Introduction
Materials and Methods
Plant Materials
Morphological Character Detection
Data Analysis
Results
Morphological Variation Between the Two Cultivated Perilla Crop Variants from Different Areas of China
Geographical Differences in Cultivated var. frutescens Found in High and Middle Latitude Areas of China
Multivariate Analysis of Quantitative and Qualitative Characteristics in Perilla variant accessions
Discussion
Morphological Variation Between Two Cultivated Types of Perilla Crop from Different Areas of China
Geographical Differences in Cultivated var. frutescens between High Latitude and Middle Latitude Areas of China
Introduction
Perilla frutescens (L.) Britt. (Labiatae) is a perennial, self-pollinated crop. Perilla crop is widely distributed in the Himalayan Mountains, Southeast Asia, and East Asia; i.e., China, Korea, and Japan (Lee and Ohnishi, 2001, 2003; Sa et al., 2015). This crop is separated into two different cultivated types on the basis of their morphology and utilization. P. frutescens var. frutescens, is an oil crop that is known by the common names ren in Chinese, deulggae in Korean, and egoma in Japanese. P. frutescens var. crispa, is a medicine or vegetable crop in East Asia. Its common names are zisu in Chinese, cha-jo-ki in Korean, and shiso in Japanese (Lee et al., 2002; Lee and Ohnishi, 2001, 2003). P. frutescens var. frutescens is generally a tall plant with large, soft seeds ( > 2 mm), green leaves and stems, and non-wrinkled leaves with a fragrance specific to var. frutescens. Conversely, P. frutescens var. crispa is smaller plant with small hard seeds ( < 2 mm), and red or green leaves and stems, wrinkly or non-wrinkly leaves, and a fragrance specific to the var. crispa (Lee et al., 2002; Lee and Ohnishi, 2001, 2003).
These two variants have been cultivated and used in East Asia since ancient times. In East Asia today, P. frutescens var. frutescens is extensively cultivated and used in Korea (Nitta, 2001; Nitta et al., 2003), although cultivated var. frutescens probably originated in China (Makino, 1961). In Korea, var. frutescens is used both as an oil crop and vegetable crop; however, var. crispa is not currently being cultivated because of its use as a Chinese medicine has decreased, although it is occasionally found as a relict in Korea (Lee and Ohnishi, 2001; Lee et al., 2002). Conversely, var. crispa is extensively cultivated and used in Japan, where its leaves are used for spicy vegetables or pickles (Nitta, 2001; Nitta et al., 2003). In East Asia, although the wild ancestor of Perilla crop has not been identified, weedy plants of Perilla crop were recently described (Nitta and Ohnishi, 1999; Lee and Ohnishi, 2001, 2003; Lee et al., 2002; Nitta et al., 2003, 2005). In East Asia, these weedy plants grow naturally and are commonly found near roadsides, wastelands, and farm fields or houses. In China, the two cultivated types of Perilla crop can be found in multiple provinces, but the main producing areas of cultivated var. frutescens are in the central and southern regions of China, such as Taiwan, Hunan, Jiangxi, Jiangsu, Anhui, Henan, and Hulunbuir City of Inner Mongolia. In addition, the weedy types of Perilla crop usually grow either on the edge of a village or field, or around farmhouses. The cultivated var. frutescens is most widely grown in the Yanbian area of Northeast China inhabited by Chinese-Koreans, followed by Changchun, Jilin, and Songyuan of Jilin province (Wu et al., 2005). In northern areas of China, seeds of cultivated var. frutescens are chiefly used as an oil crop (Tan et al., 2012), while P. frutescens var. crispa is used mainly for Chinese medicine (Meng et al., 2009). For example, the seeds and dry leaves of P. frutescens var. crispa are used in several classical prescriptions (Luo et al., 2000; Mao et al., 2010), as well as for the treatment of depression (Yu et al., 2016), relieving pain and aiding in miscarriage prevention, treatments for cough or asthma, and constipation (Yu et al., 2016). In addition, the leaves of P. frutescens var. crispa are commonly used in the preparation of crab or fish due to its use in detoxification in China for more than 2,000 years (Yu et al., 2016). However, var. crispa is cultivated on a small scale and only found in a relict form in farm fields or beside farmhouses in China.
