Introduction
Materials and Methods
Plant Materials and Heat Treatment
RNA Extraction and cDNA Synthesis
Analysis of Genes
Real-time PCR Analysis
Results
Flavonoid Biosynthesis Genes Expression in ‘Campbell Early’ Fruit After Temperature Treatment
Expression of Genes Involved in Stilbene Compounds in ‘Campbell Early’ Fruit in Response to Temperature Change
Discussion
Introduction
Grapes are a major fruit crop with about 65 million tons produced annually. Grape have high temperature adaptability (Reisch et al., 2012); however, some grape cultivars are sensitive to high temperature during the ripening season and are susceptible to fruit growth and quality damage (Lee et al., 1979. Global temperatures have recently increased due to climate change, and these increases
have occurred during grape growing seasons. Changes in temperature have affected fruit quality and induced physiological disorders including succulent growth, growth prolongation, defoliation, and fruit color failure (Lee et al., 2015; Sugiura et al., 2007).
Anthocyanin content is one of the most important determinants of grape quality and accumulates more effectively at low temperatures (Haselgrove et al., 2000; Mori et al., 2005b; Yamane et al., 2006; 2009; Jaakola and Hohtola, 2010). Moreover, anthocyanin biosynthesis is regulated by internal factors such as vacuoles, pH, sub-pigments, metal ions, and sugar substitution, as well as by environmental factors such as temperature and light intensity and quality (Kim, 1999). Moreover, initial high temperatures are responsible for berry discoloration and can significantly reduce the synthesis and accumulation of anthocyanins in grapes (Yamane et al., 2006).
Flavonoids are involved in many physiological functions including flower coloration, UV protection, and insects and pathogen protection (Chen et al., 2012). Stilbene synthase (STS) is closely related to chalcone synthase (CHS) and flavonoid biosynthesis (Halbrock and Grisebach, 1979). Stilbenes, including resveratrol, represent a small class of plant secondary metabolites derived from the phenylpropanoid pathway (Langcake and Pryce, 1977; Chong et al., 2009). Phenylpropanoid compounds comprise a large family of secondary metabolites that are involved in responses to a variety of biotic and abiotic stresses and considered types of phytoalexins, antimicrobial compounds accumulated in plants against pathogen infections. Among the phenylpropanoid antimicrobial compounds, flavonoids and STS are induced by low temperatures (Christie et al., 1994; Kim et al., 2016; 2017), pathogen attack (Dercks and Creasy, 1989), and wounding (Hahlbrock and Scheel, 1989). Three STS mRNAs were drastically altered in response to elicitors, cell wall extract of Phytophthora cambivora, in grapevine cells (Wiese et al., 1994).
The mechanism that inhibits anthocyanin accumulation at high temperatures under ripening conditions is unclear. Plants with red pigment such as grapevines downregulate genes related to anthocyanin biosynthesis in fruits in response to high temperatures (Lin-Wang et al., 2011; Moon et al., 2007; Yamane et al., 2006). Movahed et al. (2016) reported that high temperature suppressed the expression of anthocyanin biosynthesis genes, peroxidase activity, and UDP glucose flavonoid 3-O-glucosyltransferase (UFGT) enzyme activity. Neither anthocyanin accumulation nor the rate of sugar accumulation was affected by optimal temperature conditions; however, they were negatively and positively influenced by high temperature conditions, respectively (Kliewer and Torres, 1972; Mori et al., 2005b; Tomana et al., 1979).
Transcriptomic analysis provided a wealth of data at the molecular level about berry development and ripening in grapes, as well as an efficient means of identify candidate genes associated with berry quality traits and responses to different environmental stresses. The effect of temperature on metabolism in ripening grape berries has been demonstrated at the transcript level (Carbonell-Bejerano et al., 2013; Rienth et al., 2014); however, to elucidate the precise mechanism of color inhibition by high temperature in grapes, it is necessary to analyze the response of the flavonoid pathway to temperature at the transcript level in grape berries that are sensitive to high temperature (Lee et al., 1979; Mori et al., 2005b).
