Introduction
Materials and Methods
Plant Material
Experimental Design and Treatments
Preparation and Application of Chemical Mutagens
Rate of Mutation
Phenotypic Evaluation
Evaluation of Stomatal Characteristics
Flow Cytometry
Statistical Analysis
Results and Discussion
Survival and Mutation Rate
Growth of Oryzalin-induced Echeveria Cultivar Plants
Stomatal Characteristics
Flow Cytometry
Introduction
Over the past three decades, mutation breeding has been successfully applied in the ornamental plant industry (van Huylenbroeck, 2018). To alter the genetic information of a plant in a stable manner, mutagenesis is induced to produce mutants using physical, chemical, or biological methods (Forster and Shu, 2011). By modifying the phenotypic characteristics, such as the flower and leaf color, shape, and chlorophyll variegation, mutation breeding can introduce novel traits in existing plants, thereby increasing their desirability and marketability (Ascough and van Staden, 2008). Among the available tools for plant mutagenesis, the use of chemical mutagens is efficient and convenient to induce random mutations across the whole genome or within any locus or gene. For instance, chemical mutagens have been applied to induce large flowers in ornamental ginger (Prabhukumar et al., 2015), new color combinations in Delphinium malabaricum (Kolar et al., 2015), and morphological variations in the leaf and flower size and the shape of Gerbera jamesonii Hook plants (Ghani et al., 2013).
In the potted plant industry, succulents have seen sustained strong market demand due to their geometrical leaf shape, various colors, and ability to survive indoors with minimal care (Baldwin, 2013; Cabahug et al., 2018). Furthermore, owing to their aesthetic appeal and faux-like features, succulent plants with unique leaf shapes and color patterns demand higher prices (Griffiths, 2013; Cabahug et al., 2018). Natural breeding, cultivation management, and mutagenesis can be used to develop and enhance plant features. However, natural breeding requires long periods, and the altered environmental factors are subject to reversal upon seasonal changes. These factors make mutagenesis an efficient, stable, and rapid alternative (Pathirana, 2011; Cabahug et al., 2020).
Mutagenic plant induction is commonly achieved through the application of chemical mutagens. Compared to physical and biological methods, chemical mutagenesis is preferred, as mutagens are easy to handle and do not require sophisticated equipment (Oladosu et al., 2016). Colchicine and oryzalin are two commonly used chemical mutagens in ornamental crops, such as Cymbidium hybrids (Hwang et al., 2015), Hebe ‘Oratia Beauty’ (Gallone et al., 2014), and Watsonia lepida (Ascough and van Staden, 2008), and both have been found to be effective in both vegetative propagules and under in vitro conditions. In addition, both colchicine and oryzalin have been used to induce chromosome doubling in plants (Kermani et al., 2003). However, oryzalin has been reported to be more effective in inhibiting tubulin polymerization and to possess a greater affinity for plant microtubules while being less hazardous to human health than colchicine (Morejohn et al., 1987).
The proponents of the present study previously attempted the use of colchicine in various Echeveria cultivars with great success (Cabahug et al., 2020) and have pursued the exploration of whether the same effects can be achieved using oryzalin. The treatment concentration and duration of the proposed mutagens are essential considerations for achieving ideal results with mutagenesis, and the 50% lethality (LD50) dose of the mutagen is considered to provide the highest mutation rate (Leitão, 2012). To this end, the present study determined the optimal treatment concentration and duration of oryzalin in selected Echeveria cultivars and identified changes in the phenotype, stomata, and nuclear DNA content in oryzalin-induced mutants.
Materials and Methods
Plant Material
The selected succulent cultivars, specifically ‘Brave’, ‘Momotarou’, and ‘Sistar’, with fully expanded leaves, were procured from a succulent nursery in Goyang, Gyeonggi-do, Korea. The plants were transported to the physiology laboratory facility of the Department of Convergence Science, Sahmyook University in Seoul, Korea. Leaves from the three lower whorls of each plant were carefully removed from the mother plant to create clean and uniformly sized leaf-cutting propagules. The leaf cuttings were randomly selected and assigned to the respective treatments.
Experimental Design and Treatments
The present experiment followed a completely randomized design with a 5×4 factorial arrangement. With five treatment concentrations (0.2%, 0.4%, 0.6%, 0.8%, and 1.0%) and four treatment durations (3, 6, 9, and 12 h), 20 treatment combinations of oryzalin were applied. Each treatment combination was replicated five times, with ten leaf cuttings per replication, resulting in 50 leaves per treatment combination. Untreated leaf cuttings directly planted after detachment served as controls. In total, 1,050 leaf cuttings were used from each cultivar.
Preparation and Application of Chemical Mutagens
While handling any chemical mutagen, protective gear (N95 mask with extra lining, goggles, gloves, disposable arm sleeve guards, protective aprons, and lab gowns) was worn by the researchers during the preparation and treatment. A 1% stock solution of oryzalin (Surflan, Han Nong Ltd., Korea) was prepared and diluted with distilled water based on the treatment concentrations. Then, oryzalin solutions of different concentrations (700 µL) were placed in each compartment of a container. Leaf cuttings were placed upright in each compartment to ensure that the growing point of the cuttings (point of detachment) was exposed or submerged, which is the expected point of absorption. During the treatment, the containers with treated leaf cuttings were placed in a dark fume hood as a safety protocol to avoid their exposure to light. After dipping, the leaf cuttings were carefully removed from the treatment trays, air-dried, and planted in a nursery.
Rate of Mutation
The number of active Echeveria leaf cuttings that successfully produced shoots and roots was counted 12 weeks after the treatment (WAT). Putative mutants were separated into individual pots and subjected to a phenotypic evaluation. The rate of mutation was computed by determining the number of mutants divided by the number of successfully developed plants, and the obtained values was multiplied by one hundred to determine the percentage.
