quality has different effects on the growth and development of plants. There are many studies that relate the light quality with the growth of leafy vegetables, flowers, seedlings, succulent plants, medicinal plants, cereals, and herbs (Bae et al., 2017; Choi et al., 2014; Im et al., 2013; Jeon et al., 2018; Kim et al., 2018; Lee et al., 2016a; Lee et al., 2016b; Nam et al., 2018). However, there isn’t much research on the relationship between the light quality and plug seedlings of tomato widely found in Korea.
Therefore, this study was conducted to examine the effects of monochromatic and combined LEDs lighting on the growth and development of ‘Mini Chal’ tomato plug seedlings in a closed-type plant production system and to examine the method to produce the high-quality seedlings with short stems and high compactness from seeding to production in a closed environment.
Materials and Methods
Plant Materials and Growth Conditions
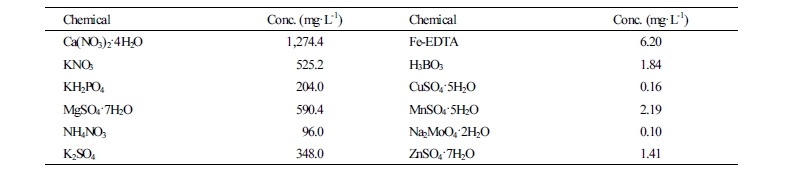
Seeds of ‘Mini Chal’ tomato (Solanum lycopersicum L.) were sown in 40-cell plug trays (54 × 28 × 4.8 cm, Bumnong Co. Ltd., Jeongeup, Korea) filled with a commercial growing medium on January 15, 2018, and grown in a closed-type plant production system (CPPS, C1200H3, FC Phoibe Co. Ltd., Seoul, Korea) at 25°C temperature, 50% relative humidity (RH), and 200 µmol·m-2·s-1 photosynthetic photon flux density (PPFD) using fluorescent lamps (FL, FHF32SSEX-D, OSRAM GmbH, Munich, Germany) for 7 days before light quality treatments. Eight days after sowing, 3 replicates of 20 seedlings per treatment were used for this experiment. The growth conditions were set with 25°C/18°C day and night temperatures, respectively, 50 ± 10% RH, and 200 µmol·m-2·s-1 PPFD on the basis of the upper leaves of the plants with a 12/12-hour light/dark photoperiod. To ensure that these settings were correct, measurements were confirmed with a temperature/RH meter (TR-72U, T&D Co. Ltd., Nagano, Japan) at all times. The light intensity was measured and adjusted at the top of the plant every 3 days using a photometer (HD2101.2, Delta Ohm SrL, Caselle, Italy). The nutrient solution was supplied by subirrigation every day at 08:30 am (Sonneveld and Straver, 1994), with pH 6.5 and 0.5 - 1.5 dS·m-1 EC (electrical conductivity) that gradually increased in concentration (Table 1). Plug trays were rearranged every day after irrigation to provide uniformly distributed light wavelengths and intensities. Seedlings were grown for 23 days after treatment (DAT) with various light treatments.
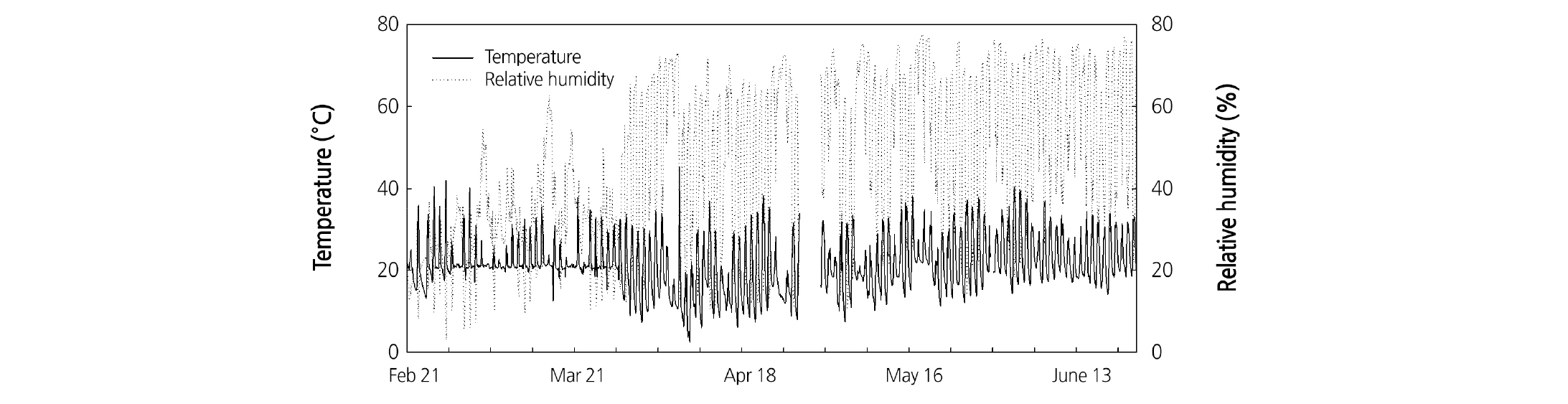
At 24 DAT, the seedlings were transplanted into slab-type rockwool media (Grodan Co. Ltd., Roermond, The Netherlands) in a Venlo-type glasshouse. Three seedlings were planted per slab. The nutrient solution was automatically drip-irrigated two to three times a day for 10 minutes (Sonneveld and Straver, 1994), and pH and EC were 6.5 and 1.5 - 2.0 dS·m-1, respectively (Table 1). The mean temperature and RH in the daytime were approximately 22°C and 40%, respectively (Fig. 1).
