Introduction
Materials and Methods
Plant materials and treatments
Determination of fruit quality attributes
Statistical analysis
Results
Shriveling and decay incidence
Changes in the weight loss rate and firmness
Changes of SSC, TA, the SSC/TA ratio, and the pH
Significance of maturity stage, and storage temperature and duration on fruit quality attributes
Discussion
Shriveling and decay incidence
Changes in weight loss rate and firmness
Changes of SSC, TA, the SSC/TA ratio, and the pH
Conclusion
Introduction
Mango (Mangifera indica L.), a subtropical evergreen fruit tree in the family Anacardiaceae (Tharanathan et al. 2006; Singh et al. 2013), is currently one of the most widely cultivated and consumed fruits worldwide. Its increasing demand is largely attributed to consumer appreciation of its distinctive aroma, sweet flavor, and high contents of provitamin A, vitamin C, and dietary fiber, qualities that align with growing interest in nutrient-dense, exotic fruits. On the production side, gradual warming in many warm temperate regions, together with advances in greenhouse cultivation technologies, has enabled commercial mango production to expand beyond traditional tropical areas (Chung et al. 2021; Lim et al. 2021; Jung et al. 2022).
Mango cultivation was adopted relatively recently in Korea, with grafted material introduced from Taiwan initially planted on Jeju Island in 1993. Since then, the cultivated area has expanded to approximately 93 ha across 228 farms, collectively producing nearly 8,700 tons annually. Expansion into mainland coastal regions has been supported by regional development programs that promote mango as a high-income alternative crop. However, mid-winter temperatures in these regions still fall below the threshold for its safe field production, making heated plastic-house systems essential to prevent cold injury (Lim et al. 2016; Lim et al. 2021).
The red-blushed mango cultivar ‘Irwin’, originally selected in Florida, USA from a ‘Lippens’ seedling, dominates mango production in Korea, as it flowers reliably under subtropical greenhouse conditions and commands a price premium in the market. Korean mango growers employ three production schedules by adjusting heating and night-cooling, using forcing (April-May harvest), standard (June to early August), and retarded (mid-August to early September) settings. Forcing incurs high energy costs, while the retarded schedule delays pruning and disrupts carbohydrate accumulation for the following crop. As a result, the standard is the most widely adopted scheduling strategy. However, according to this schedule the harvest is concentrated in mid-summer, when market prices typically drop to about half relative to early-season values. Extending the market window for standard-scheduled fruit could substantially improve profitability.
Mango fruit ripening involves coordinated changes in peel color, firmness, acidity, and soluble solids as starch and organic acids are converted into sugars and cell wall polysaccharides are hydrolyzed (Tharanathan et al. 2006). Classified as climacteric, mango fruits exhibit a burst of ethylene followed by an increase in respiration at the onset of ripening, which accelerates softening, with even trace amounts of ethylene (approximately 0.01 µL·L-1) having the potential to hasten this process significantly (Singh et al. 2013). Controlled low-temperature storage can slow both ethylene generation and respiration, but the optimum temperature range (typically around 12–13°C for two to three weeks) depends on the cultivar and maturity stage (Kader 1992; Singh et al. 2013). Fruits harvested at the green-mature stage are susceptible to chilling injury below 10°C (Mohammed and Brecht 2002), whereas fully colored fruits can tolerate cooler storage temperatures but deteriorate more rapidly. In Korea, the fruits are often harvested at an advanced stage to meet consumer preferences for flavor and aroma. However, this practice can increase the risk of fruit decay and reduce the shelf life (Ntsoane et al. 2019).
Only limited research has been conducted with regard to the quality change of ‘Irwin’ mango fruits during storage, especially of those produced from greenhouses in Korea. Although the quality change during storage has been compared according to the maturity stage of greenhouse-grown ‘Irwin’ mango fruits (Wijethunga et al. 2023), the optimal combination of maturity stage and storage temperature to extend the shelf life while maintaining premium fruit quality remains to be identified. In the present study, quality attributes in greenhouse-grown ‘Irwin’ mango fruits harvested at three maturity stages were monitored during storage at different temperatures.
Materials and Methods
Plant materials and treatments
‘Irwin’ mango trees were grown in greenhouses within a 1.65-ha orchard in Seogwipo (33°17’N, 126°36’E), Korea. Mangoes were cultivated in a heated greenhouse under a forcing cultivation system. Flowering and pollination occurred in December and harvesting was carried out on April 16. Unblemished uniform fruits were randomly harvested from approximately 2,250 trees at three different maturity stages (stage 1, 2, and 3), as determined using a color chart (Fig. 1) developed by the Research Institute of Climate Change and Agriculture, Jeju, Korea. The CIE L, a, b values, as well as the chroma and hue angle of fruits harvested at stages 1, 2 and 3 are presented in Table 3. A colorimetric analysis showed that the hue angle increased from 31.93 to 54.88 with advancing maturity, corresponding to a shift from reddish to more orangey-yellow tones. This reflects ripening-associated changes in peel pigmentation. The harvested fruits were then stored in walk-in chambers set to 8, 12, and 16°C with 90% relative humidity.
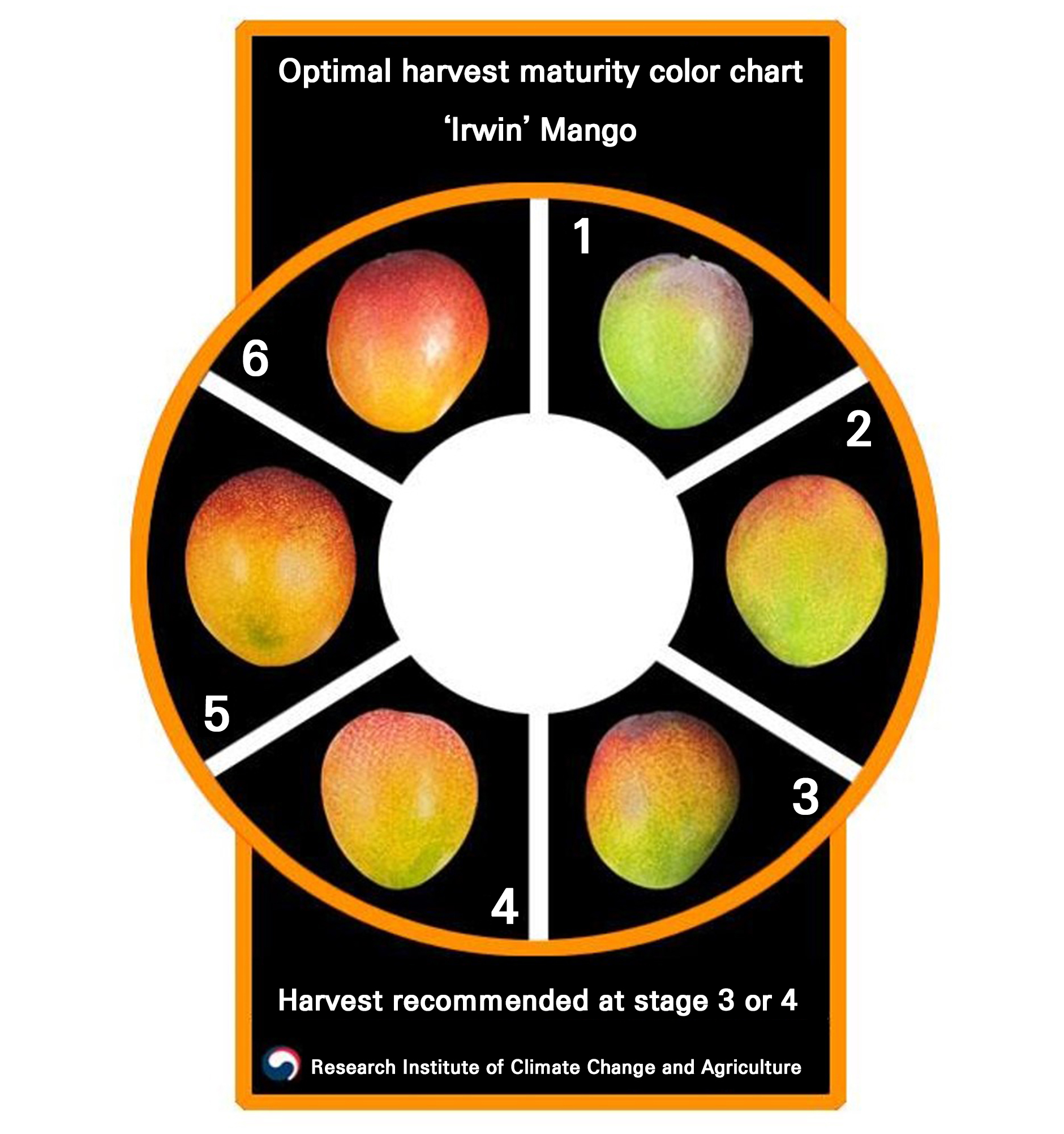
Fig. 1.
