Introduction
Early development of plant-based antibody expression
Plant-expressed antibody formats and derivatives
Therapeutic and diagnostic applications of plant-derived antibodies across diseases
Technological advances in expression efficiency
Plant species utilized for antibody expression
Glycoengineering strategies for antibody production in plant expression systems
Production of multi-target antibodies using plant expression systems
Challenges and future prospects
Discussion
Introduction
Monoclonal antibodies (mAbs), immunoglobulins that selectively bind to target antigens, are essential for the diagnosis and treatment of numerous diseases, including cancer, autoimmune disorders, and infections (Ecker et al. 2015). They have been developed as a precision medicine exhibiting high target selectivity compared to small-molecule drugs and can minimize non-target side effects (Ecker et al. 2015). The global antibody market reached approximately $266.84 billion in 2024 and is projected to surpass $670 billion by 2033, driven by double-digit annual growth. By 2024, over 120 therapeutic mAbs had received global approval, demonstrating their dominant role in the modern biopharmaceutical industry (Table 1) (Crescioli et al. 2025). Table 1 presents an overview of mAbs produced in several host systems, including mammalian, yeast, bacterial, and plant platforms. However, despite this achievement, current manufacturing platforms have numerous limitations. Hybridoma technology and CHO cell lines still constitute the industry standard. However, these processes incur high production costs, involve lengthy development timelines, and have complex infrastructure requirements. Recent studies demonstrated that plant-based expression platforms offer distinct advantages over mammalian cell culture systems, such as CHO, based on cost-effectiveness, scalability, and the production rate, attributable to their simpler infrastructure and lower cultivation requirements (Morel et al. 2024). Additionally, mammalian cell systems require strict biosafety and quality control, thereby increasing production costs (Jin et al. 2025). Therefore, a more cost-effective and scalable production system is essential. Plants are garnering interest as a promising alternative to solving these problems. Immunoglobulin (Ig)G expression in tobacco was first reported in the late 1980s (Hiatt et al. 1989), and subsequent rapid developments in plant biotechnology have facilitated the production of various antibody types, including IgG, secreted IgA, the single-chain variable region (scFv), and bispecific antibodies (De Muynck et al. 2010). For instance, plant-produced anti-CTLA-4 antibodies exhibit anti-tumor efficacy similar to the levels of commercial immune checkpoint inhibitors in animal models, and antiviral antibodies produced by N. benthamiana have exhibited strong neutralizing activity against HIV and influenza (Chen 2022). Transient expression systems using deconstructed viral vectors can achieve gram-per-kilogram yields within weeks. This speed makes them attractive platforms for rapid response strategies during pandemics (Diamos et al. 2019). However, plant systems are also associated with a number of unresolved issues. Recent advancements in glycoengineering have significantly enhanced the plant-specific glycosylation structure, yet regulatory considerations remain owing to possible immunogenicity complications (Strasser 2023). Additionally, the high cost of large-scale purification and downstream processing and the lack of international regulatory standards for plant-derived drugs present challenges that hinder commercialization (Buyel 2023). Despite the clinical evaluation of the HIV-neutralizing antibody 2G12 produced under good manufacturing practice (GMP) conditions, plant-derived antibodies have not yet achieved commercial success. Furthermore, while mAbs are essential therapeutics, there is a pressing need for platforms that enable rapid, economical, and large-scale production to satisfy the increasing demand for these antibodies (Sack et al. 2015). Plant-based expression systems possess significant potential to meet these needs. This review offers a comprehensive overview of the historical evolution and technological advancements of plant-derived antibodies, their future applications, and the persistent constraints and problems associated with their commercialization. The possibility of plant platforms as an innovative strategy for biopharmaceutical production is also examined (Fischer and Buyel 2020).
Table 1.
