Introduction
Materials and Methods
Seeds collection
Temperature requirement for embryos growth and radicles emergence
Effect of GA3 on radicles emergence of seeds
Observation of embryos growth and viability of seeds
Data analysis
Results
Temperature requirements for embryo growth and radicle emergence
Effect of GA3 on radicle emergence
Interaction between incubation temperature and GA3 concentration
Discussion
Introduction
Hepatica is a genus of herbaceous perennials in the Ranunculaceae and is native to central and northern Europe, Asia and eastern North America. It can be found in deeply shaded deciduous woodland and scrubland. It has been known that seeds are dispersed from this plant in spring and show radicle emergence in autumn (October in South Korea), but cotyledons do not emerge until early spring of next year. The color of flowers varies from dark blue to pure white bloom from late winter to following early spring, followed by leaf development. Some leaves wither away during summer and the rest remain in evergreen until the following spring (Tamura, 1993).
Hepatica nobilis occurs in eastern North America, Europe, and Japan, and Hepatica nobilis Schreber var. japonica Nakai is native to Japan (Hara and Kurosawa, 1958), where it is called ‘Yukiwariso’. In Japan, H. nobilis flowers have double petals and various colour patterns, which have been developed by breeding with wild varieties. H. asiatica Nakai was considered as a variety of H. nobilis (Hara, 1952), but now considered as a separate species (Nakai, 1937). H. asiatica Nakai is a perennial plant native to Korea and called ‘No-ru-gui’ in Korean (Lee, 1982). H. asiatica inhabits areas with deciduous trees and herbaceous plants where soils are somewhat acidic (pH 5.1-5.3). Monthly average of air temperature in these natural habitats ranged between -5 and 5°C in winter, 18 and 24°C in summer, and 10 and 15°C in spring and autumn (Lim and Sang, 1990).
H. asiatica has been studied in terms of methyl alcohol- extractable phytochemical components (Chi and Lee, 1981), growth and cultivation (Lim and Sang, 1990), taxonomic status and genetic variation (Kim and Lee, 1994), life cycle (Hong, 1995), differentiation and development of flower buds (Lee et al., 2005; Song et al., 2003), and control of flowering (Lim et al., 2007; Song et al., 2003).
However, information on seed germination are needed for mass propagation and maintenance of population for this species, which has not been reported before, except for other Hepatica species. Fresh seeds of H. nobilis Schreber (Engell, 1995) and H. nobilis Schreber var. japonica (Nomizu et al., 2004) were found to contain underdeveloped embryos. Seeds of H. americana and H. acutiloba exhibited deep morphophysiological dormancy. Seeds of H. acutiloba were also reported to have epicotyl dormancy and radicle emergence occurs in autumn at low temperatures following a period of high temperatures in summer, while cotyledons emerge in spring after a period of low temperatures during winter (Baskin and Baskin, 1985).
The objective of this study was to assess temperature requirements and the role of exogenous GA3 on embryo growth and radicle emergence of H. asiatica seeds under controlled conditions, i.e., in the laboratory and inside a non-heated plastic house to characterize seed dormancy and promote seed germination of this species.
Materials and Methods
Seeds collection
Seeds of H. asiatica were collected in May 2010 and June 2011 just before dispersion from parent plants. Plants were grown in a non-heated plastic house on the campus of Gyeongnam National University of Science and Technology, Jinju (35°10´N, 128°05´E), Korea.
Temperature requirement for embryos growth and radicles emergence
Before laboratory experiments, seeds were disinfected in 70% ethanol for 2 min, 4% NaOCl for 15 min, and then rinsed with sterilized water three times. After disinfection, 10 seeds were placed in a plastic Petri dish (9 cm × 3 cm) each filled with 40-50 mL of 0.9% agar and 600 mg・L-1 germicide (Bencide LX 150, Wonyang, Korea). The plastic Petri dishes were sealed with parafilm to control contamination and maintain constant hydration in seeds during the experiments. On 11th of May 2010, three replicates of 20 seeds each were placed in temperature-controlled incubators in the dark at three constant temperatures (10, 15, and 20°C) or a high temperature of 30°C for 2, 4 or 8 weeks followed by 15°C for 26 weeks. Seeds were monitored at 5 day intervals for 26 weeks, and those with a radicle of more than 2 mm in length were recorded as an emerged radicle.