Phenotypic Evaluation
Based on forward genetics, mutant succulents were screened using phenotypic categories, in this case leaf morphology and chimeras, compared to the control phenotype (Wu et al., 2005; Taylor, 2017). Phenotypic data were collected from the mutated succulents and divided into two categories: a) plant parameters, in this case the plant height and diameter and the leaf length, width, and thickness; and b) the plant structure, which describes the shape, edge, and apex, following the Manual of Leaf Architecture (Ash et al., 1999).
Similarly, the leaf color was determined to compare the putative mutants and controls. CIELAB color (Spectrophotometer CM-2600d, Konica Minolta Inc., Japan) was used to determine the L*, a*, and b* values or color spaces to indicate the lightness, hue, and saturation, which were plotted in a quadrant to determine the actual color. The values were interpreted with reference to the Royal Horticultural Society (RHS) color chart to determine the RHS color groups and values.
Evaluation of Stomatal Characteristics
Six treatments of‘Brave’, four treatments of ‘Momotarou’, and three treatments of ‘Sistar’ as well as the control treatment were selected for a stomatal evaluation. Mutant plants were randomly selected for each treatment, and leaves were collected from the base of each plant. The nail varnish technique (Gitz and Baker, 2009) was used to evaluate the stomatal size and density. The samples were examined under a light microscope (Olympus BX53F, Japan) at 100× and 800× magnification. To determine the stomatal density, the number of stomata was randomly counted across the surface in each case. The stomatal size was measured using ImageJ software (v 1.52a, USA).
Flow Cytometry
Flow cytometry was conducted based on the methodology described by Dolezel et al. (2007). Healthy young leaves (approximately 20 mg) from the first three whorls of succulent mutant plants were sampled to measure the DNA content. The selected leaves were chopped with a razor blade in 1 mL of ice-cold nuclei isolation buffer (Galbraith et al., 1983). The homogenate was mixed by pipetting the solution up and down to avoid air bubbles. The mixture was subsequently filtered through a 50 µm nylon mesh (Shanghai Bolting Cloth Manufacturing Co. Ltd., Shanghai, China) and then through a 30 µm nylon mesh (CellTrics Filters, Sysmex Asia Pacific, Singapore). The final homogenate was labeled and 50 µL propidium iodide (Sigma-Aldrich, Cat. No. P4170; Molecular Probes; Cat. No. P-3566) was simultaneously added to 50 µL RNase (Sigma-Aldrich, Cat. No. R5000) and placed in a 1.5 mL Eppendorf tube (Sigma-Aldrich, T9786-1000EA). The sample was incubated on ice for approximately 5–10 min before the analysis. Thereafter, the tube was placed in a CytoFLEX flow cytometer equipped with a 50 mW 488 nm solid-state diode laser (Beckman Coulter Inc., USA).
Glycine max Merr. ‘Polanka’ with a known DNA content of 2.50 pg (2C) served as the reference standard. An external type of DNA content analysis was employed, which involved successive analyses of the sample and standard. The peaks of both the standard and sample were compared using CytExpert v2.3 (Beckman Coulter Inc., USA), and the sample DNA content was computed.
Statistical Analysis
Survival data and quantitative parameters were collected, organized, and subjected to a standard analysis of variance (ANOVA) using SPSS (version 22; IBM Inc., Armonk, NY). Duncan’s multiple range test was used to compare means at the 5% significance level.
Results and Discussion
Survival and Mutation Rate
The survival rate and number of mutants of each cultivar treated with oryzalin are shown in Table 1. Although the highest survival rate percentage was observed for ‘Momotarou’, only one mutant plant was produced from each treatment (0.4% at 6 h, 0.6% at 3 h and 9 h, and 0.8% at 9 h). ‘Sistar’ control plants and those treated with 0.6% + 3 h showed a 52% survival rate with no mutants, while the majority of the oryzalin-treated mutant plants showed high mortality rates exceeding 50%. The use of 0.8% + 3 h achieved the highest mutant rate of 7.14%, followed by 0.8% and 1.0% at 3 h (5.0% for both).
Table 1.
Survival rate and produced number of Echeveria mutants of Oryzalin-treated cultivars
The ‘Momotarou’ and ‘Sistar’ leaf cuttings showed high sensitivity to oryzalin, producing only one or two mutant plants per treatment. In addition to its application as a chemical mutagen, oryzalin is considered a dinitroaniline herbicide, which may cause mortality in plants by disrupting microtubules, thereby blocking the growth of plant cells (Morejohn et al., 1987; Taiz and Zeiger, 2010). However, in some cases, particularly in cells that can recover and reassemble, this chemical may induce alterations, such as those in mutant plants (Wasteneys and Williamson, 1989), a process which may have occurred at a low incidence in the tested cultivars.
In ‘Brave’, the survival rate of oryzalin-treated plants after five treatments (control, 0.2% + 9 h, 0.4% at 6 h and 9 h, and 1.0% + 6 h) exceeded 50%. The highest mutation rate was observed in the 1.00% + 3 h (25%) treatment, followed by the 0.6% + 6 h (13.33%) and 0.2% + 6 h (11.90%) treatments. Fourteen treatments produced mutants from oryzalin.
In‘Brave’, more mutants were produced than those in the other cultivars, suggesting the greater sensitivity of this cultivar to an oryzalin application. However, no definite trends were observed based on the treatment concentration or dipping time. The highest mutation rate was recorded in the treatment at the highest concentration (1.0%), with a mortality rate of 76%. Ke et al. (2019) reported that in addition to the concentration of the mutagen under the indicated treatment time, several other factors, such as the absorption dose, must be considered, and these factors vary across chemical mutagens. Likewise, regardless of the application of mutagenic treatments, the propagation of succulents using leaf cuttings or with any other vegetative propagule is somewhat unpredictable (Tuttle, 2012).
Growth of Oryzalin-induced Echeveria Cultivar Plants
Table 2 presents the growth response of Echeveria cultivars to the oryzalin application. Based on DMRT and Tukey’s test, the oryzalin treatment significantly affected the growth and development of all cultivars.
Table 2.
