Introduction
Materials and Methods
Ginseng Samples
Moisture Content
Acidic Polysaccharide Content
Crude Polyacetylene Content
Total Polyphenols
Extract and Sample Preparation for Ginsenoside Analysis
Reverse-phase HPLC Ginsenoside Analysis
Statistical Analysis
Results and Discussion
Moisture Contents
Ginsenosides
Acidic Polysaccharide, Crude Polyacetylene and Total Polyphenol Contents
Introduction
Korean ginseng (Panax ginseng C. A. Meyer) has been used as a medicinal plant in Korea for more than 2,000 years. Different types of ginseng are categorized based on their processing method as fresh, white, or red ginseng, among others (Kim et al., 2010). Red ginseng is manufactured by steaming and drying, which enhances its shelf life and biofunctional properties. For example, ginsenoside-Rg3, panaxytriol, and arginyl-fructsyl-glucose show an increase in content, or are generated only after processing (Nam, 2005). Ginseng has many demonstrated beneficial effects, including antiinflammatory and anticancer activity, as well as being an antioxidant. The major compounds in ginseng are ginsenosides and polyacetylenes while acidic polysaccharides, phenolic compounds, fatty acids, and peptides are minor compounds (Kiefer and Pantuso, 2003; Kim et al., 2012). The majority of research on the compounds found in ginseng aimed to identify the chemical structures of ginsenosides (Hwang et al., 2005; Kim and Kang, 2009; Son and Ryu, 2009). Ginsenosides have anticancer,antioxidant, atheroprotective, antihypertensive, antifatigue, and anti-inflammatory activity (Lee et al., 2004). Polyacetylenes, the second major group of compounds in ginseng, includes panaxynol, panaxydol, panaxydiol, and panaxytriol. Polyacetylenes inhibit cancer and platelet action and improve neurite outgrowth and scopolamine-induced memory impairment (Choi et al., 2008). Polyacetylenes are widely distributed as panaxynol in vegetables such as carrots, celery, parsnips, and parsley (Christensen, 1992; Christensen and Brandt, 2006; Huang et al., 2009; Liu et al., 2007). Panaxynol is sensitive to heat and is unstable at room temperature unless dissolved in organic solvents such as hexane or petroleum ether (Herrmann et al., 2013a). Although acidic polysaccharides represent a minor ginseng component, they have major beneficial effects, including reducing blood sugar levels, strengthening the immune system, and anticancer activities. (Chang et al., 2007). Except for starch, which accounts for 20-30% of ginseng, at least 21 polysaccharides are found in ginseng, including Panax A-U, PG1-PG5, and an anti-complementary polysaccharide. Panax A-U decreases blood sugar levels, and PG1-PG5 activate biophylaxis (Do et al., 1993). Acidic polysaccharides also have antitumor, anti-complementary, and hypoglycemic activities, and also inhibit tumor toxohormone-L lipolysis. Phenolic compounds are found in most plants and have antibiotic, cancer preventative, and spasmolytic effects. Phenols are potent antioxidants, as they contain a phenolic hydroxyl group (Jo et al., 2011). Polyphenol oxidases (PPO) are widely distributed in plants, and play a role in oxygen scavenging and stress response (Meeting et al., 1993). The antioxidant activity of phenolic compounds is largely due to their redox properties. The medicinal value of phenolics has been well documented (Cai et al., 2004).
Local ginseng landraces are cultivated all over Korea, but are not clearly distinguished from one another (Rhim et al., 2010). Novel cultivars are difficult to develop, as the breeding and selection process takes at least 30 years (Lee, 2003). Novel cultivars include Chunpoong, Yunpoong, Geumpoong, and K-1 (Kwon et al., 1998, 2000; Lee et al., 2015a), all of which need to be genetically categorized (Bang et al., 2010; Seo et al., 2003). To date, there are many comparative analyses between Korean ginseng and ginseng from other countries, but few comparative analyses exist between different Korean ginsengs. In this study, we analyzed and compared the ginsenoside, polyacetylene, acidic polysaccharide, total polyphenol contents of four local landraces from Inje, Geumsan, Jinan, and Punggi, as well as four new varieties, Chunpoong, Yunpoong, Guempoong, and K-1. By comparing the component contents of the different varieties, this study can help guide decisions in cultivation, breeding, and selection.
