Introduction
Materials and Methods
Plant materials
Fruit quality attributes
Determination of total carotenoids, total phenolic, and flavonoids compounds
Determination of total antioxidant scavenging capacities
Analysis of individual phenolic compounds
Statistical analyses
Results
Fruit visual appearance and physiological disorders
Fruit quality attributes
Color responses of alginate-coated kiwifruit
Responses of phenolic compounds, carotenoids, ascorbic acid, and total antioxidant scavenging capacities
Correlation responses to alginate coating treatment
Discussion
Conclusion
Introduction
Since the commercial release of the gold-fleshed ‘Haegeum’ kiwifruit (Actinidia chinensis) cultivar, its high disease resistance during cultivation and postharvest has enabled high yields in the southern part of Korea (Kim et al. 2015). The ‘Haegeum’ cultivar has relatively high contents of total phenolics and total flavonoids compared with the other Actinidia spp., along with strong total antioxidant scavenging capacities (Lim et al. 2014; Lee et al. 2015a; Hwang et al. 2017). The phenolic compounds and total antioxidant scavenging capacities are higher in the skin and flesh tissues of the ‘Haegeum’ cultivar than in the other cultivars at harvest (Lee et al. 2015b). Additionally, the viability of neuronal PC-12 cells upon H2O2-induced oxidative stress determined using 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium bromide (MTT) assay is the highest in ‘Haegeum’ cultivar compared with the other Actinidia species (Lee et al. 2015b). Anti-dementia activity is also highly prominent in the ‘Haegeum’ cultivar, as well as in numerous Actinidia species (Lim et al. 2014; Hwang et al. 2017). Furthermore, the levels of total phenolics, total flavonoids, total antioxidant scavenging capacities, and reducing power are higher in gold-fleshed ‘Haegeum’ cultivar than in green-fleshed ‘Hayward’ cultivar during shelf life (Lim et al. 2018; Choi et al. 2022a) and long-term cold storage, along with higher level of ascorbic acid (Choi et al. 2022b). Nonetheless, fruit respiration rate and fruit weight loss are higher in ‘Haegeum’ than in ‘Hayward’ cultivar at short-term shelf life and thus flesh firmness and soluble solids content (SSC) are lower in ‘Haegeum’ cultivar (Choi et al. 2022b). By contrast, flesh firmness is lower in the ‘Haegeum’ cultivar than in the ‘Hayward’ cultivar during cold storage; however, SSC level is higher in the ‘Haegeum’ cultivar (Kim et al. 2023). Fruit quality attributes, such as firmness, weight loss, respiration, and ethylene production rate, are affected by the kiwifruit cultivar and harvest time during shelf life (Shin et al. 2020; Tilahun et al. 2020). Additionally, the cultivar ‘Haegeum’ is relatively susceptible to fruit decay during cold storage (Choi et al. 2019). Necrotic peel disorders, such as brown colored and pitted symptoms have been observed in fruit of A. arguta during cold storage (Park et al. 2025; Peng et al. 2025). Physiological storage disorders, such as fruit shriveling and decay, are strongly influenced by storage temperature in the fruit of the ‘Autumn Sense’ hardy kiwifruit cultivar during cold storage (Park et al. 2024a).
Several techniques have been developed to control or at least suppress the incidence of physiological disorders during cold storage and shelf life in kiwifruits. During both the shelf life and cold storage, fruit quality attributes, the incidence of physical and physiological disorders are differentially affected by the type of packaging containers used for hardy kiwifruit cultivars (Park et al. 2024b). For example, one of these techniques (temperature-controlling approach) entails the cold-storage of hardy kiwifruit cultivars (Park et al. 2024a). The 1-methylcyclopropene (1-MCP) technology has also been broadly used to retain the quality of ‘Hayward’ kiwifruit and ‘Cheongsan’ hardy kiwifruit during shelf life (Choi et al. 2023) and cold storage (Lim et al. 2016), respectively. Preharvest Ca-chitosan application can effectively delay the reduction of flesh firmness in cold-stored fruits of ‘Garmrok’ kiwifruit (Kumarihami et al. 2020). Nonetheless, alginate-based edible coating treatments, one of the numerous potential applications and technologies used to retain and control the incidence of physiological disorders, have been introduced to kiwifruit and applied to evaluation of the quality in fresh-cut kiwifruit at harvest (Li et al. 2017) and cold-stored hardy kiwifruit (Xiong et al. 2024). Furthermore, alginate oligosaccharide coating enhances fruit resistance to postharvest decay in kiwifruit at ambient temperatures during shelf life (Zhuo et al. 2022). Effectiveness of alginate oligosaccharide coating treatment is dosage-dependent on the control of gray mold, blue mold, and black rot in the fruit of ‘Hongyang’ kiwifruit cultivar stored at 25°C during shelf life (Liu et al. 2020).
In this study, we hypothesized that applying an alginate coating before harvest might help preserve the fruit quality attributes, modulating secondary metabolites, and reducing physiological disorders in the fruit of gold-fleshed ‘Haegeum’ kiwifruit cultivar during shelf life. Thus, the aim of this study was to assess the effectiveness of preharvest alginate coating on fruit quality attributes, total and individual phenolic compounds, ascorbic acid, total antioxidant scavenging capacity (ABTS and DPPH assays), and the incidence and severity of physiological disorders, such as fruit shriveling and decay. Additionally, we aimed to clarify the relationship between these physiological disorders and the response variables in the fruit of the gold-fleshed ‘Haegeum’ kiwifruit cultivar during shelf life.
Materials and Methods
Plant materials
The ‘Haegeum’ kiwifruit (A. chinensis Planch.) cultivar were harvested on November 13, 2024, in Haenam, Jeollanam-do, Republic of Korea (34°34'27"N and 124°52'17"E). Fruits were harvested at 176 days after full bloom (full bloom date, May 21, 2024; harvest date, November 13, 2024). Fruits were grown using conventional cultivation methods, and 30 fruits with the same conditions per treatment were randomly marked on trees of the same age and size. For alginate coating treatments at 0, 1, and 2%, sodium alginate (Sigma-Aldrich, St. Louis, MO, USA) was prepared in distilled water at concentrations of 1% and 2% (w/v). To completely dissolve sodium alginate, the mixture was autoclaved and then stirred until fully dissolved. No additional surfactants were added. The fruits were dipped once in the solution one day before harvest and then air-dried for 16 hours, while still hanging on the plant. The treated fruits were harvested and immediately transferred to the Chung-Ang University in Anseong, Republic of Korea. The first half of 15 fruits per treatment were used solely for measuring daily fruit respiration and ethylene production rates for up to 14 days, whereas the second half were used for the measurement of fruit quality attributes, including flesh firmness, SSC, titratable acidity (TA), SSC/TA, color variables of three different fruit tissues, including peel, cortex and core, as well as total and individual phenolic compounds, total carotenoids, ascorbic acid, and total antioxidant scavenging capacities assessed using assays of ABTS and DPPH, after 2 weeks of shelf life.
Fruit quality attributes
Fruit quality attributes, including fresh weight, respiration rate, ethylene production rate, color variables, firmness, SSC, and TA were evaluated according to a previous study (Lwin and Lee 2021). Fruit weight was measured by an analytical WZ-3A scale (WZ-3A, CAS Corp., Yangju, Republic of Korea) at harvest and during the 14-day shelf-life period. The color variables of three different tissues, including peel, cortex, and core, were determined by chroma meter (CR-400, Minolta Co., Osaka, Japan).
