Introduction
Materials and Methods
Plant materials, growth conditions, and mechanical stress treatments
Analysis of anthocyanin and total chlorophyll concentrations
DAPI staining and analysis of electrolyte leakages
Observation of leaf stomatal opening and closing
Statistical analysis
Results and Discussion
Pipe-induced brushing similarly triggers stomatal closure and causes less cell damage in tomato leaves compared to conventional mechanical stress treatments
Pipe-induced brushing is effective for the large-scale application of mechanical stress in tomato plants without causing severe damage
Pipe-induced brushing reduces plant growth and enhances anthocyanin accumulation with reversible damage
Pipe-induced brushing regulates mechanical stress responses across various tomato cultivars
Introduction
Applying mechanical stress to various plant species regulates the growth, development, and the synthesis of secondary metabolites, enhancing tolerance and resistance to abiotic and biotic forms of stress. For instance, when barley plants are stamped, their growth and productivity are improved (Ohtani 1950). Similarly, when tomato plants are subjected to mechanical stress treatments using clip-on stems, the plants exhibit early flowering and enhanced productivity (Castro-Estrada et al. 2024). Additionally, tomato plants subjected to rubbing treatments show enhanced accumulation of secondary metabolites such as lignin (Sabina and Jithesh 2020) and anthocyanin (Yoon et al. 2024a). Mechanical stress has also been utilized to retard the growth of tomato (Sabina and Jithesh 2020; Yoon et al. 2024a) and watermelon plants (Jeong et al. 2023). These findings suggest that mild mechanical stress plays a crucial role in plant development and in the regulation of secondary metabolism.
However, conventional methods, including rubbing or brushing, are laborious and time-consuming when applied to a large number of plants. These methods can also cause undesired tissue damage and reduced stem diameters (Yoon et al. 2024a, 2024b). Previous studies have demonstrated that pipe-induced brushing effectively activates mechanical stress responses in a large number of tomato transplants (Latimer and Thomas 1991; Baden and Latimer 1992). These earlier studies mainly focused on tomato plant growth parameters, such as the stem length and plant weight. However, they were limited in their assessment of cell damage and anthocyanin accumulation, which can also affect plant growth.
Anthocyanins are water-soluble plant pigments that are critical in regulating mechanical stress tolerance across various species, including tomato plants (Solanum lycopersicum L.). Anthocyanin accumulation in plant tissues is often triggered by environmental factors such as high light conditions (Zhang et al. 2019), temperature changes (Chalker-Scott 1999; Kiferle et al. 2015; Qiu et al. 2016), and mechanical stress (Yoon et al. 2024a). These pigments protect plant tissues by scavenging reactive oxygen species, such as hydrogen peroxide (H2O2) and superoxide (Gould et al. 2002; Barba-Espín et al. 2021), and by reducing insect herbivory (Chen et al. 2021). Anthocyanin biosynthetic genes are regulated by several transcription factors, including, SlTT8 (a tomato bHLH transcription factor) and SlAN2 (a tomato R2R3-MYB transcription factor) (Zhang et al. 2019; Yoon et al. 2024a). A previous study by the authors revealed that applying mechanical stress to tomato plants can induce the expression of SlTT8 and SlAN2, resulting in the upregulation of anthocyanin biosynthetic genes (Yoon et al. 2024a). These findings suggest that anthocyanin accumulation serves as an indicator of the mechanical stress response. We hypothesize that pipe-induced brushing can minimize cell damage and induce anthocyanin accumulation in plants. However, research thus far has not tested this.
Here, we assess pipe-induced brushing for its effects on tomato plants. Our findings show that this method can be effectively and simultaneously applied to tomato transplants, reducing plant heights and causing the accumulation of anthocyanin. This method can also minimize tissue damage compared to that which arises when using conventional methods.
Materials and Methods
Plant materials, growth conditions, and mechanical stress treatments
To investigate the responses of tomato plants to mechanical stress, four different cultivars were used: “Red Cherry” (obtained from Back to the Roots, USA), “Roma VF” (obtained from Burpee, USA), “Super Sweet 100 Hybrid” (obtained from Burpee, USA), and “Red Currant” (obtained from Burpee, USA). Seeds were germinated and grown in soil in pots (10 cm × 10 cm) at 25°C under standard long-day greenhouse conditions (a 16-h light/8-h dark photoperiod).
Two-week-old plants (height of approximately 7–10 cm) were subjected to various mechanical stress treatments at 10:00 a.m. daily for five consecutive days, and these plants were used for further analysis. The planting distance between the plants was approximately 13 cm so as not to disturb the brushing of neighboring plants. The pipe-induced brushing treatment was performed manually. The brushing speed was approximately 3 sec per tray (60 cm). One cycle consisted of a single back-and-forth movement, as previously described (Fig. 1A; Latimer and Thomas 1991).