Domestication is an evolutionary process through which domesticated plants become morphologically and physiologically divergent from their wild ancestors (Schwanitz, 1966; Harlan, 1992), and the morphological differentiation in crop species within their geographic distribution areas has been of considerable interest in the study of the evolution of crop species (Gould and Johnston, 1972; Wyatt and Antonovics, 1981). Although China has a very long cultivation history of Perilla crop, information on the morphological variation in accessions of Perilla crop collected from various regions in China has not been sufficiently collected. Therefore, the purpose of this study was to analyze the morphological variation in accessions of Perilla crop collected from different areas of China using 17 morphological characters as parameters for measuring their variation.
Materials and Methods
Plant Materials
The materials for this study consist of 87 accessions (84 cultivated var. frutescens and 3 cultivated var. crispa) collected in different provinces of China. These provinces are located in the high latitude (Northeast China) and middle latitude areas (North China and Northwest China) of China. The number of accessions and the name of the collection places for Perilla accessions collected in China are shown in Table 1 and Fig. 1.
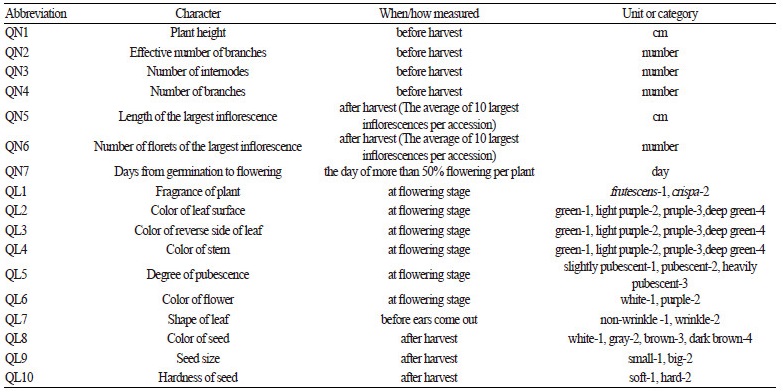
Table 1. Perilla frutescens accessions collected from northern areas of China that were surveyed for morphological trait differences.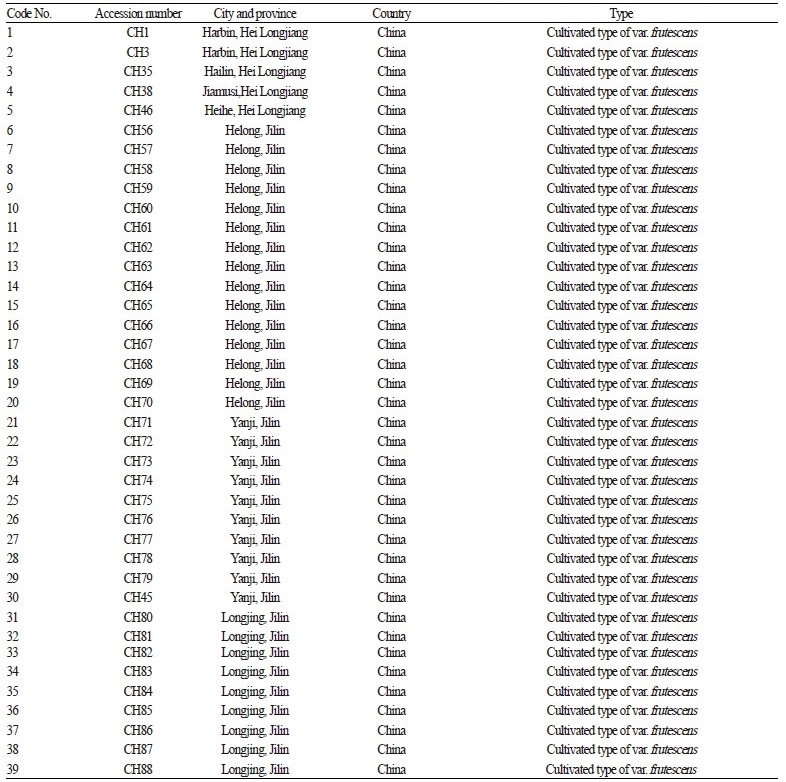 |
Table 1. Continued. zDifferent letters (a, b) within a row indicate significant differences at p <0.05. |
Fig. 1.
