Introduction
Material and Methods
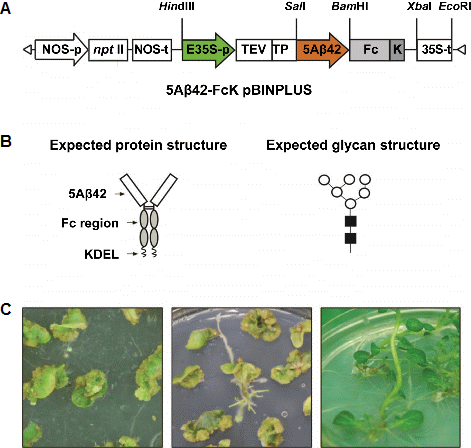
Construction of Gene Expression Cassette
Generation of Transgenic Plants
Polymerase Chain Reaction (PCR) Amplification of Genomic DNA
Southern Blotting
Northern Hybridization
SDS-PAGE and Protein Blot Analysis
Results
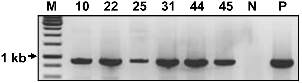
PCR to Confirm the Presence of 5Aβ-Fc Fusion DNA in a Transformed Potato
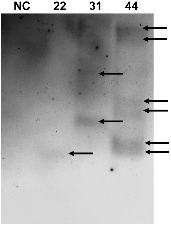
Southern Blot Analysis of the 5Aβ-Fc gene in Transgenic Potato
Transcription of the 5Aβ-Fc Gene in a Transgenic Potato
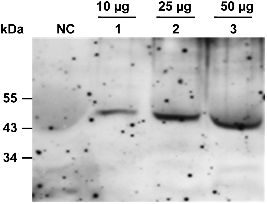
Immunoblot Analysis to Confirm Expression of the 5Aβ-Fc Fusion Protein
Discussion
Introduction
Alzheimer’s disease (AD), the most common form of dementia in the elderly is believed to be caused by the formation of amyloid plaques in the brain, which are made up of small peptides (i.e., 39-43 amino acids) called β-amyloids (Aβs) (Racchi et al., 2008; Weiner HL and Frenkel D, 2006). Mice were immunized with synthetic Aβ to produce anti-Aβ antibodies, thus, preventing the formation of amyloid plaques and protecting transgenic AD mice from AD (Morgan et al., 2000; Schenk et al., 1999). Although the results from the Aβ vaccination experiment in the transgenic mice were promising, this approach to AD patients resulted in meningoencephalitis (Nicoll et al., 2003; Orgogozo et al., 2003). Thus, the vaccine production, immunization protocols, and vaccine structures have been newly modified and improved to reduce such adverse effects (Moretto et al., 2007). Biotechnology and genetic engineering techniques have been used to produce subunit vaccines that stimulate a strong immune response. To create the subunit vaccine, the genes encoding the appropriate subunits are isolated from the genome of the infectious agent and expressed in bacteria (Ma et al., 2013), yeast (Wang et al., 2013), insects (Kanai et al., 2013), and plant host cells (Chen and Lai, 2013), which then produce large quantities of the subunit molecules. Subunit vaccines are safe for use in immunocompromised patients because they do not cause disease (Emini et al., 1986). Plants are considered to be an alternative expression system for the production of recombinant protein and oral immunization with several advantages, including low-cost production, the lack of human pathogen contaminants, and easy post-translational modifi-cation (e.g., glycosylation, an important factor in the biological activity of glycoproteins) (Agarwal, 2008; Brodzik, 2006; Ko, 2003; Ma, 2003; Richter, 2000; Tacket, 1998). We previously reported that Aβ is expressed in plants and biologically functions to induce immune responses in mice (Youm et al., 2005, 2008). Successful protective immunization requires optimized vaccine structures for proper administ-ration protocols, efficient uptake, and presentation by an antigen-presenting cell (APC) (Kreutz et al., 2012). Thus, the biologic properties of the vaccine and its expression level have been optimized in plant production systems (Lu et al., 2012). To achieve both the goals, vaccine sequences have been fused to extra sequences, such as cholera toxin B (CTB) (Langridge et al., 2010), GUS (Cueno et al., 2010), and flagellin (Cuadros et al., 2004), for use as adjuvants or for stabilization. Furthermore, the plant-made vaccine is often purified by its fusion to the multi-His tag for an injectable vaccination approach (Buyel et al., 2012). Fusion of the IgG Fc fragment to the vaccine is another approach that makes it easy to purify the recombinant vaccine expressed in plants (Lu et al., 2012). Fusion of the IgG Fc fragment to the vaccine may potentially have properties such as enhanced efficacy of the vaccination by efficiently targeting it to the APCs and increasing the half-life (Hu, 2008; Mosolits et al., 2004). Furthermore, the fusion of the IgG Fc fragment to the vaccine was shown to provide higher expression levels, better yields, and increased stability in plant expression systems (Lu et al., 2012). Indeed, the Fc domain helps to solubilize hydrophobic proteins and allows for easy detection and purification of the fusion proteins in mammalian cells (Lo et al., 1998). Stable accumulation of the recombinant protein in the endoplasmic reticulum (ER) was achieved by fusion with the KDEL ER-retention signal, which is quite advantageous in establishing high expression levels of recombinant proteins (Gradinaru, 2008; Urakami, 2008). The aim of this study was to investigate whether the 5Aβ-FcK fusion can be expressed in the transgenic potato plant.