Effect of GA3 on radicles emergence of seeds
Seeds were imbibed in distilled water or GA3 solutions of 200 or 500 mg・L-1 for 24 h at room temperature. After pretreatment of GA3, seeds were surface-sterilized by the method described above. On 11 May 2010, three replicates of 20 seeds each were placed in plastic Petri dishes on the same solidified agar described above and incubated at 10, 15, and 20°C, respectively. Observations during 26 weeks of incubation were the same as those described above.
Intact seeds of H. asiatica collected in May 2011 were imbibed in distilled water (control) or GA3 solutions of 400 or 800 mg・L-1 for 24 h at room temperature. After pretreatment, 100 seeds per treatment were placed in 15-mL plastic tubes that contained moistened sand mixed with 600 mg・L-1 germicide (Bencide LX 150, Wonyang, Korea) and these tubes were incubated at 15°C in dark incubators. For investigation, 10 seeds were picked out of the incubator at 2 week intervals for 22 weeks. Morphological changes in embryo and the extent of seeds staining were observed under a dissecting microscope (SMZ-168 series, Motic Incorporation Ltd., Hong kong) using tetrazolium chloride staining (Nomizu et al., 2004). Morphological changes in embryo were identified according to developmental stages shown in the seeds of H. nobilis Schreber var. japonica Nakai as follows: inviable seeds (IVS), globular embryo (GE), heart-shaped embryo (HSE), torpedo-shaped embryo (TSE), cotyledon-shaped embryo (CSE), and radicle emergence (RE).
Observation of embryos growth and viability of seeds
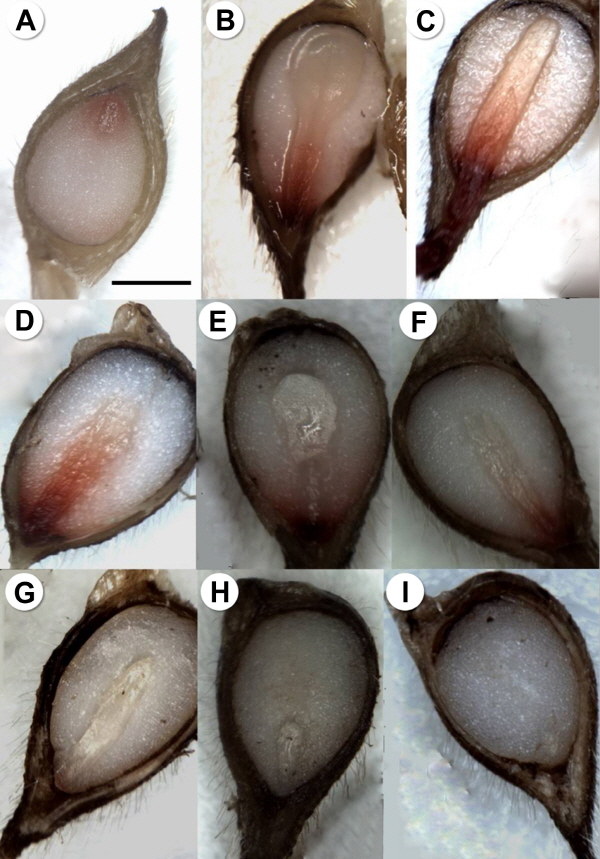
At the end of laboratory experiments and during 26 weeks of incubation, seeds viability and embryos growth were assessed using tetrazolium chloride staining and observation on morphology of embryos. Assessment for seeds viability and embryos growth were divided into five categories based on the observation by tetrazolium chloride staining (Brown, 1996) as follows: level 4, normal embryo growth and normal staining (Fig. 1B, C and D); level 3, normal embryo growth and partial staining (Fig. 1D); level 2, normal embryo growth and poor staining (Fig. 1E and F); level 1, normal embryo growth and no staining (Fig. 1G); level 0, poor embryo growth and no staining (Fig. 1H and I). Seeds with elongated embryos were regarded as seeds with normal embryo growth, but unstained or rotten seeds as inviable ones.