Collection sites of Perilla frutescens accessions that were evaluated in this study. Perilla frutescens accessions collected from high-latitude areas (Northeast China, 41°21′N--53°33′N) are shown in the upper oval; Perilla frutescens accessions collected from middle -latitude areas (North China and Northwest China, 32°18′N-41°21′N) are shown in the lower oval.
Morphological Character Detection
To assess the morphological variation of the accessions of two cultivated types of Perilla crop plants, ten individuals of each accession were grown in a field at the College of Agriculture and Life Sciences, Kangwon National University, Chuncheon, Gangwon-do in Korea. Approximately twenty seeds from each accession were sown in a nursery bed in early May and kept in a glass house for a month. Ten seedlings of each accession were then transplanted into the field in early June. We examined seven quantitative and 10 qualitative characters that were selected on the basis of the previous report by Lee and Ohnishi (2001) and evaluated at the appropriate growth stages as described in detail in Table 2 of this report. Measurements of the seven quantitative characters and observation of 10 qualitative characters were made on 10 individuals for each accession.
Data Analysis
The measurements of each trait were compared among the accessions using one-way analysis of variance (ANOVA) followed by the Student-Newman-Keuls test for multiple comparison of means among the accessions of cultivated types of var. frutescens and var. crispa collected from different areas of China. Principal component analyses (PCAs) were conducted to detect differences among accessions and to try to divide into groups the accessions. Geographical comparison of the accessions collected from high and middle latitude areas of China was also made by using univariate and multivariate analyses. These comparisons were made only for accessions of cultivated var. frutescens and var. crispa because there was no sample of weedy types of var. frutescens and var. crispa available in this study. Microsoft Excel Statistical Analysis System Program was used for univariate and multivariate analyses.
Results
Morphological Variation Between the Two Cultivated Perilla Crop Variants from Different Areas of China
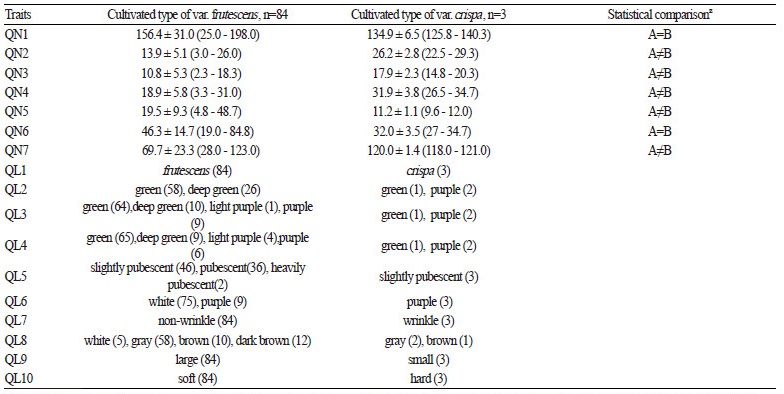
The average values, standard deviation, range of the seven quantitative traits, and morphological characteristics of 10 qualitative traits for accessions of cultivated types of var. frutescens and var. crispa were summarized in Table 3. According to the results of our quantitative traits survey, QN1 was 156.4 cm (25 - 198 cm) and 134.9 cm (125.8 - 140.3 cm) in cultivated types of var. frutescens and var. crispa accessions, respectively. QN2 was 13.9 (3.0 - 26.0) and 26.2 (22.5 - 29.3) in accessions of cultivated types of var. frutescens and var. crispa, respectively. QN3 were 10.8 (2.3 - 18.3) and 17.9 (14.8 - 20.3) in accessions of cultivated types of var. frutescens and var. crispa, respectively. QN4 was 18.9 (3.3 - 31.0) and 31.9 (26.5 - 34.7) in accessions of cultivated types of var. frutescens and var. crispa, respectively. QN5 was 19.5 (4.8 - 48.7) and 11.2 (9.6 - 12.0) in accessions of cultivated types of var. frutescens and var. crispa, respectively. QN6 was 46.3 (19.0 - 84.8) and 32.0 (27.0 - 34.7) in accessions of cultivated types of var. frutescens and var. crispa, respectively. QN7 was 69.7 (28.0 - 123.0) and 120.0 (118.0 - 121.0) in accessions of cultivated types of var. frutescens and var. crispa, respectively.
