Introduction
Materials and Methods
Plant Growth and Environments
Carbon Dioxide Treatments
Measurement
Results and Discussion
Introduction
Strawberry (Fragaria×ananassa Duch.) is one of the most popular fruits in the world and is a good source of vitamins, minerals, and anthocyanin (Cordenunsi et al., 2002). The approximate area and production yield of strawberries cultured in protected facilities in Korea is 6,403 ha and 195,964 tons (MAFRA, 2016), with and about 2,647 tons exported in 2015 (aT, 2016).
Proper light intensity, temperature, humidity, and carbon dioxide (CO2) are important components of growth environments that can affect fruit quality and yield. CO2 fertilization is commonly used on commercial farms because the CO2 saturation point of most horticultural crops is higher than the atmospheric CO2 concentration. The application of CO2 to strawberry crops was found to increase fruit sucrose content and yield up to 30-40% (Jeong et al., 1996). Previous studies provided proper ranges of CO2 fertilization in strawberry cultivation; Mortensen (1987) suggested that elevated CO2 levels of up to 700-900 mg·L-1 can be applied in winter and early spring for commercial strawberry cultivation, and Jeong et al. (1996) suggested that CO2 fertilization with 900 mg·L-1 at 20°C was an optimal condition for strawberry cultivation. Conversely, some studies found that high CO2 concentration reduced yield and fruit quality (Koch and Mooney, 1996; Lee and Lee, 1994; Yu et al., 2015). When the two strawberry cultivars ‘Benihoppe’ and ‘Frandy’ were cultivated under high CO2 concentrations in a closed ecosystem, 2,000 mg·L-1 CO2 (over saturation point, 1,500 mg·L-1) inhibited the net photosynthetic rate of ‘Benihoppe’ cultivar by 12.4% than that of ‘Frandy’ (2.3%) (Yu et al., 2015).
In the long-term (over 50 d), CO2 fertilization with 800 mg·L-1 in tomato cultivation increased the photosynthetic rate until 20 d but it had no effect ever since 30 d after the treatment and decrease more than untreated plant (Lee and Lee, 1994). The biochemical basis for the decline of photosynthesis with long-term elevated CO2 treatments is not clearly understood, although sink imbalance, carbohydrate accumulation, and negative feedback mechanisms have been proposed as models (Bowes, 1991; Stitt, 1991). an increase of 600 mg·L-1 in the CO2 level promoted leaf net photosynthesis of strawberry, but a further increase to 750 and 900 mg·L-1 decreased net CO2 assimilation, which was accompanied by a distinct decrease of optimal quantum yield and macronutrient deficiency (Keutgen et al., 1997).
Although there are numerous studies of effects elevated CO2 has on photosynthesis in plants grown in a large range of experimental conditions, little information is available on the effects of elevated CO2 concentrations have on strawberry productivity, which would benefit the strawberry industry. The aims of this study were to provide information on the effects of CO2 fertilization on the vegetative and reproductive growth and yield in commercial strawberry cultivation.
Materials and Methods
Plant Growth and Environments
The experiment was carried out in two double plastic houses [100 m (L) × 10 m (W) × 3.3 m (H)] located in Kyoungnam Sacheon-si starting on the 5th of September 2016. Nine hundred strawberry plants ‘Maehyang’ were planted in two rows with an interval of 15-16 cm on a cocopeat substrate [dust:chip = 7:3 (v/v), Kims Trade Ltd., India] placed in beds [0.3 m (W) × 80 m (L) × 0.2 m (H)] 1.0 m from the ground. The UOS nutrient solution developed for strawberry cultivation at the University of Seoul (UOS; Lee et al., 2017) was supplied 3-4 times in 2-3 min duration each day at the EC 1.0-1.2 dS·m-1 level and at pH 5.8.