Color chart for determining harvest maturity of ‘Irwin’ mango fruits. The chart was developed by the Research Institute of Climate Change and Agriculture, Jeju, Korea. Maturity was visually classified into six stages based on the peel color, and stages 3 and 4 are recommended to be suitable for the commercial harvest of table fruits.
Determination of fruit quality attributes
Fruit quality attributes, including both external and compositional traits, were determined and monitored at three- or four-day intervals during storage. Ten individual fruits were used to determine each quality attribute.
Shriveling was visually assessed using a modified rating scale developed by Nunes et al. (2007) with the following categories: 1 = none (no signs of shriveling), 2 = slight (minor signs of shriveling, not objectionable), 3 = moderate (shriveling evident, becoming objectionable), 4 = severe (definitely objectionable), and 5 = extreme (shriveled, wrinkled, and dry; not acceptable under normal conditions). Decay was visually evaluated using a modified scale based on criteria from Ketsa et al. (2000), ranging from 1 to 5; 1 = none, 2 = 0–25%, 3 = 26–50%, 4 = 51–75%, and 5 = 76–100% of the fruit surface affected.
Fruit weight (FW) was measured using an electronic scale (Jw-1-2000; ACOM, Seoul, Korea), and the weight loss rate during storage was calculated using the following formula:
Weight loss rate (%) = [(original FW–measured FW)/original FW] × 100.
Fruit firmness was measured following the method of Gunness et al. (2009) using a texture analyzer (CT-3; AMETEK Brookfield, Middleboro, MA, USA) equipped with a cylindrical probe with a diameter of 2 mm. The equatorial side of each fruit was compressed by 4 mm at a speed of 2 mm·s-1. The maximum force was recorded and expressed as Newtons (N).
The soluble solids content (SSC) was measured using a digital refractometer (HI96801; Hanna, Romania) and is expressed as °Brix. Titratable acidity (TA) was determined by titration with 0.16 N NaOH in a 5-mL juice sample according to the method described by Lim et al. (2021), and the pH of the juice was measured using a pH meter.
Statistical analysis
Statistical analyses were performed using R software (version 4.1.2) and RStudio (version 1.3). A two-way analysis of variance (ANOVA) was conducted using the ‘agricolae’ package (version 1.3.7) to assess the main effects and interactions between maturity stage and storage temperature. Mean comparisons were performed using Duncan’s multiple range test at p < 0.05.
Results
Shriveling and decay incidence
Shriveling in ‘Irwin mango fruits was observed during storage and varied greatly with both the maturity stage and storage temperature (Table 2). Shriveling was first observed on 6 DAS in fruits harvested at stage 3 and stored at 16°C. On the other hand, it was most delayed in fruits harvested at stage 2 and stored at 8°C, where shriveling appeared on 28 DAS. On 6 DAS, the shriveling incidence was significantly affected by the maturity stage, storage temperature, and their interaction. However, until 17 DAS, the maturity stage didn’t reveal a significant effect, whereas the storage temperature showed a highly significant effect. As the storage temperature was increased, the incidence of shriveling tended to increase. From 20 DAS, the maturity stage, storage temperature, and their interaction all revealed significant effects, indicating that shriveling development was also dependent on the maturity stage at later stages. Under the same temperature conditions, fruits harvested at stage 2 consistently exhibited a lower degree of shriveling compared to those harvested at other stages.
Decay in the fruits was observed during storage and varied greatly with both the maturity stage and storage temperature (Table 3). In fruits harvested at stage 3 and stored at 12°C and 16°C, decay symptoms were initially observed at 10 DAS, demonstrating the significant effects (p < 0.01) of the maturity stage, storage temperature, and their interaction. Fruits stored at higher temperatures and harvested at more advanced maturity stages revealed a higher incidence of decay. During the later storage period (after 20 DAS), all three factors, maturity stage, temperature, and their interaction, showed significant effects, resulting in relevant differentiation among the treatments. Fruits harvested at stages 1 and 2 and stored at 8°C first revealed decay symptoms at 28 DAS, indicating that early-harvested fruits stored under low temperature conditions exhibited delayed decay development. Under the same storage temperature, decay incidence decreased with an earlier maturity stage, while the decay incidence increased with a higher storage temperature within the same maturity stage.
Changes in the weight loss rate and firmness
The weight loss rate increased steadily in the fruits during storage (Fig. 2). In fruits harvested at maturity stage 1, the weight loss rates were significantly lower at 8°C than at 12 and 16°C throughout the storage period, with no significant differences between 12 and 16°C (Fig. 2A). In fruits harvested at maturity stages 2 and 3, the weight loss rates were significantly lower at 8, followed by 12 and 16°C, with the differences becoming more pronounced over time (Fig. 2B and 2C). Regardless of the storage temperature, fruits harvested at maturity stages 2 and 3 exhibited significantly higher weight loss rates than those at stage 1 (Fig. 2). By the end of the storage period, the weight loss rates at 8°C remained below 10% and were approximately half of those observed at 12 and 16°C, regardless of the maturity stage.
