Introduction
Materials and Methods
Plant materials
Cultivation conditions in the PFAL
Plant growth measurements
Analysis of anthocyanin and lutein contents
Statistical analysis
Results and Discussion
Conclusions
Introduction
Sweet potato [Ipomoea batatas (L.) Lam], a vegetatively propagated crop of the genus Ipomoea in the Convolvulaceae family, possesses leaves, vine stems, and flowers above ground, and fibrous, pencil, and storage roots below ground (Huamari 1992). Sweet potatoes are gaining attention as a functional food due to their high amounts of dietary fiber, vitamins, and minerals. Storage roots serve as a rich source of energy and are widely used for food and industrial purposes (Widodo et al. 2015). In addition, sweet potato leaves contain abundant levels of vitamins A and C (Alam et al. 2020); high levels of dietary fiber; and significant amounts of phenolic compounds, carotenoids, and other antioxidants (Johnson and Pace 2010). Thus, sweet potato leaves are utilized as salad greens in Southeast Asia and are processed into tea in Japan. According to the Rural Development Administration (RDA 2018), the essential amino acid content in sweet potato leaves is more than twice that of tubers and is considerably higher than in other leafy vegetables such as lettuce. Moreover, sweet potatoes contain high concentrations of lutein, a pigment that accumulates in the macula and contributes to visual health by preventing age-related macular degeneration and improving memory (Menelaou et al. 2006; Li et al. 2020; Martini et al. 2022). The lutein content in sweet potato leaves is reported to be more than 1.2 times higher than that of kale and approximately 4.9 times higher than that of spinach (RDA 2018). To harness the nutritional value of sweet potato leaves, cultivars intended for leafy vegetable use have been developed. In Japan, the cultivar ‘Suioh’ was bred as a leafy sweet potato with a higher total polyphenol content and more antioxidant activity than spinach, broccoli, and cabbage (Ishiguro et al. 2004). Similarly, in Korea, ‘Tongchaeru,’ a cultivar rich in anthocyanin, was developed in 2020 (KFSRF 2023).
A plant factory with artificial light (PFAL) is a highly controlled indoor cultivation system that uses artificial lighting, such as LEDs, to optimize the environmental conditions for plant growth (Mitchell and Sheibani 2020). Currently, PFALs are primarily used for cultivating fast-growing leafy vegetables such as lettuce, kale as well as herbs such as basil. It is well established that, for economic reasons, staple crops such as rice, wheat, and maize are not suitable for production in PFALs. However, Kozai (2018) suggested that crops such as potato and sweet potato could be feasible for production in PFALs if all parts, including the leaves, petioles, and stems, are utilized or salable.
Based on this, we hypothesized that leafy sweet potato, characterized by rapid growth and low vertical development, may be suitable for PFAL cultivation. Moreover, the rich contents of secondary metabolites such as anthocyanin and lutein in this plant enhance its potential value further. To evaluate this potential, we investigated the feasibility of hydroponically cultivating leafy sweet potato in a PFAL and assessed the effects of nutrient solution electrical conductivity (EC) on plant growth and quality outcomes.
Materials and Methods
Plant materials
Two sweet potato cultivars, ‘Tongchaeru’ (developed in Korea) and ‘Suioh’ (developed in Japan), were used in this study. Both cultivars are bred specifically for use as leafy vegetables. To produce uniform tissue-cultured seedlings, single nodes were excised from in-vitro-grown seedlings and sub-cultured on Murashige and Skoog (MS) solid media (MS 4.4 g·L-1, sucrose 30 g·L-1, and Gelrite 4 g·L-1) in the plant tissue culture facility of the University of Seoul. The cultured seedlings were subsequently grown for 21 days in a plant culture room under controlled conditions, with an air temperature of 25°C, a white LED light intensity level of 200 µmol·m-2·s-1, and a 16/8 h light/dark photoperiod.