Ethylene (C2H4) production and respiration (CO2) rates were measured by the following methods. In 0.92 L containers, three fruits per replicate were incubated for 1 h. There were five replications (n = 5) and 1 mL gas sample was collected in triplicate from each plastic container. To determine the ethylene production rate, the samples were injected into an 8890 GC system (Agilent Tech., Inc., Santa Clara, CA, USA) with CP-Al2O3/Na2SO4 GC column (CP7568, 50 m × 0.53 mm × 10 µm, Agilent Tech., Inc., Santa Clara, CA, USA). The setting temperature of injector, flame ionization detector (FID), and oven were set to 200, 250, and 70°C, respectively. For the respiration rate (CO2), samples were directly injected into a YL6500GC system (YL Instrument Co., Ltd., Anyang, Republic of Korea) with Porapak Q packed GC column (Supelco Inc., Bellefonte, PA, USA). The temperature of injector, thermal conductivity detector (TCD), and oven were configured to 200, 150, and 50°C, respectively. The flow rate of helium (He) was set at 30 mL min-1 for both GC instruments. The ethylene production and respiration rates were expressed as nL kg-1 s-1 and µL kg-1 s-1, respectively.
Flesh firmness was measured with a 2-mm plunger on the 2 sides of fruit at the equator portions, and setting as 2 mm s-1 for pretest and 10 mm s-1 for posttest by Brookfield CT3 texture analyzer (CT3 4500, Brookfield, Inc., Middleborough, MA, USA). The firmness was expressed in Newtons (N). Fruit juice was squeezed using a juicer to determine SSC (%) by PAL-1 refractometer (Atago Co., Ltd., Tokyo, Japan). TA (%) was measured by an auto titrator (EasyPlus, Mettler-Toledo, LLC, Columbus, OH, USA), with 0.1 M NaOH solution to titrate the juice until the pH reached 8.2 (Park et al. 2024b).
Physiological disorders were assessed by measuring fruit shriveling and peel decay as severity. Severity was quantified based on the damaged area of the fruit, using the following scale: 0 = 0%, 1 = 1%–10%, 2 = 11%–25%, 3 = 26%–50%, 4 = 51%–75%, and 5 = 76%–100% (Byeon et al. 2023; Lwin et al. 2023b).
Determination of total carotenoids, total phenolic, and flavonoids compounds
For total carotenoid concentrations, lyophilized samples (1 g) were extracted with 10 mL of 80% acetone for 2 h in the dark. The extract was then centrifuged using a Labogene 1580R centrifuge (Gyrozen Co., Ltd., Gimpo, Republic of Korea) at 3,000 rpm for 15 min at 4°C, and subsequently filtered through Whatman filter paper (No. 51, Merck KGaA, Darmstadt, Germany). The absorbance was measured at 663, 646, and 470 nm by Epoch 2 Microplate Reader (BioTek Instruments, Inc., Winooski, VT, USA). The total carotenoid concentrations were then calculated in mg per kg of dry weight using the equations of Arnon (1949) as modified by previous study (Latt et al. 2023).
For total concentrations of phenolic compounds and flavonoids, the frozen sample (0.5 g) was homogenized for 1 min with 5 mL 80% aqueous methanol, and vortexed for 1 min. The mixture was sonicated for 20 min on ice and centrifuged in the condition of 10,000 ×g and 4°C for 10 min. Supernatant was filtered using a syringe membrane filter (BS45-PV13, BioFACT Co., Ltd., Daejeon, The Republic of Korea). Total phenolic compounds were determined using the modified Folin–Ciocalteu method (Latt et al. 2025). Briefly, 0.2 mL of extract was supplemented with 0.2 mL of Folin solution and 2.6 mL of water. The mixture was then incubated at 21°C for 6 min. Subsequently, a 7% sodium carbonate (Na2CO3; 2 mL) aqueous solution was added and vortexed. After 90 min, the optical density of the mixture was measured at 750 nm by Microplate Reader (Epoch 2, BioTek Instruments, Inc., Winooski, VT, USA). After measuring absorbance, the total phenolic content was reported as milligrams of gallic acid equivalent (GAE) per kilogram fresh weight. The total flavonoid concentrations were quantified using a previously reported method with some modifications (Lwin et al. 2023a). Diluted sample (1 mL) was vortexed with 0.3 mL of 5% NaNO₃ solution and 4 mL of distilled water for 5 min. A 10% AlCl₃ solution (0.3 mL) was then introduced and the mixture was vortexed. Following a 6 min incubation, 1 M NaOH (2 mL) and 2.4 mL distilled water were added. The sample was measured at the absorbance at 510 nm using spectrophotometer (Epoch 2, BioTek Instruments, Inc., Winooski, VT, USA), and the total flavonoids contents were reported as milligrams of catechin equivalents per kilogram of fresh weight.
Determination of total antioxidant scavenging capacities
For antioxidant scavenging capacities, assays of ABTS and DPPH were held using a method developed by Re et al. (1999) with minor modifications of previous study (Latt et al. 2024). For the assays, 0.5g of frozen ground samples were mixed with 5 mL of 80% methanol and centrifuged at 3,000 rpm for 15 min at 4°C. The extract of 10 µL was mixed with the working solution (190 µL) in a microplate. The reading absorbance were 734 and 517 nm, respectively by Microplate Reader (Epoch 2, BioTek Instruments, Inc., Winooski, VT, USA). Capacities of ABTS and DPPH were calculated by previous study (Latt et al. 2024).
Analysis of individual phenolic compounds
The individual phenolic compounds were analyzed based on a previous study, with some modifications (Park et al. 2025). Briefly, 0.5 g of lyophilized samples were ground, extracted with 5.0 mL of 70% methanol containing formic acid (2%), and vortexed. The extract was sonicated for 20 min on ice and centrifuged at 10,000 ×g and 4°C for 10 min. The extract was then filtered with a PVDF syringe filter (BS45-PV13; BioFACT Co., Ltd., Daejeon, Republic of Korea). The analysis of individual phenolic compounds was performed by Ultimate 3000 UPLC instrument (Thermo Fisher Scientific Inc., Waltham, MA, USA) equipped with a UV detector (Thermo Fisher Scientific Inc., Waltham, MA, USA) and XDB-C18 column (990967-902; Agilent Tech., Inc., Santa Clara, CA, USA). The temperature condition of column was set at 30°C, injection volume 10 µL, and with the flow rate 1.0 mL min-1. Solvent A: 0.5% formic acid in HPLC-grade water and solvent B: 0.5% formic acid in acetonitrile. The mobile phase was programmed as follows: 95% A (0 min), 90% A (20 min), 78% A (40 min), 60% A (55 min), and 5% A (60 min). Individual phenolic compounds were identified and quantified according to their retention times compared to authentic standards. A regression equation was developed using the response area to predict the concentration. The value of the compounds was expressed as mg kg-1 on a dry weight basis.
Statistical analyses
For the measurements of ethylene production rate, weight loss, and flesh firmness, 15 individual fruits per treatment (n = 15) were taken, and these fruits were treated as independent biological replicates in the analysis for these continuous time points variables. The response variables were determined with the analysis of variance (ANOVA) test and mean difference were performed with the LSD test at a p < 0.05 level. For the remaining biochemical analyses (including phenolics and antioxidant capacity), 5 fruits were pooled to form one biological replicate and three replicates (n = 3) were used per treatment, and the Duncan’s multiple range test was applied to examine the effect of alginate coating treatment using SAS (v.9.3, SAS Institute Inc., Cary, NC, USA). Graphs were visualized and statistical outcomes were analyzed using SigmaPlot (v.10.0, Systat Software Inc., San Jose, CA, USA). Prior to analysis, the dataset underwent auto-scaling, wherein each variable was mean-centered and normalized by its standard deviation. Hierarchical clustering was conducted using Pearson’s distance metrics and Ward’s linkage method, resulting in the construction of a normalized heatmap matrix using MetaboAnalyst 6.0 (https://www.metaboanalyst.ca/). Finally, a correlation coefficient network was constructed to examine the interrelationships among the variables and the results were performed using MetScape (v3.1.3) integrated with Cytoscape (v3.8.2) (https://cytoscape.org/).