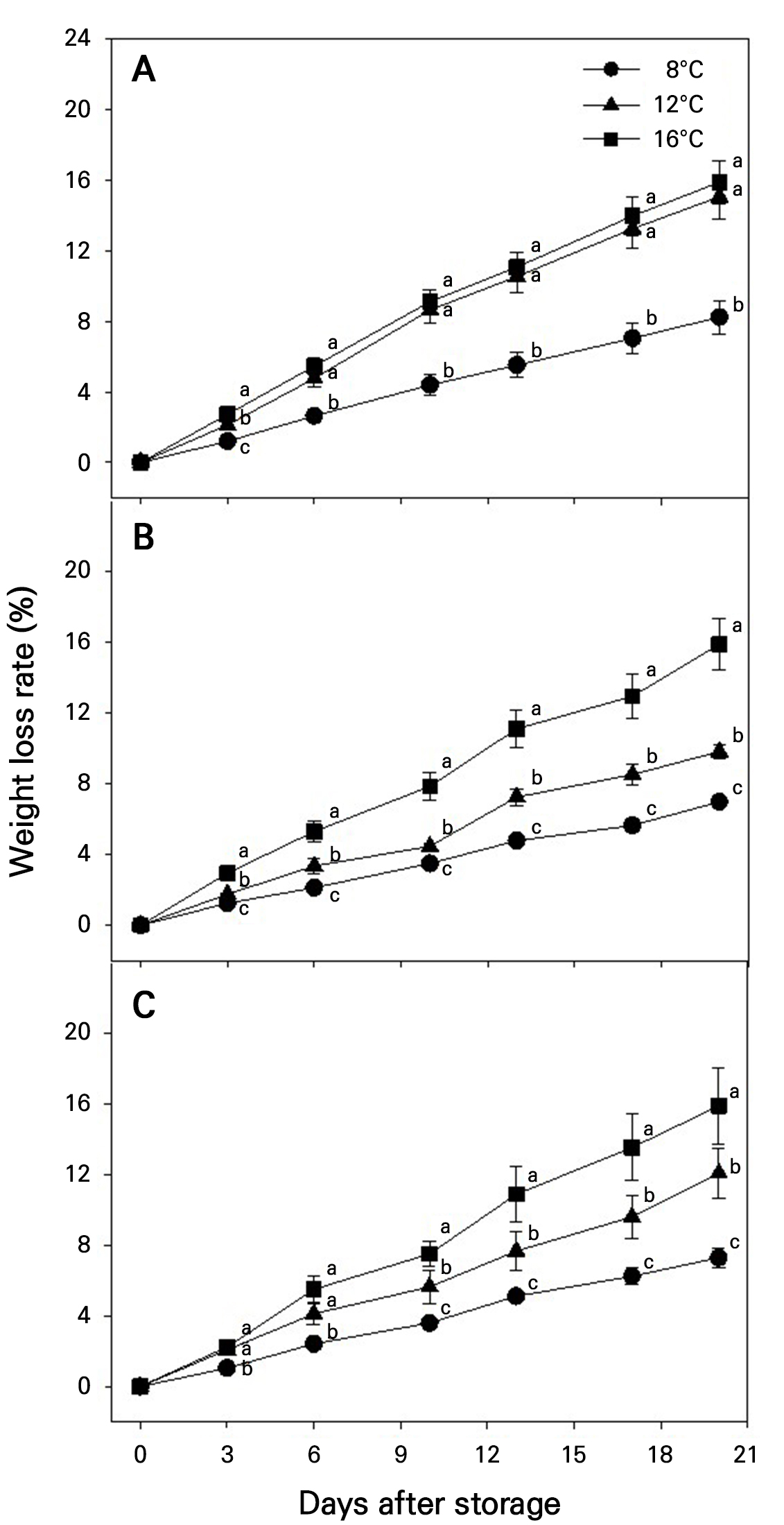
Fig. 2.
Changes in the weight loss rate during storage at different temperatures in ‘Irwin’ mango fruits (n = 5) harvested at maturity stages 1 (A), 2 (B), and 3 (C) based on the color chart in Fig. 1. Vertical bars are the standard error of the means. Different letters above the means indicate significant differences among temperature treatments at each storage day according to one-way ANOVA. *, **, and ***. Significant at p < 0.05, 0.01, and 0.001, respectively.
Overall, fruit firmness gradually declined during storage (Fig. 3). When the fruits harvested at maturity stage 1 were stored at 8°C, however, firmness decreased up to 3 DAS and remained relatively unchanged thereafter (Fig. 3A). Firmness was significantly higher at 8°C than at 12 and 16°C throughout the storage period, regardless of the maturity stage, but no significant differences were observed between 12 and 16°C (Fig. 3). By the end of the storage period, the firmness values were approximately 10, 7, and 6 N at 8°C for fruits harvested at maturity stages 1, 2, and 3, respectively, showing lower firmness in fruits harvested at more advanced maturity stages. At 12 and 16°C, in contrast, the firmness values dropped below 4 N for fruits harvested at any maturity stage.
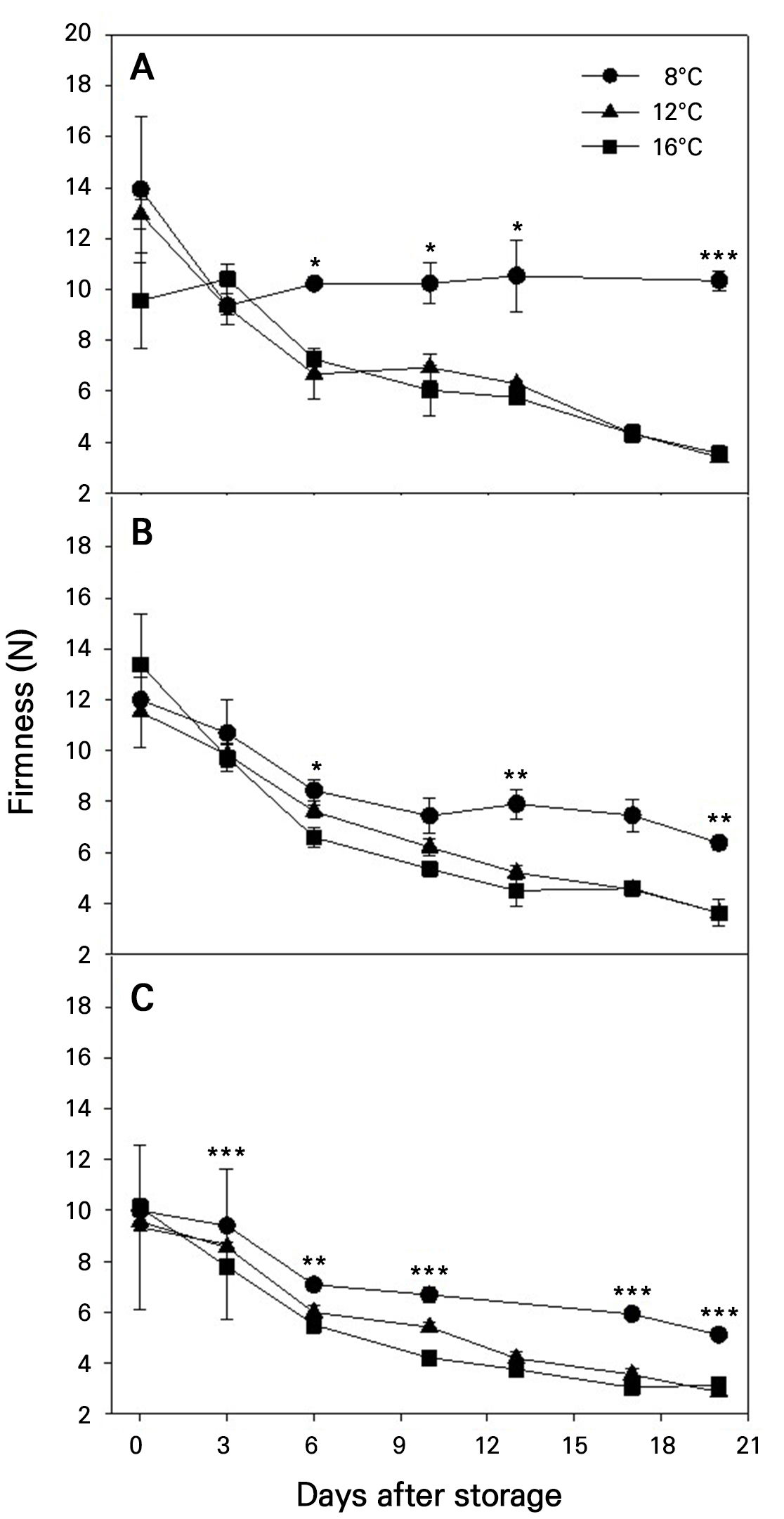
Fig. 3.