Comparison of host expression systems and representative antibodies. Antibodies produced in mammalian, yeast, bacterial, and plant hosts are listed with their corresponding antibody format, molecular target, and clinical or diagnostic indication. This table highlights the defining characteristics of each production platform and demonstrates that plant systems can generate variable antibodies comparable to those from conventional hosts. It further underscores the expanding potential of plant-based molecular farming for next-generation antibody therapeutics
| Host | Antibody | Antibody Format | Target | Indication / Application | Reference | |
| Mammalian cell | CHO cell |
Etanercept (Enbrel) | Fc-fusion protein | TNF-α | Rheumatoid arthritis, Psoriatic arthritis | Weinblatt et al. (1999) |
| CHO cell |
Adalimumab (Humira) | Human IgGκ | TNF-α | Rheumatoid arthritis, Crohn’s disease | Kempeni (2000) | |
| CHO cell |
Blinatumomab (Blincyto) | Bispecific T-cell engager (BiTE) | CD19/CD3 | Acute lymphoblastic leukemia | Bargou et al. (2008) | |
| CHO cell |
Trastuzumab (Herceptin) | Human IgG1 | HER2 receptor | HER2+ breast cancer | Carter et al. (1992) | |
| Yeast | Pichia pastoris |
Eptinezumab (Vyepti) | Humanized IgG1 | CGRP | Migraine prophylaxis | Dodick et al. (2014) |
| Bacteria | E. coli |
Ranibizumab (Lucentis) | Fab fragment | VEGF-A | Neovascular AMD | Ferrara et al. (2006) |
| E. coli |
Caplacizumab (Cablivi) | Nanobody dimer | von Willebrand factor (A1) | Autoimmune hematologic disorder | Scully et al. (2019) | |
| Plant | N. tabacum | 13F6-plantibody | Large single-chain antibody (LSC) | Ebola virus glycoprotein | Ebola virus diagnosis | Olinger et al. (2012) |
| N. tabacum | Nimotuzumab | Humanized IgG1 | EGFR | Head and neck cancer | Perez and Moreno (2014) | |
| N. tabacum | HIV-neutralizing mAb 2G12 | Human IgG1 | HIV-1 gp120 | HIV infection | Trkola et al. (1996) | |
| Zea mays | C225 (Cetuximab) | Chimeric IgG1 | EGFR | Metastatic colorectal cancer | Ludwig et al. (2004) |
Early development of plant-based antibody expression
Preliminary studies confirmed that transgenic N. tabacum could maintain antigen-binding capability by expressing full immunoglobulin chains, providing strong evidence that plants could functionally express complex mammalian proteins (Hiatt et al. 1989; Hyun-Soon et al. 2014). Subsequently, researchers successfully reconstituted secretory IgA (sIgA) by crossing four independent transgenic tobacco lines that expressed the IgA/G mixed heavy chain, κ light chain, J chain, and secretory component, respectively (Ma et al. 1995). Plant-derived sIgA specifically recognized the Streptococcus mutans antigen, indicating that plant cells could execute the assembly and secretion of complex proteins, which were generally considered possible only in mammalian cells (During et al. 1990). These ground-breaking discoveries established the viability of functional antibody expression in plants and laid the foundation for the future production of therapeutic proteins through molecular farming. Following these initial studies, several antibody types were expressed in plants between 1995 and 2000. In 1995, full-length anti-TMV IgG (mAb24) was produced in N. tabacum, confirming a reduction in virus infection rates in F1-generation plants, demonstrating in-planta antiviral efficacy of a plant-made antibody (Voss et al. 1995). In 1998, the complete form of the colorectal-cancer-targeting monoclonal antibody CO17-1A was successfully expressed and assembled in N. benthamiana through the tobacco mosaic virus (TMV) vector, indicating the possibility of plant expressions of antibodies for chemotherapy (Verch et al. 1998). In 1999, TMV-specific IgG was produced in tobacco culture cells, presenting the feasibility of transitioning to plant-cell-culture-based production systems at the whole-plant level (Fischer et al. 1999). In 2000, single-chain antibodies (scFv, T84.66) targeting the colorectal cancer marker CEA were stably expressed in grain crops, including rice and wheat. Functional antibodies were detected in leaves and seeds, indicating the feasibility of seed-based long-term storage and production strategies (Stöger et al. 2000).
Plant-expressed antibody formats and derivatives
Over the past 30 years, plant platforms have utilized various antibody structures beyond full-length IgG. IgG was initially shown to assemble and express its function in Nicotiana species and was subsequently reproduced as a tumor-targeting monoclonal antibody (e.g., CO17-1A) (Verch et al. 1998). Furthermore, sIgA was reconstituted from transgenic tobacco plants, demonstrating efficient polychain assembly suitable for mucosal immune applications (Ma et al. 1995). Beyond IgG and IgA, polymeric IgM antibodies have also been successfully assembled in N. benthamiana, confirming that high-molecular-weight immunoglobulins can be correctly folded and secreted within plant cells (Jugler et al. 2022). The scFv and Fab fragment formats were implemented in leaf- and grain-seed-based production, facilitating production and storage strategies that rely on seed storage (Stöger et al. 2000). The Fc fusion/IgG-like (e.g., scFv-Fc) format has become a standard research scaffold that efficiently performs binding and efficacy assessments through transitory methods (De Muynck et al. 2010). Polyspecificity has been extended to bispecific and similar double-target formats, which have been expressed in plants, with double binding and in vitro functions confirmed against infectious diseases and tumor targets (Morel et al. 2024). This accumulated evidence confirms that plants serve as a versatile platform for producing structurally diverse and application-specific antibody formats (Fig. 1). Notable antibody formats effectively produced in plants include full-length IgG, secretory IgA (sIgA), scFv-Fc, IgM, large single-chain (LSC) IgG, VLRB, camelid HCAb (VHH-Fc), and the Fc-containing bispecific PD-L1/CTLA-4 IgG-like antibody, collectively demonstrating the structural diversity achieved through plant molecular farming over the past three decades (Bulaon et al. 2024). After 1995, plant-derived antibody production has progressed, entering a period of significant expansion and diversification in the early 2000s. Previous studies mostly focused on elucidating the basic principles of expression potential and functional assembly; however, during this period, studies were actively conducted to implement and validate various antibody structures (e.g., scFv, Fab, sIgA, diabody) in plants (De Muynck et al. 2010). This structural diversity demonstrates that plant-produced antibodies can develop into viable treatment and diagnostic tools beyond simple experimental proteins. For instance, plant-produced sIgA has been shown to mediate mucosal immunity, indicating the possibility of preventive applications against oral or respiratory infections (Ma et al. 1998).
