Introduction
Material and Methods
Plant Materials
Observation of Flower Bud Structure
Technique for Isolated Microspore Culture
Microspore Culture for MDE Formation
Microspore Density
Observation of the Microspores Developmental Stage
Anatomical Observation of MDE Development
Statistical Analysis
Result and Discussion
Introduction
Radish (Raphanus sativus L.) is one of the most popular root and leaf vegetables in East Asia. It is a cruciferous crop that is used as both a medicinal and edible plant. Nutritionally, radish is a rich source of vitamins B and C, peroxidase, and organic isothiocyanates, and for this reason radish has been used for many medicinal purposes (Curtis, 2007). Breeding practices such as mass selection, pedigree selection, and bud pollination have been used to increase yield and to improve the plant’s medicinal value. However, conventional breeding takes a long time, so genetic engineering has emerged as an important technique for producing improved varieties of many plants (Curtis, 2011).
Isolated and in-vitro cultured microspores can divide repeatedly and develop into embryos. The production of doubled haploid plants by embryogenesis is a useful method for obtaining homozygous individuals in a short period of time because it does not require the numerous steps involved in conventional breeding programs (Babbar et al., 2004; Forster et al., 2007; Seguí-Simarro and Nuez, 2008; Takahashi et al., 2011; Touraev et al., 2001). In many plants, the frequency of embryo induction is not high enough to perform molecular and biochemical characterization, except for a few species such as rice (Cho and Zapata, 1990; Raina and Irfan, 1998; Xie et al., 1995, 1997), tobacco (Heberle- Bors, 1989; Touraev et al., 1996a, 1996c; Žárský et al, 1992, 1995), and oilseed rape (Chuong et al., 1988; Cloutier et al., 1995; Gu et al., 2004; Polsoni et al., 1988; Senratna et al., 1991). In addition, different cultivars within a same species that can generate microspore-derived embryos (MDEs) show different MDE efficiency. For this reason, microspore culture is difficult to apply in some crops. Numerous factors affect a positive embryogenic response, including genotype and the growth conditions of the donor plants, the developmental stage of the flower buds or microspores, pretreatment of the flower buds, and the culture medium. Among these factors, the genotype of the donor plant and the developmental stage of the microspores are particularly important (Babbar et al., 2004; Seguí-Simarro and Nuez, 2008; Smykal, 2000). The effect of donor plant genotype was previously reported for several crucifer species (Babbar et al., 2004), but the focus of that study was to select a variety that had the ability to induce MDE formation.
Microspores of cruciferous crops at a specific stage of development are responsive to MDE formation. A previous report suggested that the greatest response to embryogenesis is observed when microspores are isolated at the first pollen mitosis from the late uninucleate to the early binucleate stage (Binarova et al., 1997; Takahata et al., 1991; Touraev et al., 1996a, 1996b). The role of the developmental stage of pollen at the moment of culture initiation, however, is still undetermined. The developmental stage of microspore varies greatly depending on the cultivar or species, and it is difficult to determine the developmental stage by microscopy. Therefore, it is necessary to select flower buds containing microspores that have the ability to induce MDE formation. The develop-mental stage of a microspore is associated with the length of the flower bud, floral leaf, or their ratio (Babbar et al., 2004; Smykal, 2000; Weber et al., 2005). Up until now, flower buds with a longer stigma length than to that of the floral leaf have been chosen from isolated microspore cultures of cruciferous crops (Chun et al., 2011a, 2011b). However, this method is only applicable to a few varieties. There-fore, the best time to separate the microspore should be investigated to elucidate the mechanism of non-MDE formation.
The objective of this study was to show a correlation between the length or structure of a flower bud and the microspore developmental stage in R. sativus L. In addition, the factors involved in preventing embryogenesis were investigated through the observation of MDEs during culture using microscopy.
Materials and Methods
Plant Materials
Raphanus sativus L. cvs. Taebaek and Chungwoon (Monsanto Co., Seoul, Korea) were used as donor plants for isolated microspore culture. Seeds of the donor plants were sown in 50 hole plugs. When the seedlings were 5 weeks old and showing 2-3 leaves, floral differentiation was induced in a cold room at 5.0 ± 1.0°C under continuous light for 4 weeks. After floral differentiation and the beginning of generative development, the plants were transferred to a greenhouse (length × width × height = 3.0 × 7.0 × 3.0 m3) which was equipped with an air-conditioning and heating system to maintain a day/night temperature between 23.0/16.0 ± 1.0°C.