Results
Fruit visual appearance and physiological disorders
The external and internal visual appearances of the fruits were shown in Fig. 1A. Fruit decay was detected only in 0% alginate treatment during shelf life. In addition, fruit shriveling decreased with increasing the concentration of alginate coating during shelf life (Fig. 1B).
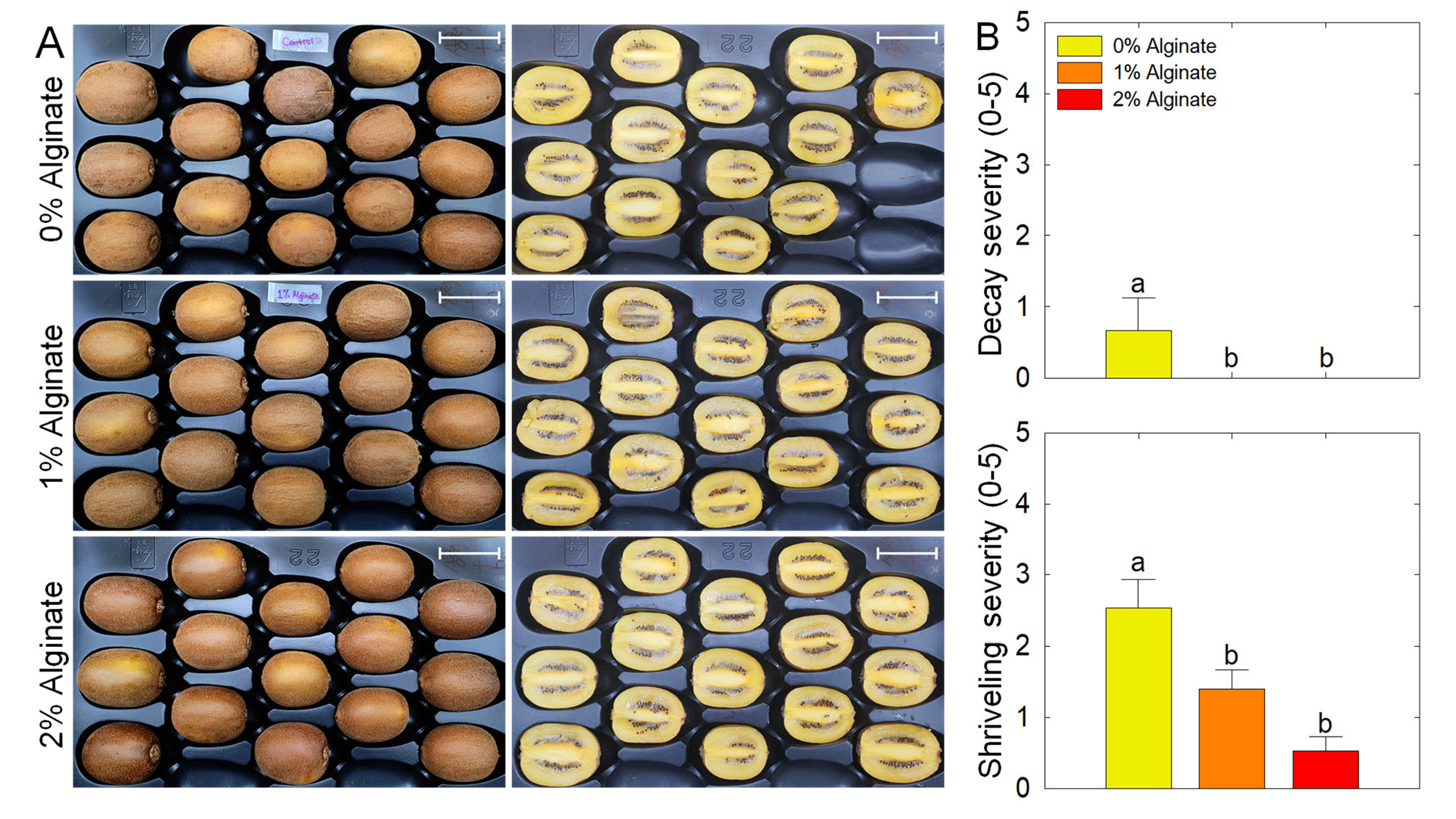
Fig. 1.
Visual appearance and the severity of fruit decay and shriveling in the fruit of ‘Haegeum’ kiwifruit coated with the different concentrations of alginate 1 day before harvest and stored at 21°C for up to 14 days (A). Severity of decay and shriveling was scored as follows: 0 = 0%, 1 = 1%–10%, 2 = 11%–25%, 3 = 26%–50%, 4 = 51%–75%, and 5 = 76%–100% of the area (B). Data indicate the mean of 15 kiwifruits as replicates (n = 15). Each letter on the bars denotes the results of the least significant difference (LSD) test at p = 0.05. The white bar on each photo indicates a length of 5 cm.
Fruit quality attributes
Fresh weight loss gradually increased with the shelf life period, regardless of the concentration of alginate. However, alginate coating reduced weight loss compared with the uncoated control fruit. No significant differences were observed in quality outcomes of kiwifruits between 1% and 2% alginate coating treatments, except for firmness. The highest peak of ethylene production rate in 0% alginate coating treatment occurred on day 11 during the 14-day of shelf life, whereas the ethylene production rate was sharply increased at the end of the shelf life in the 1% alginate coating treatment. Interestingly, the ethylene production rate was lowest in 2% alginate coating treatment during shelf life compared with the other treatments. Compared with the ethylene production rate, fruit respiration rate did not change until 6-days of shelf life, regardless of alginate coating concentration, although a clear difference exists in fruit respiration rate depending on the concentration of alginate coating. However, in the second half of shelf life, fruit respiration rate was sharply increased in 0% alginate coating treatment and peaked after 9-days of shelf life and inconsistently retained at a similar level. Fruit respiration rate gradually increased in 1% alginate coating treatment until day 13 during shelf life and then sharply declined at the end of the shelf life. Interestingly, fruit respiration rate was slow and slightly increased in 2% alginate coating treatment during the 14-day of shelf life. Nonetheless, fruit respiration rate was highest in 0% alginate coating treatment, whereas the lowest in 2% alginate coating treatment throughout shelf life of the fruits (Fig. 2A).