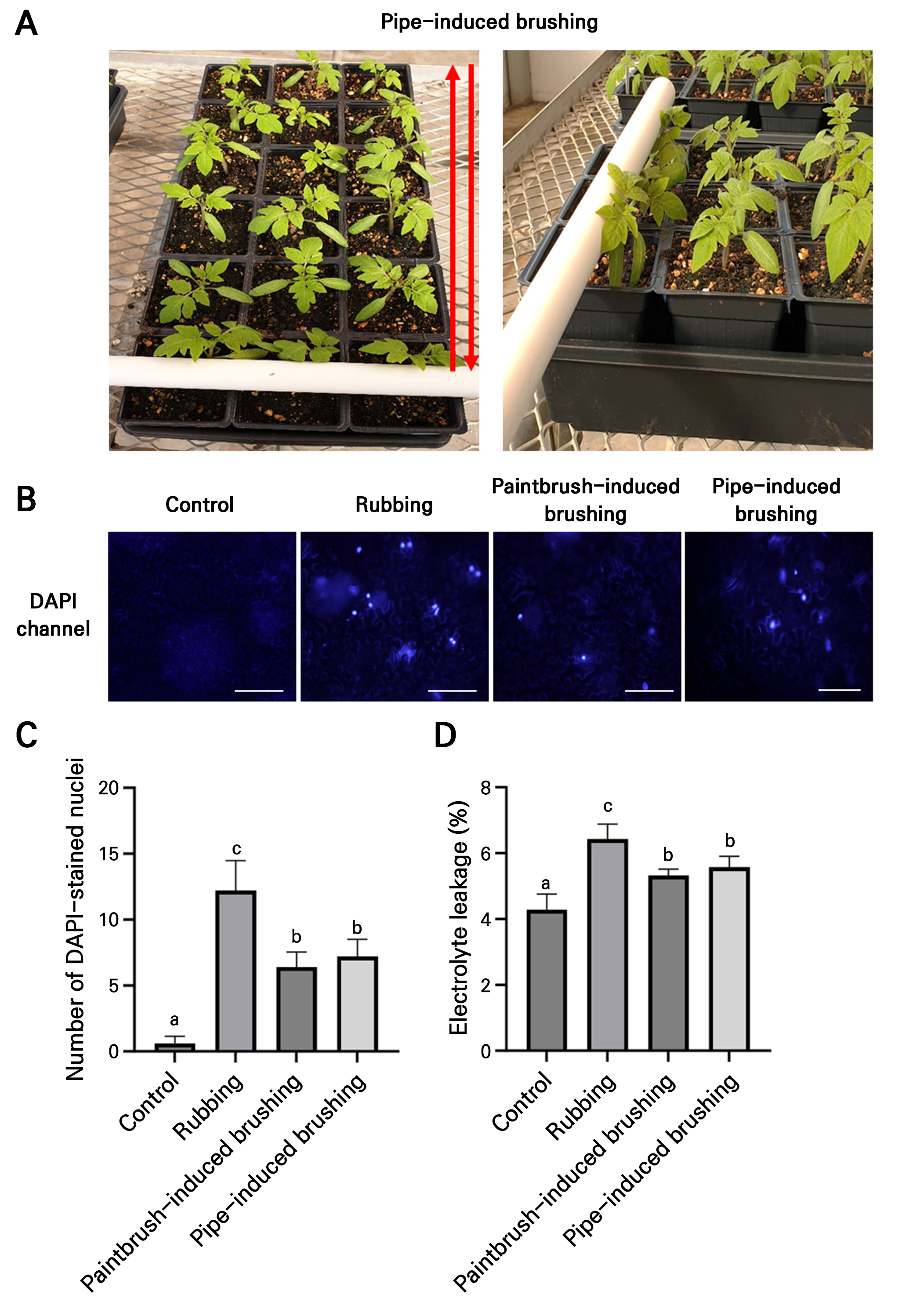
Fig. 1.
Comparison of cell damage resulting from different mechanical stress treatments. (A) Pipe-induced brushing treatment applied to “Red Cherry” tomato transplants. Red arrows indicate one cycle (a single back-and-forth). (B) DAPI staining of leaves from two-week-old “Red Cherry” tomato plants after different mechanical stress treatments. Leaves were stained with DAPI 1 min after the treatment. (C) Quantification of DAPI-stained nuclei in treated leaves. Data are presented as the means ± SD (n = 5). (D) Electrolyte leakage from the same samples. Data are presented as the means ± SD (n = 4). The statistical analysis was performed using a one-way ANOVA followed by Tukey’s test. Different letters indicate significant differences (p < 0.05) among the samples.
To examine the effects of the number of cycles of the pipe-induced brushing treatment, five, ten, and fifteen cycles were used. A randomized block design (RBD) was used for this study, with 144 “Red Cherry” plants grown in 3×6 pots per tray (i.e., a total of eight trays), with growing lasting for two weeks after germination. Each mechanical stress treatment (i.e., 0, 5, 10, and 15 cycles) was applied to two trays, resulting in 36 plants treated for each brushing cycle. These plants were grown for an additional seven days without any stress after the final brushing treatment and were then photographed. Untreated plants were also grown under identical conditions and were used as controls. Nine plants were used to measure the growth parameters of the height and stem thickness on the final day.