Observation of Flower Bud Structure
Pre-blossom buds were collected and classified into three groups based on their length as follows: 2.0 (small, S), 4.0 (medium, M), and 6.0 (large, L) ± 0.5 mm. The floral leaf, anther, and stigma were examined using a microscope (Demis-ME; Siwon Optical Technology Co., Ltd., Anyang, Korea).
Technique for Isolated Microspore Culture
Those flower buds (4.0 ± 0.5 mm in length) with a longer stigma than the length of the floral leaf were collected. The buds were sterilized in 1% sodium hypochlorite for 15 min followed by rinsing three times with sterile deionized water for 3 min. The buds were then ground in a mortar containing B5 liquid medium (Gamborg et al., 1968) supple-mented with 13% sucrose. A microspore suspension was obtained by filtration through 45-µm nylon screens. This suspension was centrifuged three times at 1,000 rpm for 3 min. After washing, the microspores were suspended in Nitsch and Nitsch (NLN) liquid medium (Lichter, 1982) supplemented with 15% sucrose. The microspore suspension (2.5 mL) was placed in a 60 × 15 mm sterile petri dish, and 0.1 mL of 1% activated charcoal was dropped onto the petri dish, which was subsequently sealed with parafilm.
B5-13% and NLN-15% media were adjusted to a pH of 6.0 and pH 5.8, respectively, using NaOH, and were filter-sterilized using a 25 µm low protein-binding membrane filter (Corning, NY, USA). The microspores were incubated in darkness at 32.5°C for 2 days prior to maintenance at 25.0°C. After 17 days of culture in the dark, the petri dish was transferred to a gyratory shaker and agitated at 70 rpm and 25.0°C.
The cotyledonary embryos were transferred directly to B5 medium supplemented with 3% sucrose and 8% agar. All MDEs were incubated to induce shoot and root regeneration. Germinated plants were transferred to artificial soil.
Microspore Culture for MDE Formation
Flower buds were collected and classified into three groups based on their length: 2.0 (S), 4.0 (M), 6.0 (L) ± 0.5 mm. The embryo yield was examined 4 weeks after microspore isolation. The embryo yield was the mean embryo number in ten petri dishes.
Microspore Density
The number of microspores was counted using a disposable hemocytometer (C-Chip DHC-NO1-5, Incyto Co., Ltd., Cheonan, Korea). A microspore with liquid medium (10 µL) was loaded into the injection area, and then the number of the microspores was counted under a microscope. The hemocytometer consisted of 9 squares, each measuring 1.0 × 1.0 mm2, and the depth of the chamber was 0.1 mm. Each square had a total volume of 10-4 mL (length × width × height = 0.1 × 0.1 × 0.01 cm3 = 10-4 cm3 = 10-4 mL). Therefore, the original number of microspores needed to be multiplied by 104.
Observation of the Microspores Developmental Stage
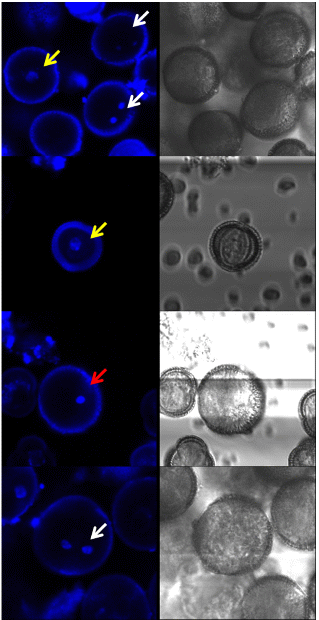
The developmental stage of the microspores was traced by fluorescence microscopy using 4’,6-diamidino-2-phenylindole (DAPI) staining. The developmental stages of the microspores were assigned as follows: early uninucleate, late uninucleate, and binucleate (Fig. 6). In the uninucleate stage, the micro-spores contained one nucleus. In the early and late uni-nucleate stages, the nucleus of the microspore was located at the center and off, respectively. In the binucleate stage, the microspores contained two nuclei.