Light Quality Treatments
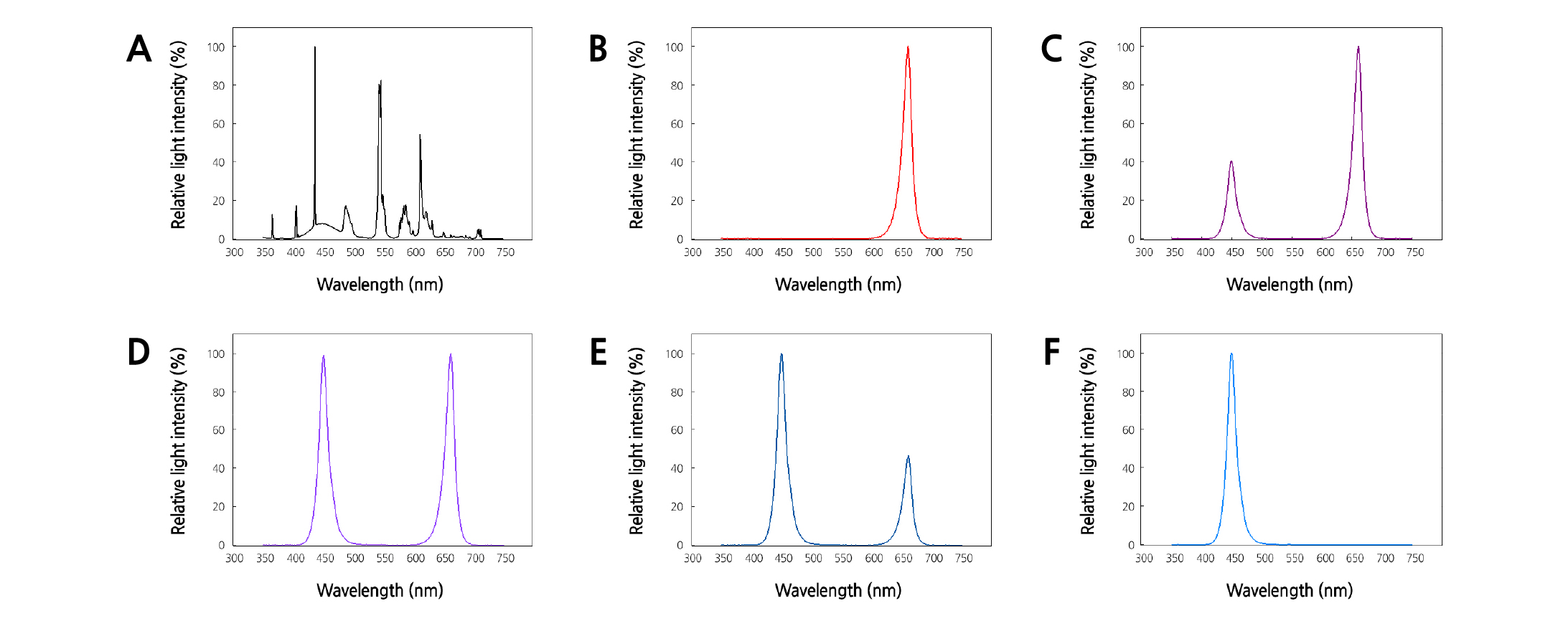
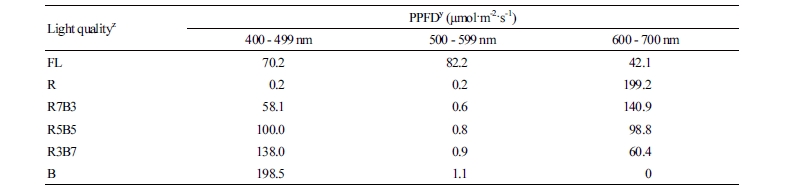
The light qualities for this experiment were sourced from FL and combinations of R and B LEDs [R:B = 100:0 (R), R:B = 70:30 (R7B3), R:B = 50:50 (R5B5), R:B = 30:70 (R3B7), or R:B = 0:100 (B) LEDs (LP1827RB-120C, L-PEC Co. Ltd., Jeonju, Korea)] (Fig. 2). These ratios are based on PPFD. To compare the effects of chemicals used to suppress plant height and to enhance the quality of seedlings at the farm readily, 150 mg·L-1 of diniconazole (Binnari, Dongbang Agro Co. Ltd., Seoul, Korea) was additionally applied one time to seedlings under FL (Dini) by foliar spray when two true leaves of a plant appeared. The light spectral distribution was measured using a spectroradiometer (ILT950, International Light Technologies Inc., Peabody, MA, USA) at five points (center and four edges) at the bottom of the tray (Fig. 2). The PPFD of wavelengths in each treatment were determined from bandwidth integration (Table 2).
Growth Characteristics
Plant growth parameters, such as the plant height, stem diameter, number of leaves, leaf area, leaf shape index, specific leaf weight (SLW), SPAD value, fresh and dry weights of stem and leaf, dry matter, compactness, and chlorophyll fluorescence (Fv/Fm) were measured at 23 DAT. The stem diameter was measured at 1 cm above the medium surface using digital vernier calipers (CD-20CPX, Mitutoya Corp., Kawasaki, Japan). The number of leaves was measured by counting all plant leaves except for the cotyledons. The leaf area was measured using a leaf area meter (LI-3000, LI-COR Inc., Lincoln, NE, USA). The SPAD value was measured on the fifth leaf from the top using a chlorophyll meter (SPAD-502, Konica Minolta Inc., Osaka, Japan). The fresh weight was measured with an electronic scale (EW 220-3NM, Kern&Sohn GmbH., Balingen, Germany). The dry weight was measured after drying the divided samples of the stem, leaf, and root for 72 hours in a drying oven (Venticell-222, MMM Medcenter Einrichtungen GmbH., Munich, Germany) at 70°C. The Fv/Fm was measured in diurnal time courses using a portable saturation-pulse chlorophyll fluorometer (PAM-2100, Heinz Walz GmbH, Effeltrich, Germany).
After transplanting, the flowering speed, position of flower clusters, and cluster height were measured at 122 days after transplanting. The flowering speed was calculated as the percentage of the flowering of the first flower in the first or second cluster in a total of 9 plants per treatment. The leaf shape index, SLW, dry matter, and compactness of tomato seedlings were calculated using the following formulae:
Leaf shape index = leaf length/leaf width
SLW (mg·cm-2) = dry weight of leaf (mg)/total leaf area (cm2)
Dry matter (%) = [dry weight of shoot (g)/fresh weight of shoot (g)] × 100
Compactness (mg·cm-1) = dry weight of shoot (mg)/plant height (cm)
Determination of Starch and Total Sugar Contents
To determine the total sugar content, all leaves of a plant were ground with liquid nitrogen at 23 DAT, stored in a deep freezer at - 70°C, and then determined using the modified method of Dubois et al. (1956). Three 0.3-g samples were taken per light treatment. The samples were mixed with 10 mL of 80% ethanol and then ground for 1 minute. After being boiled in a 60°C water bath, the supernatant was separated by centrifugation (908 × g, 20°C, 30 min). The remaining precipitate was mixed with 10 mL of 80% ethanol, boiled in a water bath at 60°C for 30 minutes, and then centrifuged in the aforementioned manner. After 2 centrifugations, the supernatant was diluted with 40 mL of 80% ethanol, 0.5 mL of 5% phenol reagent was added and vortexed, and 2.5 mL of 98% sulfuric acid was added and vortexed. After being cooled to room temperature, the absorbance was measured at 490 nm using a UV spectrophotometer, and the total sugar content was calculated using glucose as the standard.