Cultivation conditions in the PFAL
After 21 days of tissue culturing, seedlings were transplanted into hydroponic sponges and cultivated for 35 days in a PFAL. The air temperature was maintained at 25/22°C (light/dark), and the relative humidity was controlled at 75 ± 5% for the first three days after transplanting (DAT) and 65 ± 5% thereafter. White LEDs provided light intensity of 250 µmol·m-2·s-1 under a 16/8 h light/dark photoperiod, following the cultivation conditions described by He et al. (2020).
Nutrient solution electrical conductivity (EC) levels were adjusted to 1.0, 2.0, 3.0, and 4.0 dS·m-1, with the pH was maintained between 6.0 and 6.5. The nutrient solution used was formulated for leafy vegetables based on recommendations from the Japan Horticultural Experiment Station and had the following composition (in me·L-1): NO3‒ 14, NH4+ 1, P 3, K 6, Ca 8, Mg 4, and SO42‒ 4.
Plant growth measurements
Plant height, stem diameter, the number of leaves, the leaf area, SPAD values, stomatal conductance, and the fresh and dry weights of shoots and roots were measured at 7, 14, 21, 28, and 35 DAT. Stem diameter and leaf area were measured using a caliper (CD-AX/C, Mitutoyo Korea Corporation, Gyeonggi-do, Korea) and a leaf area meter (LI-3100; LI-COR Inc., Lincoln, NE, USA), respectively. SPAD values and stomatal conductance were measured using a chlorophyll meter (SPAD-502PLUS; Konica Minolta, Tokyo, Japan) and a porometer (LI-600; LI-COR Inc., Lincoln, NE, USA), respectively. Dry weights were obtained after oven-drying plant samples at 70°C for seven days.
Analysis of anthocyanin and lutein contents
At 35 DAT, leaves were harvested from each treatment group and categorized based on the presence or absence of intumescence. The leaves were freeze-dried for 24 hours and ground with a mortar and pestle.
Total anthocyanin content was determined by macerating the leaf samples in 10 mL of 95% ethanol and 1.5 N HCl (85:15, v/v), followed by storage in the dark at 5°C for 24 h. The samples were then centrifuged at 13,000 rpm for 20 minutes, and the supernatant was diluted with 95% ethanol. Absorbance was measured at 535 nm using a UV spectrophotometer (UV-2450; Shimadzu Co., Kyoto, Japan), and the total anthocyanin content was expressed as mg·g-1 DW (Murray and Hackett 1991; Xu et al. 2007).
The lutein content was analyzed using a modified method based on Choi et al. (2007) and Li et al. (2017). Ground leaf samples were mixed with 5 mL of a pyrogallol-ethanol solution and 0.5 mL of KOH and were incubated in a 70°C water bath for 30 minutes. Then, 10 mL of a NaCl solution and 6 mL of a hexane:ethyl acetate mixture (9:1, v/v) were added. After centrifugation, the supernatant was evaporated at 70°C and the residue was dissolved in 10 mL of a BHT-ethanol solution and filtered through a 0.2 µm PTFE filter. The final extract was analyzed using an HPLC system (UHPLC System, Shimadzu Co., Kyoto, Japan) equipped with a YMC-Pack Pro C18 column (4.6 × 250 mm, 5 µm; YMC Co., Ltd., Japan). Lutein was detected at 445 nm with a flow rate of 1.2 mL·min-1.
Statistical analysis
All data were statistically analyzed using SAS Enterprise Guide 9.4 (SAS Institute Inc., Cary, NC, USA). One-way analysis of variance (ANOVA) and Duncan’s multiple range test were conducted to determine significant differences among the treatment means at a significance level of p < 0.05.
