Introduction
Materials and Methods
Collection of Field Samples
Conventional RT-PCR
Cloning and Prokaryotic Expression of the CP Gene
Immunization of Chickens
Western Blot Analysis
IC-RT-PCR
Results
Conventional RT-PCR Assay
Cloning and Expression of LMoV CP
Detection of LMoV by Western Blot Using PAbs
IC-RT-PCR for Detecting LMoV
Discussion
Introduction
Lily symptomless virus (LSV), Lily mottle virus (LMoV), and Cucumber mosaic virus (CMV) are found in Korea. LMoV is widespread, whereas little spread of CMV has been reported (Lim et al., 2009). These viruses occur in the field as single or mixed infections within lily plants. The symptoms of lily viruses vary from vein clearing, leaf mottling, leaf mosaicism, chlorosis and yellow streaking, leaf curling, and brownish-necrotic spots to milder leaf symptoms. The development of mild symptoms in many cultivars prevents efficient removal of diseased plants, which may lead to an unacceptably high incidence of viruses in bulbs (Asjes, 2000). LMoV is a member of the Potyvirus genus within the Potyviridae family that has a linear single strand-positive sense-strand RNA genome of 9.6 kb containing a single large predicted open reading frame (Zheng et al., 2003). Symptoms of LMoV range from fairly mild to severe compared to those of LSV, which range from symptomless to mild. If LMoV-infected plants are also infected with LSV, symptoms can be more severe.
Conventional reverse transcription polymerase chain reaction (RT-PCR), real-time RT-PCR and enzyme-linked immunosorbent assays (ELISA) have been developed to detect lily viruses in infected leaves and bulbs. In earlier studies, RT-PCR was used to identify and detect lily viruses (Ha et al., 2008; Lim et al., 2009). Real-time RT-PCR has been used to detect and quantify plant viruses (Beuve et al., 2007; Boonham et al., 2009; Lim et al., 2010; Mortimer- Jones et al., 2009). Among these detection methods, ELISA is cost effective and suitable for rapid detection of a large number of samples. Various ELISA alternatives based on polyclonal or monoclonal antibodies have been used widely to improve detection limits and broaden strain specificity (Kong et al., 2009; Wang et al., 2010; Wu et al., 2011; Zein et al., 2007). High viral concentrations can be detected by ELISA, but it is not as useful for detecting low viral concentrations (Kim et al., 1995, 2012; Niimi et al., 1999). Preparation of a good quality antibody is important to achieve a highly sensitive ELISA, but large-scale production of mammalian antibodies is expensive and difficult.
Chicken egg yolk immunoglobulin (IgY) production has several advantages over conventional mammalian antibody production; chickens produce larger amounts of antibodies, IgY isolation is fast and simple, and egg collection is non- invasive (Dias et al., 2010; Schade et al., 2005; Spillner et al., 2012). IgY is the major antibody produced by laying hens. After immunization, IgY is secreted into the blood and transported to the egg yolk. Three immunoglobulins (IgM, IgA, and IgY) have been identified in chickens. IgM and IgA are undetectable in egg yolk, but IgY concentrations are 50-100 mg/egg yolk. One hen can produce 20 eggs per month and it is possible to obtain > 20 g IgY/year, as hens continually produce eggs for at least 10 months. The use of IgY may help lower costs of immunological testing. IgY has been used in several studies for diagnostics and therapy (Dai et al., 2013; Wen et al., 2012).
In this study, we determined whether IgY could recognize the coat protein (CP) present in leaves. LMoV CP was expressed in Escherichia coli using the pET-21d(+) expression system and then used as an immunogen for producing chicken egg yolk IgY. IgY functioned well in Western blot analyses and immunocapture RT-PCR (IC-RT-PCR).
Materials and Methods
Collection of Field Samples
Leaf and bulb samples with characteristic symptoms of lily virus infection were collected in different regions of Korea in 2012. Samples known to be infected with LMoV were obtained from the National Institute of Horticultural & Herbal Science (Suwon, Korea) and stored at -80°C for use as positive controls.
