Introduction
Materials and Methods
Experimental Site and Treatment Details
Plant Materials and Growth Conditions
Plant Sampling and Nutrient Analysis
Soil Enzymatic Activity
Rhizosphere Soil Sample Collection and Analysis of Soil Chemical Properties
Statistical Analysis
Results
Discussion
Conclusion
Introduction
Utilization of chemical fertilizers has increased worldwide due to its availability and accessibility (Graham and Vance, 2000). Intensive agricultural systems using chemical fertilizers have degraded soils by decreasing soil organic matter and reducing soil fertility (Montemurro et al., 2015). Increasing crop productivity has been the main objective in most agricultural production systems; however, other production goals, such as product quality, cost effectiveness, and environmentally friendly approaches, also need to be considered in crop production (del Amor, 2007). The application
of organic fertilizers could improve soil quality and crop yield, and is an environmentally friendly approach to reduce the use of chemical fertilizers.
Green manure is an environmental friendly organic fertilizer that has the potential to reduce the dependence on mineral fertilizers and to maintain soil organic matter content (Elfstrand et al., 2007). Leguminous green manures, such as crimson clover (Trifolium incarnatum) and residue from a hairy vetch (Vicia villosa L.) cover crop, are important nitrogen (N) sources in organic crop production (Drinkwater et al., 1998; Muchanga et al., 2017) as legumes form root nodules with symbiotic N-fixing bacteria. Hence, legume-based green manures supply N to soil. Applications of compost have long been recognized for improving soil physical properties, structure, and water holding capacity (Lavelle, 1988), and reducing the dependency on inorganic fertilizers (Maynard and Hill, 1994; Moon et al., 2018).
Soil microbes influence soil organic matter decomposition and soil enzyme activities (Nannipieri et al., 1990). Soil enzyme activities are the primary mediators of soil biological processes, including organic matter degradation, mineralization, and nutrient recycling. Decomposed organic matter serves as a nutrient source for both plants and soil microbes. Soil enzyme activities are indicators of soil quality because they are strongly connected with important soil properties, such as organic matter, physical properties, and microbial activity or biomass (Bowles et al., 2014).
Global consumption of nutrients for crop production in 2014 accounted for roughly 108 million tons of N, 47 million tons of phosphorous (P), and 38 million tons of potassium (K) (Food and Agriculture Organization Corporate Statistical Database). However, the recovery efficiency of applied fertilizers is low. For instance, Haefele et al. (2003) reported that the recovery efficiency of nutrients after rice (Oryza sativa) cultivation were 31 to 50% (the average for all sites was 36%) for N, 26 to 43% for P, and 53 to 65% for K. Large amounts of N are deposited in agricultural lands every year. The over application of N in agricultural lands leads to increased nitrate levels in ground water (Li and Yost, 2000). Optimal N fertilization management is crucial not only for improving crop production but also to protect the environment (Mosier, 2002). The objectives of this study were to assess the effect of organic and inorganic N management on red pepper (Capsicum annuum) growth, yield, soil chemical properties, and soil enzyme activities.
Materials and Methods
Experimental Site and Treatment Details
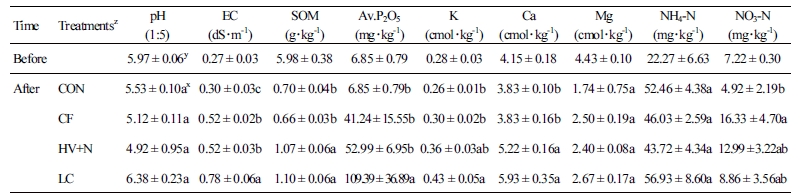
The experimental plots (4 m × 3 m) were constructed at the National Institute of Horticulture and Herbal Science research fields, located in Wanju, Korea. The plots were separated by a concrete wall about one meter deep to avoid mobilization of nutrients between the plots. The entire experimental site was covered with a polyvinyl house to exclude precipitation from the plots. The initial chemical properties of the soil used in the experimental plots, prior to the start of the experiments, are given in Table 1.