Data analysis
Percentage of radicle emergence, mean radicle emergence time (MRET) or coefficient of radicle emergence rate (CRER) were used as a parameter for radicle emergence. Percentage of radicle emergence is the proportion of radicle-emerged seeds to the total number of seeds after 21 weeks at 15°C. The MRET was calculated as
MRET = (Σ n×d ) / (Σ n)
where n is number of seeds completed radicle emergence, and d is the time (days) counting from the date of starting the radicle emergence test (day 0). CRER is the reciprocal of MRET, and expressed as percentage (Mamo et al., 2006). Viability, embryo growth and radicle emergence of seeds were analyzed by two-way ANOVA using the univariate process of general linear model (GLM) for two factors, GA3 and temperatures with three levels respectively or GA3 with three levels and various incubation time. A one-way ANOVA was employed to examine the effect of temperature or GA3 treatment on radicle emergence, MRET, CRER, embryo growth, viability, and inviable seeds. Multiple comparisons of means were made with Duncan’s test at 0.05 probability level (SPSS 7.0, USA). Relationships among variables were determined using simple linear and nonlinear regression tests with 95% confidence interval.
Results
Temperature requirements for embryo growth and radicle emergence
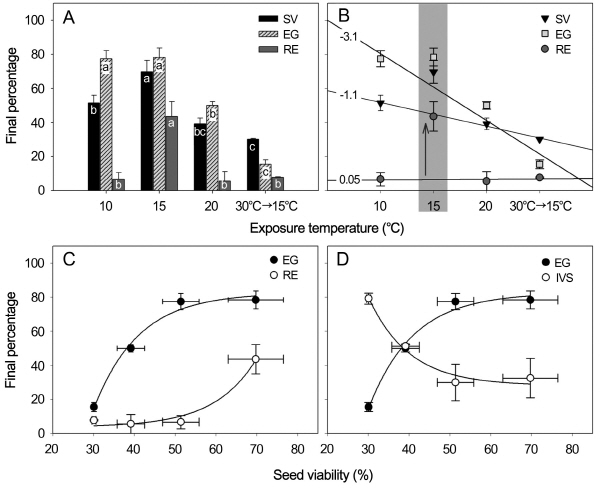
Radicle emergence of H. asiatica was less than 10% in seeds incubated at constant 10°C or 20°C for 26 weeks (Fig. 2A). Seeds exposed to constant 15°C showed more than 40% of radicle emergence, but that was decreased to 10% when seeds were warm stratified at 30°C for 4 weeks then incubated at 15°C. Both of seed viability and embryo growth were highest at 15°C (70% and 78% respectively), but they were lowest (30% and 16% respectively) in seeds incubated at 15°C following warm stratification at 30°C. Embryos were elongated inside 78% of the seeds incubated at both 10 and 15°C, but 50% of seeds at 20°C showed elongated embryos and the percentage was less (16%) in seeds incubated at 15°C following warm stratification at 30°C for 4 weeks. There were significant differences among these treatments (p < 0.05).
With an exemption of a specific response at 15°C, there were linear relationships between exposure to temperature and seed viability, embryo growth or radicle emergence (Fig. 2B). Seed viability and embryo growth had an inverse relationship with increasing temperatures (r2 = 0.98). H. asiatica seeds exposed to ≥ 20°C had a deleterious effect on embryo elongation. Embryo growth with a higher slope (-3.1) was more seriously affected by temperature than seed viability (-1.1). However, exposure to 15°C significantly affected radicle emergence, while there was no significant difference at 10, 20 or 30°C where only 6-8% seeds had emerged radicles. Embryo elongation and radicle emergence occurred in seed viability-dependent manner (Fig. 2C). However, the dependent relationships were not linear (r2 = 0.72-0.75 in linear regression test). Seed viability corresponding to 50% of maximum embryo growth and radicle emergence were approximately 35% and 62%, respectively. Also, there was an inverse relationship between embryo elongation and inviable seeds (Fig. 2D).