Results and Discussion
The growth of ‘Tongchaeru’ and ‘Suioh’ sweet potato cultivars was significantly affected by the EC of the nutrient solution. The EC 2.0 dS·m-1 treatment resulted in the highest growth across both cultivars, as reflected in the plant height, the number of leaves, the stem diameter, the leaf area, and both the shoot and root dry weights (Tables 1 and 2, Fig. 1). In contrast, growth was markedly reduced at the EC levels of 1.0 and 4.0 dS·m-1, indicating that non-optimal EC conditions induced either nutrient deficiency or osmotic stress. It is well established that the crop performance in plant factories with artificial lighting (PFAL) hydroponic systems is highly sensitive to the composition and concentration of the nutrient solution (Cho et al. 2018; Ding et al. 2018; Levine and Mattson 2021). Excessively high EC levels can impose the salinity stress, leading to reduced water and nutrient uptake, while low EC levels may result in insufficient nutrient availability, thereby limiting growth (Wu and Kubota 2008; Signore et al. 2016).
Table 1.
Growth parameters of ‘Tongchaeru’ leafy sweet potato grown under different EC conditions for 35 DAT in a PFAL
| DAT |
EC (dS·m-1) |
Plant height (cm) |
Stem diameter (mm) |
No. of leaves (/plant) |
Leaf area (cm2/plant) | SPAD value | Dry weight (mg/plant) | |
| Shoot | Root | |||||||
| 7 | 1.0 | 3.18 az | 1.84 a | 7.6 a | 15.06 b | 30.14 ab | 0.045 bc | 0.029 a |
| 2.0 | 3.20 a | 1.97 a | 7.6 a | 20.88 a | 29.12 b | 0.065 a | 0.036 a | |
| 3.0 | 2.80 ab | 1.91 a | 7.4 a | 14.03 b | 31.52 ab | 0.050 b | 0.024 a | |
| 4.0 | 2.42 b | 1.81 a | 6.8 a | 12.78 b | 32.92 a | 0.033 c | 0.018 a | |
| 14 | 1.0 | 3.42 b | 2.50 a | 9.2 ab | 48.63 c | 34.06 a | 0.148 c | 0.072 b |
| 2.0 | 5.16 a | 2.50 a | 10.4 a | 80.09 a | 35.12 a | 0.260 a | 0.109 a | |
| 3.0 | 3.76 b | 3.49 a | 9.8 ab | 58.74 b | 36.78 a | 0.176 b | 0.067 b | |
| 4.0 | 3.46 b | 2.31 a | 8.6 b | 51.83 c | 35.88 a | 0.157 c | 0.070 b | |
| 21 | 1.0 | 5.24 b | 2.67 b | 9.8 a | 96.75 c | 36.22 b | 0.419 c | 0.159 c |
| 2.0 | 7.44 a | 3.22 a | 10.8 a | 167.56 a | 41.58 a | 0.676 a | 0.242 a | |
| 3.0 | 5.68 b | 3.26 a | 10.6 a | 141.15 b | 43.28 a | 0.579 ab | 0.205 b | |
| 4.0 | 4.64 b | 3.35 a | 9.4 a | 138.51 b | 43.10 a | 0.508 bc | 0.184 bc | |
| 28 | 1.0 | 8.06 bc | 3.26 b | 11.6 a | 140.63 c | 35.58 b | 0.666 c | 0.253 c |
| 2.0 | 14.46 a | 3.89 a | 14.0 a | 300.30 a | 38.58 ab | 1.389 a | 0.479 a | |
| 3.0 | 9.00 b | 3.74 a | 11.4 a | 236.24 b | 41.98 a | 1.115 b | 0.371 b | |
| 4.0 | 6.64 c | 3.60 ab | 11.0 a | 235.13 b | 40.02 a | 0.966 b | 0.319 b | |
| 35 | 1.0 | 9.04 b | 3.27 c | 12.4 b | 128.61 c | 35.02 b | 0.728 c | 0.258 c |
| 2.0 | 16.18 a | 4.13 a | 14.2 a | 455.13 a | 42.56 a | 2.223 a | 0.634 a | |
| 3.0 | 9.96 b | 3.93 ab | 12.8 ab | 259.28 b | 42.44 a | 1.525 b | 0.451 b | |
| 4.0 | 9.98 b | 3.66 bc | 11.4 b | 213.77 b | 41.32 a | 1.141 bc | 0.362 bc | |
Table 2.