DAPI is a fluorescent stain that binds preferentially to A-T rich regions of DNA. It has been used to stain both fixed and live cells. When bound to DNA, DAPI has an absorption maximum at a wavelength of 358 nm (Tarnowski et al., 1991). DAPI was dropped directly onto the NLN medium containing microspores. The medium was then incubated in the dark at room temperature. After 1 h, a staining sample was rinsed several times and the nucleus was observed by a confocal laser scanning microscopy (TCS SP5 ADBS/Tandem; Leica Microsystems Co., Wetzlar, Germany). Image processing was carried out using a resonant scanner with an emission spectral range of 425-475 nm. The DAPI/DNA complex was excited with a 405-nm laser, and the measured range was 119.2 × 119.2 nm2.
Anatomical Observation of MDE Development
MDE development from the microspores was observed at intervals of 2 days for 30 days. The structural pattern of the microspores was observed soon after isolation using an optical microscope (Axioskop2 Plus; Carl Zeiss Co., Thornwood, NY, USA). MDEs generated from the microspores of the ‘Taebaek’ cultivar were observed using a microspore (Demis-ME; Siwon Optical Technology Co., Ltd., Anyang, Korea) 20 days after the culture was isolated.
Statistical Analysis
All results were replicated ten times. A statistical analysis was performed with SigmaPlot 12.0 statistical software. The values are presented as the average of a replicate analysis in each of the experiments followed by the standard error.
Result and Discussion
The size and ratio of the floral leaf, stamen, and stigma within the flower buds of the ‘Taebaek’ and ‘Chungwoon’ cultivars were examined by microscopy (Fig. 1). In both cultivars, the stigma was invisible since the length of the stamen was longer than that of the stigma in the S-sized flower buds. In the M-sized flower buds, the length of the stigma was longer than that of the stamen and floral leaf. The stigma was not observed in the L-sized flower buds because it was located under the floral leaf. The structure of the floral leaf, anther, and stigma according to flower bud size showed obvious differences in length and ratio, while there was no difference in the structures between the two cultivars.
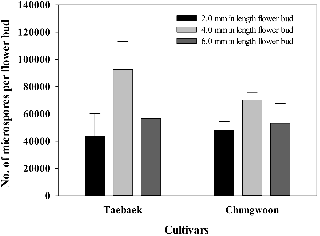
The M-sized flower buds had the highest number of microspores in the ‘Taebaek’ and ‘Chungwoon’ cultivars, followed by the L- and S-sized flower buds (Figs. 2 and 3). Furthermore, the number of microspores in the M-sized flower buds was higher in ‘Taebaek’ than that of ‘Chungwoon’, which indicated a high efficiency of MDE formation compared with ‘Chungwoon’, which cannot induce MDEs.
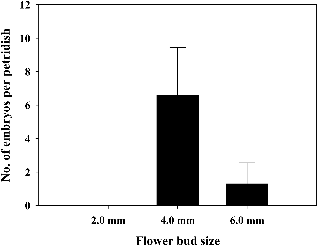
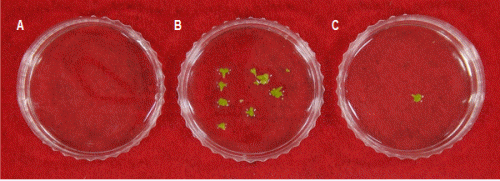
The response MDE formation rate was investigated to select the optimum bud size for isolated microspore culture. The MDE yields per petri dish in the ‘Taebaek’ cultivar were 6.6 and 1.3 for the M- and L-sized flower buds, respectively (Figs. 4 and 5). However, MDEs were not induced in the S-sized flower bud. On the other hand, ‘Chungwoon’ failed to form MDEs in S-, M-, and L-sized of flower buds.
A total of 82.1 and 81.6% of the microspores were in the late uninucleate stage in M-sized flower buds of ‘Taebaek’ and ‘Chungwoon’, respectively. The M-sized flower buds contained the greatest number of late uninucleate microspores (Table 1 and Fig. 6). The development of the microspores varied with flower bud size.