Firmness changes during storage at different temperatures in ‘Irwin’ mango fruits (n = 5) harvested at maturity stages 1 (A), 2 (B), and 3 (C) based on the color chart in Fig. 1. Vertical bars are the standard error of the means. Statistical differences among the temperature treatments at each storage day are indicated by asterisks over the means based on one-way ANOVA. *, **, and ***: Significant at p < 0.05, 0.01, and 0.001, respectively.
Changes of SSC, TA, the SSC/TA ratio, and the pH
SSC in the fruits tended to increase during storage (Fig. 4). Fruits harvested at more advanced maturity stages exhibited slightly higher SSC outcomes through all storage period. However, storage temperature had no significant effect on SSC changes in fruits harvested at any maturity stage. In fruits harvested at maturity stage 3, exceptionally, SSC values were significantly different only at 13 DAS depending on the storage temperature (Fig. 4C).
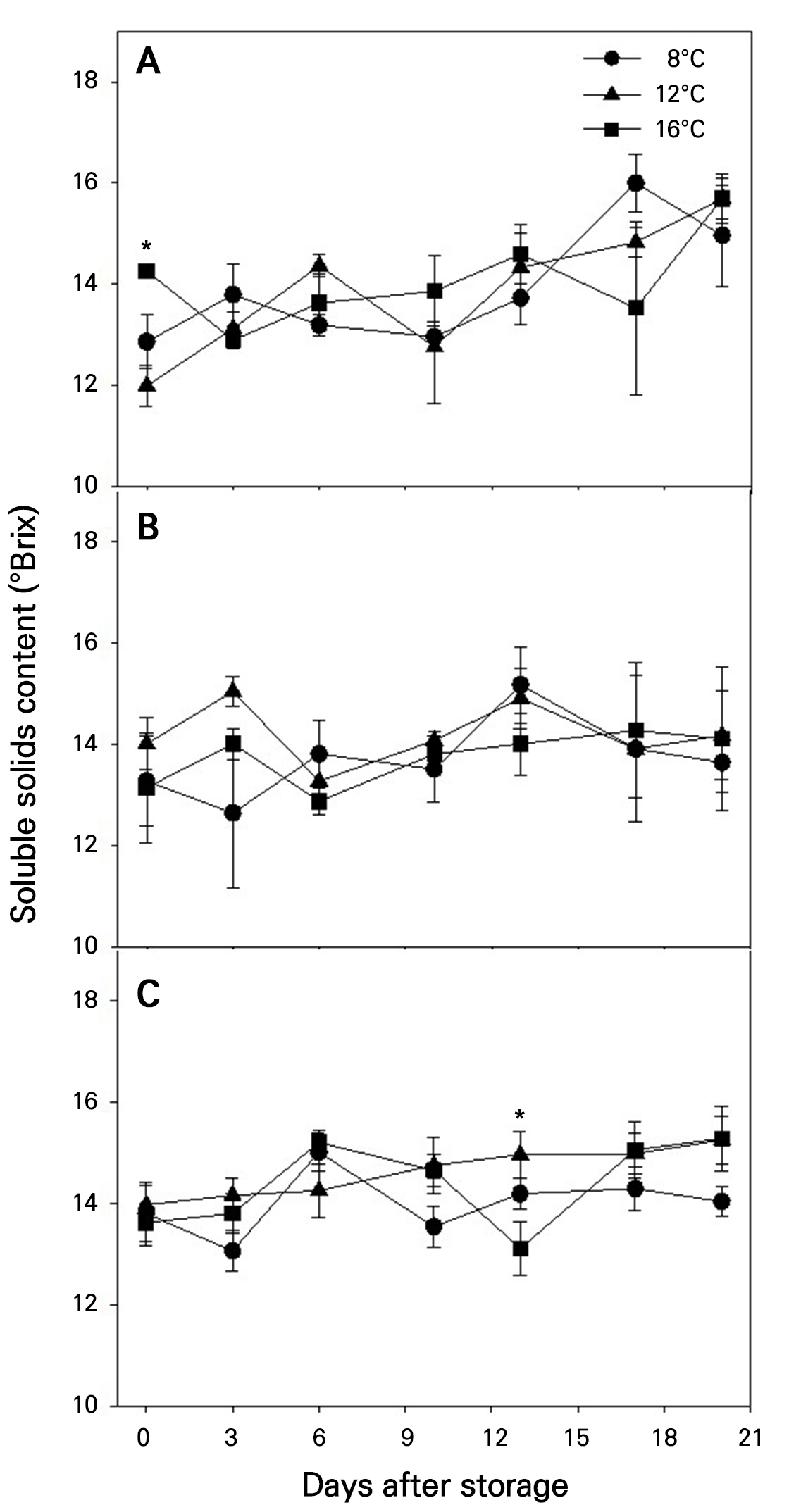
Fig. 4.
Changes in the soluble solids content during storage at different temperatures in ‘Irwin’ mango fruits (n = 5) harvested at maturity stages 1 (A), 2 (B), and 3 (C) based on the color chart in Fig. 1. Vertical bars are the standard error of the means. Statistical differences among the temperature treatments at each storage day are indicated by asterisks over the means based on one-way ANOVA. *Significant at p < 0.05.
TA in the fruits tended to decrease during storage (Fig. 5). Fruits harvested at more advanced maturity stages exhibited slightly lower TA values throughout the storage period. Although TA values were significantly different at certain DAS depending on the storage temperature, neither storage temperature nor maturity stage had significant overall effect on TA changes.
