Introduction
Materials and Methods
Plant Materials and Pre-storage Conditions
CO2 and C2H4 Production Rates
Fresh Weight Loss Rate and Gas Concentration within the Film Package
Microorganism Growth
Surface Color and Chlorophyll Content
Quality Factors
Statistical Analysis
Results and Discussion
CO2 and C2H4 Production Rates
Changes in Fresh Weight Loss Rate and Gas Concentration in the Film Package
Microorganism Growth
Surface Color and Chlorophyll Content
Changes in Asparagine Content
Changes in Firmness, Length, Off-Odor and Visual Quality
Introduction
Asparagus spears have recently become a popular food among Korean consumers, as indicated by an increase in domestic consumption (Yoo et al., 2017). However, asparagus spears currently distributed in the Korean markets have shown degradation in many qualities such as yellowing, toughness, and soft rot during storage (Yoon et al., 2017). In general, asparagus spears are known to be a crop that has a short storage life due to very high respiration and metabolic processes despite being a non-climacteric type vegetable (Zhang et al., 2012). For these reasons, many studies have aimed to suppress quality degradation during asparagus storage, including hot water immersion treatment (HWT) and modified atmosphere packaging (MAP). HWT is one of the useful methods for postharvest control of degradation and suppresses microorganism growth, color change, and chilling injury in many fruits and vegetables when briefly applied at temperatures between 43°C and 53°C (Fallik, 2004). Furthermore, heat-treated asparagus spears have shown suppression of negative geotropism (Paull and Chen, 1999), yellowing (Yoon and Kang, 2017), microorganism growth, and toughness (Chiu and Sung, 2013). MAP has a low processing cost and is easy to apply to distribution conditions, and is well known as a postharvest storage method. According to Yoon et al. (2017), use of oxygen transmission rate (OTR) films of 10,000 cc/m2·day·atm on asparagus spears can extend its storage life by more than 10 d compared to the use of perforated film by steadily maintaining appropriate CO2/O2 concentrations. Presently, there is only one specific quality study that reports single (Paull and Chen, 1999; Yoon et al., 2017) and/or combined HWT/MAP treatment for a short period (Yoon and Kang, 2017), but none of the studies have demonstrated the effects of HWT duration and MAP treatments on overall quality and shelf life for long-term storage of asparagus spears.
This study was conducted to determine how asparagus spear quality parameters are affected by complex pre-treatments with different durations of HWT and MAP for long-term storage.
Materials and Methods
Plant Materials and Pre-storage Conditions
Asparagus spears (Asparagus officinalis L., ‘Welcome’) were obtained from Yanggu-gun, Gangwon-do, Korea. Asparagus spears were immersed in 48°C water for 2, 4, 8, and 16 min, with an untreated sample used as the control. In order to reduce sample temperature, the spears were cooled as rapidly as possible by putting them into 4°C water for 5 min after HWT, except for the control, which did not treat HWT. All samples were then packed in 10,000 cc/m2·day·atm OTR film (Dae Ryung Precision Packaging Industry Co., Ltd., Gwangju-si, Korea) and the polypropylene (PP) films were processed by irradiation with a 785 nm laser at 10-15 s intervals for the MAP condition (Yoon et al., 2017). All packages were stored at 4°C and 85% relative humidity. Each package consisted of five spears and each treatment had five packages. Experiments were replicated four to six times. Representative packaged samples were selected for analysis of the respective treatments. Fresh weight loss rate and gas concentration were measured during storage, but the growth of microorganisms, surface color, chlorophyll content, and other quality factors were investigated just after storage.
CO2 and C2H4 Production Rates
The CO2 and C2H4 production rates were calculated after HWT but prior to storage. Respective samples were placed in a sealed container with a 78 mL volume at 20°C for 1 h. The CO2 production rate was measured with an infrared sensor (Checkmate, PBI, Denmark) and C2H4 production rate was measured with gas chromatography (GC-2010, Shimadzu, Japan) (Park et al., 2000).
Fresh Weight Loss Rate and Gas Concentration within the Film Package
The fresh weight loss rate was expressed as the percentage of weight lost compared to the first day during storage periods. To investigate changes of gas concentration within film packages during the storage periods, CO2 and O2 concentrations inside the film package were measured using the infrared sensor, and gas chromatography was used for measurement of ethylene content.