To compare the cell damage caused by our pipe-induced brushing and the other conventional methods tested here, we employed rubbing and brushing using a paintbrush as conventional methods. For this test, 15 “Red Cherry” tomato plants were grown, and five plants were subjected to each method. For the rubbing treatment, two-week-old plants were gently rubbed for 5 sec as previously described (Benikhlef et al. 2013; Yoon et al. 2024a). For the mechanical stress using a paintbrush, the same plants were gently brushed for 5 sec, as previously described (Verhertbruggen et al. 2013; Yoon et al. 2024b). The largest true leaves from the first branch were sampled by a hole punch and used for DAPI staining and for the observation of leaf stomatal conditions (see the “DAPI Staining and Analysis of Electrolyte Leakages” and “Observation of Leaf Stomatal Opening and Closing” subsections).
Analysis of anthocyanin and total chlorophyll concentrations
Anthocyanin concentrations were quantified as previously described (Michael and Chory 1998). Leaves were sampled on the final day. In brief, 0.1 g of leaf tissue was ground in liquid nitrogen. The ground tissue was incubated overnight at 4°C in 300 µL of methanol containing 1% HCl to extract anthocyanins under dark conditions. After incubation, 200 µL of water and 500 µL of chloroform were added to the extract, after which the mixture was vortexed to ensure homogeneity. The samples were then centrifuged at 15,000 × g for 5 min to separate the phases. A total amount of 400 µL of supernatant was transferred to a new tube and diluted with an equal volume (400 µL) of 60% methanol containing 1% HCl. The absorbance of the final extract was measured at 530 and 657 nm using a spectrophotometer (GENESYS20; Thermo Scientific, Waltham, MA, USA). Anthocyanin concentrations were calculated using the equation (A530 − A657) × 1,000 / fresh weight (FW).
Total chlorophyll concentrations were quantified as previously described (Porra et al. 1989). Leaf samples were prepared as described above, submerged in 800 µL of N,N-dimethylformamide and finally incubated in the dark at 4°C. The absorbance of the final extract was measured at 647 and 664 nm using a spectrophotometer (GENESYS20; Thermo Scientific, Waltham, MA, USA). Total chlorophyll concentrations were then calculated using the equation (17.67 × A647) + (7.17 × A664) × FW-1. Three true leaves from the first branch of three different tomato plants were analyzed to assess the anthocyanin and total chlorophyll contents.
DAPI staining and analysis of electrolyte leakages
4',6-diamidino-2-phenylindole (DAPI), which penetrates damaged cells and stains the DNA, was used to examine cell damage after the treatment. Phosphate-buffered saline (PBS; pH 7.4) containing 1 µg/mL DAPI was prepared for the DAPI staining assays (Yoon et al. 2024b). The samples were incubated for 20 min at room temperature and then washed with PBS. DAPI signals were detected using a Leica DM5500B fluorescence microscope equipped with a Leica DFC540 C camera. Five true leaves from the first branch of five different tomato plants were analyzed, and the numbers of DAPI-stained nuclei in the treated areas were counted.
For the electrolyte leakage analysis, 50 mg of leaf tissue was submerged in 10 mL of distilled water and incubated for 2 h at room temperature. The electrical conductivity (EC) was measured using an EC meter before and after autoclaving for 15 min at 121°C, obtaining EC-I and EC-II. The formula (EC-I/EC-II) × 100 was used to calculate the electrolyte leakage level (Yoon et al. 2024b). Four true leaves from the first branches of four different tomato plants were used.
Observation of leaf stomatal opening and closing
To observe the stomatal conditions, the leaf epidermis was sectioned and placed on a slide. Images of each section were captured using a Leica DM5500B fluorescence microscope equipped with a Leica DFC540 C camera. Stomata were observed using a 20× objective lens (Lu et al. 2017). To examine stomatal opening and closing, the pore diameters of untreated and treated leaves were measured using the ImageJ program (https://imagej.net/ij/). At least five true leaves were observed, and the pore diameters of ten stomatal cells were measured.
Statistical analysis
GraphPad Prism 10 was used to perform the statistical tests. A one-way ANOVA followed by Tukey’s test and a two-way ANOVA followed by Sidak’s test were used for the statistical analysis. The number of replicates and the statistical analysis methods are described in the figure legends.
Results and Discussion
Pipe-induced brushing similarly triggers stomatal closure and causes less cell damage in tomato leaves compared to conventional mechanical stress treatments
To apply pipe-induced brushing, two-week-old “Red Cherry” tomato transplants were selected. Plants at this stage were not large enough to reach neighboring plants, and brushing could be effectively applied between the plants (Fig. 1A). Plants older than this stage start producing secondary branches, which can reach neighboring plants and possibly disturb any brushing treatment.