Carbon Dioxide Treatments
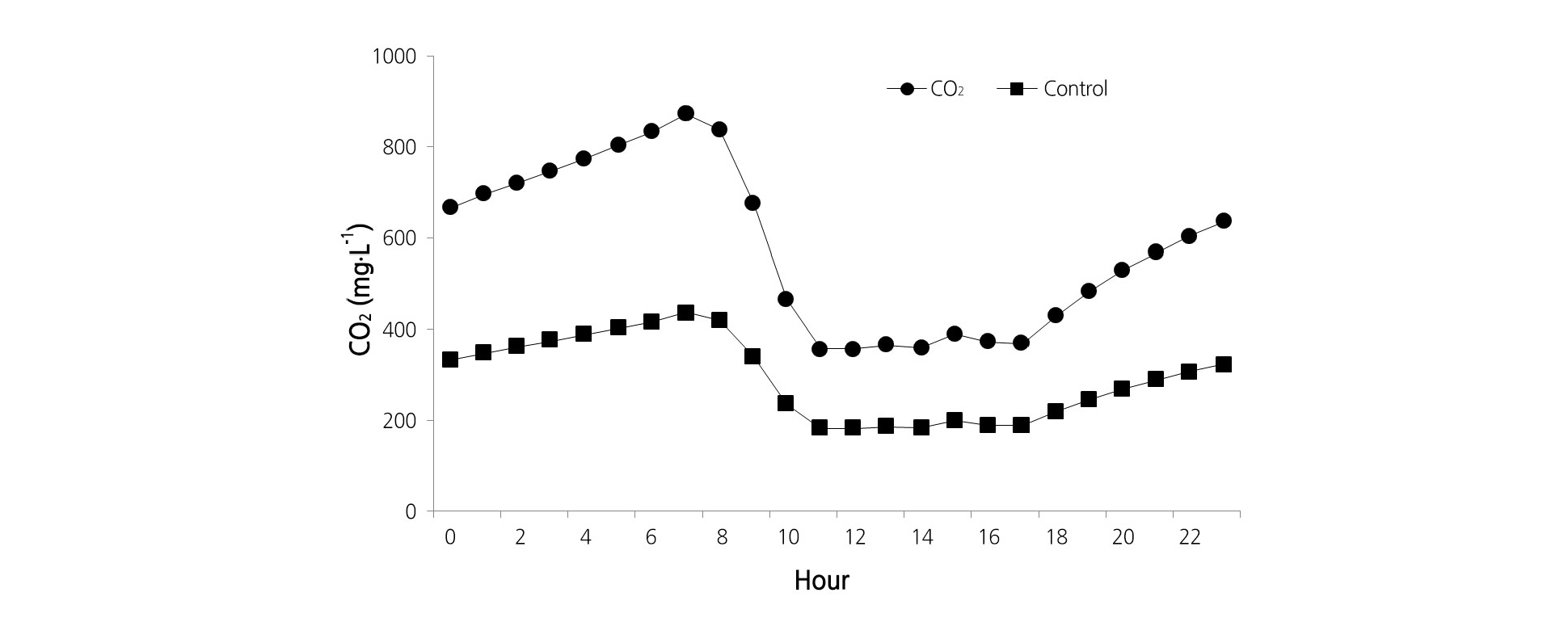
Each plastic house had air circulating and CO2 sensor systems (Mirae sensor, Seoul, Korea) to monitor the CO2 concentration. The CO2 gas was applied from the 12th of December 2016 to the 12th of March 2017. The CO2 concentration was controlled every 15 min from 7 a.m. to 9 a.m. and then every 30 min from 9 a.m. to 3 p.m. with a maintained set value of 600-800 mg·L-1 during the day (Fig. 1). The CO2 gas was automatically injected into the house from a LPG gas cylinder (2.8 ± 0.5 kPa) that was used for a gas heater system (KCH-20S, Katsura, Kyoto, Japan) and could produce 5.0 kg·h-1 CO2 gas. For the control treatment, the CO2 was applied to the house through natural ventilation when the temperature was higher than 23°C.