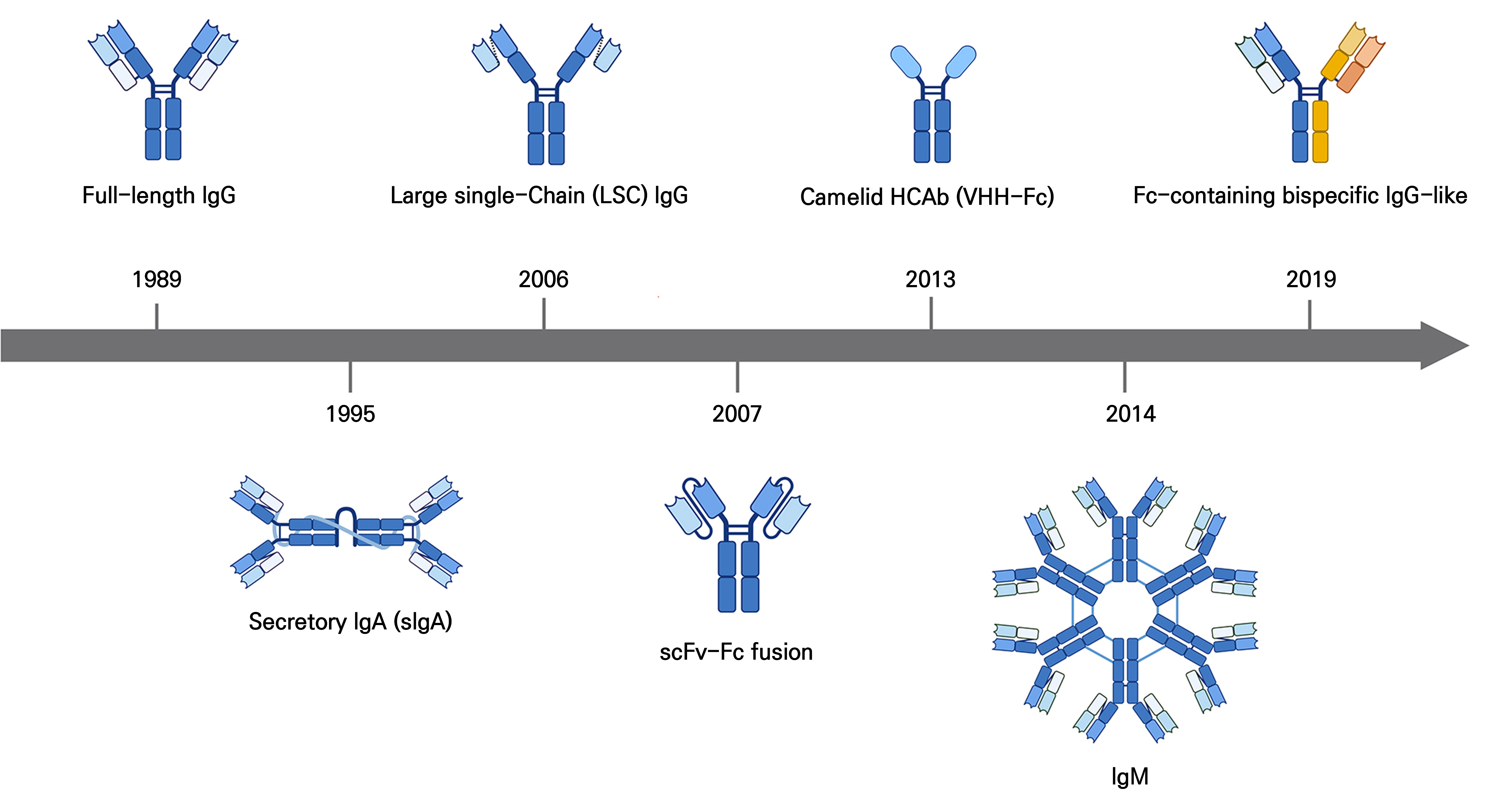
Fig. 1.
Timeline of plant-derived antibody development and format diversification (1989–2019). Key milestones include the first full-length IgG expressed in N. tabacum (1989), reassembled secretory IgA (1995), linker-secured IgG (2006), scFv-Fc fusion (2007), a camelid heavy-chain antibody (2013), IgM multimer (2014), and a bispecific antibody (2019). The figure depicts the progressive evolution of plant expression systems from proof-of-concept IgG production to structurally diverse, therapeutically oriented antibody formats (Created in https://BioRender.com).
Therapeutic and diagnostic applications of plant-derived antibodies across diseases
As plant molecular farming technologies have advanced, applications of antibodies produced in plants have gradually expanded to therapeutic, antiviral, anticancer, and diagnostic areas (Lee et al. 2024). Recombinant antibodies targeting human chorionic gonadotropin (hCG) were initially expressed in N. tabacum, demonstrating sustained immunological activity and demonstrating the feasibility of plant-based antibody production (Nandi et al. 2005). Subsequent advancements in antiviral applications include the development of plant-derived single-domain antibodies (VHH) that target Severe Acute Respiratory Syndrome Coronavirus 2 (SARS-CoV-2) spike proteins, demonstrating antiviral efficacy in vitro (Shen et al. 2023). However, advancements in anticancer research have also been significant. The initial expression of tumor-targeted complete antibody (CO17-1A) in plants laid the foundation for research on immune checkpoint inhibitors beyond simple expression studies (Verch et al. 1998). Specifically, bispecific antibodies, including Fc, which simultaneously target PD-L1 and CTLA-4 were effectively synthesized in N. benthamiana, demonstrating anti-tumor efficacy in mouse colon cancer models (Bulaon et al. 2023). The recent expansion into the diagnostic field has been remarkable, surpassing this therapeutic approach. For instance, a previous study reported a case in which an antibody expressed in plants was fused with horseradish peroxidase and used in an enzyme-linked immunosorbent assay to diagnose infectious diseases (Park et al. 2024). Additionally, serological diagnostic antigens for detecting SARS-CoV-2 infection have been produced through a plant expression system (Santoni et al. 2024). This series of studies collectively demonstrates that plant-based systems have progressed beyond preliminary proof-of-concept stages for therapeutic antibody production and are now being translated into real-world clinical therapeutic and diagnostic applications. This progression underscores the multifunctionality and significant industrial potential of plant platforms.