Materials and Methods
Construction of Gene Expression Cassette
Five times repeated Aβ (5Aβ) was fused with the Fc region of human immunoglobulin G1 (IgG1) (Fig. 1). The 5Aβ was cloned under the control of the cauliflower mosaic virus 35S promoter (containing the upstream enhancer region) into the pMBP1 vector. The 5Aβ expression gene cassette in the pMBP1 vector was cut by using the HindIII restriction enzyme and inserted into the p13Zf subcloning vector. The Fc region was amplified using the forward primer 5′-GCG GAT CCG TTG AGC CCA AAT CTT GTG ACA AA-3′ and reverse primer 5′-GCT CTA GAT CAG AGT TCA TCT TTA CCC GGG GAC AGG GAG AG-3′ containing BamHI and XbaI and fused to the 3′ end of the 5Aβ gene in p13Zf. The 5Aβ-FcK fusion-gene expression cassette was transferred to a plant expression vector pBINPLUS, yielding pBIN5AF for Agrobacterium-mediated transformation.
Generation of Transgenic Plants
Agrobacterium-mediated transformation was conducted for potato leaf explants by using the plant expression vector pBIN5AF. A. tumefaciens carrying pBIN5AF was suspension cultured in LB broth supplemented with kanamycin at 27°C for 2 d. The cultured Agrobacterium was centrifuged at 4,000 rpm for 20 min, and the pellet was resuspended in an MS (Murashihe & Skoog, 1962) liquid medium with 100 μM acetosyringone at 27°C with gentle shaking for 4 h. The potato leaves were sliced and incubated in Agrobacterium- cultured MS liquid medium with aceto-syringone (250 µg・mL-1) for 10 min while shaking. For effective transformation, the explants were placed in a co-culture medium containing 2.0 mg・L-1 of 2, 4-D. Two days later, the explants were transferred to the regeneration medium containing 2.0 mg・L-1 zeatin, 0.01 mg・L-1 NAA, 0.1 mg・L-1 GA3, 100 mg・L-1 kanamycin, 1,000 mg・L-1 carbenicillin. All plant hormones and antibiotics were filter-sterilized and added to the autoclaved medium. The explants were transferred to the fresh medium every 2 weeks intervals. The shoots generated from transformed leaf discs on selective medium were containing 50 mg・L-1 kanamycin and 1,000 mg・L-1 carbenicillin.
Polymerase Chain Reaction (PCR) Amplification of Genomic DNA
Genomic DNA was isolated from the fresh leaf tissue of transgenic and non-transgenic potato plants using a DNA extraction kit (iNtRON Biotechnology, Seoul, Korea) according to the manufacturer’s recommendations. PCR amplification of genomic DNA was performed to confirm the presence of the recombinant genes using the following primer pairs: the forward primer, 5′-GCG GAT CCG TTG AGC CCA AAT CTT GTG ACA AA-3′ and the reverse primer 5′-GCT CTA GAT CAG AGT TCA TCT TTA CCC GGG GAC AGG GAG AG-3′ PCR was performed with 38 cycles at 94°C for 60 s, 55°C for 60 s, and 72°C for 60 s.
Southern Blotting
From the transgenic plants, 30 µg of genomic DNA was digested using the SalI restriction enzyme (New England BioLabs Inc., Ipswich, MA). Isolated DNA was subjected to electrophoresis in a 0.8% agarose gel and 1× TBE buffer. Total genomic DNA was extracted according to the cetyl trimethyl ammonium bromide (CTAB) protocol described by Doyle and Doyle (1990). The genomic DNA was digested using SalI to determine the copy numbers of the DNA inserted into the independent potato transgenic lines 22, 31, and 44. The digested DNA was hybridized using a digoxigenin-labeled Fc fragment probe, which was generated using the PCR DIG Labeling Mix (Roche Diagnostics, Mannheim, Germany) with a specific primer set for the Fc gene. After overnight hybridization, the membrane was detected using the DIG Detection Kit following the manufacturer’s instructions (Roche Diagnostics). The DNA was blotted onto a positively charged nylon membrane (GE Healthcare, Piscataway, NJ) and incubated in a prehybridization buffer for 2 h at 65°C. After membrane activation, it was hybridized using a PCR- amplified and Dig-labeled specific Aβ probe at 65°C overnight. A non-radioactive method was used for the membrane detection according to the Dig Detection kit manufacturer’s protocol (Roche Applied Science, Indianapolis, IN).