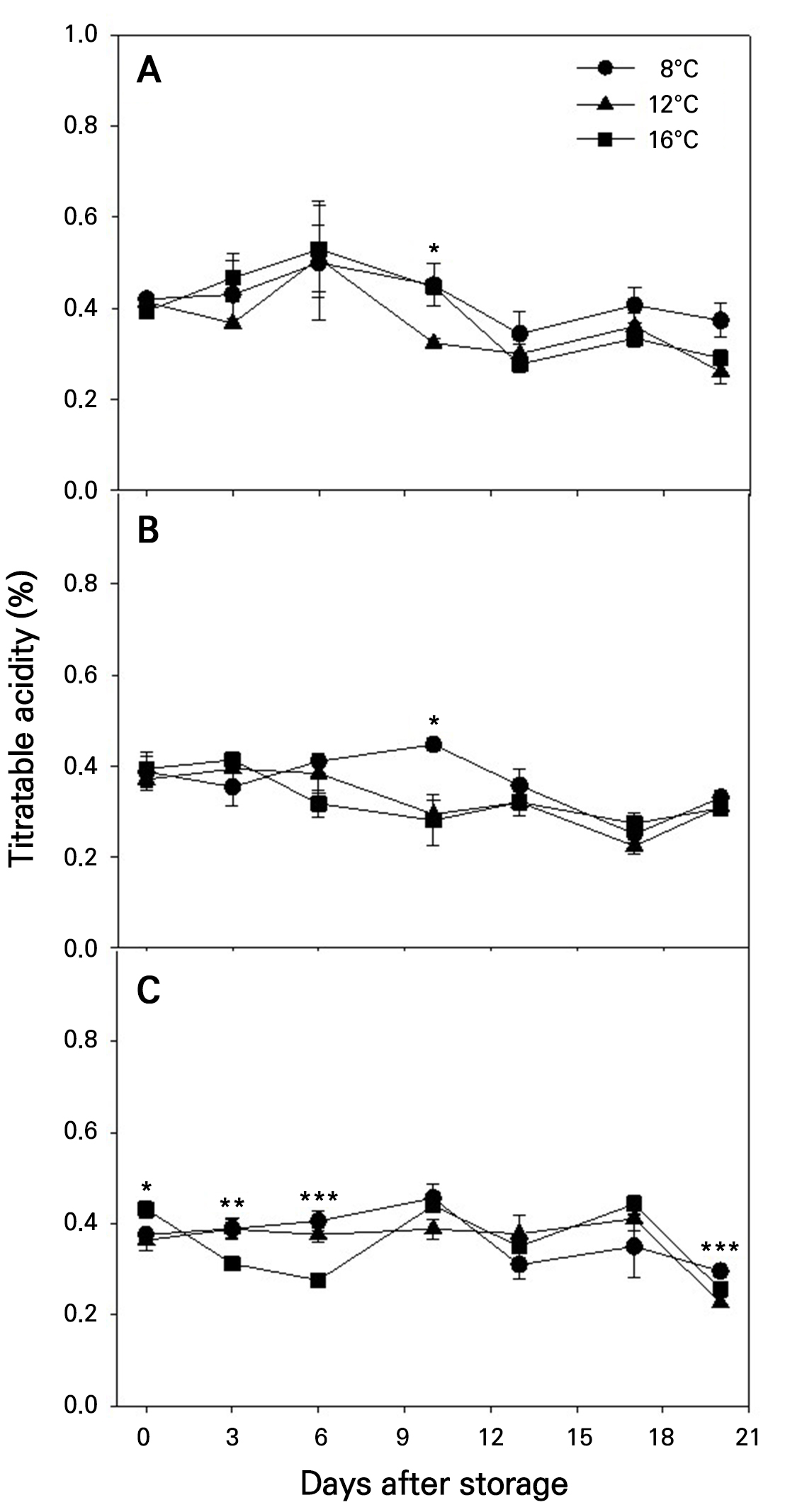
Fig. 5.
Changes of titratable acidity during storage at different temperatures in ‘Irwin’ mango fruits (n = 5) harvested at maturity stages 1 (A), 2 (B), and 3 (C) based on the color chart in Fig. 1. Vertical bars are the standard error of the means. Statistical differences among the temperature treatments at each storage day are indicated by asterisks over the means based on one-way ANOVA. *, **, and ***: Significant at p < 0.05, 0.01, and 0.001, respectively.
The SSC/TA ratio in the fruits tended to increase during storage (Fig. 6). Although these ratios were significantly different at certain DAS depending on the storage temperature, neither storage temperature nor maturity stage had significant overall effects on changes in the SSC/TA ratios.
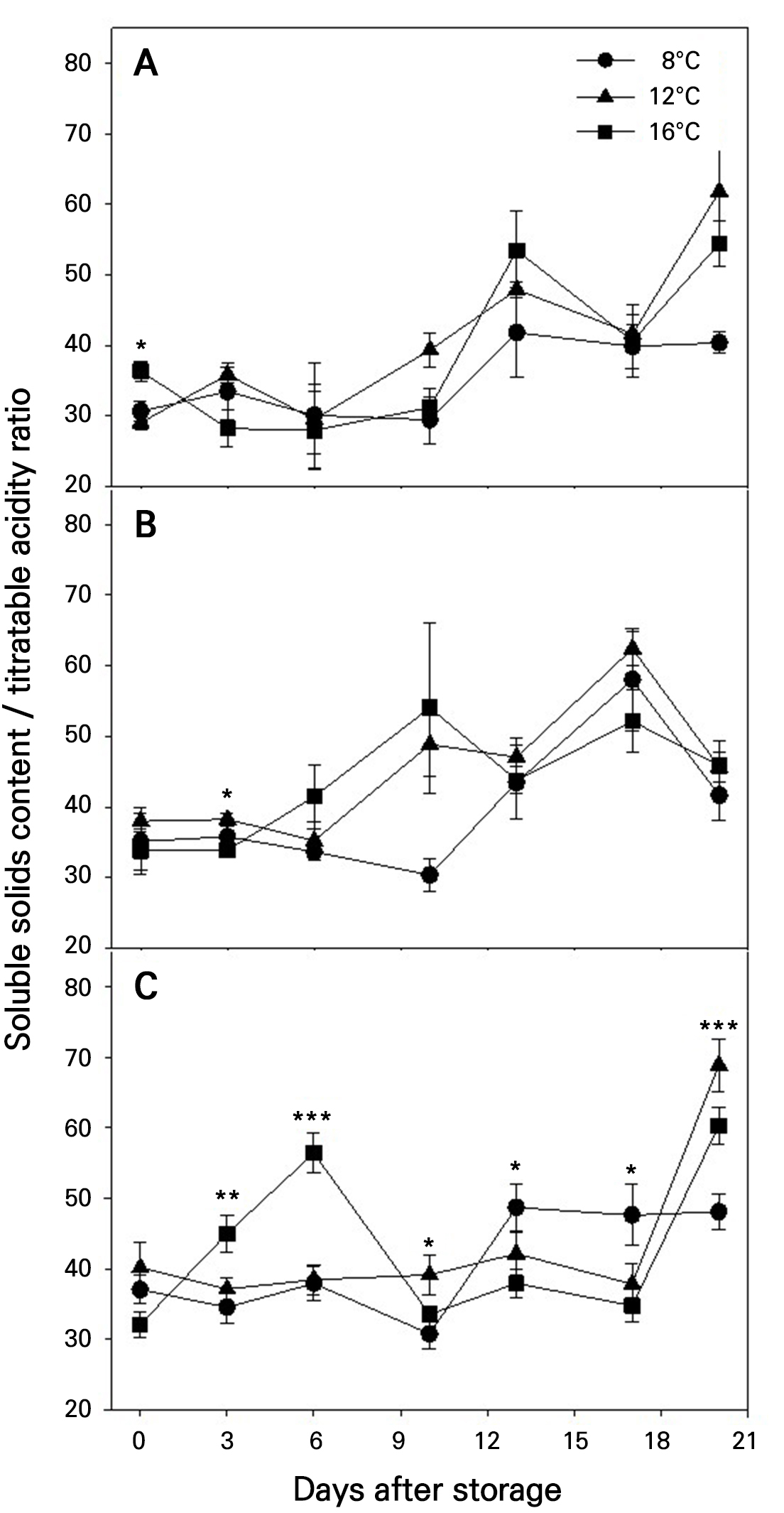
Fig. 6.