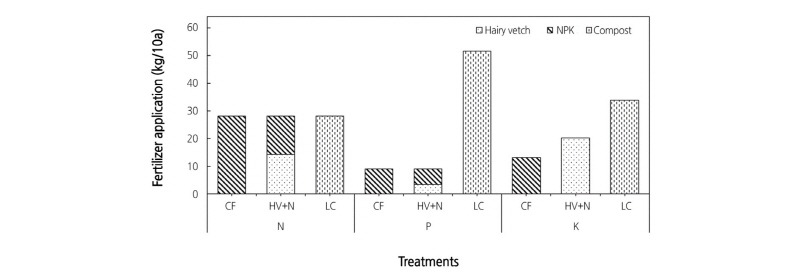
Four treatments were applied in the experimental plots in a randomized complete block design with three replications per treatment. The treatments were as follows: no fertilizer (CON), chemical fertilizer (CF), hairy vetch (Vicia villosa L.) residue and chemical fertilizer (HV+N), and livestock compost (LC). The CF treatment plots received synthetic fertilizer based on the nutrient requirements recommended by the Rural Development Administration of Korea for greenhouse red pepper production. The amount of nitrogen (N), phosphorous (P), and potassium (K) applied for the CF treatment was 28, 9, and 13 kg/10a, respectively. For the HV treatment, seeds of hairy vetch legumes were sown and allowed to grow from November until April. Hairy vetch plants were harvested and ploughed in the same plots after the nutrient contents were analyzed. In addition to HV, synthetic N and P were applied to meet the required amount for red pepper growth. Livestock compost consisted of cow manure (40%), swine manure (10%), chicken manure (5%), sawdust (40%), bark (3%), and rice (O. sativa) hull (2%). The amount of organic fertilizer applied corresponded to the N content of the chemical fertilizer. The amount of N, P, and K for each fertilizer treatment applied for 10a is given in Fig. 1.
Plant Materials and Growth Conditions
Approximately 70-day-old red pepper seedlings (Capsicum annuum L.) were purchased from Gyeonggi seedling company, South Korea. In each plot, the red pepper seedlings were transplanted in three rows with 10 plants per row. The distance between the rows was 100 cm and the distance between two plants was 40 cm. The plots were equipped with a drip irrigation system and the soil moisture content of the plots was maintained at field capacity. The plants were maintained for another 170 days in the experimental plots.
Plant Sampling and Nutrient Analysis
After 170 days, one red pepper plant from each plot was randomly selected and harvested. Leaf, fruit, stem, and root samples were collected separately. The collected samples were dried in an oven at 70°C for three days and dry weights were measured. Plant tissue macro and micro nutrient contents were measured using standard laboratory protocols. The total N content in the plant tissues was measured using a Kjeldahl analyzer (Kjeltec 8400, Foss, Sweden). Plant P content was measured by the ammonium metavanadate method. Plant K content was measured using inductively coupled plasma optical emission spectrometry (ICP-OES) (SDS-720, GBC, Australia). The plant nutrient uptake rate was determined using the equation:
Soil Enzymatic Activity
Three rhizosphere soil samples were collected from each plot 100 days after transplanting and pooled together as one replication. The collected soil samples were transported immediately to the laboratory and soil enzymatic activity assays were performed within a week. Soil dehydrogenase activity was determined following the method developed by Casida et al. (1964) using triphenyltetrazolium chloride dye. Soil β-glucosidase and phosphatase activities were determined colorimetrically as the concentration of p-nitrophenol produced after the incubation of 1 g of soil (37°C for 1 h) in 0.25 mL toluene, 4 mL of modified universal buffer (MUB; pH 6.5), and 0.025 M p-nitrophenyl-β-D-glucopyranoside or 0.115 M disodium p-nitrophenyl phosphate, respectively (Tabatabai and Bremner, 1969; Eivazi and Tabatabai, 1988). Soil urease activity was quantified colorimetrically as the NH4+ produced after incubating 5 g of soil in 2.5 mL of 0.08 M urea solution and 50 mL of 1 M potassium chloride solution at 37°C for 2 h (Kandeler and Gerber, 1988).
Rhizosphere Soil Sample Collection and Analysis of Soil Chemical Properties
Five rhizosphere soils samples (within a 10 cm radius of the roots and a 15 cm depth; approximately 100 g) were collected from each plot, pooled together, mixed, and dried after the red peppers were harvested. The dried soil samples were used for the analysis of soil chemical properties. Soil pH, electrical conductivity (EC), soil organic matter content, and available P (Av. P2O5) content were analyzed using standard laboratory protocols. Soil nitrate and ammonia contents were analyzed using a Kjeldahl analyzer. The concentrations of other nutrients, such as K, Ca, and Mg, were measured through ICP-OES.