To determine the starch content, all leaves of a plant were ground with liquid nitrogen at 23 DAT, stored in a deep freezer at - 70°C, and then determined using the modified method of Lim et al. (2003). Three 0.3-g samples were taken per light treatment. To solubilize the samples, 10 mL of 80% ethyl alcohol was added to the samples, shaken for 30 minutes, and then centrifuged at 300 × g for 30 minutes at 20°C. Ethanol (40%) was then added to the residue and centrifuged again at the same conditions. Two milliliters of 30% HClO4 and 1 mL of dimethyl sulfoxide were added to the residue and kept at room temperature for 30 minutes. After that, 2 mL of distilled water and 5 mL of H2SO4 were added and kept in a 100°C water bath for 1 hour and subsequently centrifuged again at the same conditions. To 0.5 mL of this sample, 0.5 mL of 5% phenol reagent was added and vortexed, and 2.5 mL of 98% sulfuric acid was added and vortexed. After being cooled to room temperature, the absorbance was measured at 470 nm using a UV spectrophotometer, and the starch content was calculated using glucose as the standard.
Statistical Analysis
A randomized complete block design with 3 replications and 5 plants in each replication was employed in the experiment. After transplanting, completely randomized designs with 3 replications and 3 plants in each replication were employed in a glasshouse. The statistical analysis was carried out using the statistical analysis system program (SAS 9.1, SAS Institute Inc., Cary, NC, USA). The experimental results were subjected to an analysis of variance (ANOVA) and Duncan’s multiple range tests. Graphing was performed with the SigmaPlot program (SigmaPlot 12.0, Systat Software Inc., San Jose, CA, USA).
Results and Discussion
Growth Characteristics
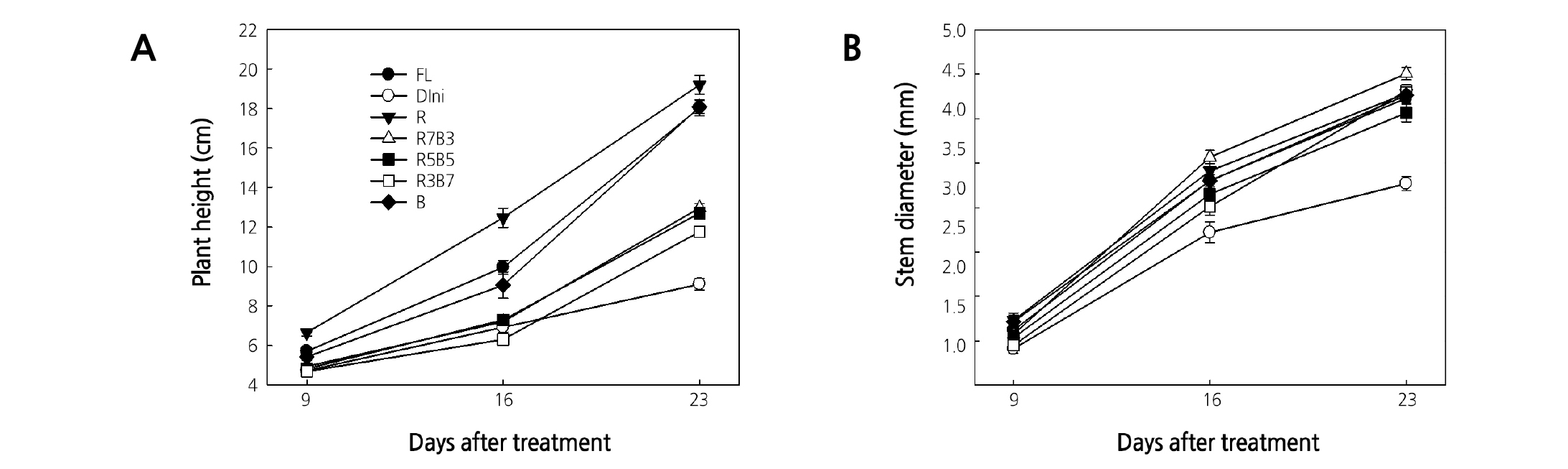
The plant height and stem diameter of tomato plug seedlings were measured 9, 16, and 23 DAT, which showed significant differences depending on the light treatment (Figs. 3 and 4). R LEDs resulted in the tallest plant height, followed by FL and B LEDs (Fig. 3A). Combined R and B LEDs treatments and Dini produced short plants. Several studies report that plant height is promoted or suppressed by R or B light, respectively (Tanaka et al., 1998; Wang et al., 2009; Johkan et al., 2010; Shimizu et al., 2011). Our results that plants grown under R LEDs were taller than plants grown under B LEDs agreed with these reports. Monochromatic R and B lights are known to have excellent light use efficiency, absorbing both wavelengths by chlorophylls a and b efficiently (Hopkins and Hüner, 2004; Terashima et al., 2009). This aligns with the results of our study, where monochromatic LEDs enhanced the height of seedlings compared to combined R and B LEDs. On the other hand, combined R and B LEDs treatments produced short plants. Similar results were observed for Korean melon seedlings, where the height of plants grown under combined R and B LEDs was smaller than the height of plants grown under monochromatic B LEDs (Lee et al., 2016a). These results suggest that combined R and B LEDs can limit seedling elongation. Despite the fact that FL contains all wavelengths between 400 and 700 nm, seedlings grown under FL were still taller than seedlings grown under combined R and B LEDs. The difference between combined R and B LEDs and FL is that FL contains green (G) light (500 - 599 nm) of 82.2 µmol·m-2·s-1 (Table 2). It is known that G light acts antagonistically to R and B light (Folta and Maruhnich, 2007). Therefore, it is considered that the G light included in the FL partially canceled the roles of the B light of 70.2 µmol·m-2·s-1 and the R light of 42.1 µmol·m-2·s-1, which make the plant short. For this reason, it is thought to be the reason that plants grown under FL were taller than plants grown under combined R and B LEDs. At 16 and 23 DAT, the stem diameter was the greatest for plants grown in R7B3 among all considered R and B LEDs combinations, and the smallest in for plants grown in Dini (Fig. 3B). A previous study reported that tomato plants grown under combined lights (combined R, B, and G LEDs) had stronger stems than plants grown under monochromatic lights (yellow, G, or R LEDs) (Liu et al., 2009). However, our result was not only different from this result, but the treatment was also different from our study. All LEDs treatments were thicker than Dini. With the exception of plants grown under Dini, there were no differences in stem diameter among plants. Therefore, treatments of combined R and B LEDs are advantageous to producing high-quality fruit-vegetable seedlings with short and thick stems.