Conventional RT-PCR
Infected leaves and bulbs were ground to a fine powder in liquid nitrogen using a mortar and pestle. Total RNA was extracted using Trizol reagent (Invitrogen, Carlsbad, CA, USA). Primers for specific detection of LSV, LMoV, and CMV were designed based on the alignment of the CP sequence of each pathogen available in the GenBank database (Table 1). RT and subsequent PCR reactions were conducted using a MJ Research PTC-150 Thermal Cycler (PE Applied Biosystems, Foster City, CA, USA) with the Maxime RT-PCR PreMix kit (Intron Biotechnology, Daejeon, Korea). The thermal cycling scheme was 42°C for 45 min, followed by 30 cycles of 95°C for 30 sec, 50°C for 1 min, and 72°C for 30 sec, with a final extension at 72°C for 7 min. The PCR products were examined by electrophoresis on 1.5% agarose gels. RT-PCR products were cloned into the pGEM-T Easy plasmid vector (Promega, Madison, WI, USA) to construct the recombinant plasmids.
Cloning and Prokaryotic Expression of the CP Gene
The LMoV CP1 forward primer (5′-CCATGGCAAATGAGA CACTTAAC-3′, NcoI restriction site is underlined), and the LMoV CP2 reverse primer (5′-CTCGAGCATAGAAATTCCAA GTAA-3′, XhoI restriction site is underlined) were used to amplify the LMoV CP gene. The RT-PCR products were cloned into a pGEM-T Easy plasmid vector (Promega). The target fragment was released from the cloned pGEM-T Easy plasmid using NcoI/XhoI and inserted into the corresponding sites of the pET21d(+) expression vector (Novagen, Gibbstown, NJ, USA). The recombinant plasmid was transformed in E. coli strain BL21 (DE3). Protein expression was induced at an absorbance of 0.3 at 600 nm using isopropyl-1-thio-β-D-galactoside (IPTG) in a final concentration of 1 mM, and the culture was incubated for 5 h with shaking at 37°C. The supernatant containing 6X His-fused CP was purified using Ni-NTA agarose beads (Qiagen, Hilden, Germany). The purified protein solution was quantified by standardization with bovine serum albumin and estimated by its molar extinction coefficient at 595 nm.
Immunization of Chickens
The purified recombinant CP was used as an immunogen and injected into 28-week-old ISA-brown chickens. Chickens were immunized on weeks 0, 2, and 4. The antigen was emulsified with an equal volume of Freund’s complete adjuvant (Sigma-Aldrich, St. Louis, MO, USA) for the first immunization. Freund’s incomplete adjuvant was similarly used for subsequent boosting immunizations. Eggs were collected 28 days from the initial immunization
Western Blot Analysis
Leaf samples from healthy or virus-infected plants were homogenized in sodium dodecyl sulfate (SDS) extraction buffer. The samples were denatured by boiling for 3 min and then separated on 12% SDS-polyacrylamide gel electro-phoresis (SDS-PAGE) gels. The proteins were then transferred to nitrocellulose membranes. The membranes were blocked in Tris-buffered saline Tween-20 (TBS-T) buffer containing 5% skim milk powder at 4°C overnight and incubated with primary polyclonal antibodies (IgG and IgY) specific to LMoV at room temperature for 2 h. After three washes in TBS-T, the membranes were incubated with horseradish peroxidase-conjugated goat anti-rabbit IgG (KOMAbiotech, Seoul, Korea) or mammalian antibody and AP-conjugated rabbit anti-chicken IgY (1:10,000) (Bethyl Laboratories, Montgomery, TX, USA) at room temperature for 1 h.
