Introduction
Materials and Methods
Observations of Alfalfa Seed Appearance
Measuring the Concentrations of Ozone Dissolved in Ozone Water and Ozone Micro-bubble Water
Seed Sterilization Treatments
Microbiological Analysis
Seed Viability Analysis
Statistical Analysis
Results and Discussion
Appearance of Alfalfa Seed
The Concentrations of Ozone Dissolved in Ozone Water and Ozone Micro-bubble Water
Comparison of Aqueous Treatments for Sterilization and Viability of Alfalfa Seeds
Introduction
In recent years, the popularity of sprouts has increased considerably due to changes in food production and eating habits. Sprouts are used as ingredients of vegetable salads and thus, are often eaten raw. However, outbreaks of human infections associated with the consumption of raw sprouts have also increased. In the late 1990’s, there were large outbreaks of food-borne illnesses caused by Escherichia coli and Salmonella spp. due to consumption of fresh vegetable sprouts in Japan and the U.S. (Michino et al., 1999; NACMCF, 1999). Sprout-borne disease outbreaks have also been reported in the U.K., Finland, Denmark, Sweden, and Canada and have involved alfalfa, cress, radish, and mungbean sprouts (Buck et al., 2003).
Seeds have been implicated as the initial inoculum source in sprout-borne disease outbreaks, though practices and conditions in sprout production may increase or decrease the extent of the microbial hazards (NACMCF, 1999; Sharma and Demirci, 2003). Therefore, seed sterilization by physical or chemical treatment is important for reducing the risks of sprout-borne disease outbreaks (Buck et al., 2003).
Physical treatments with heat or ionizing irradiation were explored for seed sterilization. Heat treatment includes exposing seeds to high temperatures for a short period because elevated temperatures kill seed-borne pathogens (Scouten and Beuchat, 2002). Ionizing irradiation has also been shown to reduce seed-borne pathogens because radiation energy can penetrate seed tissues and inactivate pathogens localized within the protected tissues (Rajkowski and Thayer, 2001). Despite their effectiveness, these methods are difficult to implement commercially because of a narrow optimum range for the temperature or irradiation and exposure time (Buck et al., 2003).
Chemical treatments with chlorine, sodium hypochlorite, and calcium hypochlorite are currently used for seed sterilization (NACMCF, 1999). However, chlorine reacts with natural organic matter to form trihalomethanes in water and trihalomethanes can cause cancer (Urano et al., 1983). Therefore, the need for the development of new sterilization technologies is increasing in order to reduce the accumulation of chemical residues and to maintain environmental and food safety (Richardson et al., 1998).
Ozone is a potential alternative for food sterilization due to its effectiveness in killing microorganisms and it offers a number of advantages over chemical treatment with chlorine compounds. The primary advantages in ozone treatment include its fast decomposition in water to oxygen, and the absence of residue or by-product (Wang et al., 2004). Researchers have previously tested the efficacy of ozonated water in the sterilization of alfalfa seeds and sprouts (Sharma et al., 2002; Singh et al., 2003; Wang et al., 2004).
Ozone has a high disinfectant activity, but it is not yet widely used due to its low solubility in water. Air-bubbles generated by air pump are 2-3 mm in diameter and they go up quickly and burst at the water surface. Therefore, the solubility of gas in water is very low. However, micro- bubbles rise more slowly to the water surface and the interior gas is completely dissolved in water because the diameters of micro-bubbles are less than 50 µm (Ikeura et al., 2011a; Takahashi et al., 2007a). Moreover, micro- bubbles have special properties such as free radical generation, self-pressurization, and negative charge. In recent times, the application of micro-bubbles in the field of food and agricultural sciences has attracted great attraction due to these properties (Ikeura et al., 2011b; Sumikura et al., 2007; Takahashi et al., 2007b).
There have been several studies on the use of ozone micro-bubbles for removing residual pesticides from vege-tables and reusing wastewater (Ikeura et al., 2011a, 2011b; Sumikura et al., 2007), however, this technique is unexplored for seed sterilization. Therefore, this study was undertaken to determine the effect of ozone micro-bubbles on seed sterilization in alfalfa sprouts. The effect of ozone micro- bubbles on seed viability was also investigated to determine the feasibility of commercial application of ozone micro- bubbles as a disinfectant in the alfalfa sprout industry.