The number of leaves was the highest for plants grown in R7B3 and the lowest for plants grown in R3B7 and B (Fig. 5A). This demonstrates that a high B light ratio reduces the formation of new leaves. The total leaf area per plant was the largest for plants grown in FL, followed in order by plants grown in R7B3 and R3B7, and the lowest was for plants grown in Dini (Fig. 5B). Despite plants grown in R3B7 having few leaves, the large leaf areas observed are thought to be more advantageous for photosynthesis. Sufficient leaf area is essential to plant growth as it is responsible for photosynthesis, fixing CO2, and accumulating starch (Tsukaya, 2004). A wider range of tendencies was observed for leaf area of plants grown under combined LEDs than under monochromatic LEDs. This demonstrates that the treatment of combined R and B LEDs on seedlings affects the growth of leaves more than stems. In ‘Cuty’ tomato plant, leaf area was greater in seedlings grown under combined R and B LEDs (R:B = 65:35 ratio) than under monochromatic R LEDs (Son et al., 2018). However, leafy vegetables such as lettuce, spinach, and komatsuna showed different results, and leaf area was wider in monochromatic R or B LEDs, except for spinach. Leaf area of spinach was similar between R and combined LEDs (Ohashi-Kaneko et al., 2007). As a result of leaf growth of spinach ‘Sushiro’ and ‘Worldstar’ irradiated with white, monochromatic R or B, and combined R and B LEDs, cell density and size of spinach leaves showed different responses as affected by light quality in two cultivars (Lee et al., 2018). Therefore, leaf growth is considered a variety and genetic characteristic. These results suggest that a proper combination of R and B LEDs is essential to the leaf growth in ‘Mini Chal’ tomato (Britz and Sager, 1990; Hoenecke et al., 1992).
Leaf shape, size, and thickness are phenotypically plastic and responsive to changes in the environment, such as light intensity and quality (Givnish, 1988; Smith and Whitelam, 1997; Yano and Terashima, 2001). The leaf shape index was the highest for plants in R and the lowest for plants in Dini (Fig. 5C). Plants exposed to other light treatments had similar leaf shape indices. A high leaf shape index signifies a long and narrow leaf, and a low leaf shape index means a short and wide leaf (Fig. 6). This indicates that R light produces long leaves, which have been reported in previous studies using lettuce (Lee et al., 2010; Son et al., 2012).
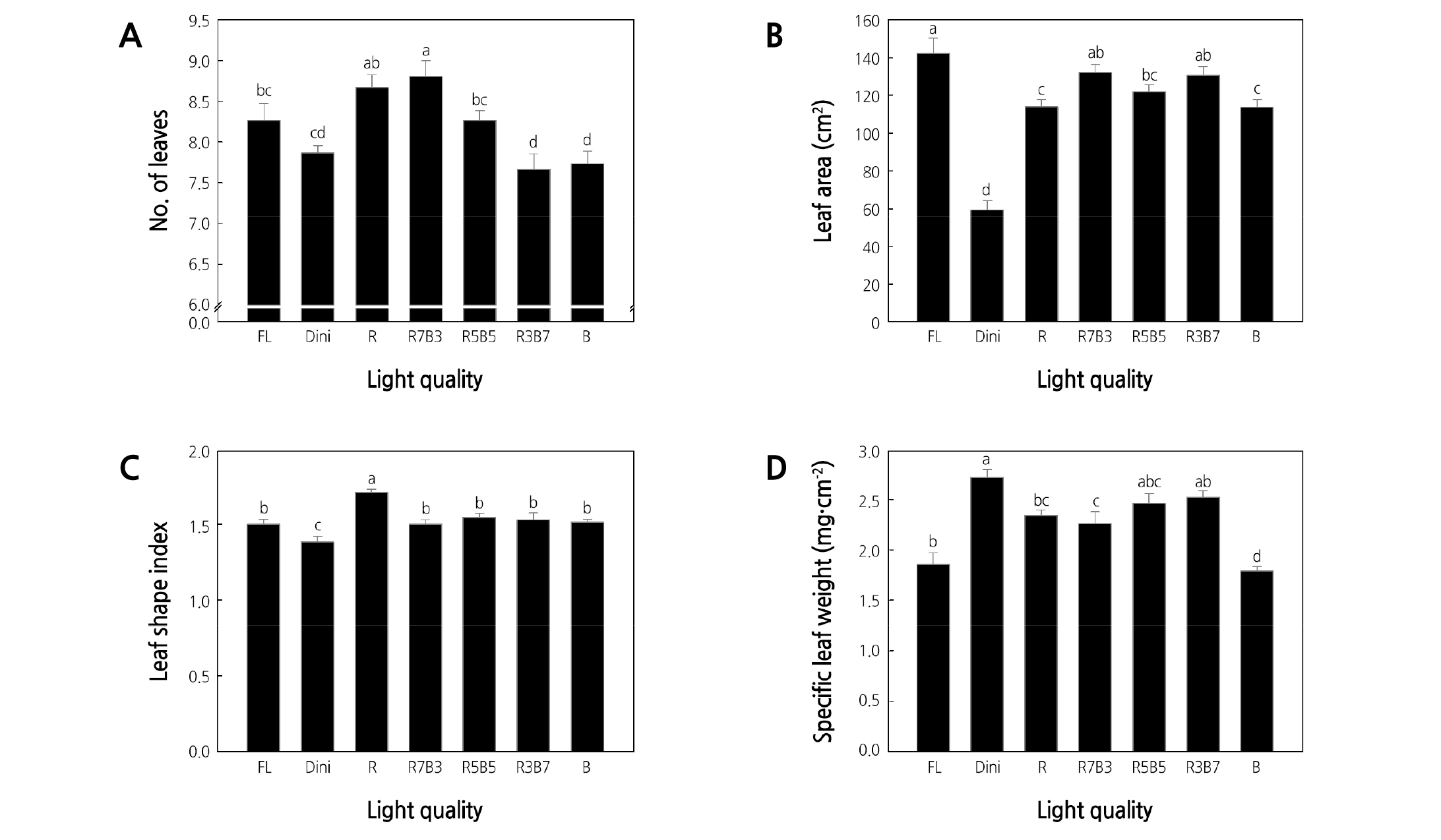
Fig. 5. The effects of the light quality on the number of leaves (A), leaf area (B), leaf shape index (C), and specific leaf weight (D) of ‘Mini Chal’ tomato (Solanum lycopersicum L.) plants measured 23 days after treatment. Refer to Fig. 2 for details on the light quality. Vertical bars indicate the mean ± S.D. (n = 15). Different letters above the bars indicate significant differences by Duncan’s multiple range test at p ≤ 0.05.
The SLW is presented as an indicator of the leaf thickness. The SLW was the greatest for plants grown in Dini, followed by plants grown in R3B7 and R5B5, and the lowest for plants grown in FL and B (Fig. 5D). Leaf thickness is an important factor for water availability in plants and is indicative of the plug seedling quality. Thin leaves quickly desiccate (Tsukaya, 2004). Dini, R3B7, and R5B5 environments produced leaves with relatively higher SLW compared to leaves of plants grown in other light treatments. The light qualities used in FL and Dini were identical, but plants showed drastically different responses to these two treatments. In contrast, leaves of plants grown under R5B5 and R3B7 were similar to leaves of plants grown under Dini. It was shown that monochromatic LEDs did not have a positive effect on the leaf growth. R7B3, with a high R light ratio, increased the number and area of leaves but resulted in low SLW and was therefore detrimental to leaf growth. In contrast, R3B7 increased the total leaf area and SLW and seems to influence leaf growth more positively, even when restricting the formation of new leaves.