Changes in the soluble solids content/titratable acidity during storage at different temperatures in ‘Irwin’ mango fruits (n = 5) harvested at maturity stages 1 (A), 2 (B), and 3 (C) based on the color chart in Fig. 1. Vertical bars are the standard error of the means. Statistical differences among the temperature treatments at each storage day are indicated by asterisks over the means based on one-way ANOVA. *, **, and ***: Significant at p < 0.05, 0.01, and 0.001, respectively.
The pH in the fruits tended to increase during storage (Fig. 7). The increase became more pronounced at higher storage temperatures regardless of the maturity stage. For example, at 20 DAS, pH values were significantly lower at 8°C than at 12 and 16°C. Additionally, fruits harvested at more advanced maturity stages exhibited slightly higher pHs throughout the storage period.
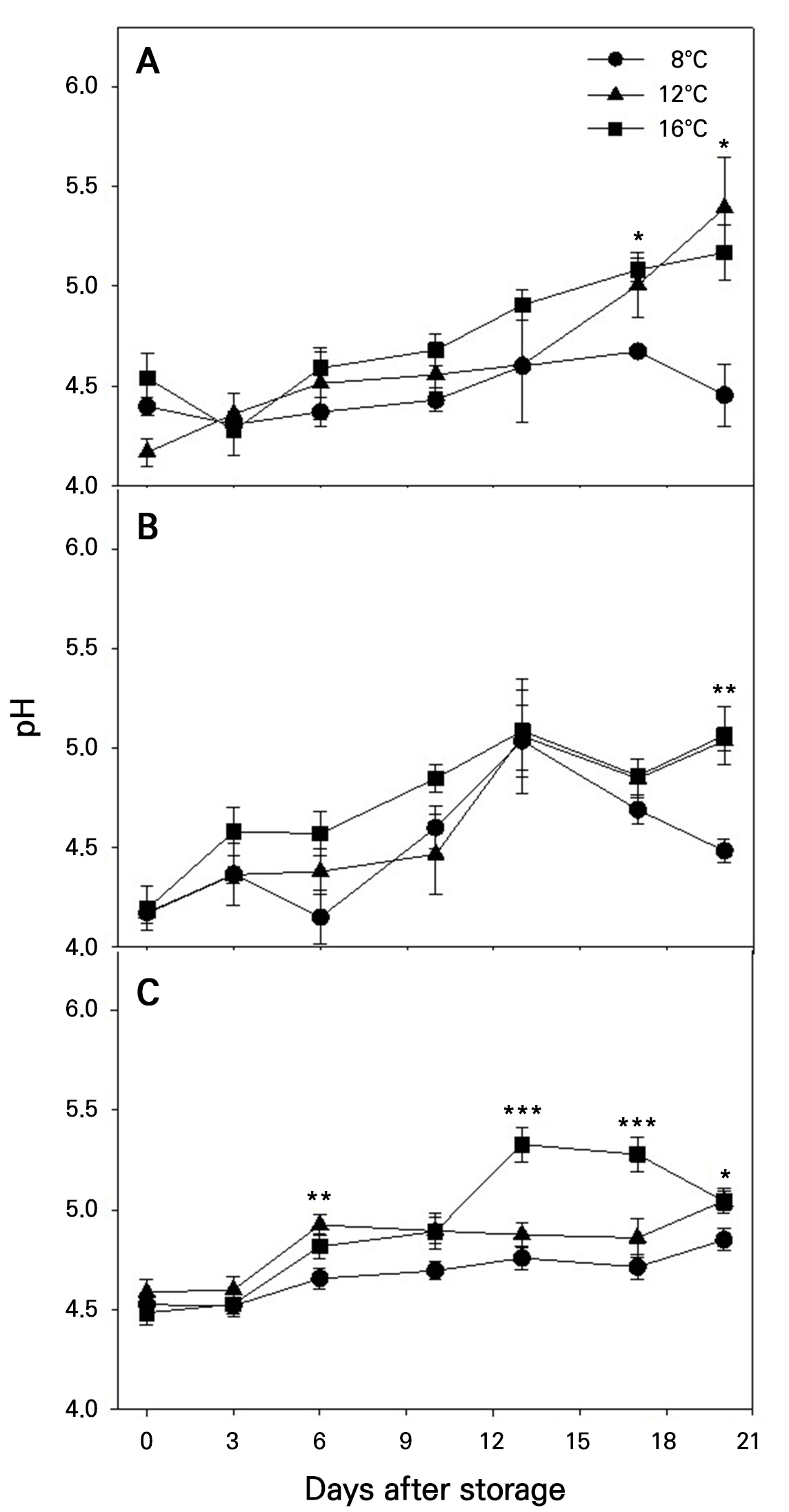
Fig. 7.
Changes of pH during storage at different temperatures in ‘Irwin’ mango fruits (n = 5) harvested at maturity stages 1 (A), 2 (B), and 3 (C) based on the color chart in Fig. 1. Vertical bars are the standard error of the means. Statistical differences among the temperature treatments at each storage day are indicated by asterisks over the means based on one-way ANOVA. *, **, and ***: Significant at p < 0.05, 0.01, and 0.001, respectively.
Significance of maturity stage, and storage temperature and duration on fruit quality attributes
A two-way mixed ANOVA was conducted to examine the main and interaction effects of maturity stage, and storage temperature and duration on the quality attributes of ‘Irwin’ mango fruits (Table 4).
The weight loss rate in the fruits was significantly affected by the main effects of the storage temperature (p < 0.001) and storage duration (p < 0.001) and by the two-way interactions of maturity stage × storage duration (p < 0.001) and storage temperature × storage duration (p < 0.001). However, the main effect of the maturity stage was not statistically significant. Fruit firmness was significantly affected by the main effects of maturity stage (p < 0.001), storage temperature (p < 0.001), and duration (p < 0.001), as well as by the two-way interactions of maturity stage × storage temperature (p < 0.01), maturity stage × storage duration (p < 0.05), and storage temperature × storage duration (p < 0.01) and the three-way interaction of maturity stage × storage temperature × storage duration (p < 0.01).
SSC in the fruits was significantly affected by the main effects of maturity stage (p < 0.001) and storage duration (p < 0.001), as well as by the two-way interactions of maturity stage × storage duration (p < 0.05). However, the main effect of storage temperature was not statistically significant. TA was not significantly affected by any of the main and interaction effects of maturity stage, or storage temperature and duration. The SSC/TA ratio was significantly affected by any main and interaction effects of the maturity stage, or the storage temperature and duration, though an effect was found for the interaction of maturity stage × storage temperature. pH was significantly affected by the main effects of maturity stage (p < 0.001), storage temperature (p < 0.001), and storage duration (p < 0.001), as well as by the two-way interactions of maturity stage × storage duration (p < 0.01) and storage temperature × storage duration (p < 0.05).
Discussion
Shriveling and decay incidence
Globally, commercially produced mango fruits are mostly intended for export, with only a small proportion allocated for domestic distribution. In the international mango trade, several quality-related challenges arise during storage, including chilling injury and anthracnose. However, the most critical concern is the potential spread of fruit flies, which necessitates quarantine treatments typically involving heat treatments such as hot air or a hot water treatment (Yimyong et al. 2011; Zhang et al. 2012), as mandated by importing countries. Shriveling in export-oriented mango fruits is a key physiological indicator of postharvest disorders, often caused by prolonged internal heating associated with a hot water treatment. In contrast, in South Korea, mango fruits are primarily distributed as premium gift items for domestic consumption and are generally marketed without undergoing a hot water treatment.