Materials and Methods
Ginseng Samples
Local landrace samples were purchased as fresh ginseng from 6-year-old plants in four provinces (Inje, Geumsan, Jinan, and Punggi). Four new varieties (Chunpoong, Yunpoong, Guempoong, and K-1) were obtained from Changsu-myeon, Pocheon-si (Gyeonggi-do, Korea). K-1 is a new cultivar developed by Gyeonggido Agricultural Research & Extension. Each sample was obtained from a different field in the same province. In all, 24 ginseng samples were obtained from three fresh ginseng roots per cultivar. Fresh ginseng was steamed at 95°C to produce red ginseng. The red ginseng samples were dried in a drying oven and ground into powder. Each sample was analyzed three times and the average value was used for statistical analysis.
Moisture Content
Water content was determined using a moisture analyzer (ML-50, A&D, Tokyo, Japan).
Acidic Polysaccharide Content
A carbazole-sulfuric acid method (Han et al., 1992) was used to analyze acidic polysaccharide content, and β-Dgalacturonic acid was used as the standard. Powder (1 g) from each ginseng sample was extracted by sonication in 10 mL distilled water for 3 h at 70°C and the centrifuged at 1,000 × g for 20 min. A 1-mL aliquot of the ginseng extract supernatant was washed with 4 mL 99.9% ethanol and centrifuged at 1,000 × g for 20 min. Samples were resuspended in 1 mL distilled water, and 0.5 mL resuspended ginseng extract was diluted 20-fold with distilled water. A 0.25-mL aliquot of carbazole (0.1% in 99.9% ethanol) and 3 mL sulfuric acid was added to resuspend the ginseng extract. The mixture was heated in a water bath at 85°C for 5 min, and cooled at room temperature for 15 min. Absorbance was measured at 525 nM with a spectrophotometer (Optizen 3220UV, Mecasys, Daejeon, Korea).
Crude Polyacetylene Content
Ginseng powder (5 g) was extracted by sonication in 200 mL 99.5% methanol at 70°C for 3 h and filtered with Whatman No. 4 paper. A 150-mL aliquot of petroleum ether was added to the extract, shaken for 1 h, and filtered once more with Whatman No. 4 paper. The filtrate was completely dried with a vacuum evaporator at 50°C and crude polyacetylenes were weighed.
Total Polyphenols
Total polyphenols were measured by the Folin-Denis method (Hong et al., 2011). A 100-mg portion of ginseng powder was extracted with 2 mL 99.5% methanol and sonicated at 4°C for 3 h. The extract was centrifuged at 1,000 × g for 20 min, and 0.5 mL extract was analyzed. Then, 8 mL distilled water and 0.5 mL 2 N Folin-Ciocalteu reagent were added to the extract, followed by incubation at room temperature for 3 min. 1 mL 20% Na2CO3 was added to the sample and incubated in the dark at room temperature for 1 h. Absorbance was measured at 725 nm (Optizen 3220UV, Mecasys, Daejeon, Korea). Gallic acid was used as the standard.