To compare cell damage between conventional methods and pipe-induced brushing, rubbing and paintbrush-induced brushing were employed as conventional methods. Five cycles were used for pipe-induced brushing in this experiment. The number of DAPI-stained nuclei was counted in each case, and electrolyte leakage in the leaf samples was measured 1 min after the treatment. The results showed that the rubbed leaves exhibited more DAPI-stained nuclei (Fig. 1B and 1C) and higher levels of electrolyte leakage (Fig. 1D) than those subjected to paintbrush or pipe brushing.
Stomatal opening and closing are crucial for environmental adaptation and a key indicator of plant stress responses (Qi et al. 2018; Samakovli et al. 2020; Zhao et al. 2021). Tomato plants were subjected to the same mechanical stress treatments, and leaves were observed 1 min after the treatment. All three treatments could rapidly induce stomatal closure (Fig. 2A and 2B). These findings suggest that pipe-induced brushing effectively triggers stomatal closure with less leaf epidermal damage compared to other conventional methods.
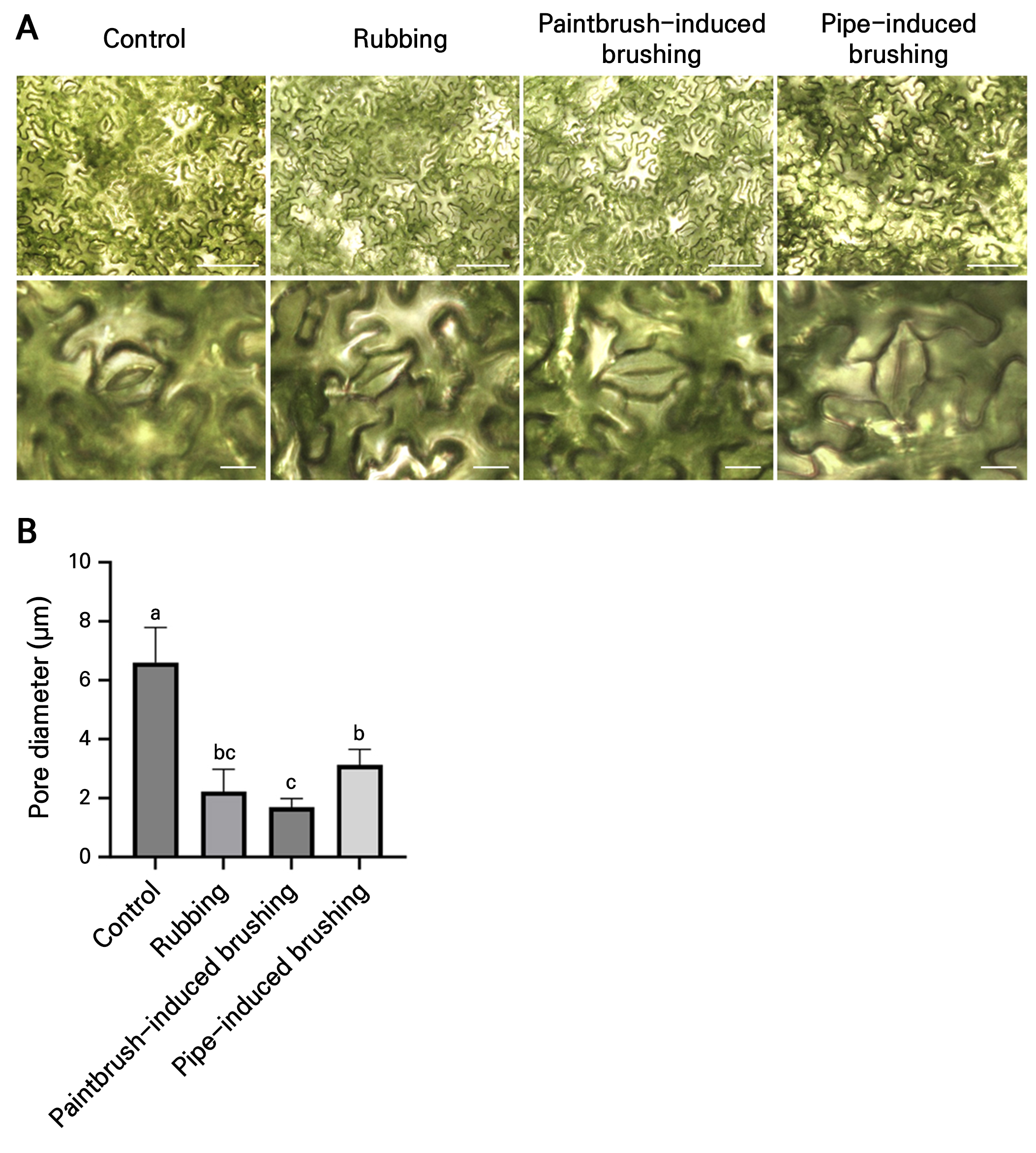
Fig. 2.