Statistical Analysis
Data were subjected to analysis of variance (ANOVA) and significant differences of the means were compared by Duncan’s multiple range test (DMRT) at p ≤ 0.05. All data were analyzed using SAS package (verson 9.2, SAS Institute, Cary, NC, USA).
Results
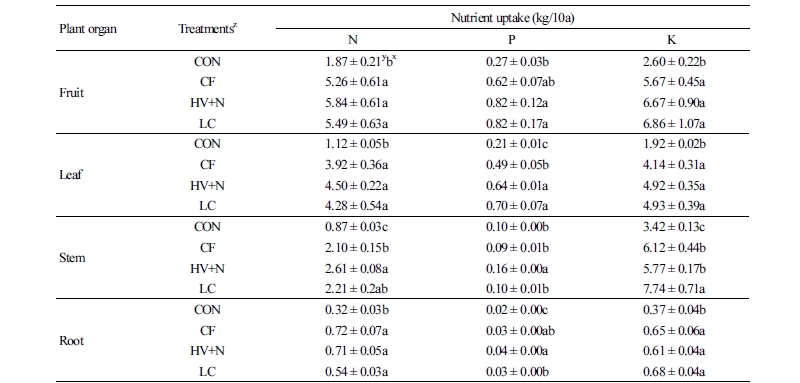
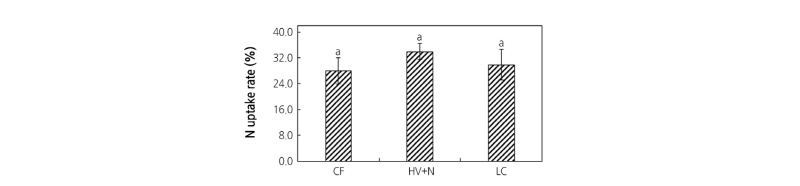
All fertilizer treatments increased plant dry matter content compared to the control. Significantly higher red pepper leaf, stem, root, and fruit dry weights were observed in all fertilizer treatments (Fig. 2) compared to the control; however, no differences were observed between the organic and chemical fertilizer treatments. Significantly higher nutrient contents of the fruit, leaf, stem, and root tissues were observed for both organic and inorganic fertilizer treatments compared to control plants (Table 2). In whole plant analyses, all fertilizer treatments showed almost two-fold higher N, P, and K uptake than in the control (Fig. 3); however, no differences were observed between the organic and chemical fertilizer treatments. Even though different sources of N were applied, it did not influence the N uptake rate by the plants (Fig. 4).
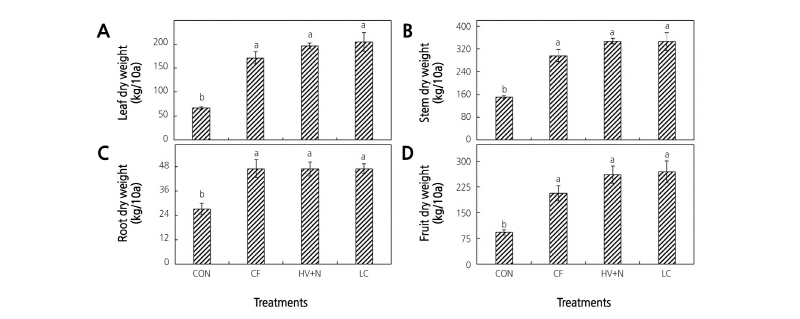
Fig. 2. Plant growth and yield response to organic and inorganic fertilizer application. (A) leaf dry weight, (B) stem dry weight, (C) root dry weight, and (D) fruit dry weight. Bars with same letters are not statistically different according to DMRT test (p ≤ 0.05). CON: control, CF: chemical fertilizer, HV+N: hairy vetch + CF, LC: livestock compost.