Technological advances in expression efficiency
Various molecular-level optimization strategies have been established to enhance transcription efficiency, translation accuracy, and intracellular stability, with the result being an increase in the production of recombinant antibodies in plants. Initially, potent promoters, including CaMV 35S and those derived from TMV or geminiviruses, were employed to markedly enhance transcriptional activity in plants of the genus Nicotiana (Amack and Antunes 2020). Similarly, to overcome codon bias discrepancies between plants and mammals, a codon optimization strategy reflecting plant-preferred codon usage was implemented, leading to a threefold increase in antibody accumulation compared to the non-optimized sequence (Buyel 2023). During the post-translational stage, the most effective strategy is to add the KDEL sequence, an endoplasmic reticulum (ER) retention signal, to the C-terminus of the antibody heavy chain to enhance intracellular stability and production efficiency simultaneously. This modification prevents the premature secretion of proteins and induces proper assembly, resulting in a production increase of approximately two- to five-fold when full-length IgG is expressed in N. tabacum (Schouten et al. 1996). These molecular optimization strategies laid the foundation for further advancements. Transient expression systems such as MagnICON®, pEAQ, and pTra now enable high-yield antibody production in plants. These systems enable high yields of several grams per kilogram of biomass within approximately one week post-infiltration and maintain productivity even during the simultaneous expression of multiple subunits (Diamos et al. 2019). This optimized platform has demonstrated practical efficacy in therapeutic antibody production; notably, the Ebola antibody cocktail ZMapp, produced under GMP conditions in N. benthamiana, achieved 100% survival in a non-human primate model (Wong et al. 2024). Through advancements in promoter design, codon optimization, ER retention through the KDEL sequence, and vector engineering, N. benthamiana has been established as a highly efficient expression platform capable of the industrial-scale production of therapeutic-grade mAbs.
Plant species utilized for antibody expression
Various plant species have been employed for recombinant antibody production, including tobacco, cereals, and even moss. In addition to traditional platforms such as Nicotiana, cereals, and moss, several horticultural crops, including Solanum lycopersicum (tomato), Capsicum annuum (pepper), Lactuca sativa (lettuce), and Brassica oleracea (broccoli), have been explored as alternative biofactory hosts (Kang et al. 2025). These species offer high biomass productivity, established agronomic practices, and the potential for edible vaccine delivery, expanding the diversity and applicability of plant molecular farming platforms. For instance, transgenic N. tabacum (tobacco) was used to produce the first plant-derived antibody to enter human clinical trials, sIgA (CaroRx®), aimed at preventing dental caries (Wycoff 2005). Although complex antibodies can be assembled in transgenic tobacco, yields are generally low, typically in the range of tens of micrograms per gram of leaf tissue (Sheshukova et al. 2016). However, a temporary expression system using N. benthamiana leaves achieved a remarkably high yield, accumulating antibodies at hundreds of milligrams per kilogram of fresh leaves (Akher et al. 2025). This rapid and efficient expression system has facilitated the plant-based production of pharmaceutical candidates, including the anti-Ebola virus monoclonal antibody cocktail ZMapp™ (Wong et al. 2024). Stable expression systems in cereal seeds, including maize and rice, have also been utilized for antibody production, enabling long-term storage and oral administration. Yields of 30–40 µg per gram of dried seed material are typical (Joung et al. 2016). The dry seed environment contributes to enhanced protein stability. Additionally, mosses such as Physcomitrella patens can be cultured in bioreactor conditions to produce antibodies with human-like glycosylation profiles, achieving a production level of approximately 300 mg/L (Ruiz-Molina et al. 2022). Furthermore, chloroplast genetic engineering in plants such as tobacco (cigarette plant) and lettuce allows very high protein expression levels reaching several milligrams per gram of leaf biomass (Table 2) (Ruiz-Molina et al. 2022). Table 2 summarizes the broad range of plant expression systems, production tissues, target subcellular compartments, and typical yields with representative examples. These chloroplast-based systems are particularly promising given their ability to produce vaccine antigens or antibody fragments for oral administration using the natural bioencapsulation process within plant cells (Joung et al. 2016).
Table 2.