Extract and Sample Preparation for Ginsenoside Analysis
The Korean Food & Drug Administration 2014 health functional food method 3-55 was used to analyze ginsenosides. Ginseng powder (1 g) was extracted with 50 mL 70% aqueous methanol at 70-80°C for 1 h. The sample was cooled and centrifuged at 1,000 × g for 10 min. The supernatant was saved in a vacuum evaporator flask. The process was repeated again using the remaining residue, and the two supernatants were combined and dried in a vacuum evaporator at 60°C. The concentrate was dissolved in 5 mL HPLC-grade distilled water and loaded on a Sep-pak C18 cartridge (Waters Inc., Milford, MA, USA) which had been activated with a pre-rinse of 5 mL HPLC-grade MeOH and 5 mL HPLC-grade distilled water. Following sample loading, the cartridge was washed twice with 5 mL HPLC-grade distilled water. The sample was eluted with HPLC-grade MeOH and filtered through a 0.45-μm membrane filter for HPLC analysis.
Reverse-phase HPLC Ginsenoside Analysis
Ginsenosides were analyzed with a Shimadzu 10 Avp HPLC system (Tokyo, Japan) equipped with a UV detector (SPD-10Avp, Shimadzu, Tokyo, Japan) and gradient pump. The detection wavelength was 203 nm. A Gracesmart column (250 mm × 4.6 mm, 5 μm; Grace, Columbia, MD, USA) was used at 40°C. A 10-μL aliquot of the sample was injected, and a mixed mobile phase of distilled water (solvent A) and acetonitrile (solvent B) was used under gradient conditions. The gradient elution was: 0-10 min, 20% B; 10-42 min, 20-30% B; 42-67 min, 30-40% B; 67-70 min, 40-47% B; 70-80 min, 47-80% B; 80-93 min, 80% B; 93-95 min, 80-20% B; 95-115 min, 20% B. The flow rates of solvents A and B were 1.2 mL·min-1. Ten ginsenosides, Rg1, Re, Rf, Rb1, Rc, Rb2, Rb3, Rd, 20(S)-ginsenoside Rg3, and 20(R)-ginsenoside Rg3, were used as standards. Ginsenoside standards were purchased from Chromadex (Irvine, CA, USA).