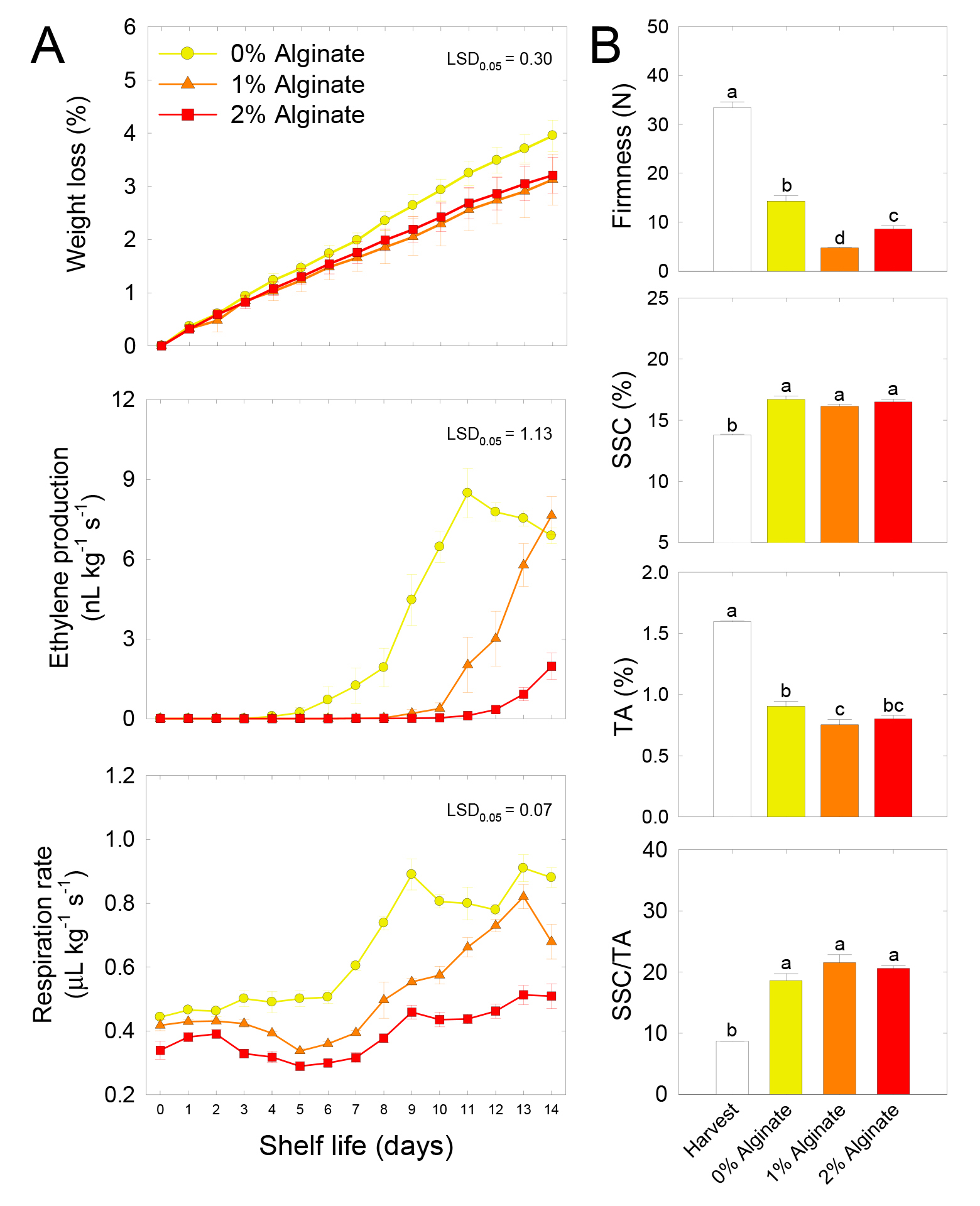
Fig. 2.
Physiological responses of fruit fresh weight loss, ethylene production rate, fruit respiration rate (A), flesh firmness, soluble solids content (SSC), titratable acidity (TA), and SSC/TA ratio (B) in the fruit of ‘Haegeum’ kiwifruit coated with different concentrations of alginate at harvest and then stored at 21°C for 14 days. Firmness data indicate the mean of 15 kiwifruits as replicates (n = 15), and the results for SSC, TA, and SSC/TA indicate the mean of three replicates (n = 3) ± standard error (SE). The least significant difference (LSD) test was used to examine significance at p = 0.05 (A). Each letter on the bars indicates the results of the LSD test at p = 0.05 (B).
Flesh firmness decreased throughout shelf life compared with that at harvest. After 14-day of shelf life, flesh firmness was highest in 0% alginate coating treatment and lowest in 1% alginate coating treatment. SSC increased during shelf life compared with the harvest time point, irrespective of alginate coating concentrations. That is, the SSC level did not differ among alginate coating concentrations during shelf life. However, TA decreased during shelf life compared with the harvest time point, and TA was highest in the 0% alginate coating treatment and lowest in the 1% alginate coating treatment during shelf life. Thus, the ratio of SSC to TA increased during shelf life compared with the harvest time point, but no statistical differences were detected among alginate coating concentrations during shelf life (Fig. 2B).
Color responses of alginate-coated kiwifruit
Peel lightness (L*) decreased during shelf life, compared with the harvest time point, regardless of the concentration of the alginate coating. Nonetheless, peel L* value was lowest in 2% alginate coating treatment during shelf life compared with the other concentrations of alginate coatings. The values of cortex and core L* responded similarly to the alginate coating treatments during shelf life. That is, cortex and core L* values were lowest in 1% alginate coating treatment compared with the other alginate coating treatments during shelf life. The values of peel and cortex chroma (C*) were lower during shelf life than at harvest, irrespective of the concentration of the alginate coating. In addition, no statistically significant differences were observed among the alginate coating concentrations. However, core C* value was higher during shelf life than at harvest and the highest in 1% alginate coating treatment than in other treatments during shelf life. The hue angle (h°) was lower during shelf life than at harvest, regardless of the concentration of alginate coating. While no statistical difference was detected in peel h°, cortex and core h° values were lowest in 1% alginate coating treatment compared with the other treatments during shelf life. Peel redness (a*) value was higher during shelf life than at harvest, whereas lowest in 2% alginate coating treatment during shelf life compared with the other alginate coating treatments. However, the cortex and core a* values increased during 14 days of shelf life compared with that at harvest, irrespective of the alginate coating concentration. Nevertheless, cortex and core a* values were highest in 1% alginate coating treatment after shelf life compared with the other alginate treatments. In addition, peel and cortex yellowness (b*) decreased during shelf life compared with that at harvest and did not differ among the alginate coating concentrations. However, core b* value was increased after shelf life and highest in 1% alginate coating treatment compared with the other treatments (Fig. 3).