IC-RT-PCR
Leaf samples from lily plants showing disease symptoms were collected, and sap was extracted from the leaf samples using extraction buffer. The sap was centrifuged at 13,000 rpm for 1 min and the supernatants were kept on ice. A 200 µL aliquot of each diluted primary polyclonal antibody (IgG and IgY) was added to PCR tubes and incubated at 37°C for 4 h. After three washes in TBS-T, 200 μL of leaf sap was dispensed into the PCR tubes and incubated overnight at 4°C. After three washes in TBS-T, the virions were disrupted at 80°C for 10 min and used for RT-PCR. The LMoV forward primer (5′-GCGTTCCAGGCAAATGAG-3′), and the LMoV reverse primer (5′-GTAAACCACACTATAACA CAAAGAG-3′) were used to amplify the LMoV gene.
Results
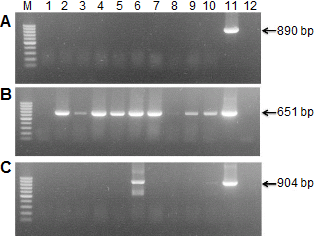
Conventional RT-PCR Assay
Leaf samples and bulbs showing characteristic symptoms of virus infection were collected from Gang-won and Chung- nam province, Korea in 2012. LSV, LMoV, and CMV were detected by RT-PCR (Fig. 1). Eighty field samples showing characteristic symptoms of virus infection were identified; four plants with LSV, 63 plants with LMoV, and two plants with CMV. LMoV predominated in our tests, whereas little spread of LSV and CMV was found. The nucleotide sequences of eight isolates were identified (six LMoV; GenBank accession numbers KC430329 to KCKC430332, KC243417, JX908883: one LSV; JX962776: one CMV, JX913865). The CP regions were determined and compared with the corresponding regions of LMoV (AJ564636), LSV (AJ516059), and CMV (AJ296154). The nucleic acid identities between reference viruses and eight isolates were 95-99%.
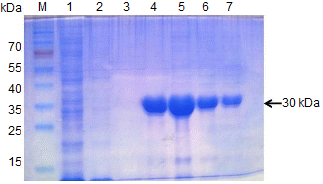
Cloning and Expression of LMoV CP
The entire LMoV cp gene was cloned from genomic RNA extracted from virus-infected lily by RT-PCR, and the sequence was confirmed using an automated DNA sequencing system. The amplified fragments were inserted between the NcoI and XhoI sites in pET-21d(+) to produce a C-terminal His-tagged CP fusion protein. The resulting plasmid was designated pET-21d(+)-LMoV-CP. SDS-PAGE revealed that the recombinant LMoV CP band with a molecular mass of 30 kDa was expressed successfully in IPTG-induced pET-21d(+)-LMoV-CP(–)-transformed bacteria but not in pET-21d(+)-transformed control bacteria. Recombinant His-tagged LMoV-CP was purified from the soluble bacteria fractions through an Ni-NTA agarose column by elution with 300 mM imidazole buffer. The eluted fraction contained a major band of the 30 kDa recombinant CP and was used to induce production of polyclonal antiserum in rabbit and chicken (Fig. 2).
Detection of LMoV by Western Blot Using PAbs
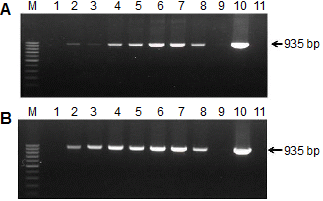
To evaluate the suitability of PAbs to LMoV, antibodies recovered from rabbit and chicken were diluted to 1:10,000 for Western blotting. Figure 3 shows that both the IgY and IgG antibody reacted with LMoV-infected plant tissue extracts and the recombinant His-LMoV CP but not with healthy samples. IgY produced slightly stronger signals than those of IgG. Although some samples (Fig. 3, lanes 2 and 3) were RT-PCR positive, the CP band was not visible in the Western blot.
IC-RT-PCR for Detecting LMoV
The IC-RT-PCR method was used to test for the presence of LMoV in field samples. PAb (100 μL) diluted at 1:100 in coating buffer was used to coat the PCR tubes, and 100 μL of leaf sap was used for detection. LMoV primers produced a single 935 bp band from LMoV-infected lily plants, but no band from healthy lily samples was amplified by IC-RT-PCR. In our Western blotting experiments, some samples were not detected with either the IgY or IgG antibody, but the same samples were amplified by IC-RT-PCR.