Measurements
Ten plants in each treatment were labeled in developing leaves and used for the measurement of leaf length and width without destruction of the plant. The photosynthetic rate was measured in four fully expanded leaves from plants in each treatment using a CO2 gas analyzer (LI-6400XRT, LI-COR Biosciences, Lincoln, NE, USA). Block temperature and standard CO2 concentration were kept at 25°C and 400 mg·L-1, respectively. The artificial photosynthetically active radiation (PAR) was gradually increased from 100 to 2,000 µmol·m-2·s-1 during all measurements.
Strawberry fruits were harvested at 2-day intervals when 60-70% of the fruits had turned red. At each harvest, yield data (fruit fresh weight) were determined. Acid and soluble sugar content was measured in 15 fruits for each treatment, which were harvested from the first, second, and third clusters using a picket brix-acidity meter (PAL-BX/ACID, Atago, Tokyo, Japan). The vertical and horizontal hardness of fruit were measured in the outer layer, about 5-10 mm apart from the fruit center using a meter equipped with a 3 mm probe (FR-5105, Rheo Meter Compac-100 II, Sun Scientific Co., Tokyo, Japan) after cutting 0.5 cm off the top and bottom of the fruit.
Fifteen fruits from the first and second clusters from fifteen plants in each treatment were labeled for the measurement of soluble carbohydrates (sucrose, glucose, and fructose). Samples were taken three times at 3-day intervals on the 6th and 13th of January 2017. The selected fruits were of similar size (average fruit length: 5-7 mm, average fruit diameter: 11-12 mm). As soon as the fruits were harvested, they were kept on dry ice prior to being freeze-dried. Then, the freeze-dried fruit was analyzed for soluble carbohydrates using HPLC (Ultimate 3,000, Dionex, Sunnyvale, CA, USA), a detector (Shodex RI-101, Showa Denko, Tokyo, Japan), sugar-pak column (300 × 6.5 mm, Waters, Milford, MA, USA), and analysis software (Chromeleon 6.0 Chromatography Data System Software, Dionex, Sunnyvale, CA, USA).
The data were analyzed for statistical differences using the SAS package (statistical analysis system, version 9.3, SAS Institute Inc.). The data were analyzed using ANOVA (analysis of variance) and t-test at 5%, *p < 0.05.