Northern Hybridization
Total RNA from the leaves of transgenic and non-transgenic potato plants was isolated using the RNAgents (Total RNA isolation system, Promega, Madison, WI) according to the manufacturer’s instructions. The concentration of isolated total RNA was determined by measuring the absorbance at 260 nm. Thirty micrograms of total RNA was denatured and analyzed by northern blotting, as previously described (Youm et al., 2008). For northern blotting, the Aβ-specific probe was amplified by PCR and labeled using the Dig Labeling kit (Roche Applied Science, Indianapolis, IN). The denatured RNA was fractionated in a 1% agarose gel by using a 1× MOPS buffer, blotted to a positively charged nylon membrane (GE Healthcare, Piscataway, NJ), and then hybridized and detected using the Dig-labeled specific Aβ probe at 68°C.
SDS-PAGE and Protein Blot Analysis
Transgenic potato plants were homogenized in 200 µL of extraction buffer, which contained phosphate-buffed saline (PBS, Sigma), 1 mM EDTA, 0.1% Triton X-100, 5 mM 2-mercaptoethanol, and 1× proteinase inhibitor cocktail (Roche Co., Germany), and quantified by the Bradford method. The extracted total soluble protein was loaded in 12.5% SDS-PAGE and then transferred to the Immobilon-P Transfer Membrane (Millipore Corp., Bedford, MA) by using the mini-Protean IITM system (Bio-Rad Labs, Hercules, CA) according to the manufacturer’s recommendations. The transferred membrane was incubated with rabbit anti- human Aβ IgG (Applied Biosystems) or goat anti-human Fcγ IgG and detected using a “SuperSignal” chemiluminescence substrate (Pierce, Rockford, IL). The detected signal of the membrane was visualized using an X-ray film.
Results
PCR to Confirm the Presence of 5Aβ-Fc Fusion DNA in a Transformed Potato
The presence of the 5Aβ-Fc fusion gene from transgenic and non-transgenic potato plants was confirmed by PCR analysis (Fig. 2). In PCR analysis, the 5Aβ-Fc transgenic gene DNA was observed in a transgenic potato plant but not in a non-transgenic potato plant (Fig. 2). KDEL, the ER retention signal sequence, was added to the C-terminus of the Fc to retain and accumulate Aβ-Fc in the ER.
Southern Blot Analysis of the 5Aβ-Fc gene in Transgenic Potato
The presence of the DNA inserted into the potato genome was assessed by Southern blot analysis. Southern blot showed that the band for the target gene DNA (i.e., 5Aβ-Fc DNA) was detected in the transgenic potato plant by using a specific probe for the target gene (Fig. 3). In transgenic line 22, a single band was observed, whereas in lines 31 and 44, multiple bands were detected.
Transcription of the 5Aβ-Fc Gene in a Transgenic Potato
Transcription of 5Aβ-Fc fusion mRNA in the transgenic lines 22, 31, and 44 was determined by northern blot analysis (Fig. 4). The RNA was extracted from the leaf tissue of untransformed and transformed potato plants. Target gene expression was shown by using the Dig-labeled system. The expression levels of 5Aβ-Fc RNA were quite different in the transgenic plants. The specific probe of the target gene was bound, which clearly showed the band in the RNA of the transgenic plant. However, there was no band in the non-transgenic plant.
Immunoblot Analysis to Confirm Expression of the 5Aβ-Fc Fusion Protein
Expression of the 5Aβ-Fc fusion protein was identified in a total soluble protein (TSP) extract from the tubers of transgenic potato plants. Ten microgram, 25 μg, and 50 μg of TSP were loaded onto a 12% SDS-PAGE. A non- transgenic potato plant was used as the negative control. The 5Aβ-Fc fusion protein was detected by an anti-human Fcγ fragment-specific antibody conjugated to horseradish peroxidase, and the expected molecular mass was observed on 50 kDa. The 5Aβ-Fc fusion protein was detected with the strongest signal in 50 μg of TSP. In contrast, the band was weakly detected in 10 μg of TSP. No band was detected in the non-transgenic potato plant (Fig. 5).