Plant antibody production organized by expression system, production tissue/compartment, and subcellular targeting. This table summarizes major plant-based platforms (transient agroinfiltration, stable nuclear transgenics, transplastomic chloroplast expression, and moss bioreactor) according to the ‘expression system’, ‘production tissue/plant species’, and ‘subcellular targeting’, alongside ‘typical yield’ ranges and ‘representative example(s)’ drawn from peer-reviewed studies
| Expression system | Production tissue/species | Subcellular targeting | Typical yield | Representative example(s) | Reference |
| Transient |
Leaf (N. benthamiana) | ER (KDEL/SEKDEL) | 0.3–5 mg/g FW | ZMapp™ (Ebola mAb cocktail) | Sainsbury et al. (2009) |
| Transient |
Leaf (N. benthamiana) | Apoplast (secreted) | 0.1–1 mg/g FW | CO17-1A full IgG (TMV vector) | Eidenberger et al. (2023) |
| Transient |
Leaf (N. benthamiana) | ER (KDEL) | per paper | DVD-Ig bispecific (PD-L1 × CTLA-4) | Bulaon et al. (2024) |
| Transient |
Leaf (N. benthamiana) | ER (KDEL) | per paper | anti-HER2 VHH-Fc (single-target benchmark) | Park et al. (2024) |
| Stable nuclear (transgenic) |
Leaf (N. tabacum) | ER (KDEL) | 10–50 µg/g FW (typical) | secretory IgA assembled by crossing | Ma et al. (1995) |
| Stable nuclear (transgenic) | Seed (rice/wheat; endosperm) | ER / protein bodies | ~30–40 µg/g DW | scFv T84.66 (CEA) in cereals | Stöger et al. (2000) |
| Moss bioreactor (transgenic) | Cell culture (Physcomitrella) | Secretory / ER | ~300 mg/L (representative) | overview of moss-made biopharmaceuticals | Decker and Reski (2007) |
| Transplastomic (chloroplast) | Leaf (tobacco/lettuce) | Chloroplast (stroma/thylakoid lumen) | several mg/g FW (typical for plastid-expressed proteins) | VHH fragment improved by thylakoid lumen targeting | Lentz et al., (2012) |
Glycoengineering strategies for antibody production in plant expression systems
Plant-specific N-glycan residues, notably core β(1,2)-xylose and α(1,3)-fucose, were initially cited for safety and efficacy concerns. However, these have been largely mitigated through host glyco-editing in Nicotiana and other platforms (Strasser 2023). In N. tabacum and N. benthamiana, the CRISPR/Cas knockout of xylosyltransferase and α1,3-fucosyltransferase (ΔXT/FT, or ΔXF) enables the production of antibodies devoid of plant-specific glycoforms, yielding human-type core structures (GnGn/G0) suitable for therapeutic applications (Jansing et al. 2019). These ΔXT/FT (ΔXF) backgrounds are now considered the standard for plant-derived mAbs. Beyond removing plant-specific glycans, glycoengineering has advanced to incorporate human-type sugar moieties to modulate antibody efficacy (Strasser 2023). The introduction of human β1,4-galactosyltransferase and a complete sialylation pathway into ΔXF tobacco facilitates the generation of antibodies bearing complex N-glycans with terminal galactose and α2,6-linked sialic acid (Fig. 2). Comparable toolkits have been developed for mucin-type O-glycosylation, enabling sialylated O-glycans on recombinant proteins in N. benthamiana (Castilho et al. 2012). In the moss Physcomitrella patens, glycoengineering has similarly enabled stable human β1,4-galactosylation, providing a contained, GMP-amenable system for producing humanized glycan structures. Plant-derived glycoforms can be engineered to enhance therapeutic efficacy outcomes. For instance, ΔXF plants produce afucosylated IgG, which improves FcγRIIIa binding and enhances antibody-dependent cellular cytotoxicity (ADCC) (Montero-Morales and Steinkellner 2018). This phenomenon has been consistently demonstrated in antiviral and oncology-relevant mAbs, with afucosylated anti-CD30 antibodies produced in the aquatic plant Lemna exhibiting superior ADCC relative to controls (Cox et al. 2006). Recent studies have demonstrated that afucosylated broadly neutralizing antibodies improve the clearance of infected cells under immunocompromised conditions, highlighting the therapeutic importance of precise glycan control. Furthermore, combining host glyco-editing with vector and process optimization enhances N-glycan site occupancy and glycan homogeneity on plant-derived IgG1, preserving a low-fucose, human-like profile that correlates with increased functional activity (Kao et al. 2024).
