Introduction
Materials and Methods
Plant Materials Used for Stock Plants
Growth of Stock Plants and Runner Plants as Affected by PPF Level
Growth of Runner Plants as Affected by Container Volumes
Data Analysis
Results and Discussion
Growth of Stock Plants and Runner Plants as Affected by PPF Levels
Growth of Runner Plants as Affected by Container Volumes
Introduction
A form of asexual reproduction in strawberries is clonal growth, in which runner plants derive from runners and remain attached to their stock plants until their roots are sufficient to support themselves (Alpert and Mooney, 1986). The runner plants originate at runner tips; their leaves grow first, and then their roots and runners are formed (Nishizawa, 1994; Saito et al., 2008). The runners between the stock plants and the runner plants transfer resources from the stock plants to the runner plants to support growth(Roiloa et al., 2007). When stock and runner plants grow in heterogeneous environments, they share resources with each other to procure insufficient resources in their respective environments (Alpert and Stuefer, 1997).
The runners in short-day cultivars grown in Korea are generated under long day conditions and high temperatures (Verheul et al., 2006; Zahedi and Sarikhni, 2016), and their production rates increase under high photosynthetic photon flux (PPF) and CO2 concentrations (Chen et al., 1997; Kim et al., 2010). Using a plant factory with artificial lighting that specializes in transplant production, in which environmental conditions can be easily controlled, is appropriate for rapid and year- round transplant production. However, it is difficult to apply the conventional strawberry propagation methods to a plant factory because the size of stock plants used in the conventional propagation method is too large. Kozai (2016) suggested that the appropriate height of transplants produced in a plant factory should be less than 15 cm to maintain adequate air current and relative humidity, while the height of stock plants used in the conventional propagation method is approximately 25 cm and the length of their leaf blade and petiole are ca. 5 and 20 cm, respectively (Kim et al., 2010; Konsin et al., 2001).
To overcome the limitations of the conventional propagation methods for transplants production in a plant factory with multilayered shelves, Chun et al. (2012) developed a propagation method called the autotrophic transplant production method (ATPM). This method is performed as follows: (1) stock plants whose size (crown diameter: ca. 5 mm) is smaller than that for conventional methods (9 -13 mm; RDA, 2013) are used, (2) runner tips that have unfolded their bract (generated from the stock plants) are fixed on medium in 150 mL pots, (3) runner plants derived from the runner tips are separated from their stock plants when their crown diameter reaches the initial size of the stock plants (ca. 20 days after placing stock plants), and (4) they are then used as new stock plants for next propagation cycle.
We hypothesized that, if all of the runner plants’ nutrients and water can be received from stock plants and unfavorable environmental conditions for runner plants do not affect their growth while they are still connected with their stock plants, the efficiency of transplant production by ATPM in a plant factory can be improved by decreasing the light intensity or container volume for the runner plants. To confirm these hypotheses, we investigated (1) the effect of different light intensities applied to both stock and runner plants on the growth of runner plants, and (2) the effect of different container volumes for runner plants on the growth of runner plants.
Materials and Methods
Plant Materials Used for Stock Plants
Strawberry plants (Fragaria × ananassa Duch. cv. Maehyang) used for stock plants were selected based on crown diameter of 5.0 ± 0.5 mm. Selected stock plants had two compound leaves and one runner with an unfolded bract. They were planted into 150 mL containers using a 32- cell cutting plug tray (Bumnong Co. Ltd., Jeongeup, Korea) filled with commercial medium (Plant World; Nongwoo Bio Co. Ltd., Suwon, Korea).
Growth of Stock Plants and Runner Plants as Affected by PPF Level
Six stock plants, planted in 150 mL containers, were arranged in a row on the bed and their runners were fixed in 150 mL containers filled with commercial medium for the formation of runner plants. The distance between plants in a row was 120 mm and between rows was 240 mm. Nine different treatments composed of three different light intensities were applied to both stock and runner plants; PPF 100 (104.8 ± 6.9 μmol∙m-2∙s-1), 200 (192.5 ± 5.6 μmol∙m-2∙s-1), and 400 (391.7 ± 12.8 μmol∙m-2∙s-1). Light intensity was controlled by changing the number of 32W cool white fluorescent lamps or using tinted films. Air temperature during the light/dark-period was maintained at 27°C/23°C, and the photo-period and CO2 concentration were 16 h and 800 μmol∙mol-1, respectively. A closed irrigation system was used in the plant factory and plants were sub-irrigated with Yamazaki nutrient solution for strawberries (EC 0.7 dS∙m-1; Yamazaki, 1982) for 10 minutes once a day. At 20 days after placing (DAP), six stock and runner plants from each treatment were selected, and the number of compound leaves and runners, crown diameter, leaf area, and dry weight were measured.