Discussion
LMoV is widespread in Korea and causes serious yield losses in lily plants. Therefore a simple, reliable, and sensitive technique to control the viral disease is required. Real-time RT-PCR has been used to rapidly detect LMoV, but this method is not suitable for large numbers of samples (Lim et al., 2010). ELISA with PAbs or monoclonal antibodies is more convenient and practical than real-time RT-PCR (Edwards and Cooper, 1985; Liebenberg et al., 2009; Tatineni et al., 2013). However, it is not easy to diagnose mixed infections with ELISA and it is not as useful for detecting low viral concentrations. In addition, preparation of a good qualitative and highly specific antibody is critical for obtaining reliable results.
In this study, the nucleic acid homologies between the different LMoV isolates were 95-99% and such high sequence conservation allows LMoV CP to be a suitable immunogen to produce IgY for detecting different LMoV isolates. The conventional method of antigen preparation is virion puri-fication following virus propagation but LMoV is difficult to purify due to its fragility and uneven distribution. In addition, lilies are often infected simultaneously by two or three different viruses. Using recombinant CP expression in a prokaryotic system does not cause these problems because it is simple, fast, and economical. Specific antiserum prepared using recombinant CP and its application to serological diagnostic tests has been successfully demonstrated for a number of plant viruses (Gulati-Sakhuja et al., 2009; Kim et al., 2012; Wu et al., 2013).
IgY, the predominant serum immunoglobulin in birds, reptiles, and amphibians, is transferred from serum to egg yolk to confer passive immunity to embryos and baby chicks. In recent years, specific IgY has received considerable attention in passive immunotherapy for infectious disease due to a number of advantages (Dai et al., 2013; Wen et al., 2012). IgY can be extracted in high yield and recovered from immunized hens over prolonged periods. The phylogenic distance of birds from mammals makes production of antibodies against antigens that may be non-immunogenic in mammals more successful in chickens. In addition, IgY binds neither the mammalian Fc receptor nor the rheumatoid factor. These differences have allowed for increasing interest in a wide variety of applications such as xenotransplantation, immunodiagnostics, and immunotherapy. Compared to antibody production in rabbits, IgY technology is economic, and very low quantities of antigen are required to obtain long-lasting and high IgY titers (Dias et al., 2010; Schade et al., 2005; Spillner et al., 2012).
In this study, the LMoV CP was expressed in E. coli, and two PAbs were produced in rabbits and chickens. A single protein band with a molecular mass close to LMoV was specifically detected by Western blotting in virus-infected leaf samples, although the CP bands were not detected in some leaf samples (Fig. 3). These findings indicate that the PAbs prepared in this study were highly specific for LMoV and IgY and that they have comparable sensitivity to a mammalian antibody. We also developed an IC-RT-PCR procedure that offers sensitive, specific, and rapid detection of LMoV in field samples. This method avoided RNA extraction and was carried out easily in a single tube. IC-RT-PCR methods have been developed for detecting many other plant viruses (Ganesh Selvaraj et al., 2009; Kim et al., 2012; Wu et al., 2011). We found that a single 935-bp band could be amplified from LMoV-infected leaf sap using the IC-RT-PCR assay, whereas no DNA band was amplified from healthy leaf sap (Fig. 4). IgY against CP has been used successfully to detect LMoV infection by IC-RT-PCR. In addition, IC-RT-PCR detected viruses in lily samples that were negative by Western blot. We speculate that a low virus concentration in some samples may have caused detection problems.
In conclusion, we successfully produced a large amount of IgY against LMoV by immunizing laying hens with recombinant LMoV CP. Using this PAb, IC-RT-PCR was established for detecting lily viruses in field samples. This method can be used for virus surveillance to help reduce economic losses in the lily industry. Our results suggest that IgY may offer a new large-scale source of low-cost antibodies.