Statistical Analysis
Data are presented as mean ± standard deviation with n = 3. A one-way ANOVA determined whether the results differed significantly. Duncan’s test was used to identify differences. The level of statistical significance was set at P < 0.05. The statistical procedures were performed using SPPS ver. 18.0 software (SPSS Inc., Chicago, IL, USA).
Results and Discussion
Moisture Contents
Moisture contents of the ginseng powder samples were 14.1-16.9% (Table 1). These values were required for calibration of the following analyses.
Table 1. Moisture content of the red ginseng powders for calibration. Results are mean ± standard deviation.
| |
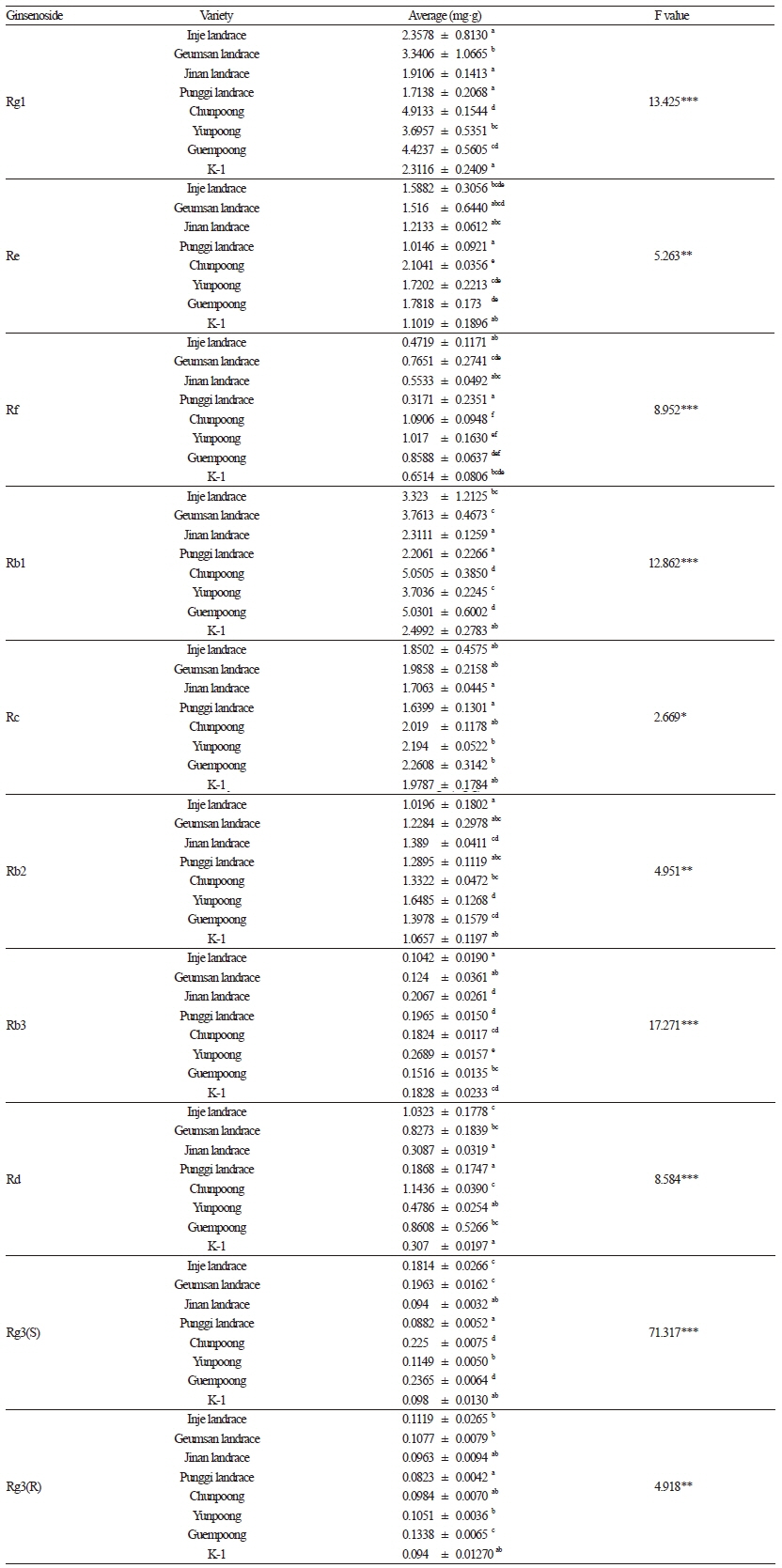
Ginsenosides
The novel varieties, including Chunpoong, Yunpoong, and Guempoong, contained more ginsenosides than did the local landraces (Inje, Geumsan, Jinan, and Punggi; Table 2). Chunpoong red ginseng contained high amounts of Rg1, Re, Rf, Rb1, and Rd, whereas Yunpoong red ginseng had high amounts of Rb2 and Rb3. Guempoong contained high amounts of the ginsenosides Rc, 20(S)-ginsenoside Rg3, and 20(R)-ginsenoside Rg3. The contents of all of the ginsenoside components were significantly different among the landraces and varieties, particularly for Rg1, Rf, Rb1, Rd, and 20(S)-ginsenoside Rg3. Table 2 shows the grouping results from Duncan’s test. The novel varieties have higher ginsenoside contents and arealso disease-resistant and high yielding (Kwon et al., 1998, 2000). In another study, Yunpoong fresh ginseng had more ginsenoside than did the other new varieties (Kim et al., 2009). Chunpoong was originally developed to produce highly valuable red ginseng (Kwon et al., 1998), and our results supported the same conclusion.