Materials and Methods
Observations of Alfalfa Seed Appearance
Alfalfa seeds (Medicago sativa L.) were fixed in FAA (35% formaldehyde:acetic acid:ethanol, 10:1:1) at 4oC for 24 hours. They were fixed with 1% osmium tetroxide in 50 mM sodium phosphate buffer (pH 7) at 4oC for 24 hours and dehydrated through a series of ethanol dilutions (30, 40, 50, 60, 70, 80, 90, 95, and 100%; 20 min each). The samples were stored overnight in 100% ethanol and further dehydrated by soaking in 100% ethanol and then in 98% isoamyl acetate for 15 minutes each. After the removal of isoamyl acetate, they were dried by critical point drying and observed using scanning electron microscopy (SEM).
Measuring the Concentrations of Ozone Dissolved in Ozone Water and Ozone Micro-bubble Water
Ozone water was generated using an air pump combined with an ozone generator (OG-030V, Ozone Engineering Co., Ltd., Incheon, Korea) into 20 L of tap water. Ozone micro- bubbles were generated in water using a micro-bubble generator (Korea EMB Technology Co., Ltd., Seoul, Korea) and an ozone generator (OG-030V, Ozone Engineering Co., Ltd., Incheon, Korea), simultaneously. We activated the generators for 5 minutes and measured the concentrations of dissolved ozone every 30 seconds in ozone water and ozone micro-bubble water.
Seed Sterilization Treatments
Five treatments were evaluated for their efficacy in sterilization of alfalfa seeds: tab water (TW), micro-bubble water (MBW), ozone water (OW), ozone micro-bubble water (OMBW), and chlorine (CL) (Table 1). MBW was generated using a micro-bubble generator (Korea EMB Technology Co., Ltd., Seoul, Korea). OW and OMBW were generated as described above. CL was prepared with diluted commercial chlorine bleach (0.5%). Alfalfa seeds were then soaked in these solutions for 5 minutes, while non-washed seeds were used as a control.
Microbiological Analysis
After sterilization, the seeds were rinsed three times with sterile distilled water and ground using a sterile mortar and pestle. One mL of ground seeds was blended in 9 mL of sterile distilled water for 30 seconds. Appropriate decimal dilutions in sterile distilled water were then plated for standard plate count, E. coli, and Salmonella spp. Standard plate count (SPC) was determined by plating on standard plate count agar, incubating at 36oC for 24 hours, and counting colony-forming units (CFU). For determination of E. coli and Salmonella spp., we used the media of Desoxycholate agar and Salmonella Shigella agar, respectively, and incubated the plates at 36oC for 48 hours.
Seed Viability Analysis
To investigate changes in vitality of alfalfa seeds after sterilization, the percentage of germination and fresh weight of alfalfa sprouts was also measured at 5 days after sowing. One gram of sterilized seeds was placed in a Petri dish and irrigated with 3 mL of distilled water every day. Petri dishes were put in the chamber setting at 23oC. Alfalfa seeds were germinated under dark condition for 3 days and after germination, alfalfa sprouts were cultivated at PPF 30 µmol・m-2・s-1 for 2 days.
Statistical Analysis
The experimental data were analyzed using SAS statistical software (SAS Institute, Cary, NC, USA). Duncan’s multiple range tests were performed to determine any significant difference (p ≤ 0.05) among the various treatments.