Effects of various mechanical stress treatments on stomatal closure. (A) Stomatal opening and closing observed 1 min after the mechanical stress treatments. Stomata were examined under a 20× objective lens (upper row). The enlarged images in the lower row show representative stomata. Scale bars: 100 µm (upper row), 10 µm (lower row). (B) Pore diameter measurements for untreated and treated leaves. Data are presented as the means ± SD (n = 10). The statistical analysis also involved a one-way ANOVA followed by Tukey’s test. Different letters indicate significant differences (p < 0.05) among the samples.
Pipe-induced brushing is effective for the large-scale application of mechanical stress in tomato plants without causing severe damage
Different numbers of cycles of pipe-induced brushing (i.e., 0, 5, 10, and 15 cycles) were tested on tomato transplants to determine a treatment that causes minimal cell damage. DAPI staining showed that the plants subjected to five and ten cycles had fewer stained nuclei 1 min after the treatment when compared with those treated with fifteen cycles (Fig. 3A and 3B). Electrolyte leakage assays were conducted under identical conditions to examine cell damage. The plants treated with five and ten cycles exhibited lower levels of electrolyte leakage than those treated with fifteen cycles (Fig. 3C). These results suggest that pipe-induced brushing induces some tissue damage at all tested cycle numbers, but treatment with fewer than ten cycles minimizes excessive cell damage.
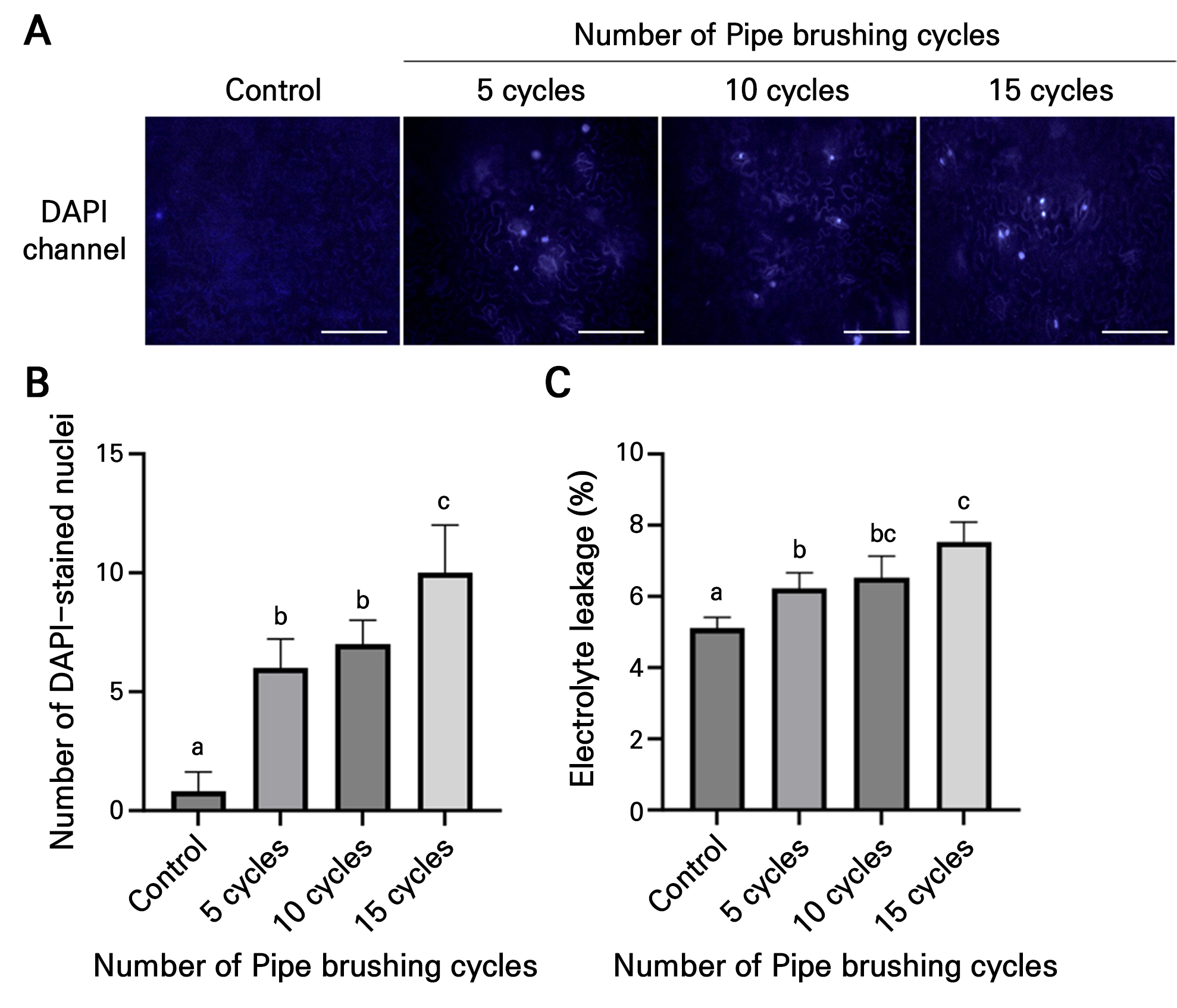
Fig. 3.