Growth parameters of ‘Suioh’ leafy sweet potato grown under different EC conditions for 35 DAT in a PFAL
| DAT |
EC (dS·m-1) |
Plant height (cm) |
Stem diameter (mm) |
No. of leaves (/plant) |
Leaf area (cm2/plant) | SPAD value | Dry weight (mg/plant) | |
| Shoot | Root | |||||||
| 7 | 1.0 | 3.16 az | 2.18 a | 6.0 a | 15.35 b | 32.06 a | 0.054 b | 0.025 b |
| 2.0 | 3.02 a | 2.28 a | 5.8 a | 21.98 a | 31.46 a | 0.084 a | 0.056 a | |
| 3.0 | 3.32 a | 2.12 a | 6.4 a | 17.84 b | 34.80 a | 0.064 b | 0.050 ab | |
| 4.0 | 3.00 a | 2.25 a | 5.8 a | 17.14 b | 34.82 a | 0.071 ab | 0.043 ab | |
| 14 | 1.0 | 2.58 b | 2.36 a | 6.2 b | 43.72 b | 34.94 b | 0.188 c | 0.060 c |
| 2.0 | 4.44 a | 2.37 a | 8.6 a | 69.54 a | 42.68 a | 0.339 a | 0.110 a | |
| 3.0 | 3.24 b | 2.41 a | 7.2 ab | 50.54 b | 41.56 a | 0.267 b | 0.091 ab | |
| 4.0 | 2.84 b | 2.35 a | 7.2 ab | 45.70 b | 42.56 a | 0.196 c | 0.074 bc | |
| 21 | 1.0 | 4.04 b | 3.23 b | 8.2 b | 89.16 c | 33.92 b | 0.574 b | 0.185 b |
| 2.0 | 6.14 a | 4.02 a | 10.6 a | 151.00 a | 39.94 a | 0.982 a | 0.297 a | |
| 3.0 | 4.34 b | 3.72 ab | 9.0 ab | 113.22 b | 40.20 a | 0.704 b | 0.203 b | |
| 4.0 | 4.36 b | 3.21 b | 9.0 ab | 119.81 b | 42.24 a | 0.671 b | 0.192 b | |
| 28 | 1.0 | 4.96 c | 3.15 a | 9.0 a | 104.55 c | 30.72 c | 0.611 c | 0.225 b |
| 2.0 | 9.46 a | 3.53 a | 11.2 a | 228.05 a | 37.84 b | 1.403 a | 0.454 a | |
| 3.0 | 7.02 b | 3.63 a | 10.8 a | 195.44 a | 42.86 a | 1.244 a | 0.377 a | |
| 4.0 | 6.62 b | 3.38 a | 9.2 a | 154.86 b | 41.40 ab | 0.913 b | 0.279 b | |
| 35 | 1.0 | 6.42 c | 3.45 b | 11.6 a | 115.30 d | 28.18 d | 0.780 c | 0.280 c |
| 2.0 | 12.21 a | 4.33 a | 12.4 a | 324.19 a | 40.06 c | 2.290 a | 0.670 a | |
| 3.0 | 8.94 b | 4.25 a | 11.2 a | 244.88 b | 43.72 b | 1.580 b | 0.435 b | |
| 4.0 | 3.16 c | 3.83 ab | 9.6 b | 191.38 c | 46.80 a | 1.330 b | 0.340 c | |
The leaf area followed a similar trend, reaching its maximum at EC 2.0 dS·m-1 in both cultivars. Notably, at 35 days after transplanting (DAT), the leaf area was 224% and 181% greater at EC 2.0 dS·m-1 compared to EC 1.0 dS·m-1 in ‘Tongchaeru’ and ‘Suioh’, respectively. The shoot and root dry weights also peaked at EC 2.0 dS·m-1 and were lowest at EC 1.0 dS·m-1. In ‘Tongchaeru’, the dry weight exhibited growth stagnation after 28 DAT under the EC 1.0 dS·m-1 treatment, suggesting that nutrient deficiency limited further biomass accumulation.