In this study, we selected differentially expressed genes (DEGs) related to flavonoid and stilbene compound synthesis in berries of ‘Campbell Early’ grapes that were exposed to high temperature, and then analyzed their expression patterns at the transcript level. We report reduced berry skin coloration in response to high temperature in this grape cultivar. This study provides meaningful molecular information that will help solve cultivation problems in grapes caused by high temperature.
Materials and Methods
Plant Materials and Heat Treatment
Healthy ‘Campbell Early’ (Vitis labruscana Bailey) grape clusters were harvested according to the modified Eichhorn-Lorenz (E-L) scheme (Coombe, 1995) and corresponded to E-L Stage after 35 (veraison) at the experimental vineyard at Yeungnam University, Gyeongsan, Korea, after which they were placed in growth chambers (Ilshin Tech., Daejeon, Korea) equipped with a temperature controller. The berries were cut off from the clusters to avoid dehydration and decay, and stored at 25°C, 30°C, and 35°C under a 16/8 light/dark photoperiod or continuous dark conditions. Berries were collected 0, 6, 12, 24, 36 and 48 h after treatment and stored at -80°C until use.
RNA Extraction and cDNA Synthesis
Total RNA was extracted from ground samples using a modified version of the method described by Chang et al. (1993). The concentration and integrity of RNA was measured using an ND-1000 Nano Drop spectrophotometer (Technologies Inc., Wilmington, DE, USA) and RNA was checked by electrophoresis in 1% agarose gels. Next, 500 ng purified RNA was reverse transcribed into first-strand cDNA using the GoScriptTM Reverse Transcription System (GoScriptTM Reverse Transcription System, Promega, Madison, WI, USA) and used as a template for PCR amplification.
Analysis of Genes
Based on the DEGs identified with transcriptome analysis, genes involved in skin coloration and the flavonoid biosynthesis pathway were selected for further study. Additionally, genes reported to be involved in the grape flavonoid biosynthesis pathway were selected and their expression pattern was analyzed. Upon transcriptome analysis of grape berries treated with high temperature, the expression patterns of 12 grape genes were identified from the flavinoid biosynthesis pathway, which included chalconeisomerase (CHI), flavanone 3-hydroxylase (F3H), dihydroflavonol reductase (DFR), leucoanthocyanidin dioxygenase (LDOX), flavonol synthase (FLS), shikimate O-hydroxycinnamoyltransferase (HCT), anthocyanidin reductase (ANR), flavanone 7-O-glucoside 2''-O-beta-L-rhamnosyltransferase (C12RT1), leucoanthocyanidin reductase (LAR), trans-cinnamate 4-monooxygenase (CYP73A), caffeoyl-CoA O-methyltransferase (CCOMP), and flavonoid 3'-monooxygenase (F3M). The primers used for real-time PCR are shown in Table 1. Stilbene gene information was described previously by Ahn et al. (2014), and the six primer sequences and information for accession and structure used are shown in Tables 1 and 2.
Table 2. Identification and structure information for STS genes
 | |
*NABIC; National Agricultural Biotechnology Information Center, RDA, Korea | |
Real-time PCR Analysis
Primer pairs were designed with the Primer3 software (http://primer3.ut.ee) and primer sequences are provided in Tables 1 and 2. Real-time PCR using first-strand cDNA synthesized from RNA extracted from temperature-treated grape berries was performed in a C1000TM Thermal Cycler (CFX96TM Real-Time System, BioRad, Hercules, California, USA) system with SYBR Premix Ex (SYBR Premix Ex Taq, TaKaRa Bio Inc., Osaka, Japan) as the fluorescent dye. Thermal cycling was performed with an initial denaturation step of 30 s at 95°C followed by 40 cycles of 95°C for 5 s, 60°C for 30 s, and 95°C for 5 s. Transcript levels were calculated using the standard curve method from triplicate data with the actin gene as an internal control and untreated berries at time zero as the expression reference sample. Data shown represent the mean fold increase in mRNA level over that in untreated berries, which were defined as the 1× expression level. Results are the means of triplicate data from three independent experiments. Bars indicate the standard deviation.