The results of the qualitative characters survey for fragrance (QL1) found that the 84 accessions of cultivated var. frutescens have their own specific fragrances, while only three accessions of the cultivated var. crispa have a specific fragrance. In terms of leaf surface color (QL2), 58 accessions of cultivated var. frutescens had green leaves and 26 accessions of cultivated var. frutescens had leaves that were a deep green color. Furthermore, the accessions of cultivated var. crispa had green (one accession) and purple (two accessions) colored leaves. When surveyed for the color of the reverse side of leaf (QL3), the accessions of cultivated var. frutescens had green (64 accessions), deep green (10 accessions), light purple (1 accession), and purple (9 accessions) colors; whereas the accessions of cultivated var. crispa had green (1 accession) and purple (2 accessions) colors, respectively. The stem color (QL4) of the accessions of cultivated var. frutescens included green (65 accessions), deep green (9 accessions), light purple (4 accessions), and purple (6 accessions) colors, while the accessions of cultivated var. crispa were green (1 accession) and purple (two accessions) only. The degree of pubescence (QL5) in the accessions of cultivated var. frutescens showed slightly pubescent (46 accessions), pubescent (36 accessions), and heavily pubescent (2 accessions) degrees, and the three accessions of cultivated var. crispa showed only slightly pubescent degree. Flower color (QL6) in the accessions of cultivated var. frutescens included white (75 accessions) and purple (9 accessions), while the flowers of all cultivated var. crispa accessions were purple in color. The shape of leaves (QL7) in the cultivated var. frutescens accessions was only the nonwrinkled (84 accessions) type, and the accessions of cultivated var. crispa showed only wrinkled (three accessions) leaves. The seed color (QL8) of the accessions of cultivated var. frutescens included white (5 accessions), gray (58 accessions), brown (10 accessions), and dark brown (11 accessions), while the accessions of cultivated var. crispa were gray (2 accessions) and brown (one accession) in color. Seed size (QL9) of the accessions of cultivated var. frutescens included the large (84 accessions) type, and the accessions of cultivated var. crispa were the small type (three accessions). Seed hardness (QL10) in the accessions of cultivated var. frutescens was found to be both soft (74 accessions) and hard (10 accessions), while all accessions of cultivated var. crispa were only the hard seed type.
Statistical comparisons of the means (p < 0.05) were as follows: Significant differences were found between cultivated var. frutescens and cultivated var. crispa for QN2, QN3, QN4, QN5, QN7. However, QN1 QN6 were not significantly different between cultivated var. frutescens and cultivated var. crispa (Table 3).
Geographical Differences in Cultivated var. frutescens Found in High and Middle Latitude Areas of China
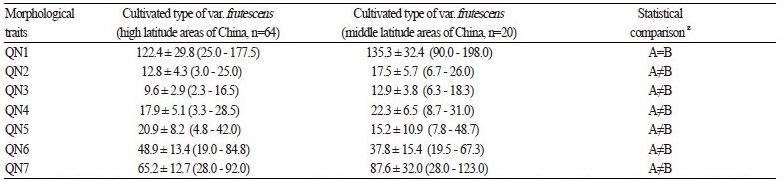
In order to understand the geographical difference between the accessions of cultivated var. frutescens from high-latitude and middle-latitude areas of China, we performed a statistical analysis of the seven quantitative characters on the accessions ofcultivated var. frutescens collected from these regions and the mean values, standard deviation, and range of the seven quantitative traits are summarized in Table 4.