Plant parameters of E. ‘Brave’ induced with Oryzalin
| Treatment | Plant measurements | Leaf measurements (mm) | |||||
| Height (mm) | Diameter (mm) | No. of leaves | Length | Width | Thickness | ||
| ‘Brave’ | |||||||
| Control | 35.40 ± 8.66zcy | 84.12 ± 5.42 b | 37.67 ± 3.84 a | 31.62 ± 1.27 b | 16.31 ± 0.55 d | 6.79 ± 0.42 e | |
| 0.2% + 6 h | 49.34 ± 3.70 a | 63.29 ± 5.07 e | 21.20 ± 2.27 c | 26.38 ± 2.45 d | 18.89 ± 1.53 c | 8.18 ± 1.48 d | |
| 0.2% + 9 h | 36.68 ± 0.00 c | 57.41 ± 0.00 f | 16.00 ± 0.00 f | 29.93 ± 0.00 c | 18.71 ± 0.00 c | 8.76 ± 0.00 d | |
| 0.4% + 3 h | 40.92 ± 1.24 b | 87.97 ± 2.40 a | 29.00 ± 2.31 d | 38.52 ± 0.31 a | 21.94 ± 1.17 b | 10.01 ± 0.87 b | |
| 0.4% + 6 h | 50.34 ± 0.00 a | 69.43 ± 0.00 d | 27.00 ± 0.00 d | 34.18 ± 0.00 a | 24.02 ± 0.00 a | 10.02 ± 0.00 b | |
| 0.4% + 12 h | 41.34 ± 0.00 b | 56.41 ± 0.00 f | 33.00 ± 0.00 b | 15.28 ± 0.00 f | 17.58 ± 0.00 c | 8.50 ± 0.00 d | |
| 0.6% + 3 h | 39.45 ± 0.00 c | 60.78 ± 0.00 e | 23.00 ± 0.00 d | 25.70 ± 0.00 d | 23.82 ± 0.00 b | 9.23 ± 0.00 c | |
| 0.6% + 6 h | 38.46 ± 2.36 c | 57.85 ± 4.61 f | 22.00 ± 1.47 d | 25.80 ± 2.22 d | 18.50 ± 0.70 c | 8.44 ± 0.49 d | |
| 0.6% + 9 h | 48.40 ± 7.80 b | 75.91 ± 6.60 c | 24.50 ± 1.50 d | 29.81 ± 2.25 c | 19.11 ± 0.77 c | 8.37 ± 2.23 d | |
| 0.8% + 3 h | 49.53 ± 0.84 b | 73.42 ± 5.60 c | 27.33 ± 2.03 d | 35.68 ± 2.91 b | 20.42 ± 2.10 b | 8.67 ± 1.46 d | |
| 0.8% + 6 h | 45.51 ± 1.17 b | 57.93 ± 5.14 f | 18.00 ± 2.00 f | 28.43 ± 3.80 c | 18.95 ± 3.09 c | 8.04 ± 0.54 d | |
| 0.8% + 12 h | 34.36 ± 0.55 c | 53.73 ± 2.77 g | 22.50 ± 0.29 d | 23.25 ± 0.08 e | 18.61 ± 0.56 c | 8.47 ± 0.02 d | |
| 1.0% + 3 h | 45.44 ± 3.10 b | 81.04 ± 3.39 b | 30.50 ± 3.77 c | 31.00 ± 1.48 b | 18.42 ± 0.91 c | 9.18 ± 1.10 c | |
| 1.0% + 6 h | 38.55 ± 5.71 c | 60.89 ± 7.33 e | 16.25 ± 1.03 f | 26.87 ± 5.24 d | 18.35 ± 2.13 c | 8.14 ± 1.22 d | |
| 1.0% + 12 h | 35.85 ± 3.80 c | 58.88 ± 9.70 d | 21.00 ± 3.46 e | 24.21 ± 2.96 e | 21.39 ± 1.22 b | 13.70 ± 4.09 a | |
| F-testx | * | ** | ** | ** | ** | ** | |
| ‘Momotarou’ | |||||||
| Control | 43.51 ± 3.33 a | 74.62 ± 4.71 a | 30.00 ± 1.00 a | 32.96 ± 2.30 a | 18.79 ± 0.68 d | 4.90 ± 0.09 c | |
| 0.4% + 6 h | 40.49 ± 0.00 b | 67.99 ± 0.00 b | 23.00 ± 0.00 b | 31.59 ± 0.00 b | 28.68 ± 0.00 a | 9.51 ± 0.00 a | |
| 0.6% + 3 h | 33.28 ± 0.00 d | 49.56 ± 0.00 d | 18.00 ± 0.00 d | 22.96 ± 0.00 d | 20.83 ± 0.00 c | 6.47 ± 0.00 b | |
| 0.6% + 9 h | 36.10 ± 0.00 c | 60.47 ± 0.00 c | 21.00 ± 0.00 c | 26.56 ± 0.00 c | 24.20 ± 0.00 b | 5.84 ± 0.00 b | |
| 0.8% + 9 h | 26.34 ± 0.00 e | 66.89 ± 0.00 b | 23.00 ± 0.00 b | 29.42 ± 0.00 b | 20.40 ± 0.00 c | 6.91 ± 0.00 b | |
| F-test | ** | ** | ** | ** | ** | ** | |
| ‘Sistar’ | |||||||
| Control | 44.07 ± 1.94 c | 80.25 ± 2.02 b | 36.00 ± 1.73 a | 32.09 ± 4.16 c | 15.77 ± 0.33 e | 6.64 ± 0.62 c | |
| 0.4% + 3 h | 44.26 ± 1.15 c | 68.23 ± 0.63 c | 24.00 ± 0.58 b | 33.51 ± 0.38 c | 18.89 ± 0.69 c | 7.68 ± 0.05 b | |
| 0.8% + 3 h | 44.26 ± 3.57 c | 80.69 ± 4.57 b | 24.00 ± 1.15 b | 42.99 ± 5.39 a | 21.16 ± 0.57 c | 8.08 ± 0.60 b | |
| 0.8% + 6 h | 36.56 ± 0.00 d | 52.44 ± 0.00 e | 17.00 ± 0.00 c | 26.47 ± 0.00 d | 17.13 ± 0.00 d | 8.18 ± 0.00 b | |
| 1.0% + 3 h | 52.78 ± 0.00 a | 90.99 ± 0.00 a | 24.00 ± 0.00 b | 37.09 ± 0.00 b | 27.35 ± 0.00 a | 9.55 ± 0.00 a | |
| 1.0% + 6 h | 46.09 ± 3.27 b | 69.69 ± 1.31 c | 21.67 ± 2.60 b | 36.05 ± 2.54 b | 23.78 ± 2.05 b | 7.96 ± 0.72 b | |
| 1.0% + 12 h | 48.78 ± 0.00 b | 60.31 ± 0.00 d | 25.00 ± 0.00 b | 31.00 ± 0.00 c | 18.91 ± 0.00 c | 7.64 ± 0.00 b | |
| F-test | ** | ** | ** | * | ** | ** | |
In the present study, succulents sprouted new plantlets at different times; hence, the plant size varied until the plants reached the optimum size. Generally, chemical mutagens cause single-nucleotide polymorphisms (SNPs), which result in point mutations (Till et al., 2007). Several chemical plant mutagenesis studies have reported that these SNPs significantly alter leaf traits, which are considered strong and visible characteristics (Sikora et al., 2011), such as the leaf shape and architecture in pepper (Arisha et al., 2015) and chickpea (Laskar et al., 2015) and the leaf hair number in Brassica rapa (Zhang et al., 2018). Therefore, we focused on the number of leaves and on leaf measurements in the present study.