Pipe-induced brushing rapidly damages tomato leaf tissues. (A) DAPI staining of leaves from two-week-old “Red Cherry” tomato transplants after brushing. (B) Quantification of DAPI-stained nuclei in treated leaves. One cycle of pipe-induced brushing means a single back-and-forth movement across the first true leaves of tomato transplants. Data are presented as the means ± SD (n = 5). (C) Electrolyte leakage from the same tomato transplants. Data are presented as the means ± SD (n = 4). The statistical analysis was performed using a one-way ANOVA followed by Tukey’s test. Different letters indicate significant differences (p < 0.05) among the samples.
Pipe-induced brushing reduces plant growth and enhances anthocyanin accumulation with reversible damage
Our previous study revealed that rubbing treatments reduced plant height and stem diameter outcomes but induced anthocyanin accumulation in tomato plants (Yoon et al. 2024a). To determine whether pipe-induced brushing triggers similar physiological responses, “Red Cherry” tomato transplants were subjected to zero, five, ten, and fifteen cycles for five days and their growth parameters and anthocyanin concentrations were measured. At all cycles, treated plants showed significantly reductions in their heights (Fig. 4A and 4B), whereas no significant differences in the stem diameter were observed between them and the controls (Fig. 4C). The anthocyanin concentrations were higher in the treated plants than in the control plants (Fig. 4D), but no significant differences in chlorophyll levels were observed in any of the cycle-treated plants compared with the control plants (Fig. 4E). However, there were no significant differences among the different cycle numbers regarding these factors (Fig. 4). These findings suggest that pipe-induced brushing with at least five cycles is enough to modulate plant growth and induce anthocyanin accumulation.
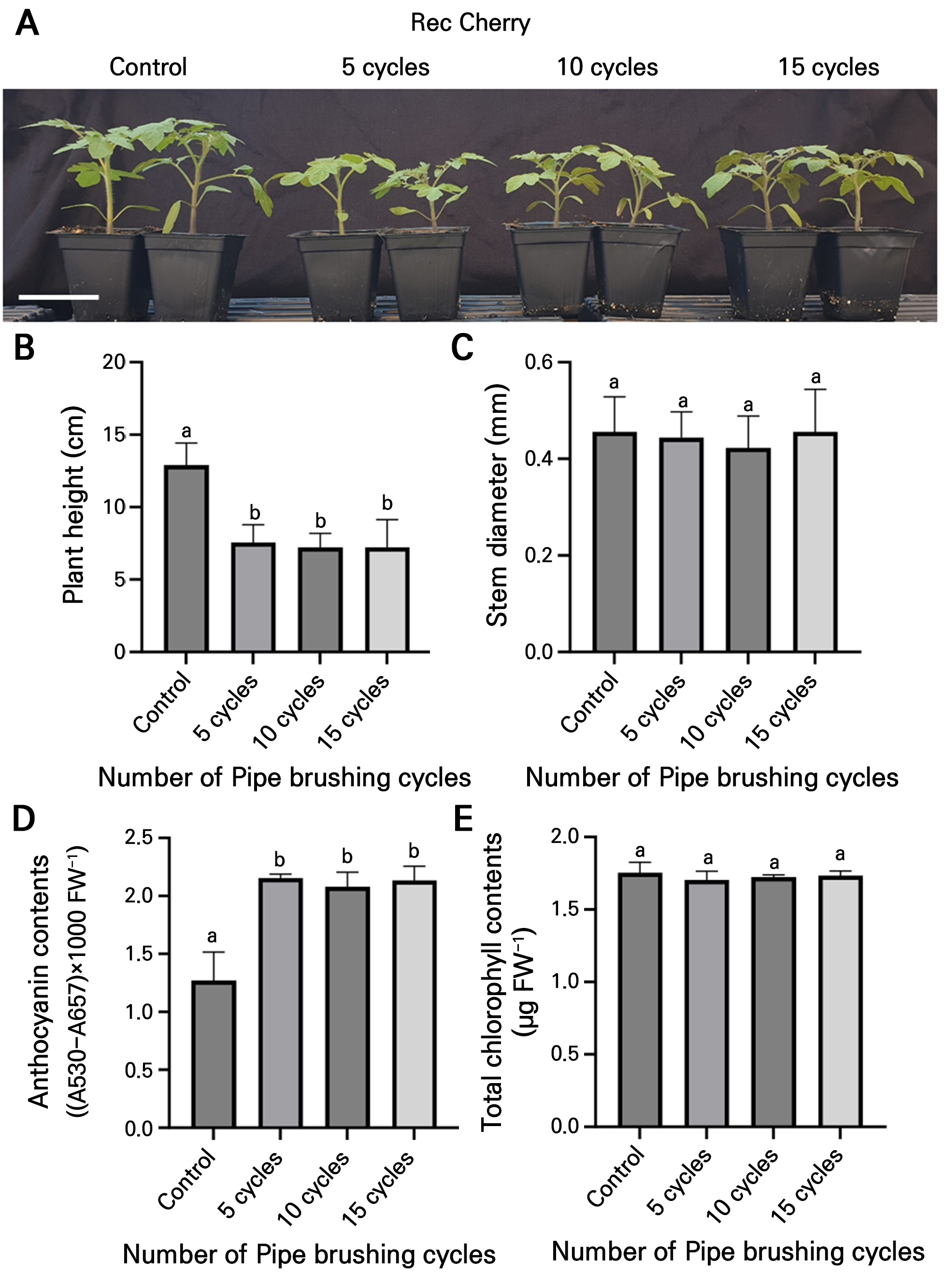
Fig. 4.