As shown in Table 4, QN1 was 122.4 cm (25 - 177.5 cm) and 135.3 cm (90.0 - 198.0 cm) in cultivated var. frutescens accessions from high and middle latitude areas of China, respectively. QN2 was 12.8 (3.0 - 25.0), and 17.5 (6.7 - 26.0) in accessions of cultivated var. frutescens from high and middle latitude areas of China, respectively. QN3 was 9.6 (2.3 - 16.5), and 12.9 (6.3 -18.3) in accessions of cultivated var. frutescens from high and middle latitude areas of China, respectively. QN4 was 17.9 (3.3 - 28.5), and 22.3 (8.7 - 31.0) in accessions of cultivated var. frutescens from high and middle latitude areas of China, respectively. QN5 was 20.9 (4.8 - 42.0), and 15.2 (7.8 - 48.7) in accessions of cultivated var. frutescens from high and middle latitude areas of China, respectively. QN6 was 48.9 (19.0 - 84.8), and 37.8 (19.5 - 67.3) in accessions of cultivated var. frutescens from high and middle latitude areas of China, respectively. QN7 was 65.2 (28.0 - 92.0) and 87.6 (28.0 - 123.0) in accessions of cultivated var. frutescens from high and middle latitude areas of China, respectively.
In addition, the results of the statistical comparison of means (p <0.05) were as follows: a significant difference was found between accessions of cultivated var. frutescens from high and middle latitude areas in China for QN2, QN3, QN4, QN5, QN6, QN7. However, QN1 was not significantly different between accessions of cultivated var. frutescens from high latitude and middle latitude areas in China (Table 4).
Multivariate Analysis of Quantitative and Qualitative Characteristics in Perilla variant accessions
In this study, principal components analysis (PCA) was used to detect the morphological variation among the accessions from different areas of China. The first and second components accounted for 36.5% and 20.3% of the total variance, respectively (Table 5). Five quantitative characters (QN1, QN2, QN3, QN4, QN7) and six qualitative characters (QL1, QL3, QL5, QL6, QL7, QL10) contributed in the positive direction on the first axis (Table 5). The other quantitative and qualitative characters were contributed in the negative direction on the first axis.
IMost accessions of cultivated var. frutescens and cultivated var. crispa were clearly separated by the first axis. In addition, most accessions of cultivated var. frutescens from high and middle latitude areas were separated by the first axis, except for several accessions (Fig. 2); namely, those from cultivated var. frutescens from high and middle latitude areas were situated to the left and right on the first axis, respectively (Fig. 2).
IResistance to Diseases and Insect Pests
The results of disease and pest infestation in ‘Kowon’ and control cultivars in the test fields are presented in Table 3. ‘Kowon’ displayed intermediate resistance against damping-off, anthracnose, and Alternaria blight, and these results were distinguishable from the control cultivars. Diseases, such as Phytophthora blight and root rot, and pests, such as mulberry mealybug and nematode infestation, were not observed, indicating a certain degree of resistance. Throughout the entire growth period, Alternaria blight is the most frequently occurring disease in all ginseng organs, including leaves, stems, and fruits (Lee et al., 2012).
Discussion
Morphological Variation Between Two Cultivated Types of Perilla Crop from Different Areas of China
The measurement of the morphological characters of plant species within the area of their geographic distribution is an essential process in the identification of varietal or subspecies differentiation in plant species (Gould and Johnston, 1972; Wyattand Antonovics, 1981). The morphological variation among accessions of plant species may be attributed to micro-evolutionary changes in different environments during the long-term evolutionary process. The process of crop domestication is generally associated with the cultivation of plant species in controlled environments (Harlan, 1992; Zohary and Hopf, 1993).