Results
Flavonoid Biosynthesis Genes Expression in ‘Campbell Early’ Fruit After Temperature Treatment
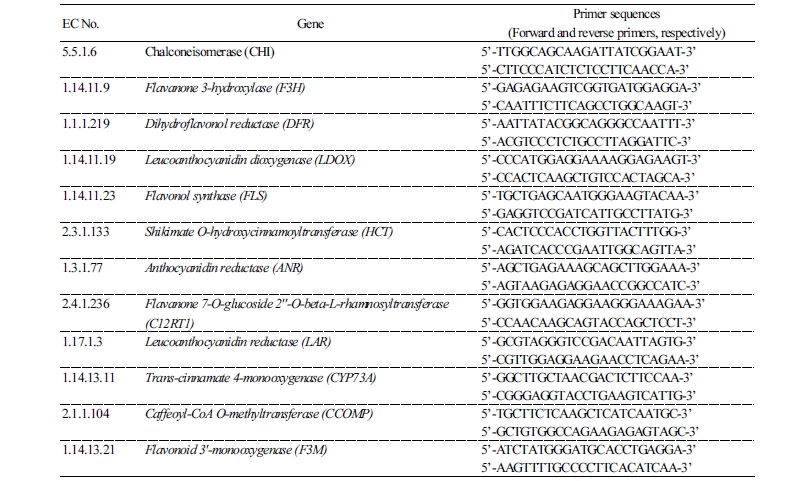
Differentially expressed genes (DEGs) involved in the flavonoid biosynthesis pathway were selected from transcriptome analysis results from grape berries treated with high temperatures (30-35°C). Among the selected DEGs, the expression patterns of CHI, F3H, DFR, LDOX, FLS, HCT, ANR, C12RT1, LAR, CYP73A, CCOMP, and F3M in temperature treated ‘Campbell Early’ grape berries harvested at ripening time were analyzed by real-time PCR. The expression of CHI, LDOX, and CYP73A genes was unchanged at all temperature conditions. Moreover, their expression was suppressed at 30-35°C relative to 25°C, and highly suppressed at 35°C(Fig. 1). The expression of FLS, HCT, and ANR genes decreased in response to all temperature treatments; the expression in berries treated at 35°C decreased sharply compared to that at 25-30°C. The expression of all other investigated genes was significantly suppressed in response to high temperature (30-35°C) treatments relative to the 25°C treatment.