|
Fig. 1. Structure of the flower buds, floral leaves, anthers, and stigmas in the ‘Taebaek’ (A) and ‘Chungwoon’ (B) cultivars by flower bud length. a, 2.0 ± 0.5 mm; b, 4.0 ± 0.5 mm; c, 6.0 ± 0.5 mm. |
Flower bud length is a simple parameter to use in selecting flower buds containing a microspore with the ability to induce an MDE (Babbar et al., 2004; Smykal, 2000). Various approaches to induce MDEs in R. sativus L. have been reported (Chun et al., 2011a, 2011b; da Silva Dias 1999; Takahata et al., 1996). Chun et al. (2011b) observed that the highest MDE yield was obtained when the microspore was isolated from buds that were 2.5-4.5 mm in length. A flower bud showing a longer length of stigma compared to that of the floral leaf is considered the standard for separating the microspore in cruciferous crops (Babbar et al., 2004; Chun et al., 2011a, 2011b; Smykal, 2002). The results from those studies are consistent with our finding of MDE formation according to the length of the flower bud. The M-sized of flower buds had the highest microspore content and showed the highest MDE yield in ‘Taebaek’. It is assumed that the microspore content of a flower bud is likely to affect MDE formation. Previous reports showed that a microspore has totipotency during the late uninucleate to early binucleate stages (Binarova et al., 1997; Touraev et al., 1996a, 1996b). Therefore, the selection of a flower bud containing microspores at the optimal stage is an important factor to increase rates of embryogenesis. Our results show that peak MDE formation in ‘Taebaek’ occurred when microspores were isolated from M-sized flower buds, which had the highest content of late uninucleate stage microspores. These results are in agreement with those of previous studies of cruciferous crops in which MDEs were obtained from microspores at the late uninucleate stage (Baillie et al., 1992; Burnett et al., 1992; Hansen and Svinnset, 1993; Telmer et al., 1992). This study found a conclusion about the correlation between the length of the flower bud and the efficiency of MDE formation based on the fact that the MDE yield was highest in M-sized flower buds, which showed high contents of late uninucleate stage micro-spores. Thus, our results can be applied to the isolated microspore culture of R. sativus L.
The processes in MDE development from a microspore are shown in Fig. 7. At the time of isolated culture, the microspore was about 10.0 ± 1.0 µm in diameter in the ‘Taebaek’ and ‘Chungwoon’ cultivars. After 10 days in micro-spore isolation, some microspores had begun to break out of the microspore wall. The microspore then divided to form a primordium, and continuous cell division occurred from the primordium in both cultivars. However after that, the microspores did not develop as MDEs, and cell division did not proceed in the ‘Chungwoon’ cultivar (Fig. 7B). Furthermore, the microspore was far too small to be seen by the human eye in the ‘Chungwoon’ cultivar. On the other hand, after the occurrence of the primordium, various developmental stages beginning from the microspore can be observed in the ‘Taebaek’ cultivar (Fig. 7A). Similar observations of the microspore to embryo have been also reported for B. napus (Yeung, 2002; Yeung et al., 1996). However, anatomical observations of MDE formation process have not been reported for R. sativus L. In general, MDE formation showed differences in response according to the genotype of the donor plant (Babbar et al., 2004; Smykal, 2002). It is too early to conclude that there is a distinct inherited difference according to the efficiency of MDE formation, since microspores isolated from ‘Chungwoon’ can be divided to form a primordium. It was found that the optimal environmental conditions for microspore to undergo embryo development after primordium formation differed between cultivars. A previous study of embryogenesis in cruciferous plants suggested that even though the frequency of MDE formation is low, it can be increased by changing the culture conditions. Temperature is a significant condition for MDE formation and further development (Binarova et al., 1997). The intensity of light and the photoperiod also affect the efficiency of MDE formation (Babbar et al., 2004). Furthermore, the frequency of MDE formation could be improved by the exchange of culture medium in some instances (Touraev, 1996c). Ethylene is one of the most important factors influencing the growth and development of microspores (Biddington, 1992; Wu et al., 2006). A previous study demonstrated that ethylene production inhibited callus development and shoot regeneration in plant tissue culture (Wu et al., 2006). The ethylene accu-mulation rate may have an effect on different developmental stages of microspores (Biddington, 1992; Gallie and Young, 2004). Further research is needed to understand the influence of ethylene. Therefore, the regulation of growth and culture conditions could be used to overcome varietal differences in MDE formation rate.
In conclusion, this study provides basic information for determining the optimum stage of bud development in order to isolate microspores based on the yield of MDEs, bud structure, microspore density, and microspore developmental stage. In addition, the developmental process during MDE formation was observed in R. sativus L. This study provides the first anatomical observations of microspores isolated from R. sativus L. Therefore, the results obtained in the present study may be applicable to isolated microspore cultures double haploid production in R. sativus L.