Results and Discussion
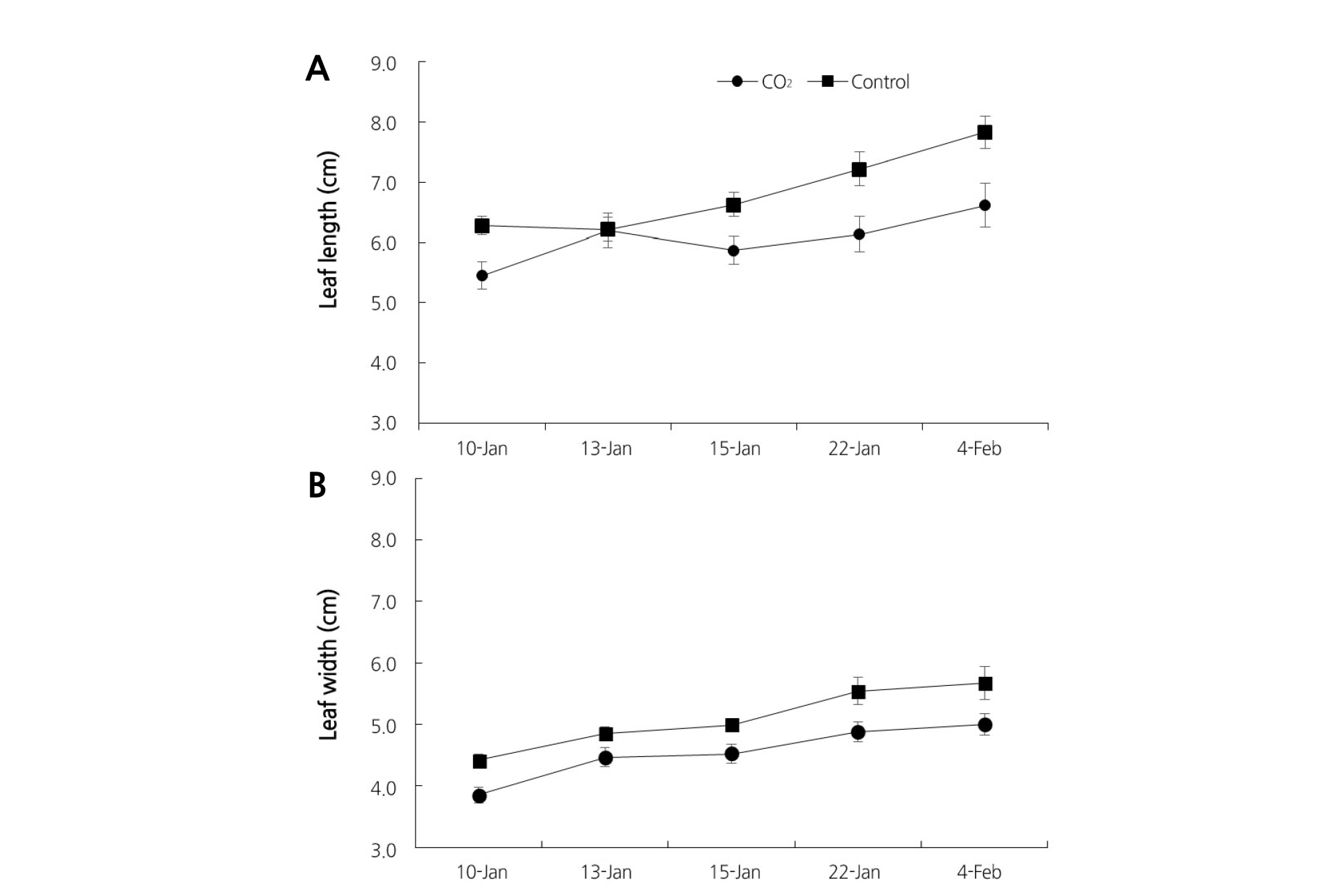
After the 30th day of CO2 fertilization, leaf lengths were 13.21% and 15.45% lower on the 10th of Jan. and 4th of Feb., respectively, with CO2 fertilization than in the control conditions (Fig. 2A). Leaf width was also significantly lower in the plants that received CO2 fertilization than that in the control. Leaf width was 12.70% and 11.81% lower on the 10th of Jan. and 4th of Feb., respectively, compared to plants that received the control treatment (Fig. 2B). This period was done at the same time vegetative and reproductive growth in strawberry.
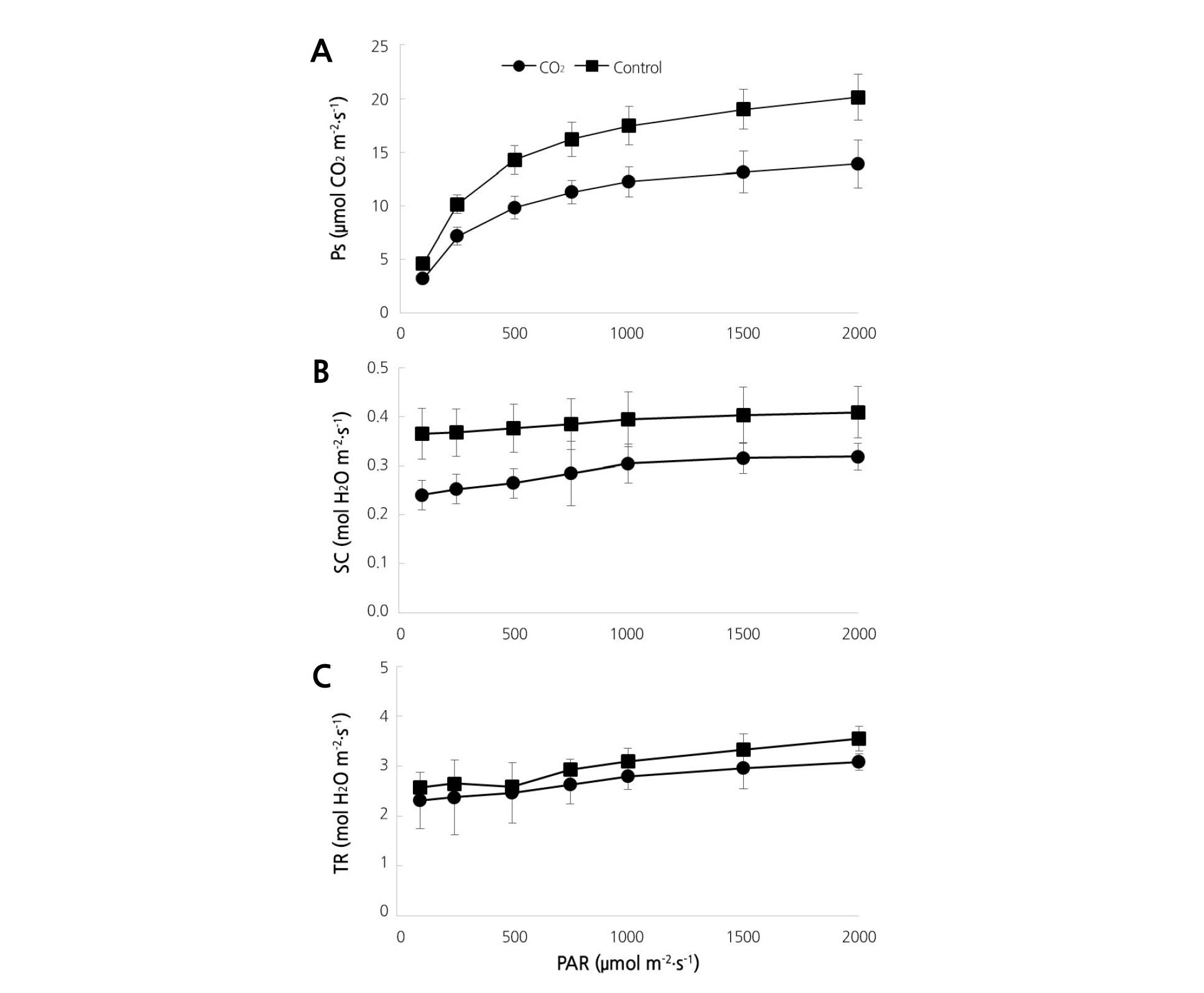
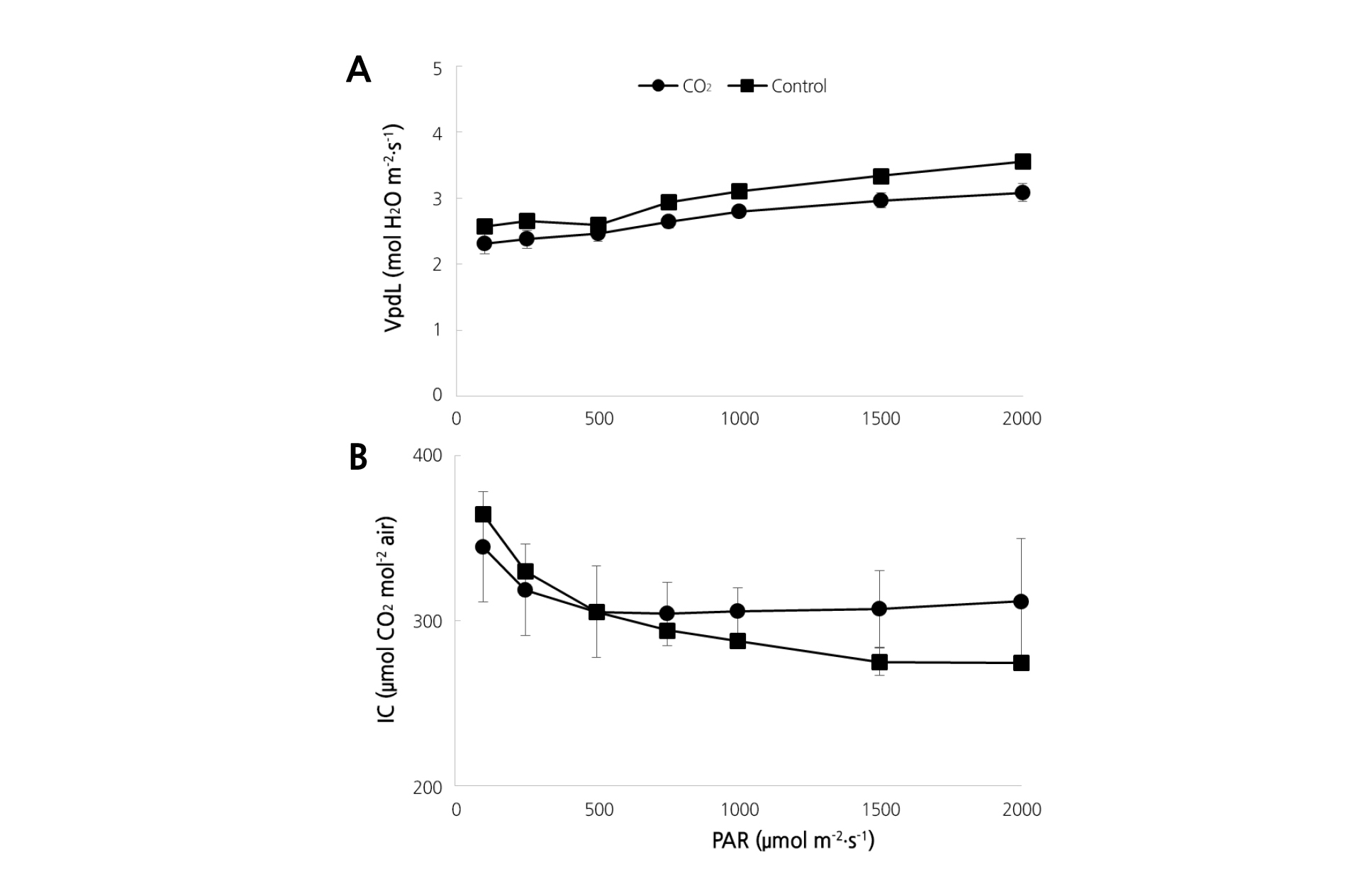
The photosynthetic rate in strawberry plants was measured by increasing photosynthetically active radiation (PAR) gradually from 100 to 2,000 µmol·m-2·s-1. We observed significantly lower rates in plants treated with CO2; rates of 30.9, 29.3, 31.1, 30.6, 29.9, 26.6, and 31.0% were measured at the PAR levels 100, 250, 500, 750, 1,000, 1,500, and 2,000 µmol·m-2·s-1, respectively (Fig. 3A). Stomatal conductivity was 22.8, 21.8, and 22.0% at PAR 1,000, 1,500, and 2,000 µmol·m-2·s-1, respectively, with CO2 fertilization, which was significantly lower than that measured in the control plants (Fig. 3B). Transpiration rates were slightly lower in the CO2 fertilization group at PAR between 100-1,500 µmol·m-2·s-1; specifically, the rate was 13.24% lower at PAR 2,000 µmol·m-2·s-1(Fig. 3C). The leaf to air vapor pressure deficit (VpdL) and the intercellular CO2 concentration were higher with CO2 fertilization compared to the control treatment (Fig. 4).
These results support previous results that were reviewed by Pospisilova and Catsky (1999), in which long-term elevated CO2 led to a decrease in stomatal conductance and transpiration rate, although the decline can be affected by environmental factors such as irradiance, temperature, and vapor pressure deficit. Lee and Lee (1994) also postulated that a decrease in photosynthesis under high CO2 (800 mg·L-1) conditions was associated with an increase of stomata resistance and decrease of transpiration rate.