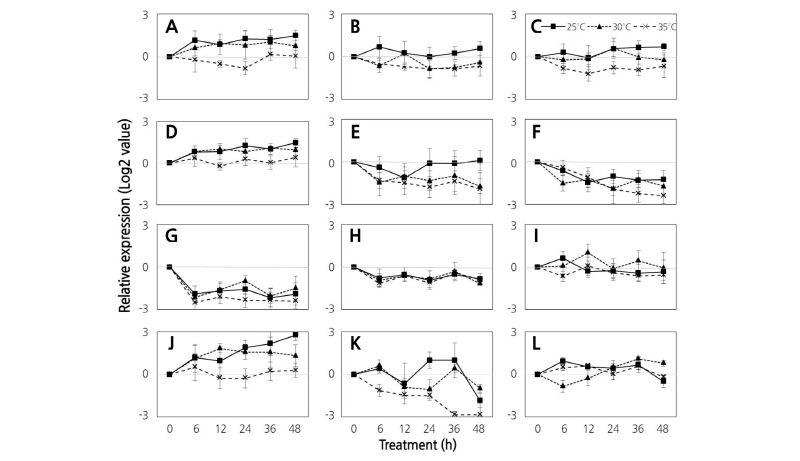
Fig. 1. Expression of genes related to flavonoid biosynthesis measured by quantitative real-time PCR analysis of ‘Campbell Early’ grapes treated with 25°C, 30°C, and 35°C under light for 0-6 h, 24 h, and 48 h; and under dark conditions for 12 h and 36 h during the initial fruit ripening stages. Results are presented as the means of triplicate data from three independent experiments. Bars indicate the standard deviation from the mean. Chalconeisomerase (A), flavanone 3-hydroxylase (B), dihydroflavonol reductase (C), leucoanthocyanidin dioxygenase (D), flavonol synthase (E), shikimate O-hydroxycinnamoyltransferase (F), anthocyanidin reductase (G), flavanone 7-O-glucoside 2''-O-beta-L-rhamnosyltransferase (H), leucoanthocyanidin reductase (I), trans-cinnamate 4-monooxygenase (J), caffeoyl-CoA O-methyltransferase (K), and flavonoid 3'-monooxygenase (L) genes.
There was not different pattern inexpression of DEGs in various temperatures between continuous dark condition and 16/8 h light/dark conditions (Fig. 2).
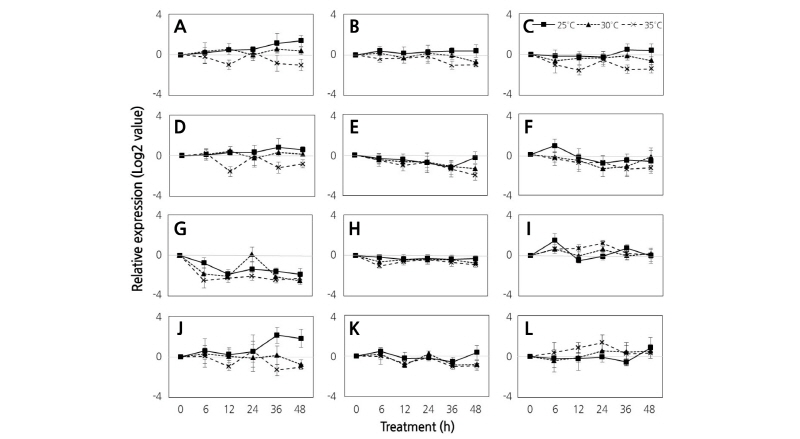
Fig. 2. Comparison of expression of genes related to flavonoid biosynthesis using quantitative real-time PCR analysis of ‘Campbell Early’ grown at 25°C, 30°C, and 35°C under continuous dark conditions during the initial ripening stages. Results are presented as the means of triplicate data from three independent experiments. Bars indicate the standard deviation from the mean. Chalconeisomerase (A), flavanone 3-hydroxylase (B), dihydroflavonol reductase (C), leucoanthocyanidin dioxygenase (D), flavonol synthase (E), shikimate O-hydroxycinnamoyltransferase (F), anthocyanidin reductase (G), flavanone 7-O-glucoside 2''-O-beta-L-rhamnosyltransferase (H), leucoanthocyanidin reductase (I), trans- cinnamate 4-monooxygenase (J), caffeoyl-CoA O-methyltransferase (K), and flavonoid 3'-monooxygenase (L) genes.
The expression of DEGs was also analyzed in completely mature ripe berries that had reached full coloration and were subjected to temperature treatments for 24 h (Fig. 3). Similar to the expression pattern of genes in the fruit at the initial ripening stage, and the expression of the DEGs was inhibited by higher temperatures compared to the low temperature control. The gene expression in berries treated with high temperatures under a 16/8 h light/dark photoperiod was highly suppressed relative to berries treated under continuous dark conditions. Moreover, the expression of most selected DEGs was inhibited at the high temperature treatment (35°C) compared to 25-30°C treatments. These results are consistent with a previous study that showed high temperature conditions inhibited or inactivated enzymes involved in the synthesis of anthocyanins in fruits (Kliewer, 1977).