The SPAD values, which reflect the chlorophyll content, exhibited cultivar-specific responses to EC. In ‘Tongchaeru’, a purple-leaved cultivar, the SPAD values remained relatively stable across the EC 2.0–4.0 dS·m-1 treatments. In contrast, ‘Suioh’, a green-leaved cultivar, tended to show increased SPAD values with rising EC levels. This trend is attributable to the enhanced nitrogen uptake under the higher EC, consistent with reports that mild salinity stress can promote chlorophyll accumulation when nutrient availability is adequate (Shah et al. 2017; Ni et al. 2011). Nitrogen is widely recognized as the key nutrient affecting the chlorophyll content and fluorescence (Zhang et al. 2003). Becker et al. (2015) reported that green lettuce exhibits greater plasticity in the photosynthetic pigment concentration than red lettuce under varying nitrogen levels.
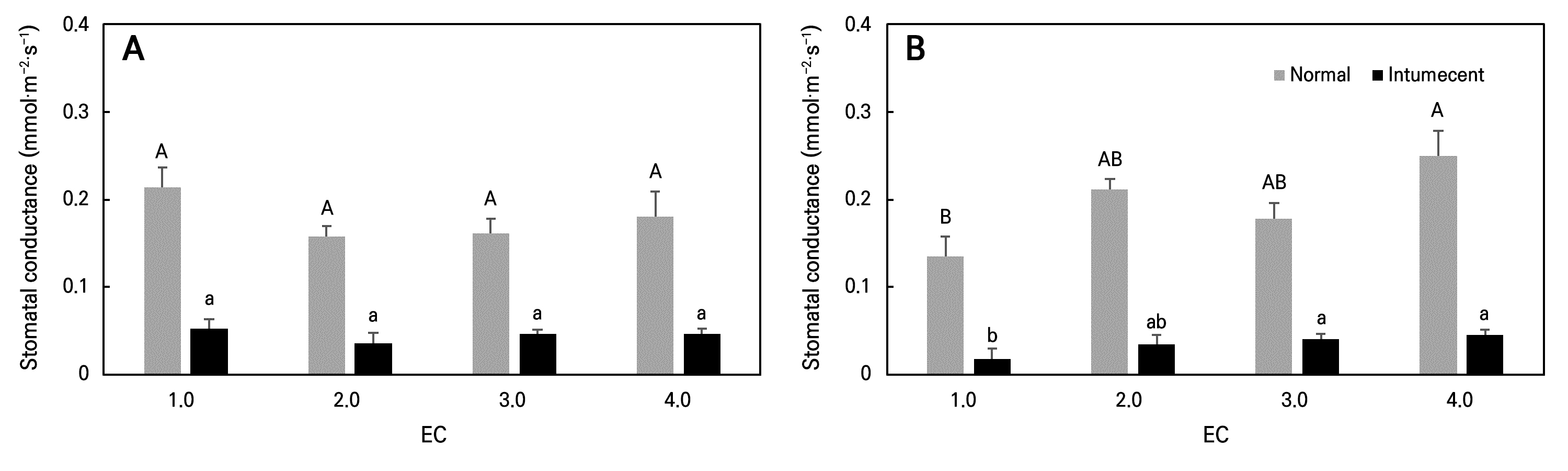
The stomatal conductance in ‘Tongchaeru’ sweet potato leaves showed no significant differences with increasing EC levels, whereas ‘Suioh’ leaves demonstrated increased stomatal conductance as EC was increased (Fig. 2). Elevated EC levels are known to increase salt stress, causing plants to reduce transpiration by closing their stomata. Previous studies have reported reductions in stomatal conductance in crops such as lettuce and tomato under high EC conditions (Wu and Kubota 2008; Adhikari et al. 2024). In contrast, we observed an opposite trend in the ‘Suioh’ cultivar. Similarly, He et al. (2024) reported a sharp increase in stomatal conductance in cucumber under high EC levels, which they interpreted as a stress-induced response aimed at preventing potential damage caused by elevated nutrient concentrations.