Table 5. Cumulative variance of the first and second principal components and loadings of seven quantitative and 10 qualitative traits on each
principal component.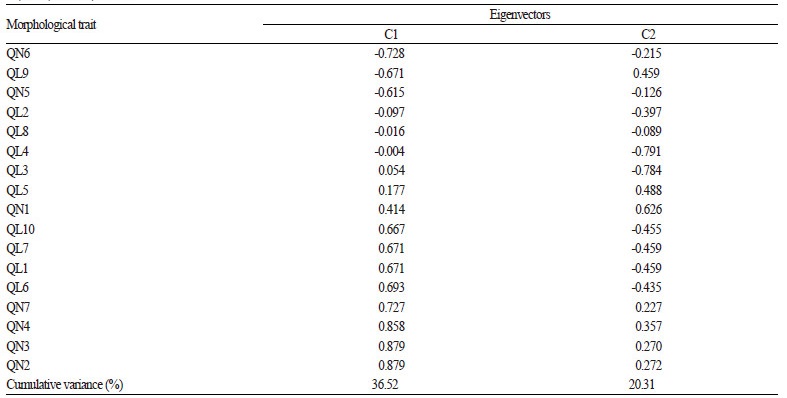 |
Fig. 2.
Fig. 2. Projection of 87 accessions of Perilla crop in the first and second principal components (PC1 and PC2). “Red squares” and “blue diamonds” represent accessions of cultivated var. frutescens from middle and high latitude areas of China, respectively; “green triangles” indicate accessions of cultivated var. crispa from middle latitude areas of China.
In this study, 87 accessions (84 cultivated var. frutescens and three cultivated var. crispa) from middle and high latitude areas of China were examined for seven quantitative characters and 10 qualitative characters. According to our results, significant differences between accessions from two cultivated types of var. frutescens and var. crispa, respectively, were shown in several morphological characters (Table 3). In addition, based on the examination of the qualitative traits, the two cultivated types of cultivated Perilla also showed morphological differences (Table 3). Therefore, these morphological traits can be considered useful characters in distinguishing the two cultivated types of Perilla crop.
In a previous report by Lee and Ohnishi (2001), they suggest that seed size, plant height, and branch number could be useful morphological traits to distinguish the two cultivated types of Perilla crop. In our study, seed size was also found to be a reliable character to differentiate between var. frutescens and var. crispa. However, the statistical comparison of the means (p < 0.05) showed that plant height between cultivated var. frutescens and var. crispa was not significantly different (Table 3). The difference between our results and the previous results of Lee and Ohnishi (2001) is probably due to the smaller population size of cultivated var. crispa used for this analysis.
According to PCA analysis, most accessions of cultivated var. frutescens and var. crispa were clearly separated by the first axis (Fig. 2). This indicates that the combination of quantitative and qualitative traits can be used to discriminate the two cultivated types of Perilla crop through multivariate analysis, consistent with the report from Lee and Ohnishi (2001). As shown in Table 5 in our study, four quantitative characters (QN2, QN3, QN4, QN6) and six qualitative characters (QL1, QL5, QL6, QL7, QL9, QL10) greatly contributed in the positive and negative directions on the first axis. Thus, these morphological characters may be considered as useful characters for distinguishing the two cultivated types of Perilla crop. China has a long history of cultivation of Perilla in East Asia. Therefore, the results of present study will provide useful information for understanding the differentiation of the two cultivated types of Perilla crop found in China.
Geographical Differences in Cultivated var. frutescens between High Latitude and Middle Latitude Areas of China
Under natural conditions, there is a close relationship between the morphological traits of local crop landraces and the habitats in which the traits have evolved and are expressed. The ecological differentiation in local crop landraces affects many traits such as the relative rate of development, resistance to biotic and abiotic stresses, edaphic responses and responses to soil fertility, and adaptation to cultivation and harvesting methods (Rao and Hodgkin, 2002). In general, adaptive genetic variation is generally considered to be quantitative and responsive even in small habitat differences, and often reacts with great sensitivity. Many studies have demonstrated that there is a clear association between population characteristics and the environment (Aston and Bradshaw, 1966; Al-Hiyaly et al., 1993; Rao and Hodgkin, 2002). Most important is that the local crop landraces are the consequence of long periods of interaction between the environment and genetic systems (IPGRI, 1994; Brush, 1995).