Fig. 3. Comparison of expression of genes related to flavonoid biosynthesis using quantitative real-time PCR analysis of ‘Campbell Early’ grown at 25°C, 30°C, and 35°C under 16/8 light/dark photoperiods and continuous dark conditions at 24 h during late berry ripening stages. Results are the means of triplicate data from three independent experiments. Bars indicate the standard deviation from the mean. Chalconeisomerase (A), flavanone 3-hydroxylase (B), dihydroflavonol reductase (C), leucoanthocyanidin dioxygenase (D), flavonol synthase (E), shikimate O-hydroxycinnamoyltransferase (F), anthocyanidin reductase (G), flavanone 7-O-glucoside 2''-O-beta-L-rham-nosyltransferase (H), leucoanthocyanidin reductase (I), trans-cinnamate 4-monooxygenase (J), caffeoyl-CoA O-methyltransferase (K), and flavonoid 3'-monooxygenase (L) genes.
Expression of Genes Involved in Stilbene Compounds in ‘Campbell Early’ Fruit in Response to Temperature Change
Expression patterns of stilbene synthesis-related genes (STS) were analyzed at the initial ripening stage of grapes subjected to various temperature treatments (Figs. 4 and 5). Expression of the resveratrol synthase (STS1) gene increased in all temperature treatments under the 16/8 h light/dark photoperiod. However, the expression of STS1 at 25°C was inhibited relative to that at 35°C. Expression of the stilbene synthase 1-like (STS14) gene decreased until 36 h of treatment, and then increased again at 48 h. Unlike the other genes evaluated, expression of STS14 was inhibited at 25°C compared to 35°C. Moreover, expression of the stilbene synthase 1-like (STS24) gene increased until 36 h, then decreased significantly at 48 h. Finally, expression of STS1a-1 (STS11), STS1a-2 (STS12), and STS1a-3 (STS13) genes, which all had similar expression patterns in the same temperature treatment, decreased over the time course and were suppressed at 35°C compared to 25°C.
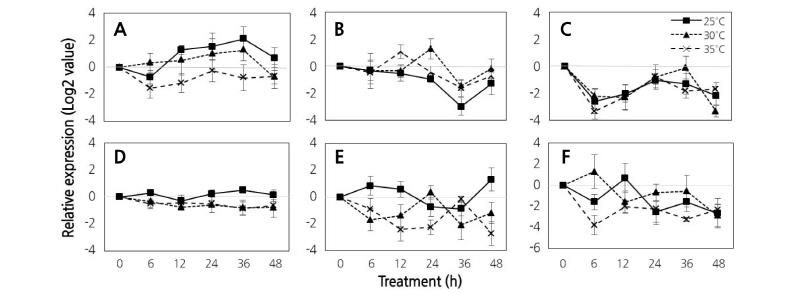
Fig. 4. Comparison of expression of STS genes using quantitative real-time PCR analysis of ‘Campbell Early’ grapes grown at 25°C, 30°C, and 35°C under light for 0-6 h, 24 h, and 48 h; and under dark conditions for 12 h and 36 h during initial ripening stages. Results are the means of triplicate data from three independent experiments. Bars indicate the standard deviation from the mean. Resveratrol synthase (A), stilbene synthase 1-like 14 (B), stilbene synthase 1-like 24 (C), STS1a-1 (D), STS1a-2 (E), and STS1a-3 (F) genes.