Growth of Runner Plants as Affected by Container Volumes
Six stock plants, planted in 150 mL containers, were arranged in a row on the bed and the distance between plants in a row was 120 mm. We set four treatments with different container volumes (21, 34, 73, and 150 mL) using 32, 50, 72, and 128-cell cutting plug trays (Bumnong Co. Ltd., Jeongeup, Korea) for runner plants. Runner tips from stock plants were fixed into containers with different volumes and cultivated for 20 days in a plant factory. The light intensity and photo-period were 210 μmol∙m-2∙s-1 and 16 h, respectively. Air temperature during the light/dark-period was maintained at 27°C/23°C and CO2 concentration was 800μmol∙mol-1. A closed irrigation system was used in the plant factory and plants were sub-irrigated with Yamazaki nutrient solution for strawberries (EC 0.7 dS∙m-1; Yamazaki, 1982) for 10 minutes once a day. At 20 DAP, 15 runner plants from each treatment were selected, and the number of compound leaves and runners, crown diameter, leaf area, and dry weight were measured. To investigate the subsequent growth of runner plants after being separated from their stock plants, ten runner plants separated at 20 DAP were transplanted into plastic pots (ø 90 mm) filled with the commercial medium and grown for 35 days in a greenhouse located in Suwon, Korea (E 127.0°, N 37.3°). 35 days after transplanting, the number of compound leaves and runners, crown diameter, leaf area, and dry weight were measured.
Data Analysis
The experimental data was analyzed using SAS 9.2 (SAS Institute Inc., Cary, NC, USA) with Duncan’s multiple range tests. Treatment differences were considered significant at a level of 5% probability.
Results and Discussion
Growth of Stock Plants and Runner Plants as Affected by PPF Levels
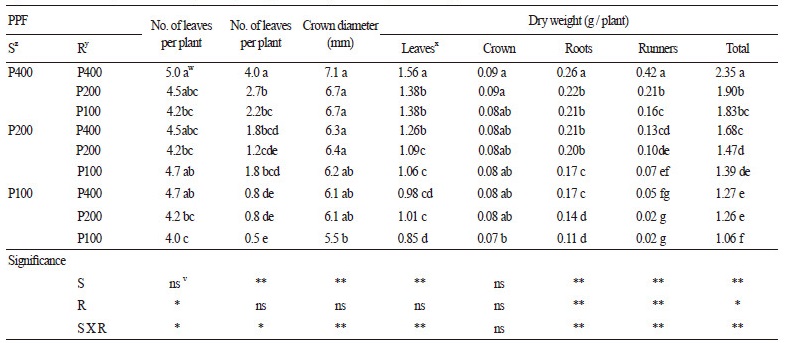
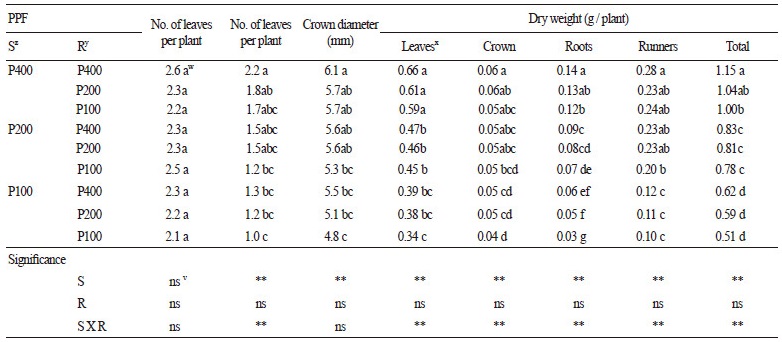
Tables 1 and 2 show the growth of stock plants and runner plants, respectively, as affected by PPF level at 20 DAP. The growth of both stock and runner plants was greatest when both were grown under 400 μmol∙m-1∙s-1, and those increased as PPF elevated. The growth of the runner plants was affected by the PPF of their stock plants regardless of their own PPF level. The dry weight of leaves in the stock plants was affected by PPF regardless of their runner plants’ PPF levels; however, their root, runner, and total dry weights were affected by the PPFs of both the stock and their runner plants. When considering the sum of the PPF applied to both stock and runner plants [ 600 (400 + 200 or 200 + 400; stock plant + runner plant), 500 (400 + 100 or 100 + 400), 300 (200 + 100 or 100 + 200)], the total dry weight of stock and runner plants increased as the PPF of the stock plants increased. Kim et al. (2010) reported that increasing PPF levels promoted the formation of runners and runner plants in a closed transplant production system until 280 μmol∙m-2∙s-1. In this study, the number and dry weight of runners derived from runner plants were not significantly affected by the runner plants’ PPF level. However, the number and dry weight of runners from stock plants increased as their PPF level increased. In clonal plants, such as strawberry, assimilates and nutrients tend to move from older to younger plants because they diffuse from higher to lower concentrations until the system equilibrates, and assimilate and nutrient pools of older plants are commonly greater than those of younger plants (Caraco and Kelly, 1991).