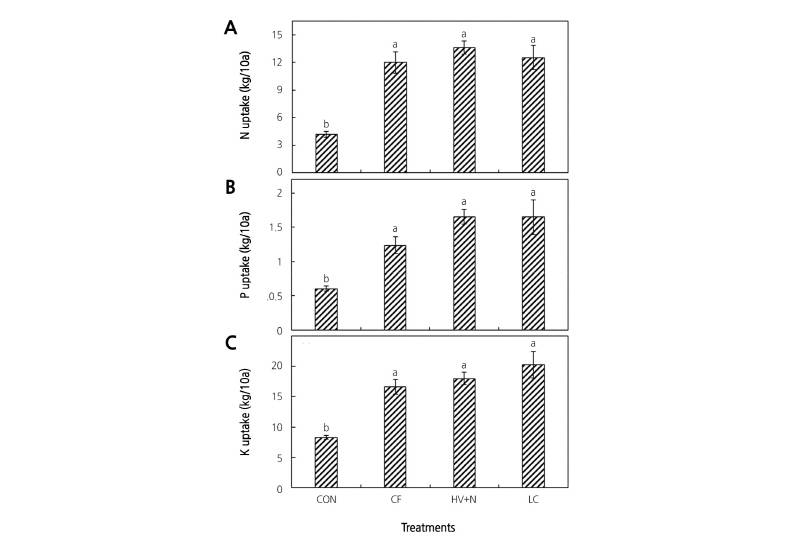
Fig. 3. Macronutrient uptake of red pepper plants under organic and inorganic fertilizer application. (A) nitrogen uptake, (B) phosphorous uptake, and (C) potassium uptake. Bars with the same letters are not statistically different according to DMRT test (p ≤ 0.05). CON: control, CF: chemical fertilizer, HV+N: hairy vetch + CF, LC: livestock compost.
Analysis of post experiment soil properties showed that the fertilizer treatments significantly influenced soil chemical properties (Table 1). Compost-applied plots had significantly higher soil EC than the CF- and HV+N-treated plots. Plots treated with organic fertilizer had significantly higher soil organic matter content than plots treated with chemical fertilizer alone. LC-treated soils had significantly higher available P than the HC+N- and CF-treated soils. Soil K and Ca contents were also significantly higher in the LC treatment, followed by the HV+N treatment. No significant differences were observed in ammonium content between the treatments. Significantly higher nitrate content was observed in the CF treatment than in the LC treatment.
Fertilizer applications significantly influenced soil enzyme activities. Significantly higher dehydrogenase activity was observed in LC-treated soils followed by HV+N-treated soils. CF-treated plots and control plots had similar dehydrogenase activities (Fig. 5A). Significantly higher urease activity was observed in the CF and HV+N treatments compared to the control (Fig. 5B). Only the HV+N treatment showed significantly higher β-glucosidase activity than the control (Fig. 5C). No significant differences were observed in phosphatase activity between the treatments (Fig. 5D).
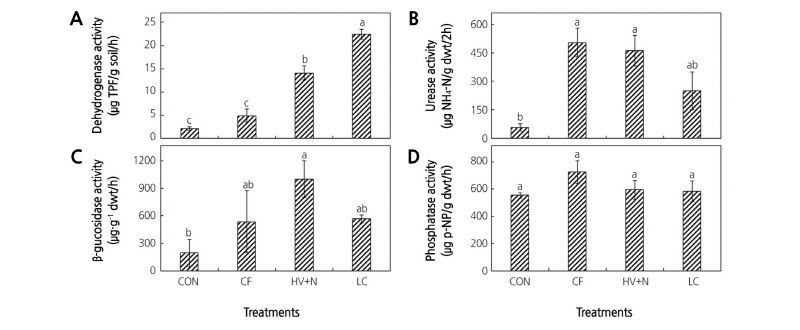
Fig. 5. Soil enzyme activities during the growth of red pepper plants under different fertilizer treatments. (A) dehydrogenase activity, (B) urease activity, (C) β-glucosidase activity, and (D) phosphatase activity. Bars with same letters are not statistically different according to DMRT test (p ≤ 0.05). CON: control, CF: chemical fertilizer, HV+N: hairy vetch + CF, LC: livestock compost.
Discussion
Adequate nutrient availability is important for crop growth and yield. Normally, crop yield is closely associated with N supply when no other nutrients are deficient in the soil (Keeling et al., 2003). Conventional CF, HV+N, and LC applications resulted in a significant increase in red pepper plant biomass and fruit yield. Combining hairy vetch and CF (the HV+N treatment) resulted in red pepper growth and yield similar to the CF or LC treatments. A previous study by del Amor (2007) also reported that conventional fertilizer, organic fertilizer, and the integration of both had a similar effect on sweet pepper yield and fruit quality. Although all fertilizer treatments improved red pepper growth and yield, the quality of the fruits is the most important measure to determine the effect of the fertilizer applications.