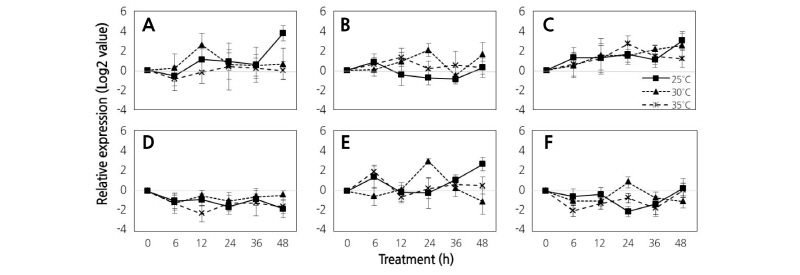
Fig. 5. Comparison of STS gene expression using quantitative real-time PCR analysis in ‘Campbell Early’ grapes that were grown at 25°C, 30°C, and 35°C under continuous dark conditions during their initial ripening stages. Results are the means of triplicate data from three different experiments. Bars indicate the standard deviation from the mean. Resveratrol synthase (A), stilbene synthase 1-like 14 (B), stilbene synthase 1-like 24 (C), STS1a-1 (D), STS1a-2 (E), and STS1a-3 (F) genes.
In continuous dark conditions, the STS1 gene was highly expressed in all temperature treatments and showed a significant increase in expression at 12 h. The expression pattern of the STS14 was was inhibited at 25°C compared to 35°C regardless of the light condition. Expression of STS24 was also inhibited at 35°C compared to 25°C. Expression of the STS24 gene increased after 6 h in response to all temperature treatments, and the expression patterns of STS11, STS12, and STS13 were similar under both 16/8 h light/dark photoperiod and continuous dark conditions.
Discussion
There have been many studies conducted to investigate the responses of fruits to increased temperatures during the growing season to counteract the environmental stresses caused by global warming. Increased anthocyanin content has been observed in ‘Pinot Noir’ grapes that were treated with abscisic acid (ABA) and matured and colored at 20°C (Mori et al., 2005a). Red ‘Aki Queen’ grapes had induced maturation and coloration in response to night temperature treatments at 5°C below the ambient temperature (Koshita et al., 2007). Summer maturity day temperatures of 30°C led to reduced marketability of grapes when compared to cultivation at 20°C (Lee et al., 1979). In apples, fruit growth was more affected by nighttime temperature than daytime temperature and coloration was promoted when nighttime temperatures were low (Reay, 1999) because the synthesis of anthocyanin, which is the main pigment in apples, was promoted at low temperatures (Ban et al., 2009). It has been reported that when anthocyanin is the main pigment in grapes, the berries are poorly colored when exposed to high temperatures at night (Kleiwer and Torres, 1972; Koshita et al., 2007). At the daytime temperature range of 15-20°C, mature grapes had a higher anthocyanin content than mature grapes grown at 30-35°C (Buttrose et al., 1971; Mori et al., 2005b). Movahed et al. (2016) reported that the grapevine VviPrx31 peroxidase gene were involved in anthocyanin degradation in ripening berries under high temperature, and that low anthocyanin content in berries at high temperature was caused by the combined impact of reduced biosynthesis and increased degradation by peroxidase activity during anthocyanin synthesis.
Flavonoid biosynthesis occurs through the phenylpropanoid synthesis pathway, which includes the activity of various enzymes such as PAL, CHS, CHI, F3H and dihydroflavonol 4-reductase (DFR) (Bogs et al., 2006; Ford et al., 1998). Expression of the genes related to flavonoid biosynthesis affects the accumulation of anthocyanins in apple skin coloration and shows an expression level that is consistent with fruit anthocyanin content (Honda et al., 2002). The genes that play an important role in anthocyanin biosynthesis can vary widely depending on species and cultivar. In the ‘Yunhongli No.1’ pear, PpCHS, PpANS, and PpUFGT are important genes for anthocyanin biosynthesis (Zhang et al., 2012). Subjecting grapes (Vitis vinifera L. × Vitis labrusca L.) to continuous high temperatures at night decreases the activity of UDP-glucose flavonoid-3-O-glucosyltransferase (UFGT) and inhibits anthocyanin accumulation in berry skins (Mori et al., 2005b).