Results and Discussion
|
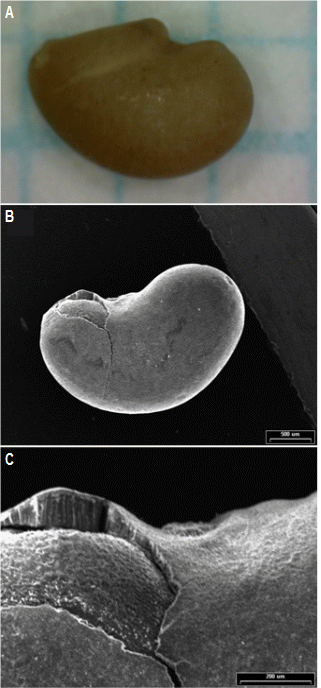
Fig. 1. The surface of alfalfa seed (A), scanning electron micro-graphs of alfalfa seed (B), and seed coat edge of alfalfa (C). |
Appearance of Alfalfa Seed
Although the visible appearance of seeds is smooth, there were many cracks and crevices on the surface of alfalfa seed when observing with a microscopy (Fig. 1). Pathogens can be harbored in cracks or crevices on the surface or between the seed coat and cotyledon of seeds (Sharma et al., 2002). Charkowski et al. (2002) observed alfalfa seeds inoculated with Salmonella enterica using microscopy and reported that S. enterica colonized the broken edges of alfalfa seed coats during the first 24 hours of seed germination.
The Concentrations of Ozone Dissolved in Ozone Water and Ozone Micro-bubble Water
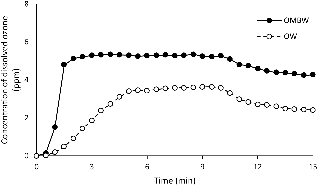
Fig. 2 shows the change in the concentrations of ozone dissolved in OW and OMBW. The maximum concentrations of dissolved ozone were 3.5 ppm at 5 minutes after the start of ozone generation and 5.3 ppm at 2 minutes after the start of ozone micro-bubble generation in OW and OMBW, respectively. In both solutions, the concentration of dissolved ozone decreased gradually with time after 10 minutes from starting the generator. Thus, the concentration of dissolved ozone was kept higher and the time to maintain the maximum concentration of dissolved ozone was longer in OMBW.
Comparison of Aqueous Treatments for Sterilization and Viability of Alfalfa Seeds
In non-washed alfalfa seeds (control), the SPC was appro-ximately 4.54 log10 CFU/mL and E. coli was detected (Table 2). There was no significant decrease in SPC between seeds washed with tap water and non-washed seeds. It indicated that washing with tap water did not affect the removal of pathogens from seeds. The treatments with MOBW and CL were most effective in reducing microbial populations, however, they did not result in complete eradication of pathogens. Salmonella spp. were not detected in all treatments and E. coli was successfully eliminated with OW, OMBW, and CL treatments. Other researchers (Sharma et al., 2002; Singh et al., 2003) have reported the effectiveness of ozone in seed sterilization and our study also shows that ozone can be used as an antimicrobial agent. However, the reduction of microbial populations by OW was significantly lower than that achieved with MOBW. The lower effectiveness of OW was probably due to the lower concentration of dissolved ozone and the inability to penetrate into damaged areas on the surface of alfalfa seeds. OMBW facilitated better penetration of ozone into the inaccessible cracks and crevices of alfalfa seeds, thus enhancing microbial decontamination.
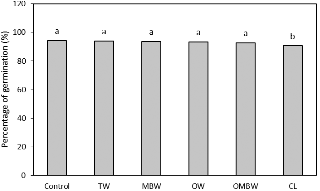
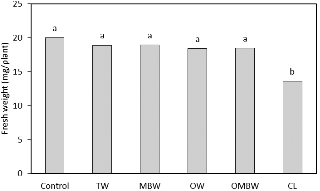
Although the sterilization effect was highest with CL treatment, the percentage of germination and fresh weight of alfalfa sprouts after CL treatment were lower than those after the other treatments (Figs. 3 and 4). Successful seed sterilization must inactivate microbial pathogens while preserving seed viability (NACMCF, 1999). Beyond microbial consideration, the use of chlorine for sterilization of alfalfa seeds creates a problem with toxic residues and by-products and also requires numerous water rinses of the seeds after sterilization.
In conclusion, OMBW was the most efficient treatment for the sterilization and preservation of germination and growth potential of alfalfa seeds. The results of this study suggest that the application of OMBW as a disinfectant for seed sterilization in the production of alfalfa sprouts is feasible. However, pathogens could not be completely eliminated in this study. There is still a need to more thoroughly investigate the dissolved ozone concentration and soaking time of OMBW for complete sterilization of alfalfa seeds.