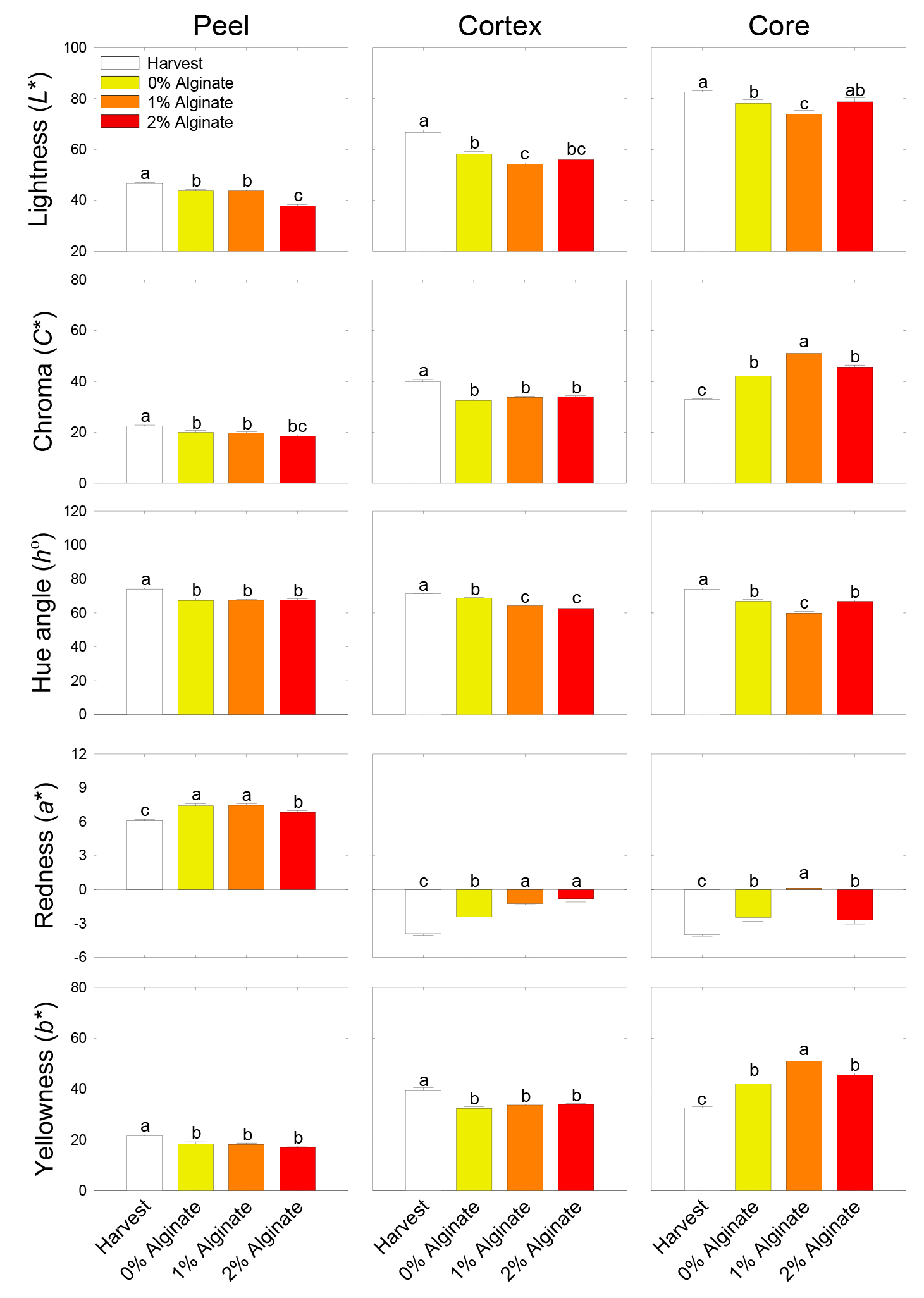
Fig. 3.
Physiological responses of fruit peel, cortex, and core color variables, lightness (L*), chroma (C*), hue angle (h°), greenness to redness (a*), and blueness to yellowness (b*), in the fruit of ‘Haegeum’ kiwifruit coated with varying concentrations of alginate 1 day before harvest and then stored at 21°C for up to 14 days. Each variable indicates the mean of fifteen kiwifruits per replicate (n = 15) ± standard error (SE). Each letter on the bars represents the results of the least significant difference (LSD) test at p = 0.05.
Responses of phenolic compounds, carotenoids, ascorbic acid, and total antioxidant scavenging capacities
Of the individual phenolic compounds, the catechin content increased during 14 days of shelf life compared with that at harvest, regardless of the concentration of the alginate coating. The level of 4-hydroxybenzoic acid was highest in 1% alginate coating treatment and lowest in 0% alginate coating treatment during shelf life. However, the gallic acid level was not affected during shelf life or by alginate coating. The contents of epicatechin and chlorogenic acid were highest in 1% alginate coating treatment during shelf life compared with the other treatments. The content of procyanidin B2 was highest in 0% alginate coating treatment after shelf life, whereas the lowest at harvest compared with the other treatments. Interestingly, the contents of total phenolic compounds and total flavonoids increased after 14-day of shelf life, regardless of the alginate coating treatment. By contrast, the content of total carotenoids was highest in 1% alginate coating treatment after shelf life but lowest at harvest. The content of ascorbic acid was lowest at harvest and increased after shelf life, with the highest value observed in the 0% alginate coating treatment. The responses of the total antioxidant scavenging capacities, assessed using ABTS and DPPH assays were lowest at harvest and then increased after 14-day of shelf life, regardless of the alginate coating treatment (Fig. 4).
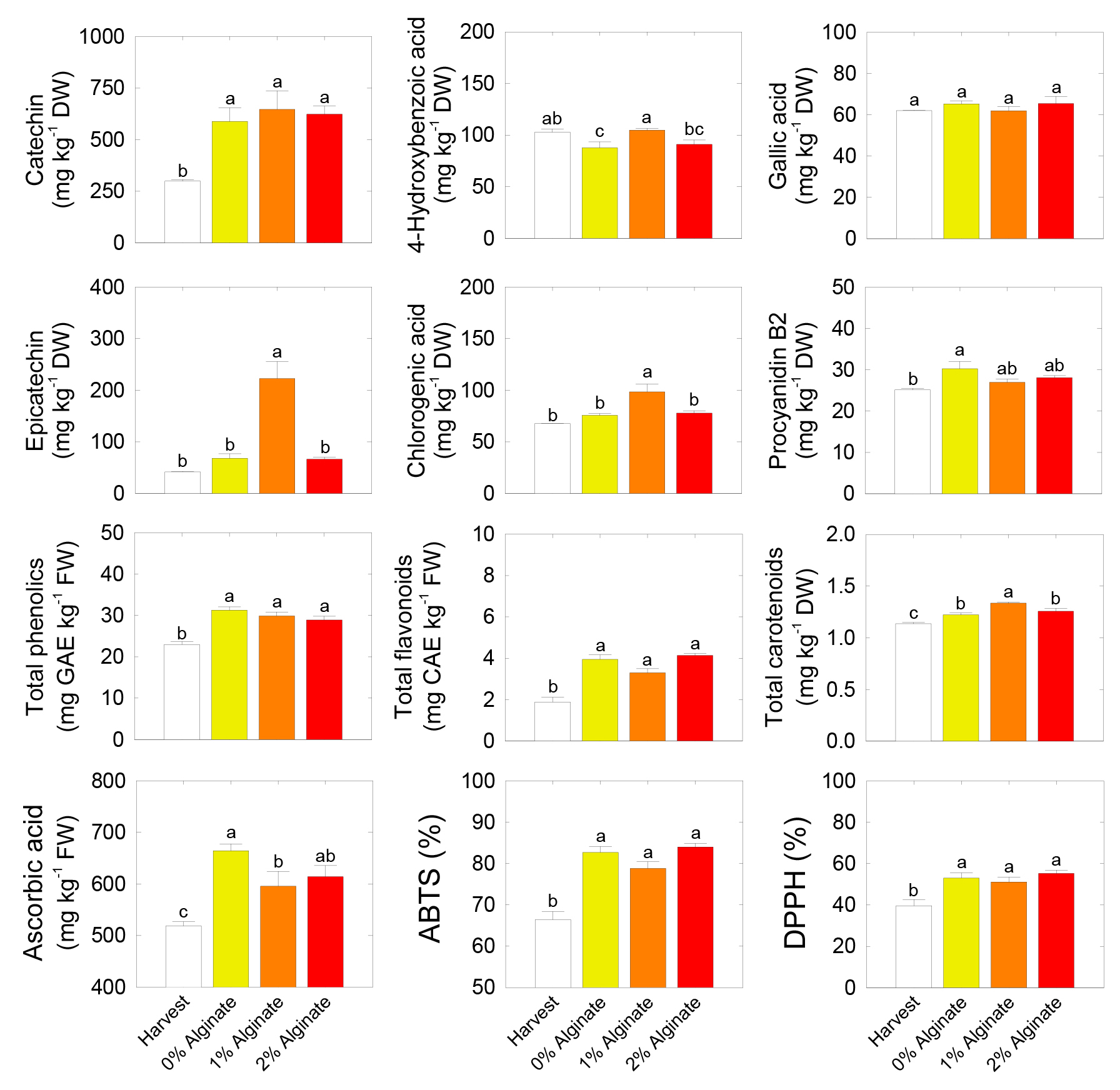
Fig. 4.