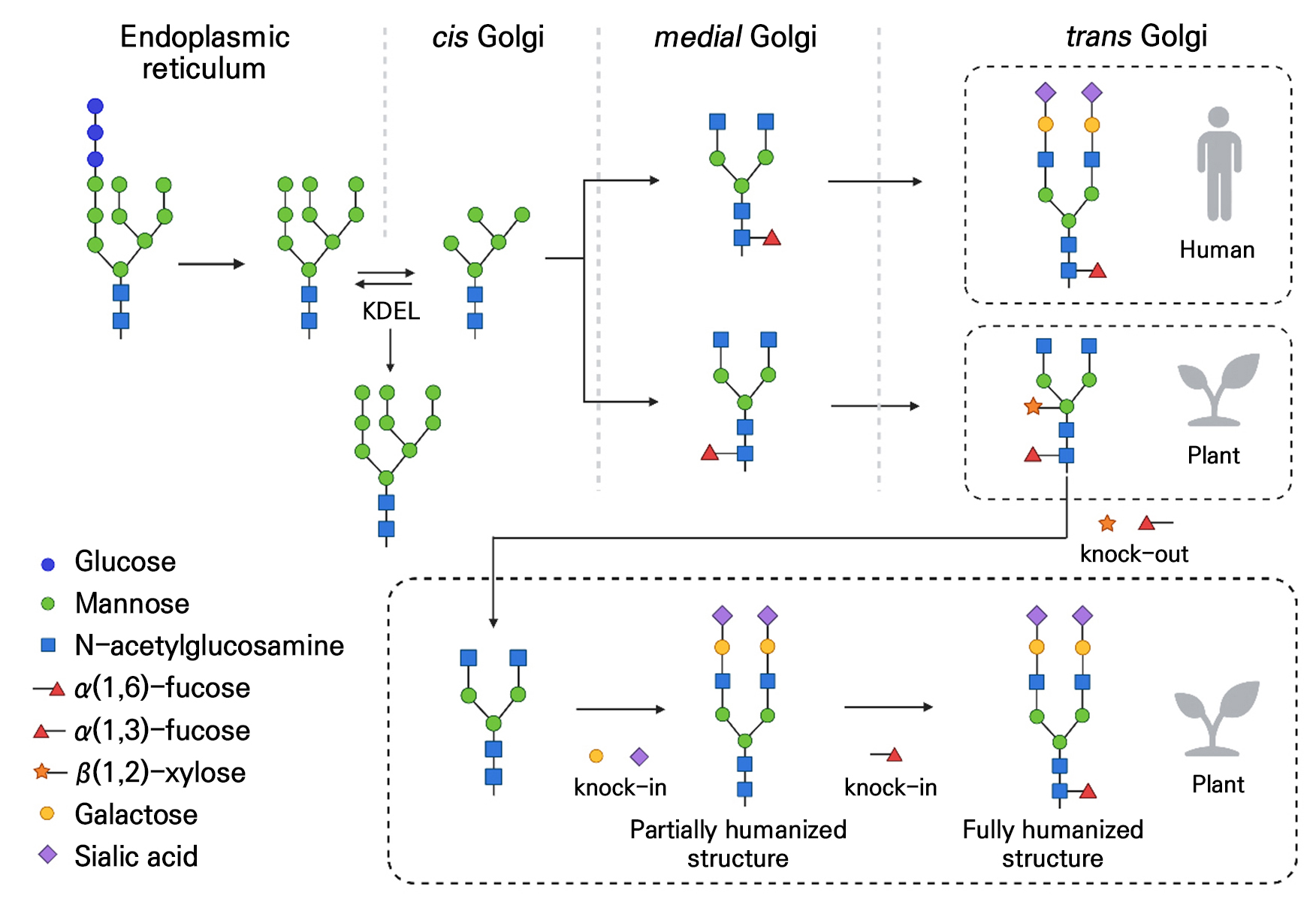
Fig. 2.
N-glycan maturation in plant cells and glycoengineering toward partially and fully humanized structures. Attachment of a KDEL retention signal confines antibodies in the endoplasmic reticulum, preventing Golgi-mediated processing and yielding high-mannose glycans. Transport through the Golgi produces plant-specific β1,2-xylose and α1,3-fucose residues, which can elicit immunogenicity and reduce FcγR binding. The knockout of XT/FT genes (ΔXT/FT) and the expression of human β1,4-galactosyltransferase yield partially humanized glycans (GnGn + Gal), enhancing stability and minimizing plant-type epitopes. The further addition of full human sialylation pathways generates fully humanized complex glycans. It should be noted that α1,6-linked fucose, typical of human glycans, has little impact on ADCC activity, whereas α1,3-fucose from plants markedly reduces Fc receptor binding.
Production of multi-target antibodies using plant expression systems
A key advantage of plant platforms is the ability to cross transgenic lines to stack antibody genes, enabling the simultaneous production of two or more antibodies within a single F1 plant. However, early F1 co-expression revealed heavy and light chain mispairing, commonly referred to as chimerism, which can compromise antibody binding and functionality (Jamal et al. 2012). To suppress chimerism, researchers have employed linker-fused formats. For instance, a large single-chain IgG fuses the variable light domain to the heavy chain through a flexible linker, ensuring correct pairing within one polypeptide. Additionally, light-chain-independent formats such as camelid VHH Fc and lamprey VLRB Fc inherently prevent the cross-assembly of heavy and light chains. These designs have facilitated successful dual-target antibody production in an individual plant. Moreover, transient co-infiltration with multiple replicons and Agrobacterium strains enables the simultaneous high-titer expression of several antibody components, allowing the rapid generation of antibody cocktails or dual-specific antibodies without stable genetic stacking (Fig. 3) (Eidenberger et al. 2023). The clinical emergence of bispecific antibodies has further spurred plant-based implementations incorporating Fc-engineering techniques, including knobs-into-holes, electrostatic steering, and common light-chain architectures, to promote correct heterodimerization and dual binding. Plant-derived bispecific prototypes have demonstrated functional efficacy against viral and oncology targets (such as dengue–chikungunya bispecific designs and checkpoint-targeting antibodies with in vivo efficacy) (Bulaon et al. 2023). Taken together, crossing-based gene stacking, anti-chimerism molecular designs, and multi-gene transient expression capabilities establish plant systems as a practical and scalable platform for the rapid and reproducible production of multi-target antibody formats.