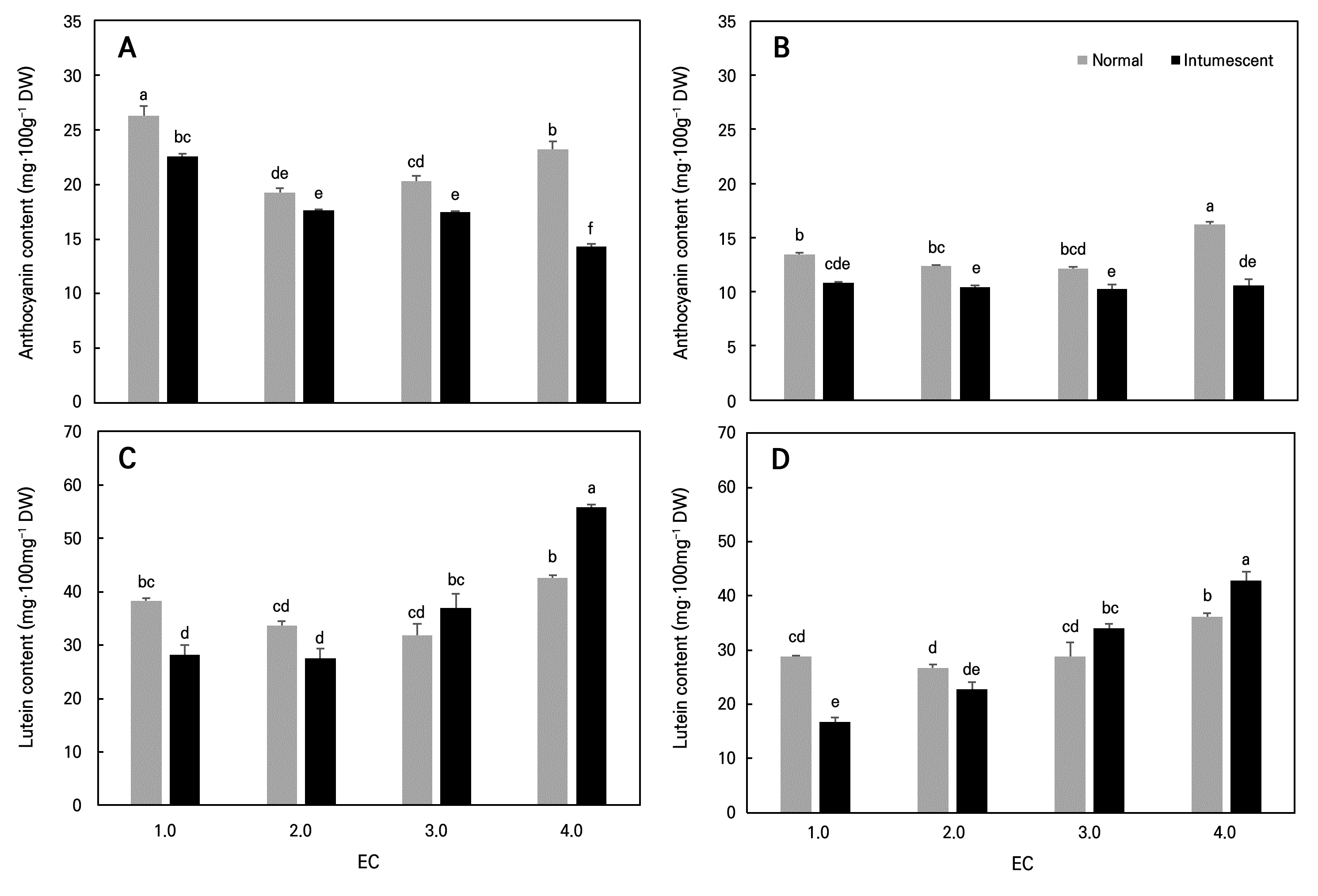
In ‘Tongchaeru’, the highest anthocyanin content was observed under the EC 1.0 dS·m-1 treatment, followed by EC 4.0 dS·m-1. In contrast, in ‘Suioh’, the highest anthocyanin content was found under the EC 4.0 dS·m-1 treatment, with no significant differences among the EC 1.0 to 3.0 dS·m-1 treatments (Fig. 3). A previous study of hydroponically grown arugula reported similar results, finding that optimal EC conditions increased the biomass yield but reduced anthocyanin accumulation (Yang et al. 2021). In general, abiotic stress conditions are associated with decreased total chlorophyll levels and increased anthocyanin accumulation, as the latter is a well-documented protective response to nutrient stress (Akula and Ravishankar 2011; Hörtensteiner and Kräutler 2011). Nitrogen deficiency, in particular, has been shown to induce the expression of key enzymes involved in the anthocyanin biosynthetic pathway (Delgado et al. 2004). Although numerous studies have demonstrated that anthocyanin biosynthesis is promoted under nitrogen-deficient conditions based on the carbon/nitrogen (C/N) balance hypothesis, it is generally accepted that excessive nitrogen availability suppresses anthocyanin production (Stewart et al. 2001; Larbat et al. 2012). Therefore, the increased anthocyanin content observed under high EC conditions in this study is likely a stress-induced response to osmotic stress caused by nutrient oversupply, rather than a direct effect of nitrogen availability.
Lutein, a carotenoid abundant in the stems and leaves of sweet potato, showed significantly higher accumulation in the normal leaves of both cultivars under the EC 4.0 dS·m-1 treatment, whereas no significant differences were observed at the lower EC levels (Fig. 3). Nitrogen availability plays a critical role in chloroplast development and carotenogenesis (Dhami and Cazzonelli 2020). Lichtenthaler and Verbeek (1973) reported that nitrogen deficiency in barley led to a decrease in carotenoid contents, including lutein. This reduction is associated with the inhibition of the initial step of carotenoid biosynthesis, as low nitrogen availability negatively affects the phytoene synthase–mediated condensation of geranylgeranyl pyrophosphate (GGPP). Kopsell et al. (2007) reported enhanced lutein accumulation in hydroponically grown kale under elevated nitrogen concentrations. Similarly, several studies have shown that a sufficient nitrogen supply promotes the accumulation of lutein and β-carotene in various vegetable crops (Boskovic-Rakocevic et al. 2012; Reif et al. 2012; Reif et al. 2013).