Responses of individual phenolic compounds, total phenolic compounds, total flavonoids, total carotenoids, ascorbic acid, and total antioxidant scavenging capacity (ABTS and DPPH assays) in the fruit of ‘Haegeum’ kiwifruit coated with different concentrations of alginate 1 day before harvest and then stored at 21°C for up to 14 days. Each variable indicates the mean of three replicates (n = 3) ± standard error (SE). Each letter on the bars represents the results of the least significant difference (LSD) test at p = 0.05.
Correlation responses to alginate coating treatment
To assess the relationships among all response variables and physiological disorders, such as fruit shriveling and decay, a correlation coefficient heatmap matrix combined with hierarchical cluster analysis was used, as illustrated in Fig. 5. One major and one minor positive correlation cluster were detected, and these two positive correlation clusters indicated a negative correlation. In the major positive correlation cluster located in the upper right region, fruit shriveling, fruit respiration rate (CO2), procyanidin B2, DPPH and ABTS assays, total flavonoids, catechin, ascorbic acid, total phenolic compounds, SSC, fruit weight loss, SSC/TA, peel a*, and ethylene production rate (C2H4) were positively correlated. In the minor positively correlated cluster located at the bottom left, TA, flesh firmness, peel h°, peel C*, peel b*, and peel L* were also positively correlated with one another (Fig. 5).
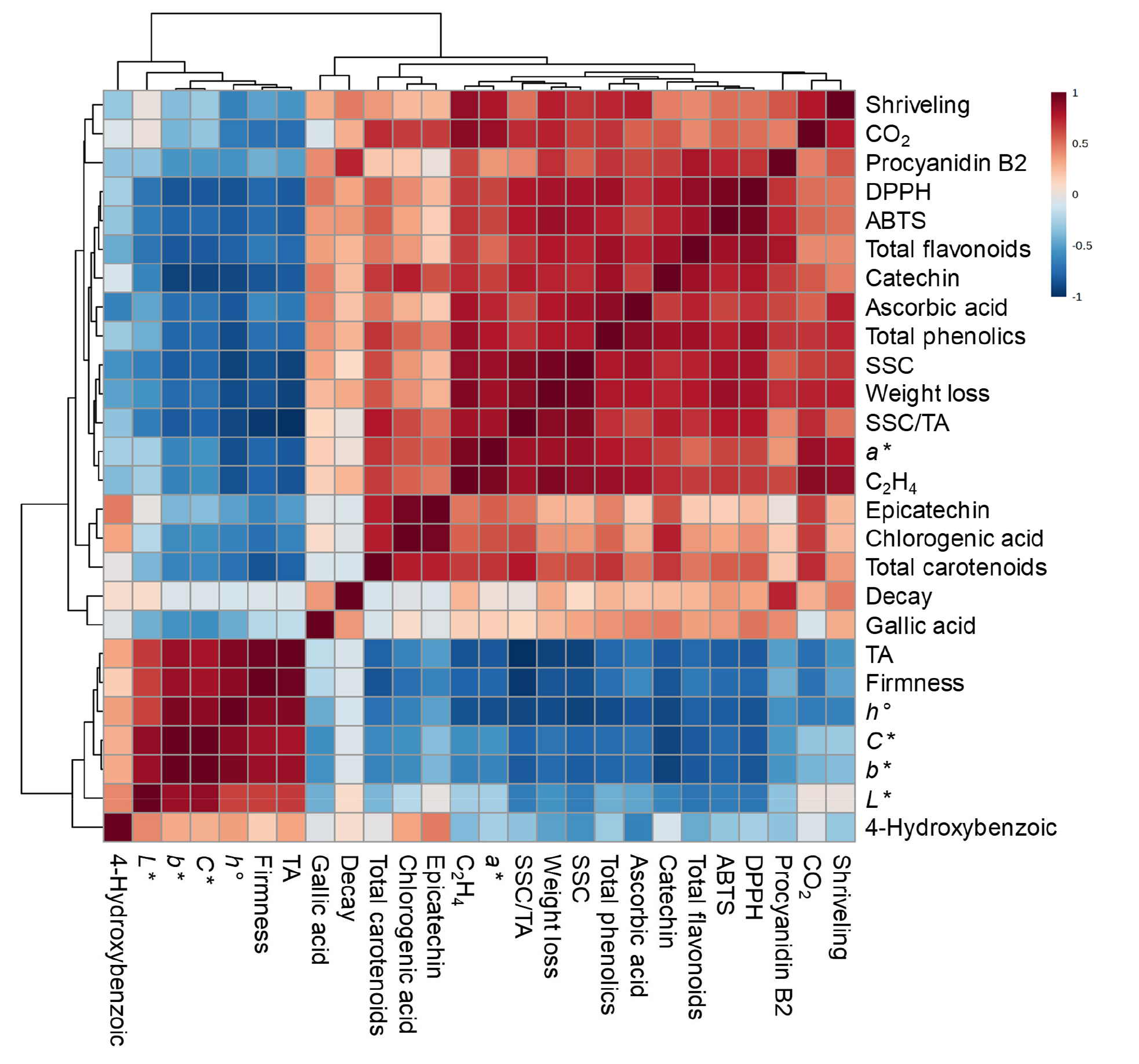
Fig. 5.
Pearson’s correlation coefficient heatmap with a hierarchical cluster analysis of fruit physiological attributes, peel colour variables, individual phenolic compounds, total phenolic compounds, total flavonoids, total carotenoids, ascorbic acid, and total antioxidant scavenging capacity (ABTS and DPPH assays) in the fruit of ‘Haegeum’ kiwifruit coated with different concentrations of alginate 1 day before harvest and then stored at 21°C for up to 14 days. The figure legend indicates correlation coefficient (r) values ranging from ‒1 to 1. Red and blue boxes indicate positive and negative correlation between variables, respectively.
For a more robust elucidation of the physiological changes of the kiwifruits throughout shelf life, all the variables were used as input to generate a Pearson’s correlation coefficient network with r ≥ |0.70| as shown in Fig. 6. Fruit decay was positively correlated only with procyanidin B2. Fruit shriveling was positively correlated with ascorbic acid, total phenolic compounds, weight loss, ethylene production rate, fruit respiration rate, and peel a*. In addition, SSC was positively correlated with weight loss, however, negatively correlated with peel h° and TA. SSC/TA was negatively correlated with flesh firmness and TA, which were positively correlated with each other. DPPH was positively correlated with peel b*, peel C*, ABTS, and catechin levels (Fig. 6).