Effect of GA3 on radicle emergence
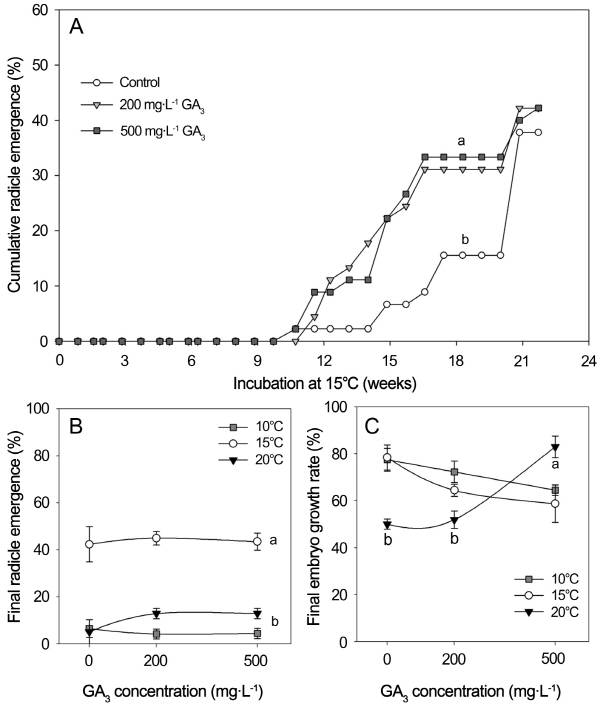
When seeds imbibed in 0 (distilled water, control), 200 or 500 mg・L-1 GA3 solution were incubated at 15°C for 26 weeks, radicle protrusion occurred significantly earlier in seeds treated with GA3 than the control (p < 0.05, Fig. 3A). In seeds imbibed in 200 or 500 mg・L-1 GA3 solution, radicle emergence increased to approximately 10% after 12 weeks of incubation at 15°C and rapidly reached 33% within 16 weeks. This resulted in a significant difference in the marginal mean of radicle emergence for each time throughout the experiment between GA3 and water-imbibed seeds (p < 0.05, Fig. 3A). MRET was decreased by 24 h of seeds soaking in 500 mg・L-1 GA3 and CRER was increased significantly from 5.4 to 6.4% per week (p < 0.05, Table 1). Thus, GA3 stimulated the speed of radicle emergence. However, seeds imbibed in distilled water and GA3 solutions reached 38 and 42% radicle emergence after 22 weeks of incubation at 15°C, respectively (Table 1). These final percentages of radicle emergence were not significantly different (p > 0.05).
In seeds incubated at 10 or 20°C, final percentages of radicle emergence was very low (< 10%) and not significantly affected by any GA3 concentration (p > 0.05, Fig. 3B). However, the embryo growth inhibited at 20°C was signifi-cantly stimulated by imbibition in 500 mg・L-1 GA3 up to a level of embryo growth more than at 10 or 15°C (Fig. 3C).
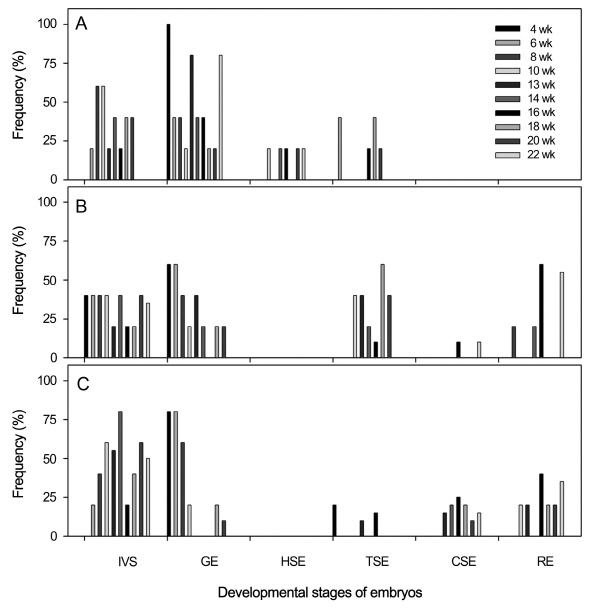
In seeds imbibed in GA3 solution (400 or 800 mg・L-1) for 24 h, morphological changes of embryos in moist sand during 22 weeks incubation at 15°C are represented in Fig. 4. Radicles did not emerge from seeds imbibed in distilled water (control) (Fig. 4A), unlike those incubated on just agar at 15°C (Fig. 3A). Globular embryos developed into the heart-shaped or torpedo-shaped embryos, but cotyledon-shaped embryos were not observed even after 22 weeks of incubation (Fig. 4A). In contrast, imbibition in GA3 solutions stimulated embryo growth and radicle emergence. In seeds imbibed in 400 or 800 mg・L-1 GA3 solution, radicles began to emerge between 8 and 10 weeks after incubation at 15°C (Fig. 4B and C) and continued to emerge by 20-60% until 22 weeks of incubation. Cotyledon- shaped embryos and emerged radicles appeared more frequently in seeds treated with 800 mg・L-1 than 400 mg・L-1 GA3 solutions, but globular embryos appeared less frequently in seeds treated with both GA3 solutions than in the control. Heart-shaped embryos were not observed in seeds treated with both GA3 solutions, but torpedo-shaped and cotyledon- shaped embryos increased. Imbibition in 800 mg・L-1 GA3 was more effective in stimulating embryo growth and radicle emergence than 400 mg・L-1 GA3 (Fig. 4C). Seeds imbibed in distilled water and 400 mg・L-1 GA3 lost their viability by 30 and 34%, respectively throughout the experiment. However, inviable seeds increased to 44% when imbibed in 800 mg・L-1 GA3.