The Perilla accessions used in this study were collected in Northeast, Northwest, and North China. These regions represent the diversity in ecological and geographic factors, such as latitude, altitude, moisture, rainfall, temperature, and topographic features that might have a strong influence on the evolution and development of Perilla species. Previous research has found that the formation of different morphological traits is related to geographic origin. Lee and Ohnishi (2001) reported that most accessions of cultivated var. frutescens from China had significant differences from the var. frutescens accessions from Korea and Japan, including differences in seed size, leaf size, plant height, internode number, and flowering time. In this study, many accessions of cultivated var. frutescens from different regions in China also had significant variation in quantitative traits such as branch number (QN2, QN4), internode number (QN3), floret number (QN6), length of largest inflorescence (QN5), and flowering time (QN7) (Table 4). Most accessions of cultivated var. frutescens from high latitude areas of China (Northeast China) produced shorter plants than those accessions from middle latitude areas. Furthermore, accessions of cultivated var. frutescens from high latitude areas had longer primary inflorescence with more florets and shorter a flowering time. Growing days and temperatures in high latitude regions of China (Northeast China) are much shorter and lower than those of middle latitude areas of China. Therefore, these traits (i.e. lower plant height and shorter flowering time) may be adaptations to the relatively harsh environment and a selection pressure of cultivation by local farmers in high latitude regions. In East Asia, the flowering time of Perilla accessions were classified into three types: early-maturity (flowering time < 100 days), middle-maturity (100 days < flowering time < 130 days, and late-maturity (flowering time > 130 days) (Lee and Ohnishi, 2001). In this study, all accessions of cultivated var. frutescens from Northeast China fell into the early-maturity type with a flowering time ranging from 28 days to 92 days. For accessions of cultivated var. frutescens from middle latitude areas of China, nine accessions were of the middle-maturity type with a flowering time of 100 days to 130 days. The other accessions were early-maturity type. In addition, three accessions of cultivated var. crispa were identified to be middle-maturity type with the flowering time ranging from 118 to 121 days.
According to Lee and Ohnishi (2001), all accessions of both cultivated and weedy types of var. frutescens collected in South China belong to only the late-maturity type (flowering time >130days). These results indicate that latitude with geographical distribution may be a critical factor that affects the flowering time in Perilla species because Perilla crop is well known as one of the representative short-day crops. In addition, the difference in flowering time is also related to farmer selection. The duration of cold weather with temperatures lower than 0°C generally lasts for six months in Northeast China. Furthermore, the temperature range (20 - 26°C) that is suitable for the growth of Perilla accessions is much shorter than that of South China and middle latitude areas of China. Therefore, these Perilla accessions with shorter flowering times are more likely to be selected for by local farmers in Northeast China. In our study, seven of the 10 accessions were collected in the provinces of Northeast China and had a short flowering period (28 days).
According to the PCA analysis, most accessions of cultivated var. frutescens from high and middle latitude areas were clearly separated by the first axis, except for several select accessions (Fig. 2). As shown in Fig. 2, these accessions were collected from high latitude areas and did not clearly separate from those accessions from middle latitude areas of China. This may be a result of the overlapping distribution of Perilla accessions originating from different regions with different environmental features. In addition, gene flow has a significant effect on the differentiation and population structure of plants (Duminil et al., 2009). Gene flow may occur easily through seeds, pollens, and other natural approaches in plain areas and human activities (Meng et al., 2015), especially business activities. For example, Perilla seeds from high latitude areas may have migrated to northwestern areas of China or other middle latitude areas through human business activities, like as expansion cultivation area and sales of Perilla seeds by humans. After being introduced into different areas, similar morphological traits of the local Perilla accessions would be formed under the selection from new environmental stress and local farmers. Due to the lack of human interference, the plant species collected in mountainous, high altitude terrains are usually revealed to have high genetic diversity and a large range for differentiation of morphological traits among and within populations (Meng et al., 2015; Chen et al., 2016; Zhang et al.,2016). These factors probably caused the accessions from different areas to be ambiguously separated in the PCA analysis.
Morphological characteristics of Perilla accessions from high and middle latitude areas of China have been illustrated in this study. Our findings will contribute to better understanding of the morphological variations in Perilla accessions from different regions of China and will also be useful for evaluating Perilla germplasm resources for the utilization of these accessions in breeding and new cultivar development.