The number of leaves was significantly reduced in the ‘Brave’ (16–33 leaves), ‘Momotarou’ (18–23 leaves), and ‘Sistar’ (17–25 leaves) mutants compared to the numbers in their respective controls (37.67, 30, and 36 leaves). The leaf length was significantly affected by the chemical treatment, albeit without significant trends in ‘Sistar’ and ‘Brave’. Meanwhile, in ‘Momotarou’, the leaf length in the controls (32.96 mm) was significantly longer than that in the mutants (22.96–31.59 mm). Differences in the width and thickness between the control plants and mutants were more evident. Leaf cuttings treated with oryzalin produced wider leaves in‘Brave’ (24.02–18.35 mm), ‘Momotarou’ (28.68–20.40 mm), and ‘Sistar’ (17.13–27.35 mm) than their respective controls (16.31, 18.79, and 15.77 mm). This trend was also evident in the leaf thickness data; as such, the lowest thickness value was found in the ‘Brave’ controls (6.79 mm), and this value was significantly lower than that in the mutants (8.18–13.70 mm). Meanwhile, the highest thickness value was found after the 1.0% + 12 h treatment. Similar trends were observed for ‘Momotarou’ and ‘Sistar’, with control plants producing significantly thinner leaves (4.90 and 6.64 mm, respectively) than mutants (5.84–9.51 and 7.64–9.55 mm, respectively).
Previous studies have reported that mitotic inhibitors, such as oryzalin, induce chromosome doubling in ornamental plants, which in turn results in polyploidization (Castro et al., 2003; Pliankong et al., 2017). Polyploidization has provided beneficial effects for many valuable plants including fruit, vegetable, and ornamental crops (Chen et al., 2021; Cho et al., 2021; Kim et al., 2022). Mutants exhibiting fewer, shorter, and thicker leaves in Echeveria cultivars reflect compactness or compact growth—a desired characteristic in ornamental potted plant production (van Huylenbroeck, 2018). Similar results have been observed in ornamental Alocasia (Thi et al., 2004), Limonium sinuatum (Mori et al., 2021), and Rosa spp. (Kermani et al., 2003), with these results showing that oryzalin-treated plants tended to produce rounder, broader, and thicker leaves or even an altered leaf apex or shape in general. Upon observing and comparing phenotypic traits based on illustrations and descriptions from Ash et al. (1999), we found that the leaf apex shape in ‘Brave’ and ‘Momotarou’ was altered from acuminate (controls, Fig. 1.1-2B) to cuspidate (mutants, Fig. 1.1-2D) and that the leaf shape was visibly altered from linear (controls, Fig. 1.1-2A) to obovate (mutants, Fig. 1.1-2C). Similarly, in ‘Sistar’, the leaf apex shape was altered from oblanceolate (controls, Fig. 1.3B) to cuneate (mutants, Fig. 1.3D) and the leaf shape was altered from round (controls, Fig. 1.3A) to obtuse (mutants, Fig. 1.3C).
Regarding the leaf blade colors, significant differences in certain color traits were noted in the CIELAB readings between the controls and the mutants (Table 3). The color readings are indicated as *L (black, 0 to white, 100), *a (green, negative values to red, positive values), and *b (blue, negative values to yellow, positive values) values. In ‘Brave’, the L* values were significantly higher in mutants, corresponding to lighter colors, while in‘Momotarou’ and ‘Sistar’, the *L values were significantly lower in mutants, signifying darker tones. Moreover, regardless of the significant differences in the a* and b* values, no changes in the RHS values or greyed-green (189A) and yellow-green (147A) color groups were noted in‘Brave’ and ‘Momotarou’. However, in ‘Sistar’, control plants were categorized into a significantly different color group (148B, yellow-green), which was a lighter color group, from the oryzalin-treated mutants (189A and 191A, greyed-green). Previous studies of chemically mutated species have reported consistent results, with the mutant cultivars exhibiting darker green leaf tones in roses (Kermani et al., 2003) and colchicine-induced Echeveria cultivars (Cabahug et al., 2020). According to Manzoor et al. (2019), the darker leaf color is an effect of polyploidization in ornamental plants. In‘Brave’, the amount of epicuticular wax or powder on the leaves was significantly increased is the mutants; this powdery material plays an important role in light absorbance and photosynthesis (Mulroy, 1979; Feakins and Sessions, 2010), and the abundance of this material may result in lighter colors.