Microorganism Growth
Approximately 3 g fresh sample was placed in sterilized water and mixed using a stomacher (Powermixer, B&F Korea, Gimpo-si, Korea) set at the highest speed for 3 min. The mixture was diluted to the 105, and cultivated on Petrifilms for 24-36 h. Microbial growth, which included total bacteria, yeast, mold, and E. coli numbers, was investigated using 3M Aerobic Count Plate Petrifilm (3M Co., St Paul, MN, USA) as previously described (Choi et al., 2015).
Surface Color and Chlorophyll Content
Change in the color of the tip and stem of asparagus spears was measured with a color meter (CR-400, Minolta, Japan) to determine the degree of yellowing as a hue angle value (Yoon et al., 2017). To calculate the total chlorophyll content, 1.0 g of the tips and stems of treated asparagus was dissolved in 10 mL methanol, and incubated at 4°C for 48 h to extract the chlorophyll. The absorbance of these extracts was measured at 642.5 and 660 nm using a UV-VIS spectrophotometer (UV mini model 1240, Shimadzu, Japan). Total chlorophyll content was calculated using the following equation (AOAC, 1997):
Total chlorophyll (mg·mL-1) = 7.12 × A660+16.8 × A642.5
Quality Factors
Electrolyte leakage was measured to determine the level of cellular tissue damage following the protocol described (Kang and Park 1998). 0.6 g pieces of tip sample were submerged in 0.4 M mannitol (25 mL) for 3 h and then the elution solution was measured immediately using an EC meter (Cyberscan PC 300, Eutech instruments, Singapore). All samples were frozen and thawed twice. Electrolyte leakage was expressed as the percentage of total leakage. Measurement of the asparagine content was performed as described previously (Sheng et al., 1993; Borek et al., 2017) with slight modification. Asparagus spear tips were briefly homogenized in 0.05 M Tris-HCl buffer, pH 7.5 [9 mL for 3 g of fresh weight (FW)]. The supernatant was then mixed into a 0.05% ethanolic ninhydrin solution in a ratio of 1:9. These mixtures were incubated at 37 for 3 h and the absorbance was measured at 340 nm (UV mini model 1240, Shimadzu, Japan). Asparagine content measurements were conducted with six biological replicates. A rheometer (Compac-100, Japan) was used for assessing stem firmness. The no. 5 (∅3.0 mm) probe was used for measurement at a speed of 1.0 mm·s-1. The length of asparagus spears was measured with a measuring tape and the rate of growth was converted into a percentage compared to the first day. Visual and off-odor qualities were evaluated with sensory evaluations conducted by five skilled panelists using the method described by Yoon et al. (2017) and graded these qualities on a scale from 1 to 5. With respect to the visual quality grade, the best grade was 5, sellable grade was 3, and the complete disposal grade was 1. The evaluation grade for off-odor was evaluated as 0 for minimal off-odor and 5 for a high level of off-odor.
Statistical Analysis
Basic statistical analysis was conducted using Microsoft Excel 2013 and IBM SPSS statistics V21.0 was used for Duncan’s multiple range tests.
Results and Discussion
CO2 and C2H4 Production Rates
The CO2 and C2H4 production rates in asparagus spears were slightly increased after HWT, but there were no statistical differences between the control, 2 min, and 4 min treatments (Fig. 1A and B). The CO2 and C2H4 production rates in asparagus spears were known to be 270 mg·kg-1·hr-1 (Kays and Paull, 2004) and 2.1-4.9 µL·kg-1·hr-1 (Haard et al., 1974) at an ambient temperature, respectively. In our experiments, the average production rates were 184 mg·kg-1·hr-1 of CO2 and 4.0 µL·kg-1·hr-1 of C2H4. According to Siomos et al. (2010), CO2 production by white asparagus spears was not particularly influenced by HWT. In this study, however, C2H4 production slightly increased in the 8 and 16 min treatments. Budde et al. (2006) reported that ethylene production by peaches increased with extended heat treatment in the air was applied to the fruits. Mature green tomatoes treated at 48°C hot water showed an increased ethylene production, whereas tomatoes treated at 39 and 42°C had a similar production rate as the 27°C treatment (McDonald, 1999). These reports supported the idea that asparagus spears treated with short-term HWT such as 2 and 4 min treatments did not affect CO2 and C2H4 production rates, whereas 8 and 16 min treatments increased their rate of production.