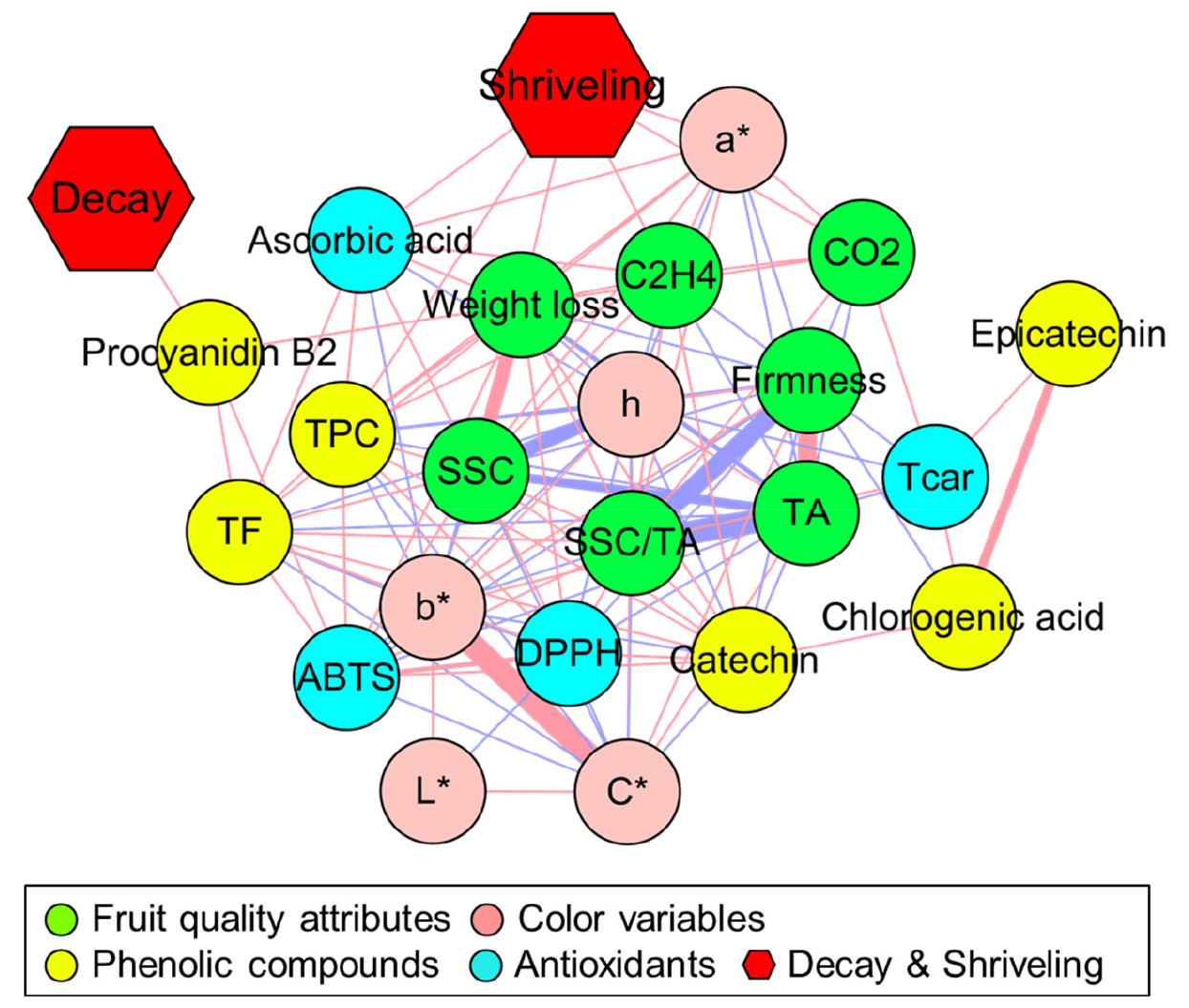
Fig. 6.
Pearson’s correlation coefficient network (r ≥ |0.70|) among fruit quality attributes, color variables, total and individual phenolic compounds, ascorbic acid, and total antioxidant scavenging capacity (ABTS and DPPH assays), and physiological disorders (fruit shriveling and decay) in the fruit of ‘Haegeum’ kiwifruit coated with different concentrations of alginate 1 day before harvest and then stored at 21°C for up to 14 days. Red- and blue-colored lines represent positive and negative correlation, respectively, and the thickness of lines indicates the significance of the correlation coefficient (r) among variables.
Discussion
Applying an alginate coating treatment one day before harvest suppressed the fruit respiration rate during shelf life, with greater suppression observed at higher alginate concentrations. Additionally, the ethylene production rate was reduced, and the climacteric peak of the ethylene production rate was delayed depending on the concentration of the alginate coating treatment. Thus, the reduction in weight loss declined slightly as shelf life increased (Fig. 2). Previous study reported that the 0.5% alginate coating treatment reduces fruit respiration rate and thus slightly delays the fresh weight loss in ‘Kosui’ pear during shelf life (Hira et al. 2022). Furthermore, the fruit respiration rate responded dose-dependently to alginate coating treatment in the fruit of the sweet cherry cultivar ‘Sweetheart’ during shelf life after cold storage (Díaz-Mula et al. 2012). During shelf life, alginate coating treatment not only drives a reduction in ethylene production and fruit respiration rates, but it has also been shown to delay the climacteric peak of ethylene production and fruit respiration rates in ‘Rambo’ tomatoes (Zapata et al. 2008). Additionally, alginate coating treatment is involved in the reduction of fruit respiration rate and then the delay of the climacteric peak in the fruit respiration of ‘French’ plum during cold storage, thereby contributing to reducing fresh weight loss while ensuring that flesh firmness is retained (Li et al. 2022). Thus, the alginate coating treatment could be directly involved in the reduction of fruit respiration rate and the delay of the climacteric peak in fruit respiration rate by inhibiting oxygen contact with the kiwifruit and suppressing any oxidative biochemical reactions during shelf life. In addition, the ethylene production rate was proportional to the concentration of the alginate coating, as shown by the fruit respiration rate. Therefore, the reduction and delay in ethylene production and fruit respiration rates could be strongly associated with the reduction in fresh weight loss by alginate coating treatment in ‘Haegeum’ kiwifruit during shelf life.
Alginate coating reduced the incidence and severity of fruit shriveling and decay during shelf life (Fig. 1). Although fruit decay severity was only slightly detected in 0% alginate coating treatment, no symptoms of fruit decay were detected in both 1% or 2% alginate coating treatments during shelf life. However, the severity of fruit shriveling decline in proportion to the concentration of alginate coating treatment in the fruit of ‘Haegeum’ kiwifruit during shelf life (Fig. 1B). The disease incidence is reduced by alginate oligosaccharide coating treatment in the fruit of ‘Bruno’ kiwifruit during shelf life (Zhuo et al. 2022). In addition, alginate coating treatment reduces the incidence of fruit decay in cold-stored fresh-cut slices of ‘Hayward’ kiwifruit (Manzoor et al. 2021). That is, alginate coating treatment limited the direct exposure of the kiwifruits to direct air, thereby directly contributing to the reduction in the incidence of fruit decay during shelf life. Along with the results of fruit decay incidence, the severity of fruit shriveling gradually declined with an increase in alginate coating concentration in the fruit of ‘Haegeum’ kiwifruit during shelf life. As shown in Fig. 2A, fresh weight loss was clearly different between the alginate-coated fruit and the uncoated control fruits. Thus, the severity of fruit shriveling was proportional to the concentration of alginate coating applied. Preharvest alginate oligosaccharide treatment reduces the incidence and severity of fruit cracking by reducing water stress in the fruit of ‘Wanhongzhu’ sweet cherry cultivar during shelf life (Xie et al. 2025). The incidence and severity of fruit shriveling are highly linked to the coating materials and package types in cold-stored blueberries and blackberries (Olmedo et al. 2025). That is, preharvest alginate coating treatment could directly reduce the fruit respiration rate and thus decrease fresh weight loss, thereby suppressing the incidence and severity of fruit shriveling during shelf life.