Pipe-induced brushing effectively triggers mechanical stress responses in tomato plants. (A) Phenotypes of three-week-old “Red Cherry” tomato plants seven days after undergoing five, ten, or fifteen cycles of pipe-induced brushing. Scale bar = 10 cm. (B, C) Plant height (cm) and stem diameter (mm) on the final day. Data are presented as the means ± SD (n = 9). (D, E) Anthocyanin and chlorophyll contents in tomato leaves on the final day. Data are presented as the means ± SD (n = 3). The statistical analysis was performed using a one-way ANOVA followed by Tukey’s test. Different letters indicate significant differences (p < 0.05) among the samples.
We also examined and tracked cell damage after consecutive brushing treatments at various time points (See “Treatment Phase” in Table 1). During the brushing period, the level of electrolyte leakage in all cycle-treated plants was higher than that in the control plants (Table 1). However, all cycle-brushed plants fully recovered seven days after the final brushing (Table 1). These results indicate that applying up to fifteen cycles for five consecutive days does not cause severe damage to tomato leaves.
Table 1.
Tracking in electrolyte leakage in Red Cherry tomato leaves during consecutive pipe-induced brushing and after the treatment
| Test | Treatment Phase | Days | Control | 5 cycles | 10 cycles | 15 cycles |
| Electrolyte leakage (%) | Initial | Day 0 | 4.0z ± 0.5y | |||
|
Brushing Treatment (Days 1‒5) | Day 1 | 3.7 ± 0.5 ax | 5.7 ± 0.7 b | 6.2 ± 0.2 b | 7.6 ± 1.0 c | |
| Day 3 | 3.9 ± 0.6 a | 6.0 ± 0.3 b | 6.0 ± 0.6 b | 6.3 ± 0.3 b | ||
|
Recovery period (Days 6‒12) | Day 6 | 3.6 ± 0.3 a | 5.3 ± 0.4 b | 5.6 ± 0.3 b | 6.0 ± 0.8 b | |
| Day 12 | 4.1 ± 0.6 a | 3.9 ± 0.5 a | 3.7 ± 0.5 a | 3.8 ± 0.2 a | ||
Pipe-induced brushing regulates mechanical stress responses across various tomato cultivars
To examine the effects of pipe-induced brushing on other tomato cultivars, three tomato cultivars: “Roma VF,” “Super Sweet 100 Hybrid,” and “Red Currant,” were selected and treated with five cycles of pipe-induced brushing. The treated plants exhibited reduced plant heights and similar stem diameters compared to the controls (Fig. 5A–5C). Electrolyte leakage assays showed that none of the treated tomato plants were severely damaged during the treatment periods (Fig. 5D). Anthocyanin concentrations were higher in all the treated plants than the control plants (Fig. 5E). These results suggest that pipe-induced brushing with five cycles can be effectively applied to various tomato cultivars.