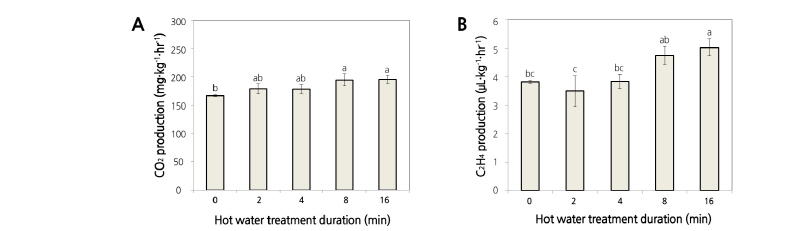
Fig. 1. Effect of hot water immersion treatments at 48°C for 2, 4, 8, or 16 min on the CO2 (A) and C2H4 (B) production rates in asparagus spears that were treated for 1 hr at 20°C were monitored. All the values are expressed as the means ± SE of three biological replicates. The different letters indicate significant differences at the 5% level for Duncan’s multiple range test.
Changes in Fresh Weight Loss Rate and Gas Concentration in the Film Package
The rate of fresh weight loss was less than 0.3% in all treatment groups of asparagus spears, and there was no statistical difference among the treatments (Fig. 2A). Since the maximum allowable weight loss rate in asparagus spears during storage is 8.0% (Kays and Paull, 2004), it is estimated that there was no deterioration of quality due to water loss during storage of samples treated with hot water. The C2H4 concentration during storage rapidly increased after the first day of storage and then it stayed relatively constant at about 1 µL·L-1 with continued storage. This may be caused by stimulated endogenous ethylene synthesis from the physical stress applied to the delicate tissue of asparagus spears during the early storage processes, such as handling and packaging (Kato et al., 2000). All treatments resulted in an approximate 0.9 µL·L-1 of C2H4 and there were no statistical differences between treatment groups on the final day of storage (Fig. 2B). Lee et al. (1995) suggested that ethylene biosynthesis is inhibited under controlled atmosphere (CA) and modified atmosphere (MA) storage conditions and that high CO2 concentration serves as a competitive inhibitor of C2H4. The CO2 concentration within the package during storage was maintained at about 4-5% regardless of HWT (Fig. 2C). The O2 concentration with all treatments rapidly decreased on the first day of storage, but it was maintained above 16.5% at later storage time points (Fig. 2D). In most of the cases, the CA and MA conditions suitable for asparagus storage include a CO2 concentration of 5-12% and an O2 concentration of more than 5% (Kader, 2002). In the conventional experiments of MAP treatments on asparagus spears, the deterioration in quality due to anaerobic respiration was observed at an O2 concentration of 3% or less (Baxter and Waters, 1991; Gariepy et al., 1991). Each of the CO2 and O2 levels in all treatments satisfied the range of suitable MA conditions for asparagus spears. Although CO2 and C2H4 production increased with long-term HWT (8 and 16 min), fresh weight loss and gas concentrations within the packaging did not show significant differences among the treatment or control groups. We therefore, concluded that proper HWT did not affect the metabolism of asparagus spears during storage.
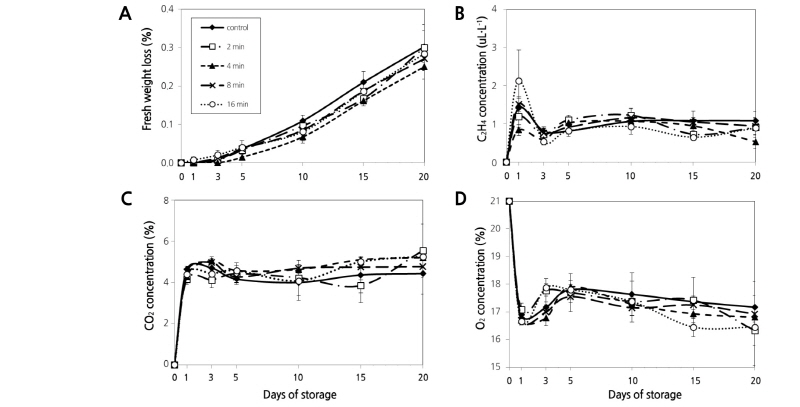
Fig. 2. Changes in loss rate of fresh weight (A) and concentrations of C2H4 (B), CO2 (C), and O2 (D) in asparagus spears treated hot water immersion at 48°C for 2, 4, 8, or 16 min and packaged with oxygen transmission rate (OTR) film (10,000 cc/m2·day·atm) for MAP at 4°C for 15 d. Data are means ± SE of four biological replicates.