Table 3.
Color readings (CIELAB values) of Oryzalin-induced Echeveria mutants
| Treatment | CIELAB color reading | RHS value | RHS color group | ||
| L* | a* | b* | |||
| ‘Brave’ | |||||
| Control | 45.94 ± 2.57zcy | –4.58 ± 0.23 | 9.40 ± 3.80 | 189A | Greyed-Green |
| 0.2% + 6 h | 55.74 ± 1.90 b | –4.63 ± 0.13 | 8.86 ± 31.15 | 189A | Greyed-Green |
| 0.2% + 9 h | 49.97 ± 1.48 b | –4.14 ± 0.10 | 5.41 ± 0.78 | 189A | Greyed-Green |
| 0.4% + 3 h | 50.90 ± 2.83 b | –5.03 ± 0.65 | 12.75 ± 0.99 | 189A | Greyed-Green |
| 0.4% + 6 h | 53.77 ± 5.49 b | –4.80 ± 0.51 | 4.96 ± 1.47 | 189A | Greyed-Green |
| 0.4% + 12 h | 53.97 ± 6.95 b | –4.72 ± 0.62 | 9.16 ± 0.88 | 189A | Greyed-Green |
| 0.6% + 3 h | 49.76 ± 5.72 b | –4.72 ± 0.20 | 7.49 ± 1.18 | 189A | Greyed-Green |
| 0.6% + 6 h | 59.70 ± 1.52 a | –5.80 ± 0.10 | 10.74 ± 1.22 | 189A | Greyed-Green |
| 0.6% + 9 h | 47.34 ± 3.06 b | –4.31 ± 0.11 | 8.74 ± 1.39 | 189A | Greyed-Green |
| 0.8% + 3 h | 56.91 ± 1.81 b | –4.63 ± 0.78 | 7.47 ± 1.04 | 189A | Greyed-Green |
| 0.8% + 6 h | 51.21 ± 0.96 b | –5.06 ± 0.18 | 8.70 ± 2.86 | 189A | Greyed-Green |
| 0.8% + 12 h | 60.32 ± 2.05 a | –5.06 ± 3.9 | 7.85 ± 1.54 | 189A | Greyed-Green |
| 1.0% + 3 h | 58.64 ± 2.53 a | –5.74 ± 0.23 | 9.46 ± 2.19 | 189A | Greyed-Green |
| 1.0% + 6 h | 55.47 ± 1.06 b | –4.90 ± 0.03 | 11.38 ± 1.25 | 189A | Greyed-Green |
| 1.0% + 12 h | 47.21 ± 3.60 b | –1.88 ± 2.09 | 7.94 ± 0.53 | 189A | Greyed-Green |
| F-testx | ** | NS | NS | ||
| ‘Momotarou’ | |||||
| Control | 27.93 ± 4.03 a | –6.22 ± 1.74 b | 14.75 ± 0.67 a | 147A | Yellow-Green |
| 0.4% + 6 h | 26.65 ± 3.68 b | –4.67 ± 0.28 a | 5.35 ± 0.41 b | 147A | Yellow-Green |
| 0.6% + 3 h | 26.51 ± 1.50 b | –4.56 ± 0.43 a | 6.28 ± 1.60 b | 147A | Yellow-Green |
| 0.6% + 9 h | 26.02 ± 2.34 b | –4.50 ± 0.24 a | 7.01 ± 1.37b | 147A | Yellow-Green |
| 0.8% + 9 h | 26.06 ± 4.19 b | –5.24 ± 0.02 a | 7.45 ± 0.56 b | 147A | Yellow-Green |
| F-test | ** | ** | ** | ||
| ‘Sistar’ | |||||
| Control | 55.52 ± 0.97 a | –5.62 ± 0.14 | 18.87 ± 2.15 a | 148B | Yellow-Green |
| 0.4% + 3 h | 51.77 ± 2.24 b | –5.29 ± 0.14 | 9.49 ± 1.38 d | 189A | Greyed-Green |
| 0.8% + 3 h | 43.05 ± 2.94 c | –5.06 ± 0.33 | 12.53 ± 1.35 c | 189A | Greyed-Green |
| 0.8% + 6 h | 43.10 ± 2.67 c | –5.94 ± 0.27 | 15.71 ± 1.28 b | 189A | Greyed-Green |
| 1.0% + 3 h | 53.00 ± 1.08 b | –5.13 ± 0.11 | 12.89 ± 1.79 c | 191A | Greyed-Green |
| 1.0% + 6 h | 50.63 ± 0.86 b | –5.34 ± 0.87 | 11.51 ± 1.62 c | 189A | Greyed-Green |
| 1.0% + 12 h | 50.61 ± 1.79 b | –4.36 ± 0.32 | 10.20 ± 1.32 d | 189A | Greyed-Green |
| F-test | ** | NS | * | ||
Stomatal Characteristics
To compare controls and mutants, the two highest treatment concentrations and longest treatment durations that produced mutant plants were selected for a stomatal analysis. Using the methodology proposed by Gitz and Baker (2009), the stomatal size and density as well as the epidermal cell density were analyzed (Table 4) and found to have been significantly affected by the application of oryzalin in the tested succulent cultivars.
Table 4.