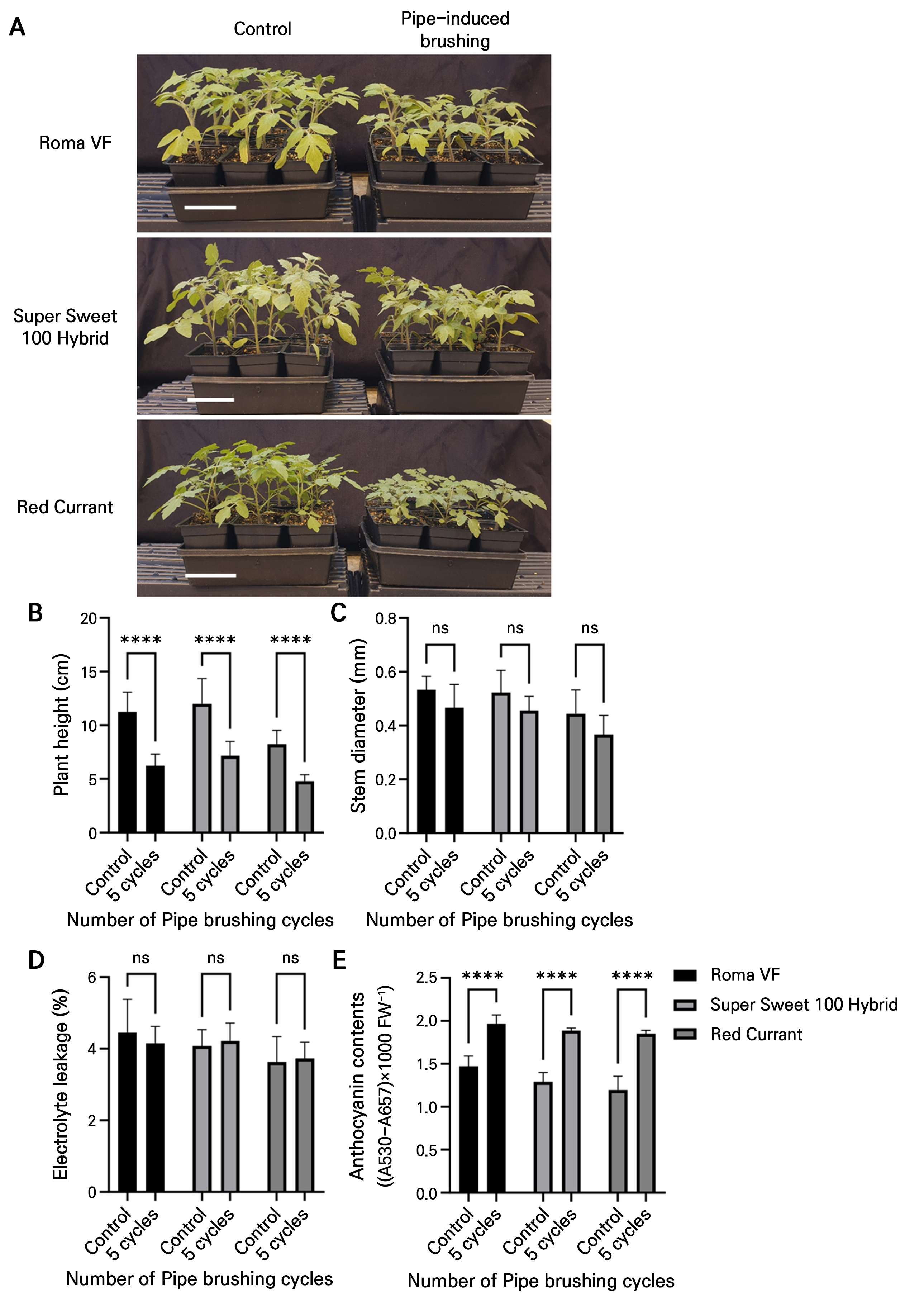
Fig. 5.
Five cycles of pipe-induced brushing effectively induce mechanical stress responses in various tomato cultivars. (A) Phenotypes of three-week-old tomato plants seven days after undergoing pipe-induced brushing (five cycles). Scale bar = 10 cm. (B, C) Plant height and stem diameter on the final day of the experiment. Data are presented as the means ± SD (n = 9). (D) Electrolyte leakage in the treated tomato leaves. Data are presented as the means ± SD (n = 4). (E) Anthocyanin content in the treated tomato leaves. Data are presented as the means ± SD (n = 3). The statistical analysis was performed using a two-way ANOVA followed by Sidak’s test: asterisks (****) indicate significant differences (p < 0.0001), and “ns” indicates no significant difference compared to the control sample.
Applying mechanical stress to tomato plants has been shown to promote early flowering and enhance productivity (Castro-Estrada et al. 2024). In this study, we show that pipe-induced brushing can effectively and rapidly activate plant mechanical stress responses for a large number of tomato transplants at once. These findings suggest that pipe-induced brushing could serve as an alternative to plant growth regulators by enhancing productivity.
However, the precise mechanisms regulating these responses at the cellular level remain unclear. We recently characterized the function of a tomato bZIP transcription factor, VSF-1, in response to hypo-osmotic stress, which mimics mechanical stress (Yoon and Tsugama 2025). In that study, VSF-1 localizes in the cytoplasm under steady conditions but transiently accumulates in the nucleus when plant cells are exposed to hypo-osmotic stress conditions, regulating plant growth. We also identified that VIP1, a VSF-1 homolog in Arabidopsis thaliana, has a similar function and confers mechanical stress tolerance in Arabidopsis leaves (Yoon et al. 2024b). Cytoplasmic calcium increased under both hypo-osmotic and mechanical stress conditions (Beffagna et al. 2005; Benikhlef et al. 2013). Calcium signaling can induce nuclear accumulation of VSF-1 and VIP1 under such stress conditions (Tsugama et al. 2018; Yoon and Tsugama 2025). These findings suggest that calcium signaling is a crucial factor in the regulation of plant growth via VSF-1 under mechanical stress in tomato plants.
In the future, we aim to determine the physiological functions of VSF-1 in tomato plants by using pipe-induced brushing treatments. This approach should provide insights to improve tomato growth and/or stress tolerance with VSF-1 for tomato cultivation operations.