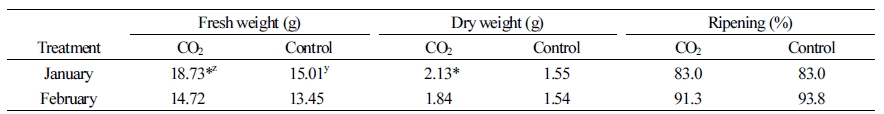
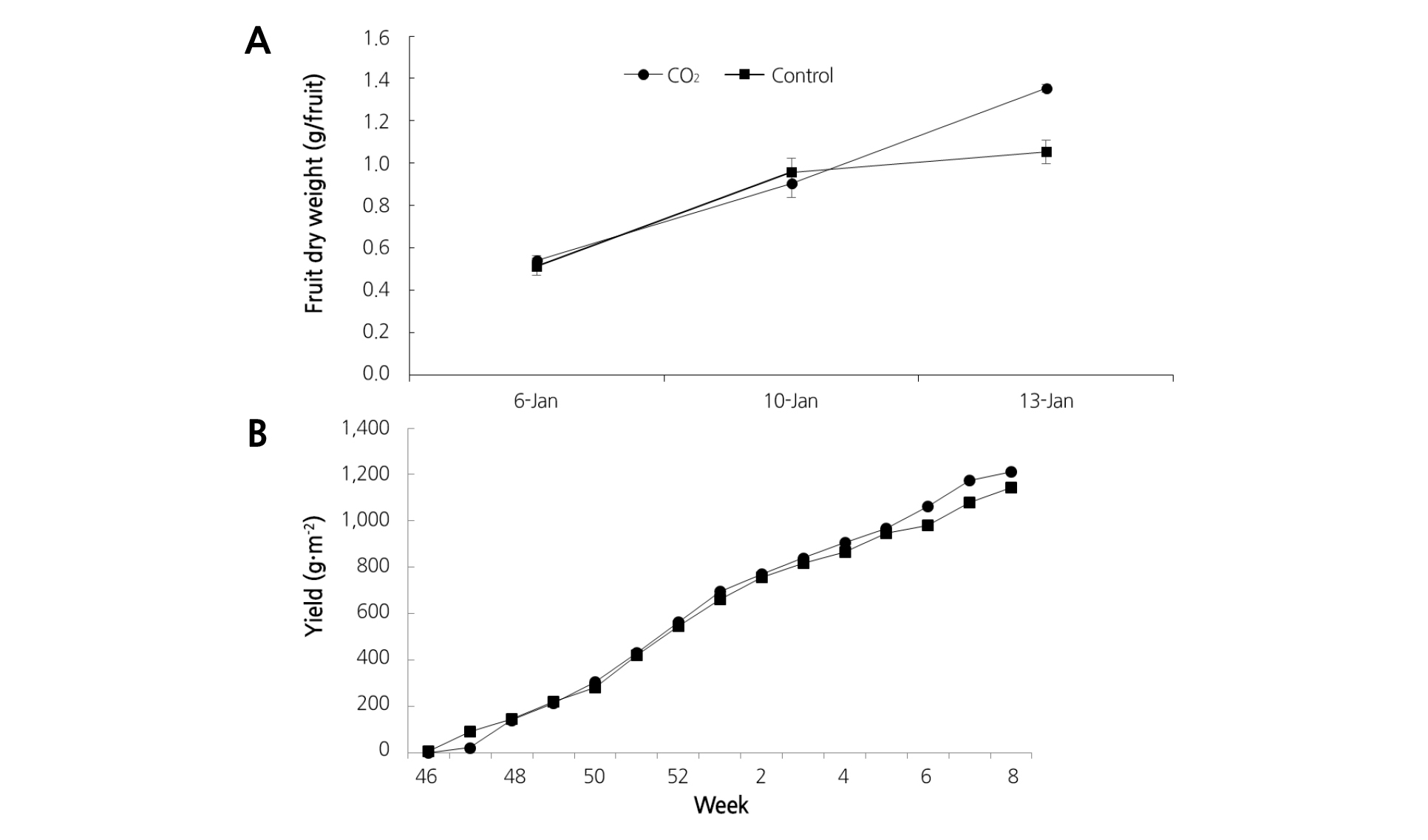
The fresh and dry weights of fruits with CO2 fertilization were both significantly higher (19.9% and 27.3%) than the untreated controls in January, but not in February (Table 1). There was also no significant difference in fruit ripening between the treatment and control groups (Table 1). Total yield was higher at 6 to 8 weeks when the CO2 was applied (Fig. 5), but it was not significant.