The SPAD value, indicative of the chlorophyll content, was the highest for plants in Dini. Seedlings grown in R5B5 and R3B7 had higher SPAD values compared to that of seedlings in all other environments (Fig. 7). Seedlings grown under B LEDs had the smallest SPAD value. It was previously thought that B light enhances chlorophyll biosynthesis, supported by observations in lettuce, cucumber, and spinach (Matsuda et al., 2007; Hogewoning et al., 2010; Lee et al., 2010). However, the SPAD value of plants increased with increasing B light ratio up to 70% in this study, while 100% B light resulted in the lowest SPAD value. In ‘Sunmang’ lettuce, the SPAD value increased with increasing B light proportion up to 47%, up to 59% in ‘Grand Rapid TBR’ (Son and Oh, 2013), and up to 35% in ‘Cuty’ tomato. These results suggest that a proper ratio of R and B LEDs, rather than B light alone, improves the SPAD value of seedlings (Son et al., 2018). Also, this result seems to be related to the leaf thickness, where a similar trend was observed (SLW, Fig. 5D). Several studies report that the SLW is positively correlated with the chlorophyll concentration (Buttery and Buzzell, 1977; Buttery et al., 1981; Hesketh et al., 1981). It is possible that appropriate combinations of R and B LEDs (R5B5 and R3B7) increased the leaf thicknesses and led to increased SPAD values.
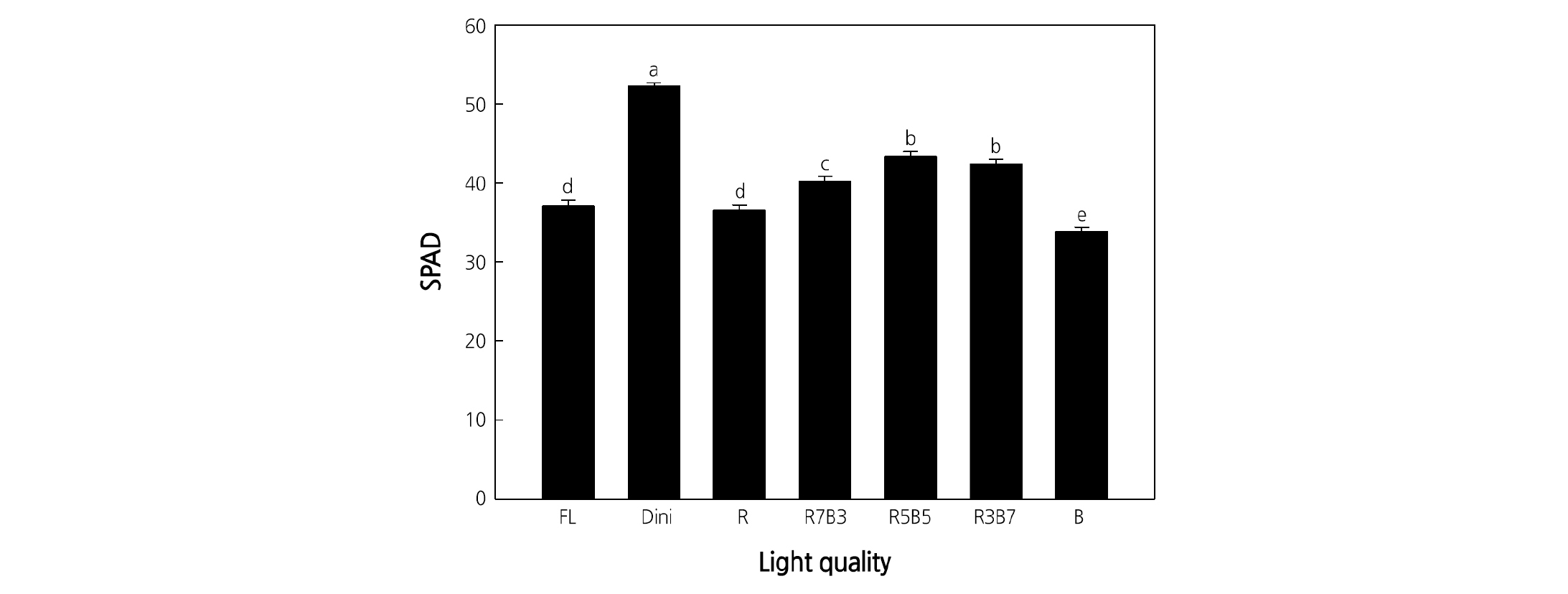
Fig. 7. The effects of the light quality on the SPAD value of ‘Mini Chal’ tomato (Solanum lycopersicum L.) measured 23 days after treatment. Refer to Fig. 2 for details on the light qualities. Vertical bars indicate the mean ± S.D. (n = 15). Different letters above the bars indicate significant differences by Duncan’s multiple range test at p ≤ 0.05.
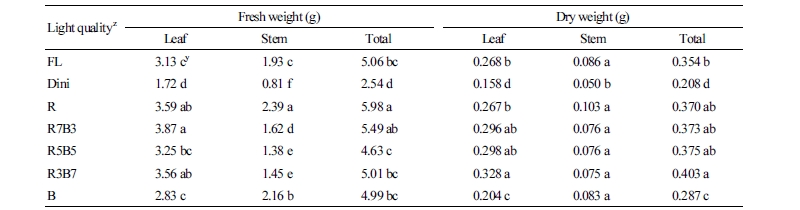
The fresh and dry weights of leaves and stems of ‘Mini Chal’ tomato plants are shown in Table 3. Total fresh weight of seedlings was the greatest in R LEDs. R7B3 produced plants with the greatest fresh leaf weight, followed by R and R3B7. Of all LEDs environments considered, R resulted in the greatest, and R5B5 and R3B7 resulted in the lowest fresh stem weight. R light promoted both stem and leaf developments. By contrast, R3B7 had the highest fresh leaf weight despite the lowest fresh stem weight. Fresh leaf and stem weights were the lowest in Dini of all considered environments. Several studies indicate that R light increases the fresh and dry shoot weights in various crops, such as lettuce, ice plant, and perilla (Nishimura et al., 2009; Johkan et al., 2010; Shimizu et al., 2011; Kim et al., 2016), as R light activates the phytochromes that promote plant growth by expanding leaves and elongating stems and petioles (Johnson et al., 1994; Mockler et al., 1999; Franklin and Quail, 2009; Kebrom and Mullet, 2016). For this reason, R light promoted the growth of seedlings and increased the fresh weight. However, seedlings grown in R5B5 and R3B7, which had low fresh stem weights, showed similar total dry weights as seedlings grown under R LEDs, which had high fresh stem weights. These results indicate that both R5B5 and R3B7 produce more compact seedlings because they produce seedlings with similar total dry weights as seedlings with higher stem fresh weights.