Previous reports found that in pepper and tomato (Solanum lycopersicum), the N concentration of fruits were significantly higher with chemical fertilization than with organic fertilization (Clark et al., 1999; Colla et al., 2002; Herencia et al., 2007). This is due to the fact that conventional chemical fertilizers supply N in a form that is immediately available to plants; whereas in organic fertilizers, the N supply is dependent on the decomposition rate of the organic matter (Lopez et al., 2013). Some researchers have confirmed that nitrate levels in vegetable crops are higher when N is supplied in an inorganic form than in an organic form (Bourn and Prescott, 2002; Williams, 2002). However, our results showed that the CF, LC, and HV+LC treatments resulted in a similar N content in all parts of the plant, including fruit, regardless of the N source. The N uptake rate by red pepper plants also showed no differences despite the differences in the N source. Our results confirm the findings by Ortega et al. (2016) that organic and conventional chemical fertilizers have similar effects on pepper growth.
Although compost-applied soils received over a two-fold higher P application than the CF or HV+N treatments, no differences in plant P uptake were observed between the fertilizer treatments. This result suggests that over application of nutrients doesn’t necessarily increase plant nutrient uptake. Chang et al. (2007) reported that applying compost in amounts greater than the required rate did not increase vegetable yield. A similar study by Hernandez et al. (2016) reported that compost application did not affect lettuce (Lactuca sativa) growth during the first crop. Our results also showed no considerable differences in red pepper yield between chemical fertilizer and compost applications after the first cropping. Similarly, despite the different amount of K applied in each treatment, no differences were observed in K uptake between the fertilizer treatments.
The fertilizer applications influenced soil chemical properties. Although all the treatments received the same amount of N, the nitrate content was higher in the chemical fertilizer-applied soil than in the organic fertilizer-applied soils. This result confirms the findings that chemical fertilizer increased the nitrate content in the soil (Hepperly et al., 2007; Bar-Yosef, 2017). The HV+N application increased the soil organic matter content more than the CF application alone. Previous studies (Ouedraogo et al., 2001; Birkhofer et al., 2008; Selvakumar et al., 2018) and our results confirmed that compost application increases the soil organic matter content. Although P and K contents were higher in the compost than in the CF, the P and K contents of the plants were similar in both treatments, and the compost application resulted in increased soil-available P and K after red pepper harvest.
Dehydrogenases play an important role in the biological oxidation of soil organic matter. The dehydrogenase activity results of this study suggest that the compost application significantly increased the microbial activity in the soil compared to the CF application (Hernandez et al., 2016). The activities of other hydrolases involved in the cycles of important plant nutrients, such as urease (N cycle), β-glucosidase (C cycle), and phosphatase (P cycle) (Tabatabai, 1994), are used as indicators for microbial-mediated soil functions (Sinsabaugh, 1994). Higher urease activities measured in the CF- and HV+N-applied soils suggest that increased soil urease activity is related to the application of mineral N as urea. Green manure increases soil enzyme activities by stimulating the soil microbial biomass (Piotrowska and Wilczewski, 2012). The hairy vetch residue application increased β-glucosidase activity in the soil, which suggests that hairy vetch provides specific substrates for this enzyme or stimulates its synthesis. A previous study by Stott et al. (2010) found a positive correlation between β-glucosidase activity and degradation of plant material. Degrading plant material releases simple sugars for use by microbial populations, thus increasing microbial activity. Despite the higher P content in compost, no differences in phosphatase activity were observed in this study. In a similar study by Hernandez et al. (2016), they reported that compost application did not influence phosphatase activity during the first crop; however, higher activity was observed during the second crop. Our results and the findings of Hernandez et al. (2016) suggest that the decomposition of compost increases over time, thereby increasing the release of nutrients over time.
Conclusion
Our results demonstrate that hairy vetch residue with addition of a chemical fertilizer, and the compost application, improved plant growth and yield similar to that observed from a chemical fertilizer application. Unlike chemical fertilizer, the hairy vetch residue and compost application improved soil chemical properties by increasing the soil organic matter and available P. In addition, organic fertilizers, such as hairy vetch residue and compost, increased soil biological activities, especially dehydrogenase and beta-glucosidase. These results suggest that organic fertilizers can be as effective at improving pepper plant growth, and yield as chemical fertilizers. These findings also suggest that hairy vetch residue and compost can be used as an alternative to chemical fertilizers to reduce the use of chemical fertilizer without losses to crop yield.