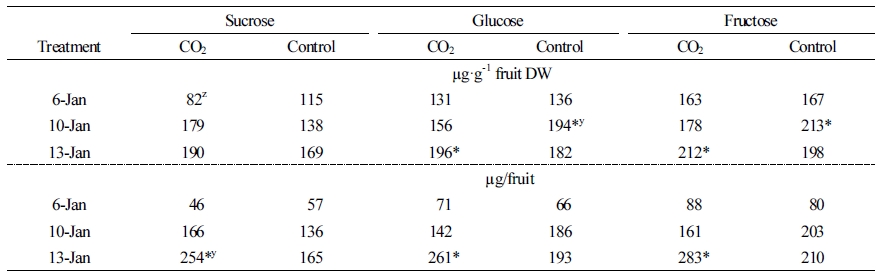
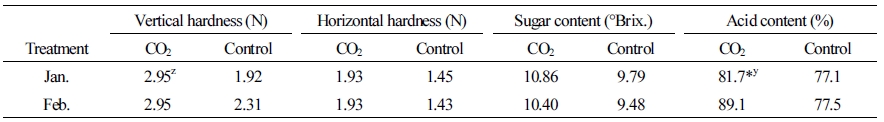
When the fruit carbohydrate concentration was measured, the initial sucrose concentration was lower in the plants treated with CO2 than in the control plants. However, at the second and last time points, this concentration was higher, about 41 µg·g-1 (30 µg/fruit) and 21 µg·g-1 (89 µg/fruit), respectively, than the control. Both glucose and fructose were also higher, measuring 14 µg·g-1 (68-73 µg/fruit) at the final time point (Table 2). Vertical and horizontal fruit hardness and soluble sugar content of plants grown in the CO2 fertilization conditions were slightly higher than those in the control conditions, although these differences were not statistically significant. The acid content of fruit grown in the CO2 fertilization was higher than the control when measured in January (Table 3).
These data support a previous study in which CO2 fertilization induced reproductive growth and improved fruit quality and yield (Nilsen et al., 1983). Our results show that the CO2 fertilization during hydroponic strawberry cultivation decreased leaf growth, which is consistent with the lower photosynthesis, stomatal conductivity, and transpiration rate; and the higher fruit carbohydrate content and fruit yield. In contrast, photosynthesis was more active in the control plants, which also had higher stomatal conductivity and transpiration, consistent with the greater leaf growth and lower fruit yield.
Soluble sugar is the most critical factor for fruit quality, 99% of which in strawberry fruits is sucrose, glucose and fructose (Montero et al., 1996). Reduced leaf growth accompanied by lower photosynthesis, transpiration, and stomatal conductivity with CO2 fertilization may have improved fruit growth by increasing photosynthates and water translocation to the fruit. CO2 fertilization in strawberry increased sugar content but decreased organic acid content (Jeong et al., 1996; Wang et al., 2003). These results indicate that CO2 has an effect on carbohydrate and organic acid metabolism. On the other hand, ‘Maehyang’ strawberry increased not only fruit hardness and sugar content but also acidity in CO2 fertilization compared to those in the non-CO2 control treatment after harvest (Choi et al., 2017).
Some studies suggest that feedback inhibition of photosynthesis with the long-term CO2 fertilization can restrict root growth and lead to nutrient insufficiency, carbohydrate accumulation and, subsequently, diminished photosynthetic capacity, which usually results from a decreased Rubisco activity (Bowes, 1991; Sicher et al., 1995).
Although long-term CO2 fertilization in the present study affected photosynthetic activity and transpiration rate, the CO2 concentration we applied was not as high as that used in previous studies. Thus, we speculate that the long-term CO2 fertilization of a protected strawberry crop can reduce vegetative growth and improve reproductive growth and fruit quality.