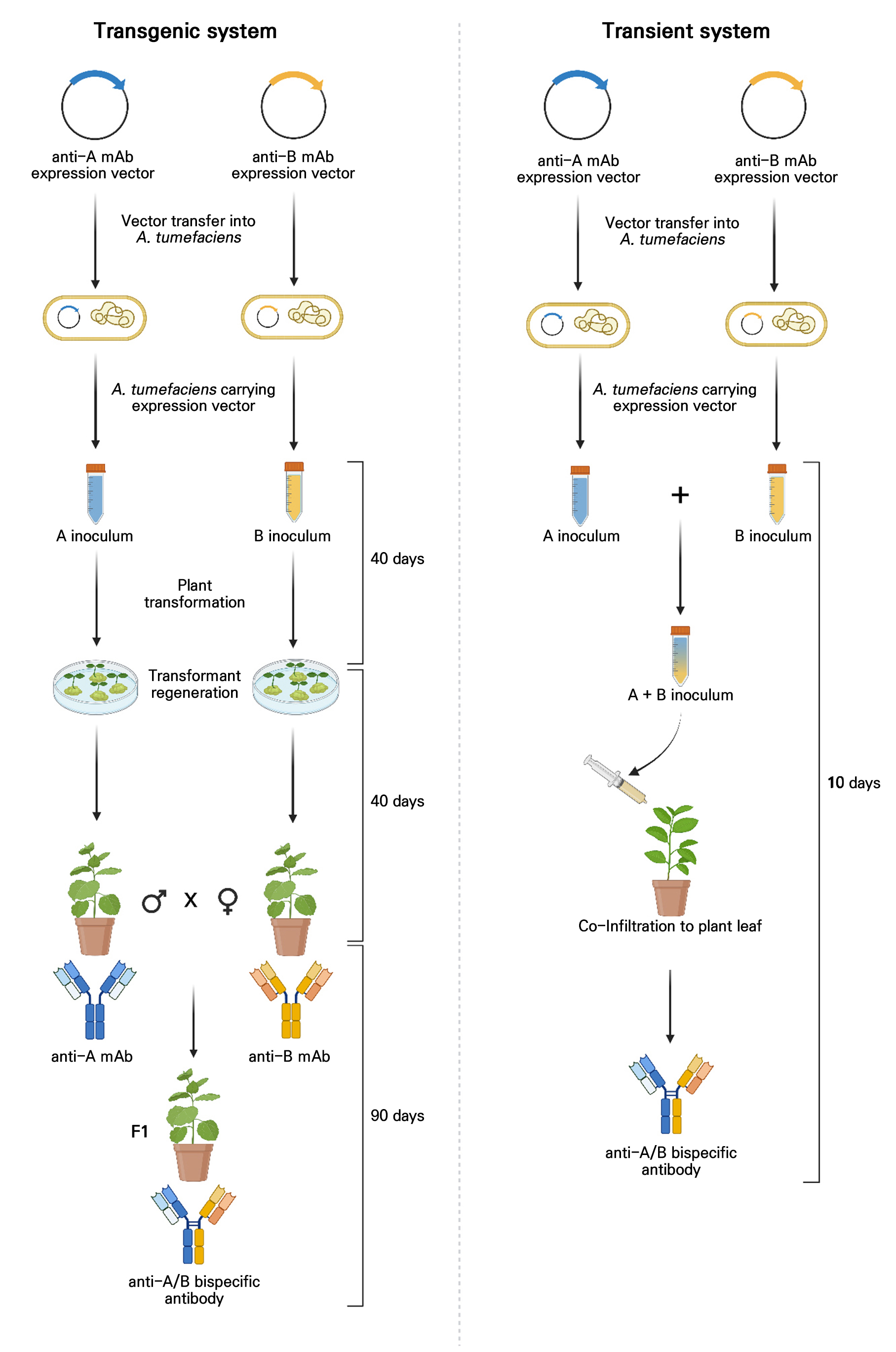
Fig. 3.
Transgenic and transient approaches for bispecific antibody production in plants. Two representative plant-based strategies for bispecific antibody production. In the transgenic approach, independent parental lines expressing each monoclonal antibody are crossed to produce F1 hybrids capable of assembling bispecific molecules. However, the transient co-infiltration method involves the mixing of Agrobacterium cultures with different antibody constructs for rapid, short-term expression in N. benthamiana leaves.