Fig. 1 shows that proportion of each organ in total dry weight of the stock plants. Proportion of leaves decreased as the PPFs applied to both stock and runner plants increased while that of crown decreased as PPF for stock plants increased. Strawberry plants connected to each other may overcome light deficiency by translocating assimilates from a plant with a high supply to a plant with a low supply by enhancing the leaf growth of the plants exposed to higher light intensity (Roila et al., 2007). However, assimilates produced by the runner plants might be used for establishment of them until 20 DAP as they could not grow independently, and they might attract assimilates and nutrients from connected stock plants due to younger than the stock plants (Alpert, 1996; Friedman and Alpert, 1991; Savini et al., 2008). As assimilates increase under higher light intensities (Kinet et al., 1985) and runner plants under lower PPF might attract more assimilates from their stock plants, the proportional weight of leaves in the stock plants might be higher when the PPF applied to the runners is lower.
Proportion of runner enhanced as the PPFs on both the stock and runner plants increased (Fig. 1), and the dry weight of their runners also promoted as both PPFs increased (Table 1). However, proportion of the runner plants was not significantly different by both PPF levels, while total dry weight of them increased when only the PPF for stock plants increased (Table 2). Runner growth depends on the amount of surplus resources for propagules because a new runner appears only when plants have sufficient resources (Alpert, 1991). Therefore, propagation rate will enhance as PPFs on both the stock and runner plants until ca.400 μmol∙m-2∙s-1, and runner plants under unfavorable environment conditions such as low light intensity by shading can be grow sufficiently by receiving assimilates or nutrients from stock plants growing under favorable environment conditions.
Fig. 1.
Proportion of each organ in the total dry weight of stock plants in ‛Maehyang’ strawberry as affected by the light intensity applied to stock and runner plants 20 days after placing in a plant factory. Letters, a-e indicate significant differences according to Duncan’s multiple range test at p ≤ 5% (n = 6)
Growth of Runner Plants as Affected by Container Volumes
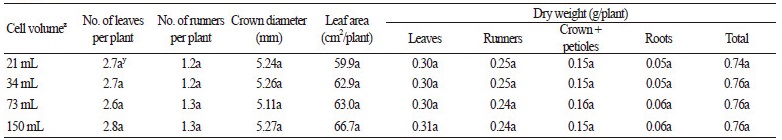
We found that the growth of runner plants was dependent on the light intensity applied to the stock plants but not on the light intensity applied to the runner plants. We hypothesized that using small volume containers for runner plants, which would decrease the distance between stock and runner plants and might decrease the light intensity for runner plants due to shading, would not negatively affect their growth. To confirm this hypothesis, we investigated the effect of different container volumes on the growth of runner plants. The numbers of leaves and runners, crown diameter, leaf area, and dry weight of runner plants were not significantly affected by container volume (Table 3). We fixed runner tips whose bract had just unfolded and where the root had not yet been established. The roots emerged after approximately 6 DAP and grew as much as possible until being separated from the stock plant at 20 DAP (Park et al., 2008). Until this point, they might receive water and mineral nutrients from their stock plants due the immaturity of their own roots (Alpert and Mooney, 1986; Friedman and Alpert, 1991). Moreover, as strawberry is clonal, runner plants connected to their stock plants can overcome stress induced by environmental conditions through support from the stock plants (Caraco and Kelly, 1991). In particular, a potential water gradient may exist from a stock plant to a runner when the runner plant has not rooted (Alpert and Mooney, 1986). As roots of the runner plants grew for just 14 days and runner plants can receive water from their stock plants, those grown in the smallest containers (21 mL/cell) did not show a reduction in growth due to the small container volume.
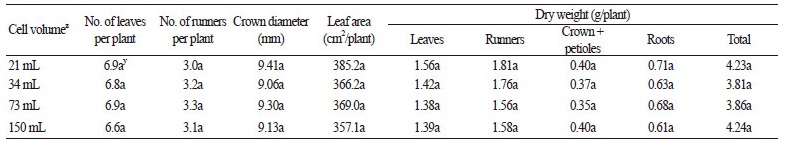
Runner plants separated from their stock plants at 20 DAP were transplanted into pots and grown until 35 days after transplanting. Table 4 shows growth and development of runner plants 35 days after transplanting. The growth of runner plants after transplanting into larger containers was not reduced by decreasing the container volume before separating. Therefore, until 20 DAP, runner plants connected with their stock plants can be grown in 21 mL containers, which will both reduce the quantity of medium consumed and increase space efficiency when strawberry transplants are produced in a plant factory.
In this study, we confirmed that the growth of runner plants is primarily affected by the environmental conditions of the stock plants, and not the conditions (light intensity, container volume) of the runner plants prior to separation. Based on these results, the efficiency of strawberry transplant production by ATPM in a plant factory can be dramatically improved by reducing the resources provided to runner plants (i.e. light energy, medium quantity, space utilization, etc.) prior to separation.