Adaxial stomatal characteristics of Oryzalin-treated Echeveria cultivars
| Treatment | Stomata density | Epidermal cells | Stomata size (µm) |
| ‘Brave’ | |||
| Control | 9.00 ± 1.15z ay | 64.33 ± 1.45 a | 20.22 ± 0.30 b |
| 0.8% + 6 h | 6.67 ± 0.33 b | 51.67 ± 2.90 b | 27.64 ± 0.09 a |
| 1.0% + 6 h | 6.67 ± 0.33 b | 53.33 ± 2.40 b | 28.35 ± 0.02 a |
| F-testx | ** | ** | ** |
| ‘Momotarou’ | |||
| Control | 20.67 ± 1.45 a | 124.33 ± 2.33 a | 17.76 ± 0.64 b |
| 0.6% + 9 h | 8.33 ± 0.33 b | 86.33 ± 1.86 b | 21.69 ± 0.32 a |
| 0.8% + 9 h | 6.67 ± 0.67 b | 78.00 ± 3.21 b | 21.41 ± 0.15 a |
| F-test | ** | ** | ** |
| ‘Sistar’ | |||
| Control | 8.33 ± 0.67 a | 94.33 ± 0.33 a | 18.61 ± 0.40 c |
| 0.8% + 6 h | 7.67 ± 0.33 b | 87.67 ± 1.76 b | 20.74 ± 0.39 b |
| 1.0% + 6 h | 7.00 ± 0.00 b | 81.00 ± 1.53 b | 27.18 ± 0.83 a |
| F-test | ** | ** | ** |
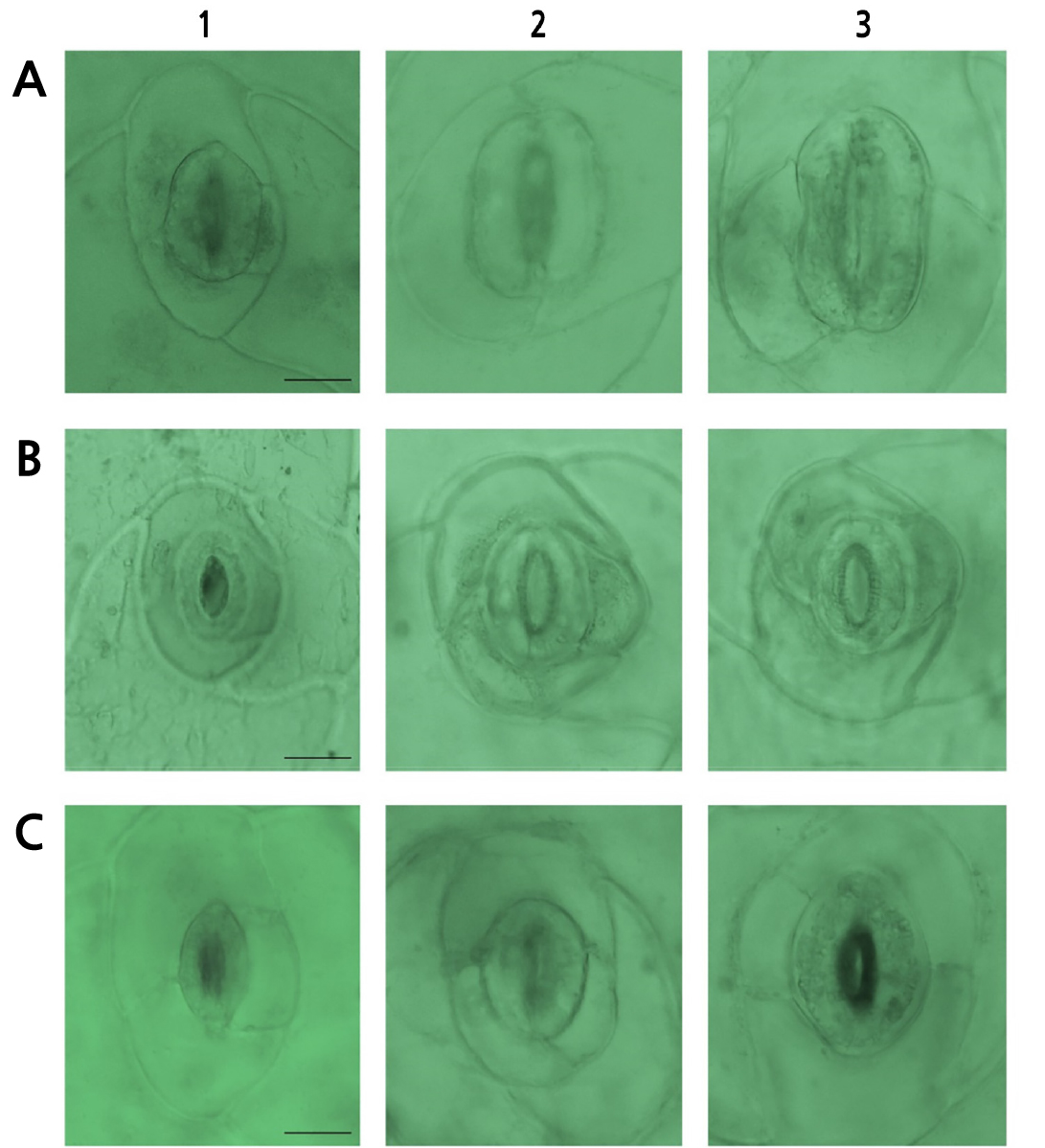
For ‘Brave’, the controls (Fig. 2.A1) showed the highest stomatal (9.00) and epidermal cell (64.33) density levels but the smallest stomatal size (20.22 µm), whereas the mutants (Fig. 2.A2-3) showed the largest stomatal size (27.64–28.35 µm). These trends in the stomatal density (Fig. 2) and stomatal size (Fig. 3) were consistent throughout all cultivars.