Storage temperature is one of the most critical factors influencing the postharvest quality and marketability of horticultural crops. Even a 10°C increase above the optimal storage temperature can accelerate fruit deterioration by two to three times (Kader 2008). For instance, shriveling was shown to develop in approximately 73% of ‘Gold 9’ kiwifruit stored at 20°C for ten days, whereas no such symptoms were observed in fruits stored at lower temperatures up to three weeks (Burdon et al. 2014). The development of shriveling was primarily associated with water loss, and its severity was closely correlated with reduced fruit firmness (Burdon et al. 2014). Hesami et al. (2021) also reported that shriveling in Indian jujube fruits stored at a high temperature was due to rapid weight loss caused by transpiration.
In the present study, shriveling in ‘Irwin’ mango fruits during storage occurred earlier at higher storage temperatures and was more pronounced in fruits harvested at more advanced maturity stages (Table 1), aligning with the findings of previous research. However, at 8°C, the initial appearance of shriveling occurred earlier in fruits harvested at maturity stage 1 compared to those at stage 2 (Table 2). As storage progressed, the effect of the maturity stage on shriveling became more pronounced, especially in fruits harvested at stages 1 and 3. Notably, fruits harvested at stage 1 tended to develop shriveling more rapidly than those harvested at stage 2, suggesting that water loss is not the only factor contributing to shriveling. This is attributable to the fact that the fruits harvested at maturity stage 1 experienced chilling injury at 8°C. Mango fruits are known to respond differently to low storage temperatures depending on their maturity stage, with less mature fruits being more susceptible to chilling injury (Zhao et al. 2008; Maul et al. 2012). Mango fruits harvested before reaching optimal maturity have also been reported to have shorter shelf lives due to increased shriveling during storage at low temperatures (Patel et al. 2014). Similarly, in carambola fruits stored at low temperatures, shriveling was associated with water loss from the fruits and manifested as chilling injury accompanied by surface browning (Imahori et al. 2021).
Table 1.
CIE L, a, and b values and the chroma and hue angle of fruits harvested at stages 1, 2 and 3
| Maturity stagez | CIE L | CIE a | CIE b | Chroma | Hue |
| Stage 1 | 46.84 ± 6.40y | 13.13 ± 6.03 | 23.32 ± 9.62 | 28.14 ± 7.18 | 31.93 ± 19.12 |
| Stage 2 | 47.14 ± 5.09 | 21.80 ± 5.70 | 25.17 ± 5.82 | 33.87 ± 5.13 | 40.86 ± 10.75 |
| Stage 3 | 44.35 ± 3.20 | 23.34 ± 1.59 | 17.45 ± 4.27 | 29.92 ± 2.74 | 54.88 ± 6.88 |
zThree different maturity stages were determined based on the color chart presented in Fig. 1.
Table 2.
Shriveling incidence during storage at different temperatures in ‘Irwin’ mango fruits harvested at maturity stages 1, 2, and 3 based on the color chart in Fig. 1
| Maturity stage (A) | Storage temperature (°C) (B) | Days after storage | |||||||||
| 0 | 3 | 6 | 10 | 13 | 17 | 20 | 25 | 28 | 31 | ||
| Stage 1 | 8 | 1.0 az | 1.0 a | 1.0 b | 1.0 b | 1.0 c | 1.1 c | 1.6 c | 2.2 c | 3.0 c | 4.5 ab |
| 12 | 1.0 a | 1.0 a | 1.0 b | 1.1 b | 1.6 b | 2.3 a | 3.1 a | 3.8 ab | 4.4 ab | 4.8 ab | |
| 16 | 1.0 a | 1.0 a | 1.0 b | 1.2 b | 1.6 b | 2.2 a | 2.6 ab | 3.9 ab | 4.6 a | 4.9 ab | |
| Stage 2 | 8 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 c | 1.0 c | 1.0 d | 1.0 d | 1.6 d | 2.4 c |
| 12 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 c | 1.0 c | 1.0 d | 2.5 c | 3.4 c | 4.4 b | |
| 16 | 1.0 a | 1.0 a | 1.0 b | 1.2 b | 2.0 a | 2.5 a | 3.3 a | 4.2 a | 4.7 a | 5.0 a | |
| Stage 3 | 8 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.1 c | 1.4 bc | 2.4 ab | 3.3 b | 3.7 bc | 4.5 ab |
| 12 | 1.0 a | 1.0 a | 1.0 b | 1.1 b | 1.7 b | 2.0 b | 3.0 a | 3.5 b | 4.2 ab | 4.9 ab | |
| 16 | 1.0 a | 1.0 a | 1.3 a | 1.7 a | 2.1 a | 2.6 a | 3.3 a | 4.0 ab | 4.8 a | 5.0 a | |
| Significance | A | ns | ns | * | ns | ns | ns | ** | *** | *** | *** |
| B | ns | ns | * | ** | *** | *** | *** | *** | *** | *** | |
| A × B | ns | ns | ** | ns | ns | ns | ** | *** | ** | *** | |
Fruit decay incidence during storage followed a trend similar to that of shriveling, with decay occurring earlier at higher storage temperatures and progressing more rapidly in fruits harvested more advanced maturity stages (Table 3). These results are consistent with previous work finding that mango fruits soften more quickly under warm, humid conditions, facilitating pathogen entry and microbial growth (Sivakumar et al. 2011). In acerola fruits, similarly, a negative correlation was observed between peel color development and decay incidence, indicating increased susceptibility to decay as ripening progresses (Ribeiro and de Freitas 2020). Accelerated decay at higher storage temperatures has also been reported in other fruits, including passion fruit (De Arruda et al. 2011) and persimmon (Velho et al. 2011).
Table 3.
Decay incidence during storage at different temperatures in ‘Irwin’ mango fruits harvested at maturity stages 1, 2, and 3 Based on the color chart in Fig. 1
| Maturity stage (A) | Storage temperature (°C) (B) | Days after storage | |||||||||
| 0 | 3 | 6 | 10 | 13 | 17 | 20 | 25 | 28 | 31 | ||
| Stage 1 | 8 | 1.0 az | 1.0 a | 1.0 b | 1.0 b | 1.0 b | 1.0 b | 1.0 c | 1.0 d | 1.8 d | 2.3 c |
| 12 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 b | 1.0 b | 1.0 c | 1.6 c | 2.7 cde | 3.6 b | |
| 16 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 b | 1.0 b | 2.9 a | 4.0 a | 4.2 ab | 4.9 a | |
| Stage 2 | 8 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 b | 1.0 b | 1.0 c | 1.0 d | 1.1 e | 1.4 d |
| 12 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 b | 1.0 b | 1.2 bc | 1.6 c | 2.5 cd | 3.9 b | |
| 16 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 b | 1.1 b | 1.3 bc | 1.5 c | 2.4 cd | 3.6 b | |
| Stage 3 | 8 | 1.0 a | 1.0 a | 1.0 b | 1.0 b | 1.0 b | 1.2 b | 1.5 bc | 2.4 b | 2.8 c | 3.7 b |
| 12 | 1.0 a | 1.0 a | 1.0 b | 1.1 b | 1.2 b | 1.6 b | 1.8 b | 2.5 b | 3.5 b | 4.4 ab | |
| 16 | 1.0 a | 1.0 a | 1.0 a | 1.8 a | 1.9 a | 2.2 a | 2.7 a | 3.8 a | 4.7 a | 5.0 a | |
| Significance | A | ns | ns | ns | ** | * | *** | *** | *** | *** | *** |
| B | ns | ns | ns | ** | ns | ** | *** | *** | *** | *** | |
| A × B | ns | ns | ns | ** | * | ns | ** | *** | ns | ** | |
Changes in weight loss rate and firmness
The weight loss rate in the fruits during storage increased steadily with higher storage temperatures and with the increase more pronounced in fruits harvested at more advanced maturity stages (Fig. 2). However, the effect of storage temperature was found to be significantly greater than that of maturity stage (Table 4). Furthermore, the increases in weight loss rates during storage (Fig. 2) appeared to be closely associated with the incidence of shriveling (Table 2). When the weight loss rates in stored fruits reached approximately 6–8%, the initial appearance of shriveling was observed regardless of the maturity stage or storage temperature. This range may represent the critical threshold for shriveling incidence in ‘Irwin’ mango fruits during storage.