Discussion
Our data showed that the 5Aβ protein could be fused to the Fc of human IgG1 and expressed in the potato plant. In this study, transgenic potato plant lines expressing 5Aβ-FcK were established via 5Aβ expression. Both PCR and northern blot analyses showed that randomly tested transgenic plants, each containing the 5Aβ-FcK transgene, expressed the transgene transcript. The immunoblot analysis showed 5Aβ-FcK protein expression in transgenic plants. Among the transgenic lines tested by northern blot analysis, 5Aβ-FcK transgenic plant line 22 had the strongest transcrip-tion level. This high expression of 5Aβ-FcK can be explained by the single copy number of the 5Aβ-FcK gene in line 22. In contrast, lines 31 and 44 had multiple copy numbers of the 5Aβ-FcK gene. These lines showed low transcription signals. Western blot analysis was conducted to confirm the expressions of the 5Aβ-FcK fusion protein, indicating that the 5Aβ-FcK fusion proteins were stable in line 22. Fusion of the Fc fragment to 5Aβ has the obvious advantage of an easy, one-step purification procedure by using the protein a method.
As a source of the recombinant 5Aβ-Fc antigen, soluble Fc fusion proteins have become valuable reagents for immunotherapy and laboratory investigations. Furthermore, the immunoadhesion antibody-like protein design resulted in soluble protein accumulation in plant tissues. It is expected that 5Aβ-FcK would have high-mannose glycan structures. In previous studies, Fc fusion with KDEL had high-mannose glycan structures (Lu et al., 2012). The KDEL ER retention signal efficiently retained the glycoproteins, thus, yielding proteins with high mannose (Lee et al., 2013). The antigen- antibody complex can enhance the efficiency and effectiveness of vaccination by targeting the vaccine to APCs. No study has shown that the common Fc-receptors on mouse APC interact efficiently with human IgG Fc. In addition, the oligomannose-type N-glycan structure would be expected to cause an enhanced immune response through the mannose receptor (MR) on macrophages and dendritic cells, which then recognizes the oligomannose of GA733-FcK (Ahlén et al., 2012).
According to a previous study (Lee et al., 2013), the ER-type glycans on glycoproteins, which are also known as oligomannose glycan-types, can further make the glycoprotein immunogenic, thus, producing IgG against the oligomannose glycosylated protein itself. In fact, some studies have shown that the Fc fusion protein can efficiently prevent or treat disease (Hu, 2008; Tobinick and Gross, 2008). The capability of the Fc region to induce an effective immune response has been suggested because dendritic cell-based antitumor immunotherapy is an efficient method for cancer therapy (Armstrong et al., 2002; Liu et al., 1998; Mosolits et al., 2004; Syrengelas and Levy, 1999). In addition, post- translational modification occurs in the ER. The KDEL can act as an autonomous retrieval signal and is introduced at the C-terminus for optimum targeting, with accumulation of interesting genes in transgenic plant glycosylation (Agarwal et al., 2008; Petruccelli et al., 2006). N-glycans of mammalian glycoproteins produced in transgenic plants differ from their natural forms (Lerouge, 1998). If the transgenic protein is directed toward and retained in the ER, then 80% of the total N-glycans attached to ER-resident proteins in the transgenic plant would be high-mannose-type N-glycans (Helenius and Aevi, 2001; Triguero, 2005). The use of the ER-retention signal has been proposed to restrict glycosylation of plant-derived antibodies or the vaccine to only high- mannose-type N-glycans (Triguero, 2005). However, in this study, the effect of Fc and KDEL on the expression levels and glycosylation patterns could not be investigated since we did not include 5Aβ without Fc or 5Aβ-Fc. Future studies need to be conducted by testing these recombinant proteins with 5Aβ-FcK.
In this study, the data clearly showed the expression of a 5Aβ-FcK fusion vaccine candidate in potato plant expression systems. Fusion of the Fc fragment of human IgG to the C-terminus of 5Aβ and the ER retention KDEL in 5Aβ-FcK is an ideal strategy to easily purify recombinant 5Aβ vaccine candidate proteins. Moreover, it enhances the accumulation of the recombinant proteins, which contain oligomannose for comparable immunogenicity of the non- KDEL-tagged mammalian-derived proteins, in plant expression systems. Here, we did not study the addition of the KDEL ER retention signal to the 5Aβ-Fc fusion protein to enhance the expression of the fusion protein in the potato plant. In the future, we will investigate whether the ER retention signal allows control over the subcellular localization of the 5Aβ-Fc fusion protein and whether different glyco- structural patterns of 5Aβ-FcK biological activities are generated in the potato plant.