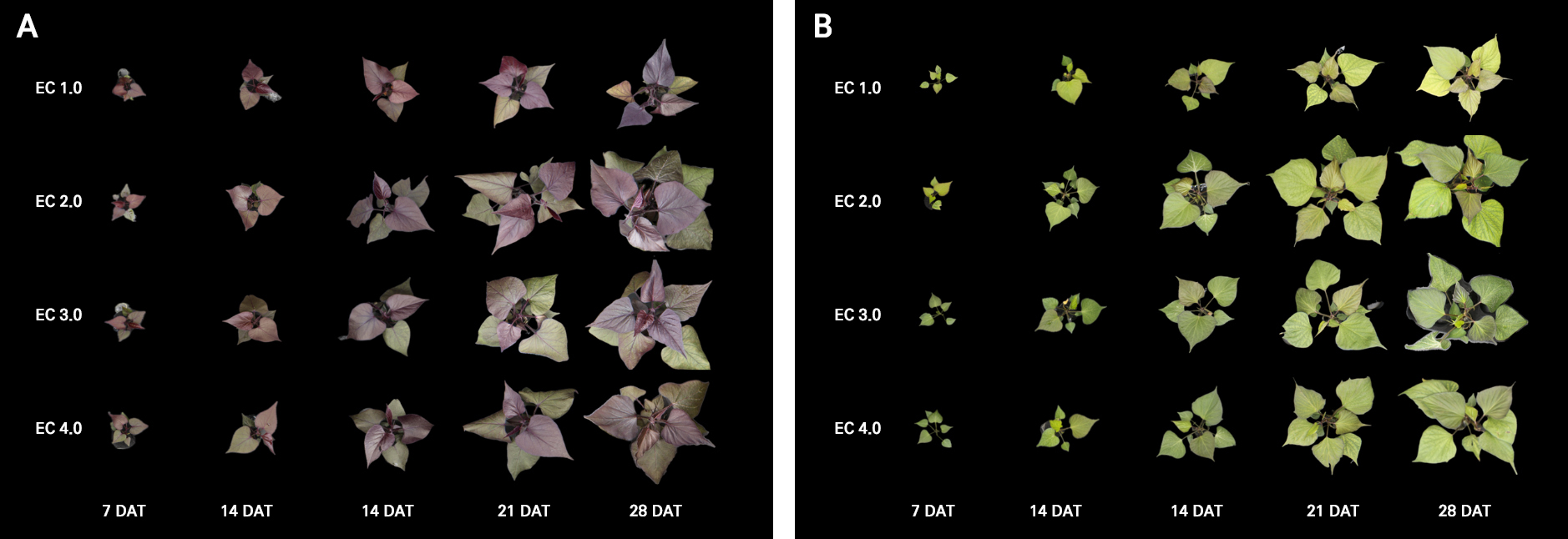
During the cultivation of leafy sweet potato in a PFAL, intumescence symptoms, which have been previously reported in Convolvulaceae crops (Williams et al. 2016), were observed. These symptoms appeared in all plants after transplanting, with more severe expression in ‘Suioh’ compared to ‘Tongchaeru’ (Fig. 4). Although the exact cause of intumescence remains unclear, it is considered a type of physiological disorder that arises in controlled environmental conditions (Sita et al. 2024). To assess the physiological impact of intumescence, measurements of the stomatal conductance and pigment contents were also conducted with symptomatic leaves. Stomatal conductance was lower in intumescent leaves compared to normal leaves, and the anthocyanin content was also reduced (Figs. 2 and 3). Under the high EC treatment, the lutein content in intumescent leaves was higher than that in normal leaves. Intumescence-induced cuticle damage may reduce leaf water retention, leading to stomatal closure to prevent further water loss (Suzuki et al. 2020). The reduced anthocyanin content is attributable to the downregulation of genes related to secondary metabolism and flavonoid biosynthesis (Wu et al. 2017), while the increased lutein content under high EC conditions may result from more severe osmotic stress in intumescent leaves caused by cuticle damage.
Conclusions
This study provides a foundational scientific basis for the cultivation of leafy sweet potato in PFALs. Both ‘Tongchaeru’ and ‘Suioh’ cultivars exhibited optimal growth at an EC level of 2.0 dS·m-1; however, the highest contents of functional compounds were observed when the EC was 4.0 dS·m-1. In addition, intumescence symptoms observed during the cultivation period were found potentially to affect photosynthetic activity and secondary metabolite biosynthesis. These findings offer a comprehensive physiological and biochemical basis for optimizing nutrient solution EC levels to enhance both biomass productivity and phytochemical quality in leafy sweet potato cultivated in PFAL systems. Future research focusing on different nutrient solution compositions and dynamic EC control approaches during the cultivation period may further improve crop productivity and nutritional quality, thereby contributing to the stable production and utilization of leafy sweet potato in PFALs.