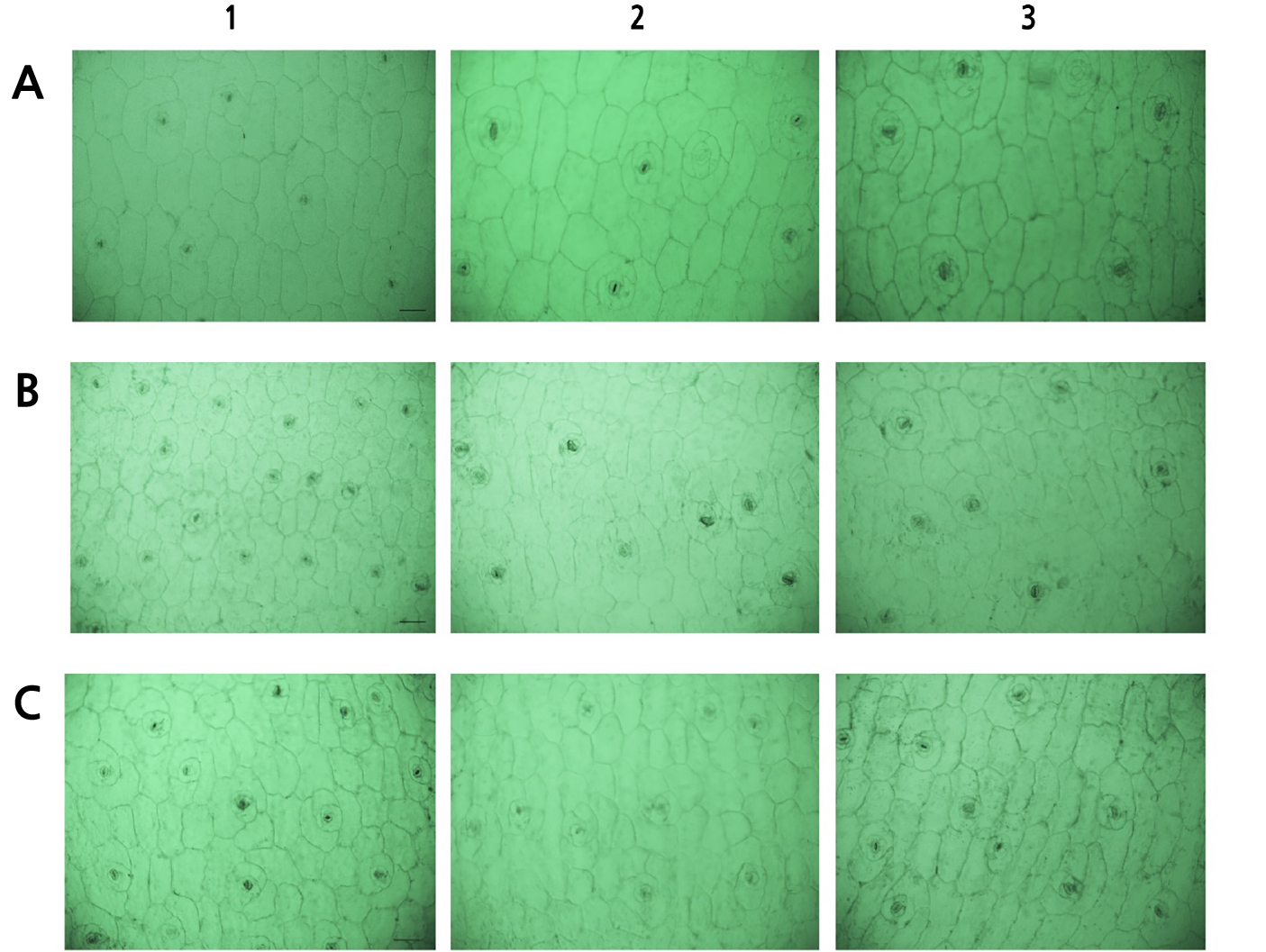
Fig. 2.
Comparison of stomata and epidermal cell density levels among oryzalin-treated Echeveria cultivars. (A) ‘Brave’, (B) ‘Momotarou’, and (C) ‘Sistar’, which were treated oryzalin with: A1–C1, controls; A2, 0.8% + 6 h; A3, 1.0% + 6 h; B2, 0.6% + 9 h; B3, 0.8% + 9 h; C2, 0.8% + 6 h; and C3, 1.0% + 6 h (100× magnification, Scale Bars = 50 µm).
Oryzalin-mutated ornamentals, such as roses (Kermani et al., 2003), sea lavender (Mori et al., 2021), and Alocasia (Thi et al., 2004), have been reported to show wider leaves, lower stomatal density, and larger stomata values, all of which are attributed to polyploidization. Along with leaf ovalization (Thao et al., 2003) and stomatal enlargement (Gantait et al., 2011), these morphological characteristics are consistent with the consequences of chromosome doubling. In a previous study, Eisinger et al. (2012) suggested that anti-microtubule mutagens, such as oryzalin, directly affect the formation and functions of stomata.
Flow Cytometry
Flow cytometry, as a widely used tool to establish nuclear DNA content in plants, is considered an effective tool during a mutant analysis to compare control and untreated plants with mutants (Xu et al., 2010; Lertsutthichawan et al., 2017; Yesmin et al., 2022; Deepo et al., 2023). In the present study, flow cytometry was used successfully to confirm the differences between succulent cultivar mutants and their respective control plants, as shown in histograms in Fig. 4.