In a study of apples and grapes, high temperature treatments of 30-35°C decreased anthocyanin content and the expression of anthocyanin biosynthesis-related genes (Azuma et al., 2012; Lin-Wang et al., 2011). In our present study, gene expression was suppressed at 35°C compared to 25°C, which is consistent with the results reported by Kliewer (1977), who found that the synthesis of enzymes involved in anthocyanin synthesis were inhibited or inactivated at high temperature in grapes.
Expression of the CHI gene depends on the fruit developmental stage and is regulated at both transcriptional and translational levels. In the grape berry, expression of the CHI gene can contribute to the accumulation of total flavonoids (Wang et al., 2012). Ubi et al. (2006) reported that the MdF3H and pDFR genes were expressed during UV-B and temperature treatment in apple skins, while expression of F3H was increased at low temperatures (Ban et al., 2007). In addition, MdCHS, MdANS, and pUFGluT genes were weakly expressed at high temperatures (27°C) and with or without UV-B treatment. The accumulation of caffeoyl-CoA O-methyltransferases (CCoAOMTs) transcripts was increased in berry skins, and VvCCoAOMT was found to be a multifunctional O-methyltransferase that can contribute to the methylation activity of grape fruit anthocyanins and is particularly useful under drought stress conditions (Giordano et al., 2016).
The FLS gene degrades anthocyanidins by converting dihydrokaempferol, it is also a DFR substrate that is used to produce kaempferol, a kind of flavonol. FLS is also a regulatory gene such as the MYBs. Anthocyanin biosynthesis is regulated by transcription complexes consisting of MYB, a basic helix-loop-helix (bHLH), and WD40 repeat protein, which activate structural genes in the flavonoid pathway (Koes et al., 2005).
Stilbene synthase genes are known to belong to the multigene family of grapes. In V. vinifera ‘Optima’, at least seven STS genes have been reported (Schröder et al., 1988; Wiese et al., 1994). In the present study, the gene expression patterns of six STS genes were analyzed in grapes treated with high temperatures during the initial ripening stage . The STS genes are involved in fruit coloration and their expression is regulated through the competition for substrates with those of products of genes such as the CHS gene (Ahn et al., 2015; Park et al., 2010; Richter et al., 2006).
Anthocyanin levels increase in grapes exposed to direct light, regardless of the ambient temperature (Spayd et al., 2002), as well as in grape rinds with increasing fruit sugar content (Zheng et al., 2009; El-Razek et al., 2010). In particular, there is a highly significant correlation between the total soluble solid/acidity ratio and the anthocyanin content regardless of the grape variety (Matsumoto et al., 2007). The poor quality of grapes and wine caused by cultivation in high temperatures is due to the decreased total anthocyanin concentration in berries (Movahed et al., 2016). Taken together, these results suggest that inhibition of grape berry skin color development by high temperature exposure is caused by the combined responses of various pathways to anthocyanin accumulation.
In our study, changes in gene transcript levels in ‘Campbell Early’ grapes exposed to high temperatures were related to flavonoid and stilbene synthesis during berry skin color development were evaluated. Analysis of the expression of these selected genes indicated that these genes were differentially regulated in response to high temperatures. The expression of most of the tested genes was lower in response to high temperature treatment at 35°C, and the expression of CHI, LDOX and CYP73A genes was significantly inhibited at 35°C. The high temperature treatment during veraison significantly reduced the expression of genes related with the synthesis and accumulation of anthocyanins. Moreover, expression of STS1a-1 (STS11), STS1a-2 (STS12), and STS1a-3 (STS13) genes than STS1 was suppressed at 35°C compared to 25°C. Taken together, the results of this study will be useful to future investigations of the molecular mechanisms of resistance to high temperature and the development of grape varieties that are tolerant to high temperature conditions.