Challenges and future prospects
Despite significant progress, several impediments to the commercialization of plant-derived antibodies remain. Foremost among these is regulatory inconsistency. Unlike mammalian cell-based medicines, which benefit from decades of established guidelines, plant-derived medicines lack internationally harmonized standards (Menary et al. 2020). The approval of the Covid-19 vaccine produced in plants in Canada exemplifies both the potential and vulnerability of the field. Global regulatory discrepancies have delayed broader commercialization, and management of genetically modified plants (GMOs) alongside compliance with GMP standards remains complex (Kim et al. 2016; Bobo 2024). Plant-specific glycosylation also presents challenges. Plant N-glycan contains β(1,2)-xylose and α(1,3)-fucose residues which are absent in mammals, raising immunogenicity concerns (Ko et al. 2008). Furthermore, the lack of terminal sialylation can reduce the serum half-life and effector function. Although glycoengineering approaches, such as human-type glycan production, polysialylation, and afucosylation, have largely solved these problems, ensuring glycan consistency and reproducibility on a large scale remains an important regulatory challenge (Strasser 2023). Economic feasibility is another critical consideration. While the upstream production cost of plant-based antibodies is estimated to be less than $100 per gram, this excludes downstream purification expenses. Beyond economic advantages, Nicotiana-based platforms also offer environmental benefits. Because plants rely primarily on photosynthesis and renewable inputs, they require significantly less energy than fermenter-based mammalian systems. Studies have shown that plant-based systems reduce the overall carbon footprint of biopharmaceutical production and align with sustainability goals through lower water usage, CO2 fixation, and potential integration with circular bioeconomy models (Buyel 2019). Large-scale filtration, chromatography, and viral clearance are required to extract protein from plants, substantially increasing overall costs. Establishing plant factors with a closed GMP system demands a high initial investment, though integration with renewable energy and circular economy initiatives may yield long-term cost savings. Maintaining batch-to-batch consistency in large-scale production remains challenging. Although the N. benthamiana transient expression system achieves high yields at the g/kg biomass level, environmental variables such as light, humidity, and temperature significantly impact protein yield and quality (Sack et al. 2015). To address these limitations, controlled environment agriculture (CEA) systems—such as greenhouse cultivation, hydroponics, and vertical farming—are increasingly applied to plant-based biomanufacturing. In these settings, factors such as the LED light quality, temperature and humidity control, and CO2 enrichment can be precisely manipulated to enhance biomass production and stabilize protein expression (Akher et al. 2025). Such horticultural engineering approaches support the emerging concept of ‘horticultural biomanufacturing’, where high-value recombinant proteins are produced under optimized conditions. Enclosed vertical farming systems can mitigate some environmental fluctuations, but this comes at the expense of increased operational costs. Additionally, the global supply chain for seeds, vectors, and agroinfiltration reagents is still underdeveloped. Overcoming these limitations will be critical to fully realize the commercial potential of plant-derived antibody technology. Continued investment, process automation, and the establishment of a clear regulatory framework are essential. Furthermore, close collaboration between academic researchers and regulatory bodies will accelerate the progress in this field. Overcoming these challenges will enable plant-based systems to emerge as a mainstream biomanufacturing platform.
Discussion
Among the versatile platforms for mAb production, plant molecular agriculture has steadily advanced. Plants can now express a diversity of antibody formats, including traditional IgG, sIgA, IgM, Fc-fusion, and bispecific constructs, often achieving high yields (Fig. 1) (Hiatt et al. 1989). Among the host species, N. benthamiana has emerged as the central platform for rapid and high-titer manufacturing through optimized transient expression systems, routinely delivering gram-level yields per kilogram of biomass within a few weeks (Giritch et al. 2006). These developments have established plants as a robust platform for applications requiring speed, format flexibility, and a scalable supply. These advancements span gene design, vector optimization, and host-supporting technologies. The use of strong promoters, plant-optimized codons, and ER-retention tags (KDEL/SEKDEL) enhances protein accumulation and assembly efficiency (Ko et al. 2003). Deconstructed viral vectors (MagnICON and pEAQ) tailored specifically for transient expression enable multigram yields per kilogram over a short timeframe (Sainsbury et al. 2009). Complementary modalities offer complementary benefits: stable transformation in tobacco and cereal enables long-term seed storage and stock maintenance. Seed-based systems offer ambient stability and support oral delivery. Moss bioreactors allow precise culture control, while chloroplast engineering significantly enhances protein accumulation in leaf biomass (Daniell et al. 2002). This modality diversity allows for the tailoring of production strategies to target product profiles that prioritize speed, yield, stability, and cost-effectiveness. Quality attributes have evolved in parallel. Initial safety concerns related to plant-specific N-glycans (core β1,2-xylose; α1,3-fucose) were addressed by glycoengineering: the ΔXT/FT background eliminates plant sugars, while the introduction of human glycosyltransferases installs β1,4-galactose and α2,6-sialic acid, generating human-compatible glycans (Fig. 2). Specifically, the platform facilitates the functional tuning of glycan structures: fucosylation knockout in the ΔXF line enhances FcγRIIIa binding and ADCC, whereas sialylation and galactosylation can modulate stability or confer anti-inflammatory properties (Castilho et al. 2012). Therefore, glycan composition in plants is no longer a limitation but a versatile design space for aligning mechanisms of action. Plants are also suitable for multi-target antibody production. Initial crossings of independent transformation lines revealed heavy/light-chain mismatches (chimera). However, chimeric-resistant designs (large single-chain IgG, common light chain, VHH-Fc, and VLRB-Fc) and Fc heterodimerization strategies (knobs-into-holes, electrostatic steering) now enable correct antibody assembly (Eidenberger et al. 2023). In transient mode, the co-infiltration of multiple Agrobacterium strains or replicons permits the rapid generation of antibody cocktails and bispecific without stable stacking (Fig. 3). Collectively, these options position plant platforms as a practical platform for research in the field of next-generation multiple-target antibodies on topics ranging from discovery-stage prototypes to long-term scalable production.