Acidic Polysaccharide, Crude Polyacetylene and Total Polyphenol Contents
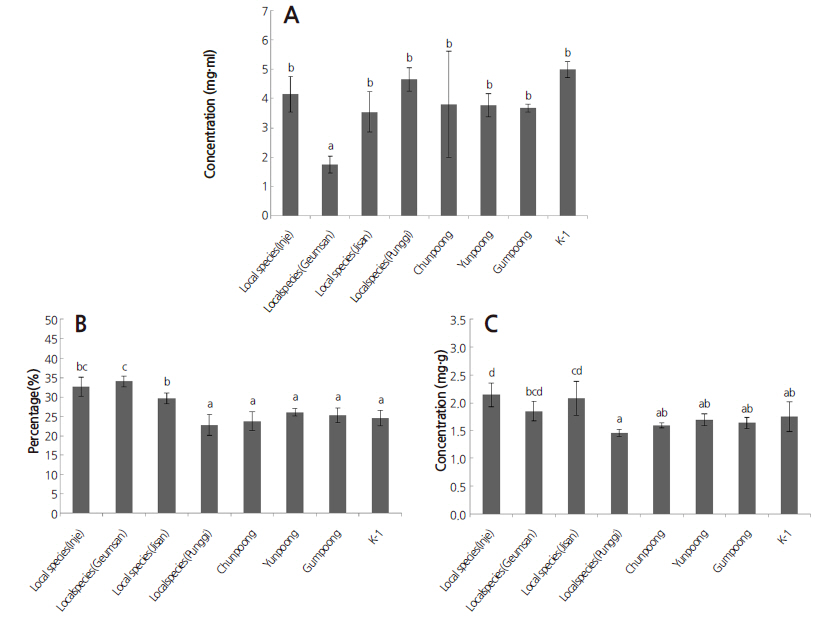
Mean acidic polysaccharide, crude polyacetylene, and total polyphenol contents are shown in Fig. 1. Acidic polysaccharide contents ranged from 4.99 mg·mL-1 in K-1 to 1.75 mg·mL-1 in the Geumsan landrace. Most samples contained similar amounts of acidic polysaccharides except the Geumsan landrace, which was significantly different from the others.
Crude polyacetylenes are easily isolated with petroleum ether because they are lipophilic (Herrmann et al., 2013b). Crudepolyacetylene contents were significantly different between the samples. The samples were divided into three groups based on the Duncan’s test results in Fig. 1. The first group was composed of the Punggi landrace, Guempoong, Yunpoong, Chunpoong, and K-1. The second group included the Geumsan landrace, for which the powder was 33.99% crude polyacetylenes and the Inje landrace (32.67%). The third group contained the Inje landrace as well as the Jinan landrace (29.65%). Crude polyacetylene content ranged from 33.99% in the Geumsan landrace to 22.78% in the Punggi landrace.
Total polyphenol content divided the ginsengs into four groups, and some samples could be placed in more than one group. The Inje landrace contained the highest total polyphenol content of 2.14 mg·mL-1, and was not significantly different from the Jinan landrace (2.08 mg·mL-1) or the Geumsan landrace (1.85 mg·mL-1). The Punggi landrace contained 1.46 mg·mL-1 total polyphenols and was not significantly different from Guempoong, Yunpoong, Chunpoong, and K-1.
We analyzed four local landraces (Inje, Geumsan, Jinan, and Punggi) and four new varieties of gingseng (Chunpoong, Yunpoong, Guempoong, and K-1) for active compounds. Ten types of ginsenosides (Rg1, Re, Rf, Rb1, Rc, Rb2, Rb3, Rd, 20(S)-ginsenoside Rg3, and 20(R)-ginsenoside Rg3), acidic polysaccharides, crude polyacetylenes, and total polyphenols were analyzed. The differences we observed may derive from environmental factors such as soil, temperature, and rainfall as well as genetic differences between the cultivars.
To date, there have been many publications describing the active components of different ginsengs (Ahn et al., 2008; Lee et al., 2015b), but there is a lack of research on differences in ginsenoside and non-saponin component contents of red ginseng between landraces and new varieties. This study will prove useful for cultivating, breeding, and selecting improved ginseng varieties based on their active components.