Microorganism Growth
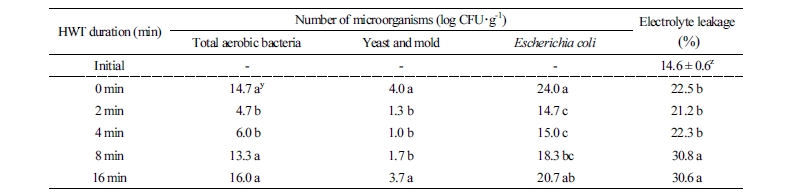
Many microorganisms, such as aerobic bacteria, mold, and Escherichia coli, can cause asparagus spears to decay during the entire post-harvest process (Chiu and Sung, 2013). Sterilization against microorganisms is one of the important factors for maintaining asparagus quality. All microorganism numbers were evaluated on the final day of our experiment and as a result, both the 2 min and 4 min treatments resulted in low aerobic bacteria (average 5.4 log CFU·g-1), yeast and mold (average 1.2 log CFU·g-1), and Escherichia coli (average 14.9 log CFU·g-1) compared to control, 8 min, and 16 min treatments (Table 1). It was considered that this was an effect on inhibiting microorganism growth due to complex pre-treatments with the appropriate HWT for 2 min and 4 min treatments and MA conditions (i.e., CO2 level of about 5%, O2 level more than 16.5%). Similarly, Chiu and Sung (2013) reported HWT microorganism inhibition in asparagus spears; however, 8 min and 16 min treatments did not show any effect compared to the control group. We suggest that tissue damage, including the incidence of soft rot and high electrolyte leakage, were due to relatively longer heating treatments (Table 1). Tissue damage can lead to microorganism infection (Kader, 2002) and in this experiment, electrolyte leakage in asparagus spears increased after storage compared to the initial value (Table 1). A high level of electrolyte leakage was found in 8 and 16 min treatment groups, followed by the control, and 2 min and 4 min treatments. High electrolyte leakage indicated damage to the cellular tissue (Wardowski et al., 1973). HWT might sterilize the surface of agricultural products (Sapers, 2001) and these results suggest that high electrolyte leakage could cause secondary damage that led to bacterial growth and off-odor (Tables 1 and 2). Thus, HWT of more than 8 min generated damage within the tissue and offset any positive heat-treatment effects against microbe infection and tissue damage.
Surface Color and Chlorophyll Content
The color change in stored asparagus spears is a major factor for U.S. market standards (Kader, 2002). Yoon et al. (2017) reported that stem yellowing during long-term storage is a major factor in visual quality degradation. The hue angle and chlorophyll content were measured in asparagus spear tips and stems in order to identify the degree of yellowing. While only the chlorophyll content in the tips of asparagus heat-treated for 2 min was significantly different from the control, hue angle and chlorophyll content were both consistently high in plants from the 2 min and 4 min treatment groups on the final storage day (Fig. 3A and B). Generally, many horticultural products are subject to color change and yellowing due to aging and ethylene production (Kader, 2002), and decreased chlorophyll is associated with senescence and C2H4 activity in asparagus (Wang et al., 1971) and broccoli (Cho et al., 2009). Chlorophyll decreased in asparagus spears processed with 10 µL·L-1 C2H4 (Lee, 2015). In this experiment, all treatments steadily maintained an approximate C2H4 concentration of 1 µL·L-1 during storage, but the degree of yellowing differed depending on the treatment group, suggesting that 1 µL·L-1 C2H4 is not able to affect asparagus spears’ chlorophyll degradation. On the other hand, the yellowing may have been caused by the duration of HWT for more than 8 min. Koukounaras et al. (2009) reported that heat treatment to rocket leaves at 50°C for 20-40 sec reduced chlorophyll degradation without heat damage during 8°C storage, but yellowing and heat damage were accelerated when the heat treatment duration was longer and the temperature was higher. Furthermore, heating treatment of 4 min inhibited chlorophyll degradation in asparagus spears (Yoon and Kang, 2017). The 8 and 16 min treatments might be associated with a reduction in chlorophyll in asparagus spears, which may be related to tissue damage (Table 1).
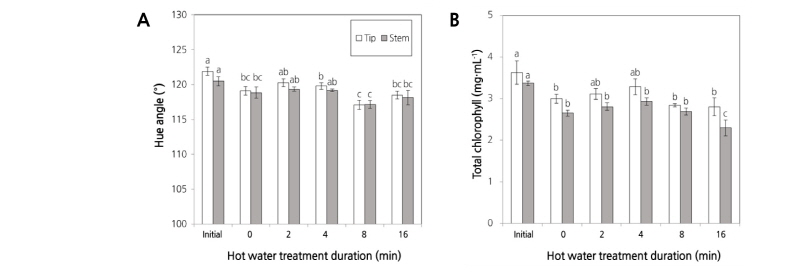
Fig. 3. Effect of hot water immersion treatments at 48°C for 2, 4, 8, or 16 min on the hue angle (A) and total chlorophyll (B) in asparagus spears packaged with oxygen transmission rate (OTR) film (10,000 cc/m2·day·atm) for MAP at 4°C for 15 d. All the values are expressed as the means ± SE of four replicates. The different letters indicate significant differences at the 5% level for Duncan’s multiple range test.