Interaction between incubation temperature and GA3 concentration
Two-way ANOVA analysis showed significant effects of temperatures between 10 and 20°C on radicle emergence, embryo growth, seed viability, and incidence of inviable seeds of H. asiatica after 26 week of incubation (p ≤ 0.05), but not for GA3 concentration (p > 0.05) (Table 2). However, the interaction between incubation temperature and GA3 concentration on embryo growth were significant (p < 0.001) (Table 2). Embryo growth of seeds imbibed with 500 mg・L-1 GA3 was significantly greater than in seeds imbibed in distilled water or 200 mg・L-1 GA3 at 20°C, but not for seeds incubated at 10 and 15°C (Fig. 3C).
Discussion
Seeds of H. asiatica have an underdeveloped embryo when dispersed from a parent plant (Fig. 1A), just like other Hepatica spp. including H. Americana (Martin, 1946), H. acutiloba (Baskin and Baskin, 1985) and H. nobilis Schreber (Engell, 1995; Nomizu et al., 2004). Stratification at 30°C for 2-8 weeks before incubating at 15°C decreased seed viability, led to inhibition of embryo growth, and resulted in very low radicle emergence (Fig. 2A). A temperature of 30°C might be too high to express a stratification effect on H. asiatica, possibly due to much lower soil temperature during May to July. In a previous study on H. nobilis Schreber var. japonica, incubation at 25°C did not promote embryo elongation either (Nomizu et al., 2004). Even a short period of warm stratification at 30°C (2 weeks) showed a deleterious effect on radicle emergence at 15°C, but warm stratification at medium temperature of 20-25°C might help expedite embryo growth and radicle emergence at 15°C.
In previous studies on seeds of Eranthis hiemalis (Ranun-culaceae), embryos started rapid development when transferred to low temperatures after ripening at 20-25°C for 3 weeks (Frost-Christensen, 1974). In natural habitats of H. asiatica, monthly mean summer temperatures were recorded between 18 and 24°C, and during autumn when radicle emergence occurred were around 10-15°C (Lim and Sang, 1990). That is consistent with the optimum temperature for radicle emergence of H. asiatica seeds found in the present experi-ment where radicles emergence reached 38% only at 15°C incubation. Radicle emergence in seeds of H. nobilis also occurred at this temperature (Nomizu et al., 2004). As shown in seeds of many Ranunculaceae plants (Atwater, 1980; Frost-Christensen, 1974; Nomizu et al., 2004), embryo elongation of H. asiatica seeds was rapidly completed between 10 and 15°C. The present study also showed that embryo growth was more susceptible to temperature (Fig. 2B) and elongation was significantly inhibited when the temperature increased above 20°C (Fig. 2A and B). The highest dependence of embryo growth on temperature (Fig. 2B) could have relevance to previous reports where temperature ranges suitable for embryo development were found to vary for each developmental embryo stage (Atwater, 1980; Frost-Christensen, 1974; Nomizu et al., 2004).