Dini, R5B5, and R3B7 produced seedlings with the greatest dry matter, while R and B LEDs environments produced plants with the low dry matter amounts (Fig. 8A). Kitaya et al. (1998) defined high-quality seedlings as ones with a high dry matter and low shoot/root ratio, specific leaf area, and hypocotyl length. Seedlings grown in R5B5 and R3B7 satisfy this definition.
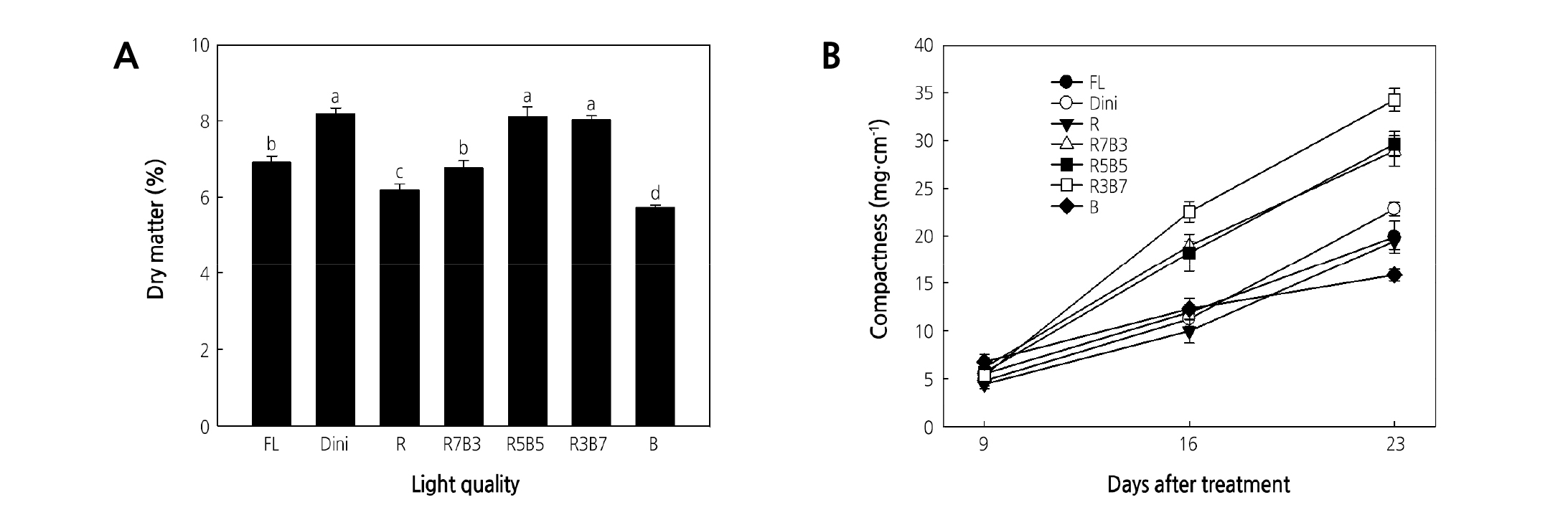
Fig. 8. The effects of the light quality on the dry matter (A) and compactness (B) of ‘Mini Chal’ tomato (Solanum lycopersicum L.) measured 9, 16, and 23 days after treatment. Refer to Fig. 2 for details on the light quality. Vertical bars indicate the mean ± S.D. (n = 15). Different letters above bars indicate significant differences by Duncan’s multiple range test at p ≤ 0.05.
Seedling compactness is the ratio of the dry shoot weight to plant height and indicates the seedling quality. A high compactness indicates that a plant has a short stem and heavy dry weight. At 16 and 23 DAT, R3B7 produced the most compact plug seedlings, followed by R7B3 and R5B5 (Fig. 8B). FL, Dini, R, and B produced seedlings with low compactness. Results of this study indicate that R LEDs increase the fresh weight but decrease the dry matter and compactness of seedlings (Table 3), while R3B7 results in a high dry shoot weight, increased dry matter, and compactness of seedlings. Combined R and B light environments, and R3B7 especially, increases the dry matter and compactness of seedlings.
Chlorophyll fluorescence analysis is a nondestructive method to analyze the physiological state of plants by quickly and easily assessing the effects of various environmental stresses on the photosynthetic tissues of plants (Kang et al., 2008). It was reported that the higher the chlorophyll fluorescence value, the more efficient the photosynthesis and transfer of sugar into the plant (Calatayud et al., 2006). When plants are subjected to extreme stresses, PS II activity is reduced and the Fv/Fm value decreases due to photoinhibition (Demmig and Björkman, 1987; Schreiber and Bilger, 1993; Long et al., 1994). In healthy plants, the Fv/Fm value is reported to be about 0.83 (Choi et al., 2004). Chlorophyll fluorescence (Fv/Fm) of seedlings grown under FL, Dini, R, R7B3, R5B5, R3B7, and B was 0.804, 0.815, 0.794, 0.791, 0.800, 0.800, and 0.810, respectively, and was the greatest for seedlings in Dini and the lowest for seedlings in R7B3 (Fig. 9). Others showed similar results. Also, the difference between the highest and lowest Fv/Fm values was only 0.024. These results are considered to be due to the similar conditions of seedling cultivation in the closed-type plant production system and the fact that the intensity of each light treatment was not high enough to cause differences in plant stresses.