Unlike the outcomes of alginate coating on ethylene production and fruit respiration rates, flesh firmness, SSC, TA, and the SSC/TA ratio did not consistently respond to the concentration of preharvest alginate coating treatment in ‘Haegeum’ kiwifruit during shelf life (Fig. 2B). In this study, alginate-coated fruits were placed at ambient temperature for up to 14 days to evaluate the physiological responses of ethylene production and fruit respiration rates during the 2-week period, along with the response of fresh weight loss. Thus, the preharvest alginate coating treatment may be relatively less effective on the physiological responses of flesh firmness, SSC, TA, and SSC/TA ratio as major fruit quality attributes after 14 days of shelf life. During the shelf-life period, the climacteric peaks of ethylene production and fruit respiration rates appeared in the middle of the shelf life, irrespective of the concentration of the alginate coating treatment. This means that fruit quality attributes are highly affected in the middle of the shelf-life period. Therefore, unlike the fresh weight loss results, the ethylene production rate, fruit respiration rate, flesh firmness, SSC, TA, and SSC/TA ratio were less consistent with the preharvest alginate coating treatment at the end of shelf life.
The contents of total phenolic compounds and total flavonoids did not differ significantly among alginate coating concentrations during shelf life. In particular, the contents of carotenoids, catechin, 4-hydroxybenzoic acid, epicatechin, and chlorogenic acid were highest in the 1% alginate coating treatment during shelf life, compared with the other concentrations (Fig. 4), suggesting that 1% alginate may be more effective in maintaining specific defense-related phenolic compounds. While total phenolic compounds and total flavonoids were generally upregulated after 14 days of shelf life, compared with harvest, regardless of alginate concentration, this increase may partly reflect water loss-driven concentration effects on a fresh-weight basis or stress-induced biosynthesis during storage. In contrast, the levels of individual phenolic compounds showed inconsistent changes over 14 days relative to harvest, indicating that different phenolics may respond differentially to alginate-induced storage related stress. Therefore, instead of focusing on compounds that did not differ significantly among treatments, the discussion emphasizes those individual phenolic compounds that increased under the 1% alginate treatment and are most likely to contribute to the maintenance of fruit quality and the reduction of decay and shriveling during shelf life. However, the levels of total phenolic compounds do not differ between untreated control and 2% alginate coating treatment in cold-stored fresh-cut ‘Flor de Invierno’ pears (Oms-Oliu et al. 2008a) and ‘Piel de Sapo’ melon (Oms-Oliu et al. 2008b). Nevertheless, the contents of total phenolic compounds and total anthocyanins responded differently to the dosage levels of alginate coating treatment in the cold-stored fruit of the sweet cherry cultivar ‘Sweetheart’ (Díaz-Mula et al. 2012). The 2% alginate spray coating treatment tends to delay the reduction of soluble phenolic compounds in cold-stored strawberries (Peretto et al. 2017). However, 2% alginate coating treatment drives the delay in the reduction of total phenolic compounds and total flavonoids contents in cold-stored fruit of ‘Allahabad safeda’ guava cultivar (Nair et al. 2018). Additionally, 1.5% alginate coating treatment plays a key role in maintaining fruit quality by reducing the levels of reactive oxygen species (ROS), including superoxide anion radicals, thereby contributing to delaying the reduction of total phenolic compounds and, subsequently, the accumulation of malondialdehyde (MDA) in cold-stored fruit of ‘French’ plum cultivar (Li et al. 2022). Although no statistical difference of total phenolic compounds and total flavonoids was detected among the alginate coating treatments during shelf life, the increase in certain individual phenolic compounds could be associated with the maintenance of fruit quality and the reduction of the incidence and severity of fruit decay and shriveling during shelf life.
While fruit decay was only positively correlated with procyanidin B2, fruit shriveling was positively correlated with ascorbic acid, total phenolic compounds, fresh weight loss, ethylene production rate, fruit respiration rate, and peel a* (Fig. 6). During shelf life, fruit decay was only detected in 0% alginate coating treatment at the end of shelf life but neither in 1% alginate coating nor in 2% alginate coating treatments (Fig. 1B). Additionally, the content of procyanidin B2 was highest in 0% alginate coating treatment during shelf life compared with the other alginate coating treatments (Fig. 4). However, the severity of fruit shriveling exhibited a gradual response to the concentration of the alginate coating treatment and decreased as the coating concentration increased during shelf life (Fig. 1B). Ascorbic acid, total phenolic compounds, fresh weight loss, ethylene production rate, fruit respiration rate, and peel a* responded similarly with respect to the concentration of the alginate coating treatments during shelf life. The alginate coating treatment reduced ethylene production and fruit respiration rates, resulting in a reduction in fresh weight loss. In addition, retaining fruit quality in terms of fresh weight loss and fruit respiration rate derived from the alginate coating treatment would be effective for the response to water-soluble compounds, such as ascorbic acid and phenolic compounds. Thus, alginate coating treatment directly delayed the incidence and severity of fruit shriveling during shelf life. Therefore, an increase in the severity of shriveling could directly affect the responses of water-soluble compounds, including ascorbic acid and total phenolic compounds.
The most unexpected result was that alginate-coated fruits, particularly the 1% treatment, exhibited lower firmness and TA, compared to the uncoated control after 14 days of shelf life, despite reduced ethylene production rate, respiration rate, and weight loss. This outcome contradicts the general expectation that coatings maintaining a low ethylene and respiration rate could delay ripening and firmness and organic acid content. Several physiological interpretations may explain this counterintuitive pattern. First, severe dehydration and surface shriveling in control fruits likely increased penetration resistance during firmness measurement, artificially elevating the firmness despite more ripened fruits. In contrast, alginate-coated fruits maintained turgor and resulting in more accurate measurement of their true physiological firmness. Second, for TA measured on a fresh-weight basis, water loss in control fruits may have caused a concentration effect, yielding apparently higher TA values, although absolute organic acid content had declined to a similar or greater extent. Additionally, alginate coatings may have altered internal atmosphere conditions (slightly reduced O2 and elevated CO2), potentially accelerating organic acid metabolism through modified TCA cycle activity or stress-induced respiratory. We acknowledge as a key limitation that firmness and TA were measured only at harvest and at the end of shelf life. This limited temporal resolution may have missed critical intermediate-stage physiological transitions, preventing a more precise mechanistic interpretation of these divergent patterns. Future studies should include more frequent sampling intervals (e.g., 3-day intervals) to better capture the dynamics of firmness reduction and organic acid depletion during storage and shelf life transitions. These alternative interpretations provide a more physiologically coherent explanation for the observed results and strengthen the overall results of this study.
Conclusion
Preharvest alginate coating treatment reduced the severity of fruit decay and shriveling in ‘Haegeum’ kiwifruit during shelf life. Among the fruit quality attributes, fresh weight loss, ethylene production and fruit respiration rates were reduced by preharvest alginate coating treatment during shelf life and then showed an inverse response to increasing alginate coating concentrations. Nonetheless, flesh firmness, SSC, TA, and SSC/TA ratio, as well as other fruit quality attributes, were not significantly different after preharvest alginate coating treatment at the end of the 14-day shelf life. The levels of catechin, total phenolic compounds, total flavonoids, total carotenoids, and ascorbic acid were higher during shelf life than at harvest, along with the total antioxidant scavenging capacity assessed using ABTS and DPPH assays. Throughout shelf life, the concentrations of 4-hydroxybenzoic acid, epicatechin, chlorogenic acid, and total carotenoids were highest in 1% alginate coating treatment compared with the other coating treatments during shelf life. While fruit decay was only positively correlated with procyanidin B2, fruit shriveling was positively correlated with ascorbic acid, total phenolic compounds, fresh weight loss, ethylene production rate, fruit respiration rate, and peel a* value during shelf life. Overall, our results showed that preharvest alginate coating treatment could be involved in reducing ethylene production and respiration rates. Moreover, the treatment also caused a decline in fresh weight loss, along with alterations in phenolic compounds, ascorbic acid, carotenoids, and total antioxidant scavenging capacity, thereby contributing to controlling the severity of fruit decay and shriveling in ‘Haegeum’ kiwifruit.