In the present study, 15°C was much more effective than other temperature conditions (10, 20 and 30→15°C) in breaking dormancy of H. asiatica seeds with a significant positive effect on seed viability. Given the inverse relationship between embryo growth and incidence of inviable seeds (Fig. 2D), it seems likely that embryonic cell death occurs as a result of retarded embryo growth. Factors inhibiting embryo development may cause cell death of embryo. Embryo growth and radicle emergence were affected by seed viability (Fig. 2C). Seed viability corresponded to 50% of maximum radicle emergence was higher than that for 50% of maximum embryo growth. This might be caused by a higher turgor pressure to puncture the endocarp during radicle extension than that during embryo elongation con-sidering that extension of embryo or radicle is a turgor-driven process (Bewley, 1997).
H. asiatica seeds, maintained at 15°C, developed into cotyledon-shaped embryo with subsequent radicle emergence, whereas seeds held at 10°C elongated as much as at 15°C, but no emergence of radicle was seen. GA3 pretreatment caused an increase in radicle emergence, but not in the final percentage of radicle emergence at 15°C. Also, addition of GA3 hardly stimulated radicle emergence at 10 or 20°C (< 13%) (Fig. 4B), but remarkably promoted embryo growth at 20°C (Fig. 3C). These results indicate that the role of GA3 in H. asiatica seeds is related to embryo growth, but not to radicle emergence.
Radicle emerged optimally only at a specific temperature of 15°C, which indicates that temperature plays an important role in regulating radicle emergence. However, radicles did not emerge from the water-imbibed seeds incubated in moist sand even at the optimum temperature (15°C) and embryos growth was retarded (Fig. 4A) unlike those on plain agar plates. Meanwhile, in non-imbibed seeds, radicles emerged from the seeds in moist sand at 15°C although radicle emergence occurred infrequently with little population (20%, data not shown). Such a difference between solidified agar and moist sand could be explained by a previous report where it was found that the highest germination (42%) of fresh seeds of Rosa bractita was obtained when seeds were sown in sphagnum moss, while the lowest percentage (1.8%) was obtained when seeds were sown on filter paper (Anderson and Byrne, 2007). Seeds in moist sand might also be affected by less available oxygen due to a difference in air contact and empty volume of container (approximately 7 and 100 mL, respectively). Like seeds of H. asiatica in moist sand, germination of rose seeds was also affected by water imbibition, which was decreased by 22% and 16% when seeds were incubated after leaching in sphagnum moss and agar, respectively. The inhibitory effect of water imbibition on radicle emergence could be due to a rapid leakage of solutes required for radicle emergence as mentioned in an earlier study (Larson, 1968).
Present and previous reports imply that incubation medium and water imbibition of seeds are important factors in radicle emergence or germination of seeds. Water-imbibed seeds that did not have emerged radicles in moist sand dramatically responded to GA3, which stimulated embryo elongation, and promoted radicle emergence up to maximal 66% at the concentration of 400 mg・L-1 GA3 solution. However, increased GA3 concentration (800 mg・L-1) showed a tendency to increase inviable seeds despite its stimulation of radicle emergence. Increased inviable seeds caused by GA3 were observed in coffee seeds (da Silva et al., 2005).
There are many reports on the stimulatory effect of gibberellic acids during seed germination (Bewley, 1997; Karssen et al., 1989). Although embryo growth of H. asiatica seeds was completed at 10°C or by GA3 pretreatment at 20°C, the elongated embryo was not allowed to have an emerged radicle at these temperatures. Thus, it can be concluded that exogenous GA3 promotes embryo elongation, but not radicle emergence of H. asiatica seeds. GA3 is thought to speed up radicle emergence of seeds at 15°C by promoting embryo growth, which also can be explained by the result of embryo elongation stimulated by GA3 in moist sand (Fig. 4).
The most significant finding of this study is that H. asiatica seeds have two separate parts of dormancy for embryo and radicle. The break of first part of dormancy lead to a fully developed embryo, which occured equally well at 10 and 15°C and the dormancy at 20°C was overcome by pretreatment with GA3. However, the break of second part of dormancy lead to radicle emergence more effectively only at 15°C and this part of dormancy cannot be overcome by pretreatment with GA3 at lower or higher temperatures (10 and 20°C). These results indicat that H. asiatica seeds have morphophysiological dormancy.