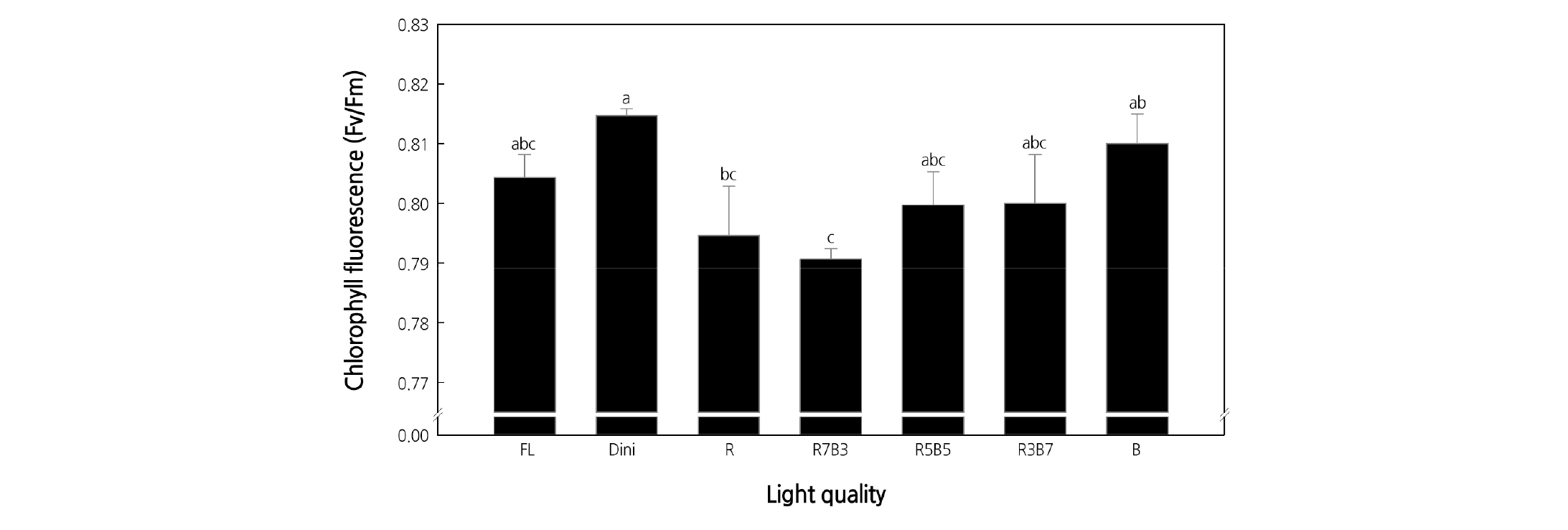
Fig. 9. The effects of the light quality on the chlorophyll fluorescence of ‘Mini Chal’ tomato (Solanum lycopersicum L.) measured 23 days after treatment. Refer to Fig. 2 for details on the light quality. Vertical bars indicate the mean ± S.D. (n = 3). Different letters above bars indicate significant differences by Duncan’s multiple range test at p ≤ 0.05.
Total Sugar and Starch Contents
The total sugar and starch contents of tomato plug seedlings are presented in Fig. 10. Tomato plants accumulate more starch and hexose than sucrose. The amount of starch accumulation was higher than the amount of hexose accumulation (Goldschmidt and Huber, 1992). The starch formed in the mesophyll cells by photosynthesis is called the assimilation starch, which can be saccharified and consumed in other parts of the crop (Fondy and Geiger, 1980). Plants grown under combined R and B lights had high total sugar content of leaves and stems, while plants grown under B LEDs had the lowest such total sugar content. Plants in R5B5 had the highest leaf and stem starch contents, while plants in B showed low leaf and stem starch contents. Combined R and B environments resulted in higher total sugar and starch contents in plants than monochromatic LEDs did in our study. Ficus elastic plants had the highest starch content when grown under combined R and B supplemental lights (R:B = 1:1 energy ratio) (Heo et al., 2010). Also, Doritaenopsis plants grown under red plus blue LEDs had significantly high levels of carbohydrates (starch, sucrose, glucose, and fructose) (Shin et al., 2008). This suggests that an appropriate combined R and B LEDs environment leads to a large accumulation of total sugar and starch. In our study, combined LEDs produced plants with high total sugar content, with R5B5 leading to especially high starch content in plants.
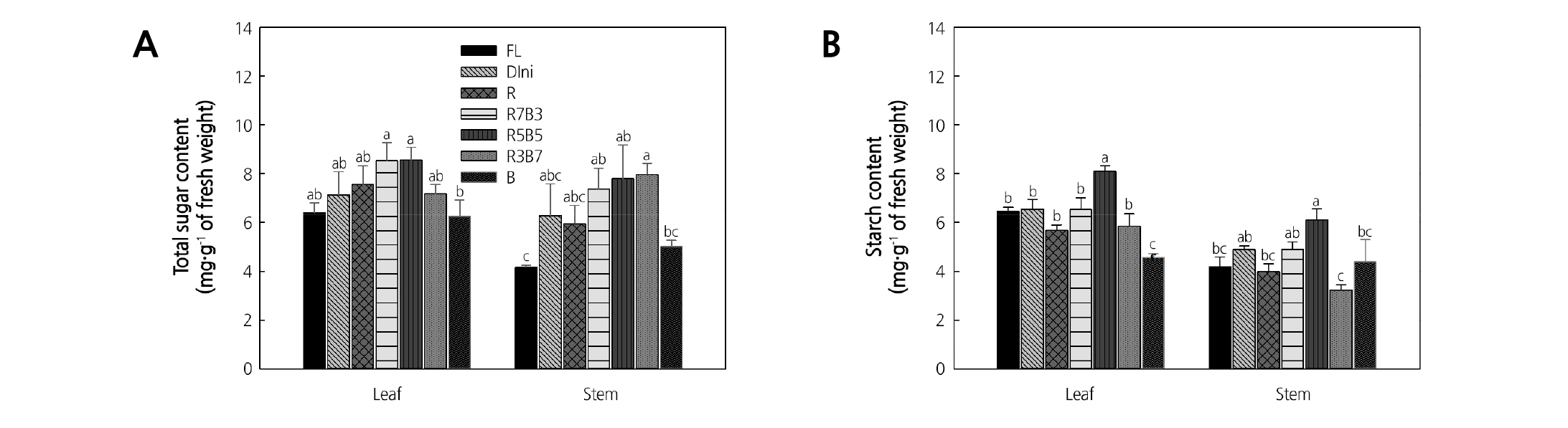
Fig. 10. The effects of the light quality on the total sugar content (A) and starch content (B) of ‘Mini Chal’ tomato (Solanum lycopersicum L.) measured 23 days after treatment. Refer to Fig. 2 for details on the light quality. Vertical bars indicate the mean ± S.D. (n = 3). Different letters above bars indicate significant differences by Duncan’s multiple range test at p ≤ 0.05.