Table 4.
Significance of maturity stage, and storage temperature and duration with quality attributes of ‘Irwin’ mango fruits
| Variable | Significance | |||||
| Weight loss rate | Firmness | SSC | TA | SSC/TA | pH | |
| Maturity stage (A) | nsz | *** | *** | ns | ** | *** |
| Storage temperature (B) | *** | *** | ns | ns | * | *** |
| Storage duration (C) | *** | *** | *** | ns | *** | *** |
| A × B | ns | ** | ns | ns | ns | ns |
| A × C | *** | * | * | ns | ** | ** |
| B × C | *** | ** | ns | ns | * | * |
| A × B × C | ns | ** | ns | ns | * | ns |
Weight loss during storage is common in many horticultural crops and has been reported to vary depending on the cultivar and the maturity stage at harvest (Nunes and Emond 2007). In ‘Alphonso’ mango fruits ripened at 20, 25, 30°C, and ambient temperatures (25–33°C), the latest shriveling appearance was observed at 20°C, likely due to reduced moisture loss at lower temperatures (Patil et al. 2016). Shriveling in fruits during storage reportedly begins once the weight loss rate reaches a certain threshold. For example, mango fruits stored at 20°C and 85–95% relative humidity reached a weight loss rate of 1.5–4% within two to five days, during which over-ripening, shriveling, and softening occurred, leading to a decline in marketability (Nunes and Emond 2007). Liu et al. (2014) reported that when the weight loss rate of mango fruits reaches 10%, they lose their original fresh taste and marketability. These thresholds vary depending on the species and cultivar. It can be assumed that differences in fruit size and tissue structure lead to variations in the exposed surface area of the peel (Nunes and Emond 2007). Also, in ‘Exp 15’ papaya fruits, softening and over-ripening were observed at a weight loss rate of 4.5%, which was identified as the storage threshold (Nunes and Emond 2007).
Fruit firmness declined progressively during storage with increasing storage temperatures and with the decline more pronounced in fruits harvested at more advanced maturity stages (Fig. 3). At 8°C, however, firmness remained relatively unchanged in fruits harvested at maturity stage 1 (Fig. 3A). Changes in firmness during storage were strongly influenced by both maturity stage and storage temperature, as well as by their interaction (Table 4).
Fruit firmness is regarded as a key indicator of softening during both ripening (Jha et al. 2013; Chea et al. 2019) and storage (Jha et al. 2010; Hossain et al. 2021). Firmness typically decreases gradually as ripening progresses or during prolonged storage. This decrease in firmness is primarily attributed to the hydrolysis or depolymerization of cell wall polysaccharides, in this case, cellulose, hemicellulose, and pectin, by the action of cell-wall-modifying enzymes like pectin methyl esterase, polygalacturonase, and β-galactosidase (Yashoda et al. 2006; Chen et al. 2015; Chea et al. 2019; Bambalele et al. 2021).
Changes of SSC, TA, the SSC/TA ratio, and the pH
SSC in the fruits tended to increase slightly (Fig. 4), while TA showed a slight decrease during storage (Fig. 5). These changes were more pronounced in fruits harvested at more advanced maturity stages (Figs. 4 and 5). The effect of maturity stage on SSC changes was statistically significant, whereas its effect on TA changes was not (Table 4). In contrast, storage temperature had no significant effects on either SSC or TA changes (Table 4), although lower storage temperatures generally tend to slow such changes. These findings suggest that ripening progresses more actively during storage in fruits harvested at a more advanced maturity stage. Additionally, the SSC/TA ratio appeared to increase during storage (Fig. 6), but neither maturity stage nor storage temperature had a significant overall effect on this change (Table 4).
During fruit ripening or storage, SSC typically increases as starch is hydrolyzed into sugars of glucose, fructose, and sucrose (Baloch and Bibi 2012; Singh et al. 2013). In contrast, TA decreases primarily due to the conversion of organic acids into sugars and their utilization in respiratory metabolism (Srinivasa et al. 2002; Yashoda et al. 2006; Baloch and Bibi 2012). In ‘Irwin’ mango fruits, the starch content was shown to decrease to very low or undetectable levels as ripening progresses, with sucrose becoming the predominant sugar (Ito et al. 1997). Concurrently, citric acid, the major organic acid in most mango cultivars, gradually decreases (Ito et al. 1997). A similar trend was observed in ‘Alphonso’ mango fruits, where the levels of starch, pectin, cellulose, and hemicellulose decreased during ripening, while SSC increased (Yashoda et al. 2006). A negative correlation between SSC and TA during ripening or storage has also been reported in ‘Langra’ and ‘Samar Bahisht Chaunsa’ mango fruits (Baloch and Bibi 2012).
The pH in the fruits tended to increase during storage, with the increase being more pronounced at higher storage temperatures and in fruits harvested at more advanced maturity stages (Fig. 7). The pH change was significantly influenced by both maturity stage and storage temperature, but not by their interaction (Table 4).
A similar pH increase during fruit ripening or storage has often been reported in other mango cultivars, for example, ‘Khirshapat’ (Islam et al. 2013), ‘Langra’ (Baloch and Bibi 2012; Islam et al. 2013), and ‘Samar Bahisht Chausa’ (Baloch and Bibi 2012). This pH increase is attributable to the conversion of organic acids into sugars (Baloch and Bibi 2012; Shivashankar 2014), which accelerates with an increase in the temperature (Kumar and Singh 1993; Rathore et al. 2007).
Conclusion
This study demonstrated that both harvest maturity stage and storage temperature significantly affect the postharvest quality of greenhouse-grown ‘Irwin’ mango fruits. Higher storage temperatures accelerated weight loss, shriveling, and decay, particularly in fruits harvested at more advanced maturity stages, while fruit firmness and acidity declined more rapidly under these conditions.
Importantly, the results here demonstrated that fruits harvested at maturity stage 2 could be safely stored at 8°C without causing chilling injury (CI), allowing for a significantly extended shelf life compared to fully matured fruits. This finding offers a practical postharvest strategy that enables distribution over a longer period. It helps to prevent market oversupply and the associated price drops during the peak harvest season.
By identifying the physiological threshold for shriveling (around 6–8% of weight loss) and demonstrating quality retention at earlier harvest stages under low-temperature storage conditions, this study provides a scientific basis for optimizing harvest timing and storage practices. These results contribute not only to preventing price drops during the oversupply season but also to enhancing the sustainability of mango production in Korea.