Changes in Asparagine Content
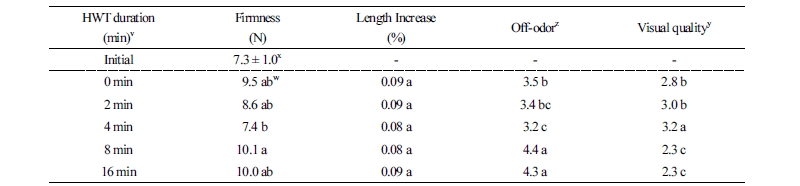
Asparagine content decreased in a time-dependent manner during storage after HWT, with the highest content measured in the control and the lowest content in the 16 min treatment group on the final storage day (Fig. 4). It is well known that denaturation of most amino acids occurs during the heat treatment process (Lee, 1995), and asparagine accumulation of harvested asparagus is strongly related to temperature (Hurst et al., 1998). Consistent with these previous results, we found that asparagine denaturation caused by heat treatment increased with treatment time.
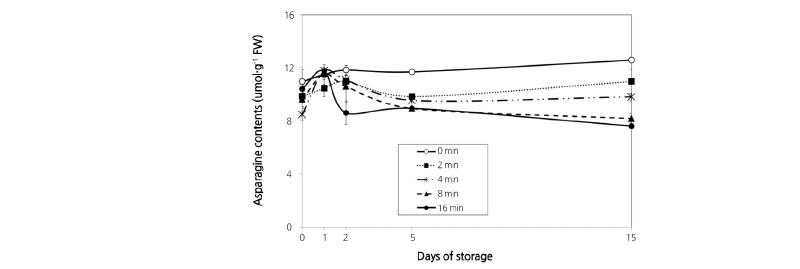
Fig. 4. Changes in asparagine content in asparagus spears treated hot water immersion at 48°C for 2, 4, 8, or 16 min and packaged with oxygen transmission rate (OTR) film (10,000 cc/m2·day·atm) for MAP at 4°C for 15 d. The regression equation was y = 0.2278x - 0.0015 (R2 = 0.9911). Vertical bars represent ± SE (n = 6).
Changes in Firmness, Length, Off-Odor and Visual Quality
Stem firmness is associated with asparagus toughness; the firmness of asparagus spears is increased by lignification of the tissue during storage. We found that the pre-storage and 4 min treatment spears had similar firmness (Table 2). Asparagus spears pretreated with 1 µL·L-1 C2H4 for 24 h before storage had enhanced lignification of tissues, which was due to the activity of phenylalanine ammonia-lyase (PAL) and peroxidase (POD) (Liu and Jiang, 2006). Chiu and Sung (2013) reported that PAL and POD activities were inhibited by the 5 min HWT. The asparagus spears in all treatments slightly increased in length after storage compared to the pre-storage values, these changes were not statistically significant (Table 2). The visual quality was examined during the storage period and maintained a saleable level of higher than 3 points with the 2 min and 4 min treatments for 15 d, while the asparagus in the 8 and 16 min treatment groups lost commercial value (Table 2). In the 8 min and 16 min treatment groups, the incidence of soft rot and progression of yellowing were estimated to be the cause of the deterioration in marketable quality. However, the 2 min and 4 min treatment groups had appropriate CA and MA conditions to inhibit yellowing, which is a key factor visual quality in asparagus spears. The off-odor in the treated asparagus was examined on the final storage date and was the highest in the 16 min treatment group (Table 2). Soft rot and mold can affect off-odor generation in broccoli (Cho et al., 2009), similarly, our results showed that the more soft rot appeared, the more severe off-odor was produced on the asparagus tips.
Based on the overall results, we suggest that the complex pre-treatments of asparagus spears with HWT for 2 to 4 mins and MAP with 10,000 cc/m2·day·atm of polypropylene OTR film had a positive effect on maintaining quality factors than the other treatment conditions tested. The HWT for more than 8 min induced softening of the tissue, which resulted in secondary damage such as microbial infection and degradation of chlorophyll and asparagine content.