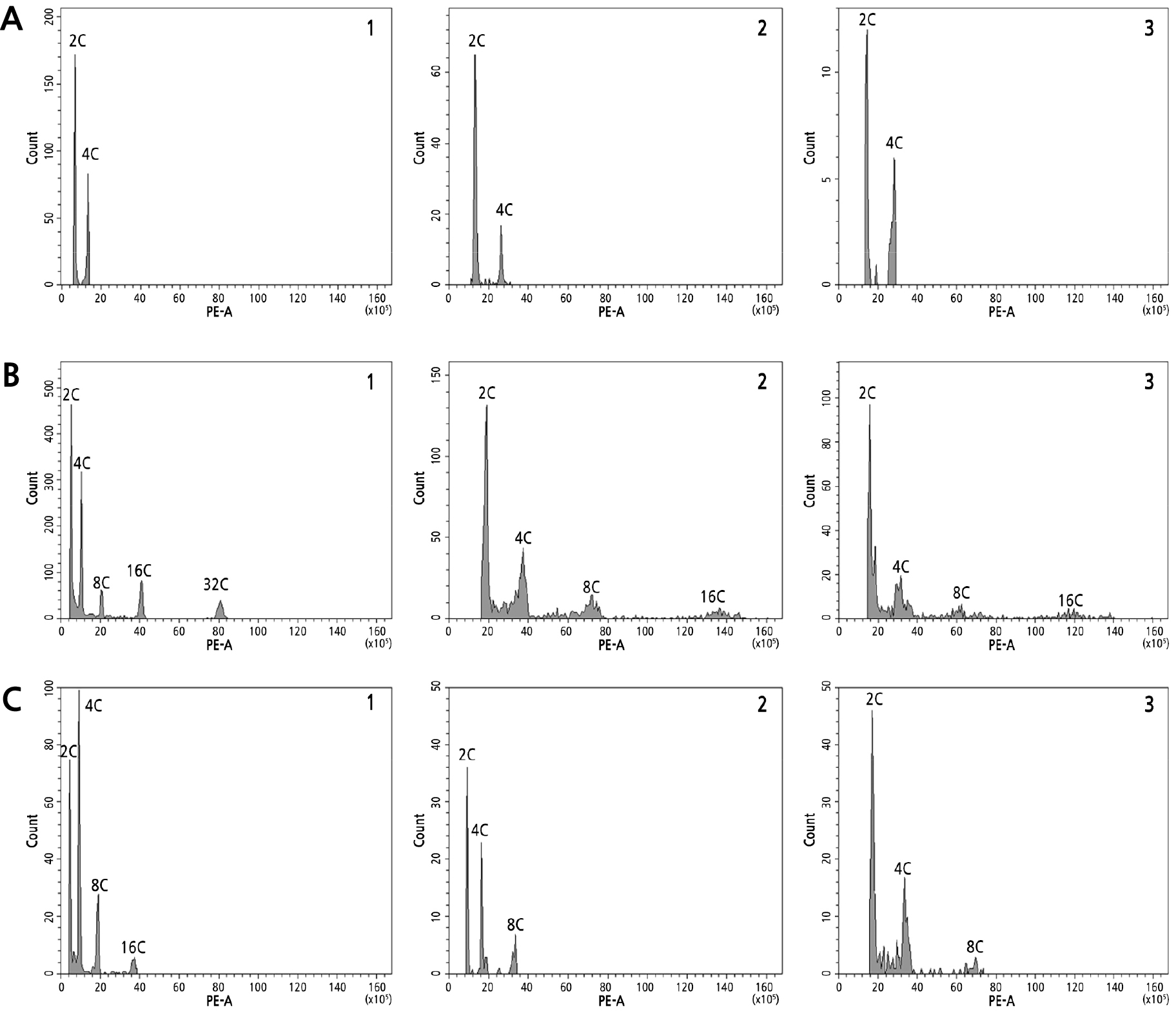
Fig. 4.
Histogram of nuclear DNA contents among oryzalin-treated Echeveria cultivars. (A) ‘Brave’, (B) ‘Momotarou’, and (C) ‘Sistar’. A1–C1, control, showing increased values compared with oryzalin-treated mutants; A2, 0.8% + 6 h; A3, 1.0% + 6 h; B2, 0.6% + 9 h; B3, 0.8% + 9 h; C2, 0.8% + 6 h; and C3, 1.0% + 6 h.
Inthe ‘Brave’ controls, the 2C peak was detected on channel 8, while in oryzalin-induced mutants treated at 0.8% and 1.0% for 6 h, the peak was noted on channel 16, implying the doubling of the nuclear DNA content. A similar trend was noted in‘Momotarou’, with the 2C peak at channel 10 in controls and at channel 20 in putative mutants in the two treatments. Meanwhile, in‘Sistar’, the 2C peak was noted at channel 8; however, in the putative mutants, this peak was detected at channel 12 after the 0.8% + 6 h treatment and at channel 20 in the 1.0% + 6 h treatment case. These results are consistent with observations in other oryzalin-induced ornamentals, such as gerbera (Tosca et al., 1995), Buddleja (Dunn and Lindstorm, 2007), and Scutellaria barbata (Yue et al., 2011). Previous studies have shown that an increase in the nuclear DNA content implies chromosome doubling, which results in the creation of polyploids (Doyle and Coate, 2018). Furthermore, oryzalin, as an antimitotic agent, induces polyploidy and has often been applied as an alternative to colchicine given that it can more effectively produce polyploids and is less toxic to animals and humans due to its greater binding affinity for plant tubulins (Morejohn et al., 1987; Dhooghe et al., 2010).
In addition to the shifts in the 2C peak locations, ‘Momotarou’ and ‘Sistar’ exhibited endoreduplication between the controls and mutants. However, in ‘Sistar’ mutants, the endoreduplication cycle was reduced despite the doubling of the nuclear DNA content. Cell polyploidization can also be achieved through endoreduplication, wherein the chromosomal DNA replicates in the absence of mitosis (Joubès and Chevalier, 2000). Each cycle is performed between the S and G phases. Various Echeveria species displaying endopolyploidy (4C–64C) were reported in a study by Palomino et al. (2021); the authors suggested that endopolyploidy plays a vital role in the genus Echeveria as a coping mechanism for these plants in fluctuating environments, which are stressful for plants. However, in‘Sistar’, the reduction may be caused by oryzalin as an antimitotic agent, hindering further normal progression of the cycle.
In summary, oryzalin successfully produced mutants in all Echeveria cultivars, albeit with a higher mutation rate in ‘Brave’. The putative mutants exhibited polyploidization, which conferred upon them certain characteristics, in this case fewer yet larger and thicker leaves with generally darker tones. A further analysis of the stomata revealed that oryzalin-induced mutants possessed larger but fewer stomata and a lower epidermal cell density. In addition to phenotypic and stomatal characteristics, polyploidization was confirmed using flow cytometry, which revealed increased or doubling of the nuclear DNA content of the mutants. Further research congruent with the present study will involve a karyotypic analysis coupled with these evaluation tools to produce succeeding generations of mutant plants. Our findings will guide the development of new cultivars of succulents and other related species with the desired traits through the application of oryzalin.