Development of Tomato Plants After Transplanting
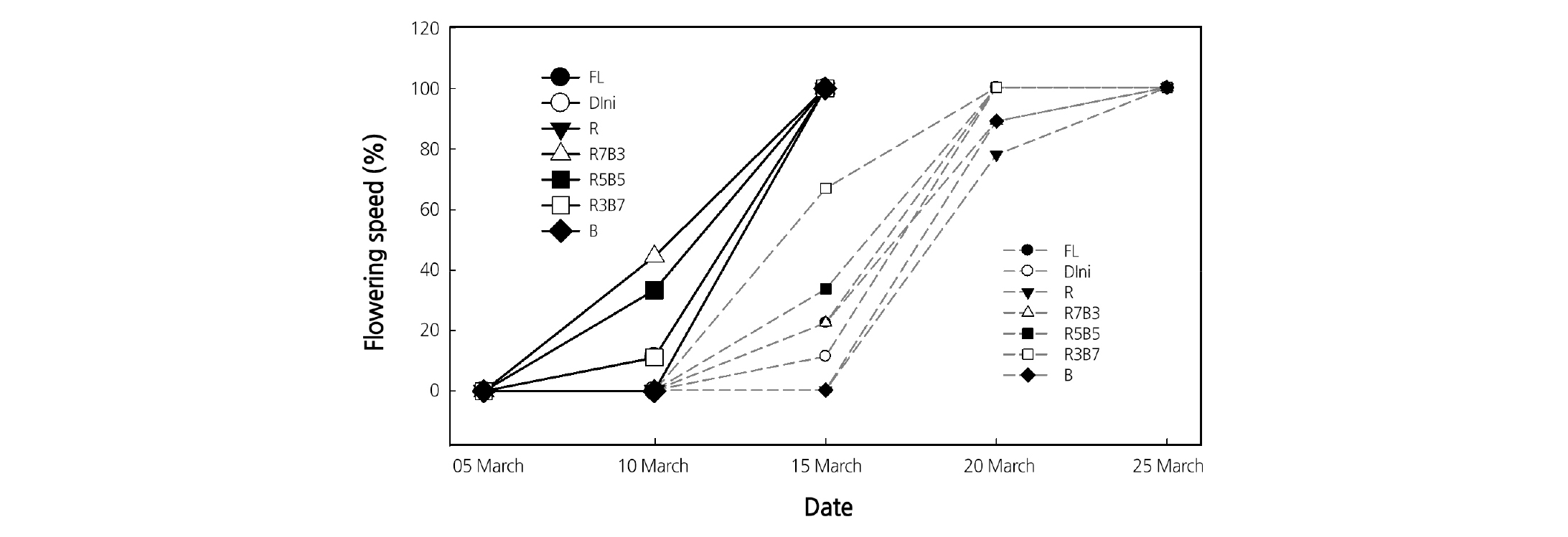
The total flowering speeds after transplanting of tomato plants grown under different light qualities during the seedling stage are presented in Fig. 11. R7B3 and R5B5 accelerated the flowering speed of the first cluster. In the second cluster, R3B7 induced the fastest flowering followed by R5B5. Kim et al. (1999) reported that raising the daily mean temperature, nitrogen concentration, and NH4+:NO3- ratio of the nutrient solution accelerated the flower bud formation of tomato seedlings. Our study suggests that the rate of flowering is a response induced by the light quality because the other environmental conditions were kept constant during the seedling period.
Several studies demonstrate a strong relationship between the flower development and carbohydrates (Rodrigo et al., 2000; Jean and Lapointe, 2001; Ruiz et al., 2001; Iglesias et al., 2003). Usually, cell division and synthesis of RNA and proteins in flower buds are promoted when plants are in flower bud formation (Bodson, 1977), and shoot tips act as a strong utilizing sink and tend to consume, rather than store, carbohydrates (Peng and Iwahori, 1994). Therefore, a high level of carbohydrates is needed for flower bud differentiation and development. Our results indicate that combined R and B LEDs environments lead to a high accumulation of total sugar and starch and promote flowering speed (Figs. 10 and 11).
The position of flower cluster and the cluster height of tomato plug seedlings at 122 days after transplanting are presented in Fig. 12. The light environment did not seem to significantly affect the position of the first and second flower clusters (Fig. 12A) but markedly differentiated the cluster height of the first and second clusters (Fig. 12B). Plants grown in FL had the greatest, and plants grown in Dini, R3B7, and R5B5 had the lowest first cluster heights. In the second cluster, plants grown in FL were the highest and plants grown in R5B5 were the lowest. In our results, the plants grown in combined LEDs environments were shorter at 23 DAT than those grown in monochromatic LEDs environments (Fig. 3). However, the differences in plant height were smaller at 122 days after transplanting than at the seedling period. These results indicated that the growth environment after transplanting was the same in all treatments.
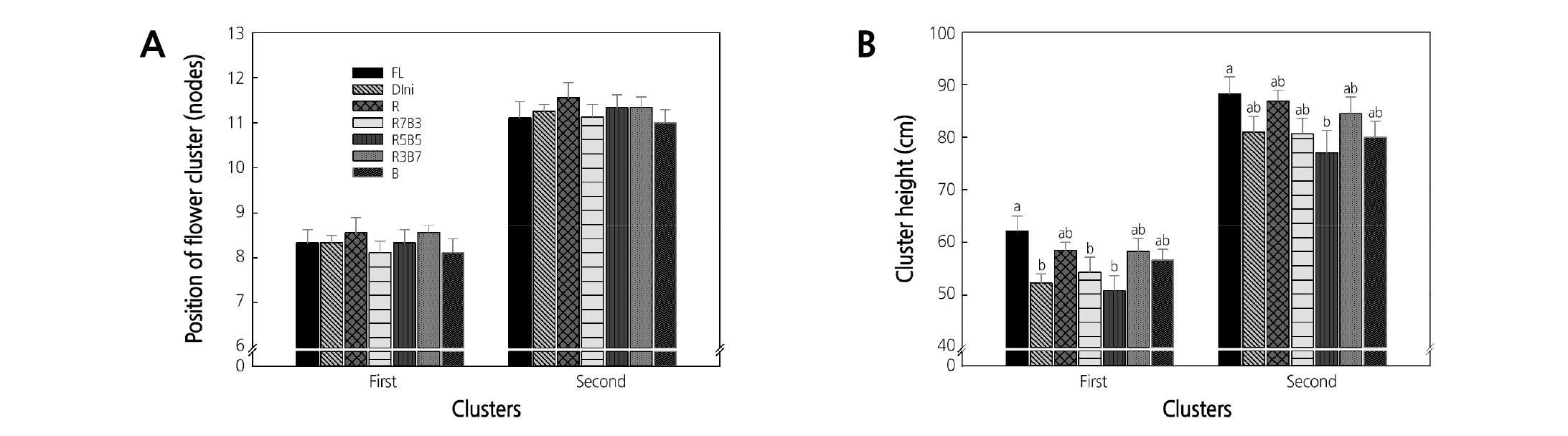
Fig. 12. The effects of the light quality on the position of flower cluster (A) and cluster height (B) of the first and second fruit setting nodes of ‘Mini Chal’ tomato (Solanum lycopersicum L.) measured 122 days after transplanting. Refer to Fig. 2 for details on the light quality. Vertical bars indicate the mean ± S.D. (n = 9). Different letters above the bars indicate significant differences by Duncan’s multiple range test at p ≤ 0.05.
There were negligible differences in the flower cluster positions and cluster heights. Consumers prefer short and compact seedlings because short seedlings have a low cluster height after transplanting, requiring less labor at harvest. However, the light environment during the seedling period did not significantly affect the growth after transplanting. Results presented in this experiment indicate that cluster heights do not differ after transplanting. However, if different light environments cause differences in stem elongation, they might make significant differences in cluster heights after transplanting.
Conclusion
In conclusion, the growth and development of ‘Mini Chal’ tomato seedlings were influenced by different light treatments. These findings suggest that R3B7 is an ideal environment for producing high-quality seedlings. It was also demonstrated that high-quality seedlings can be produced in a closed-type plant production system using a combination of LEDs, which can be widely applied in the agricultural industry.