Introduction
Materials and Methods
Plant Materials
Irradiation Methods
Statistical Analysis
Results and Discussions
Effect of Ionizing Radiation on Seed Germination
Effect of Ionizing Radiation on Embryo Malformation Rate
Effect of Radiation on Embryo Survival Rate
Half-lethal Dose of Radiation for Lavender Seeds
Mutation Rates and Mutation Spectra of Radiations
Discussion
Conclusion
Introduction
Ion beams have been widely used as highly effective mutagens in plant breeding, as they yield higher mutation rates and a wider mutation spectrum than do other traditional low linear energy transfer (LET) ionizing radiations, such as electron beams, X-rays, and gamma rays (Okamura et al., 2003). Ion beams differ from gamma rays in terms of their action on living organisms; the mutating effect of ion beams is dependent on the LET and ion type used (Tanaka et al., 1997; Hase et al., 2002). Tanaka et al. (1997) investigated the ionizing effects of He, C, Ar, and Ne ions over a wide LET range from 17 to 549 keVㆍμm-1 and of electrons with a LET of 0.2 keVㆍμm-1. They reported that plant ecotypes had different sensitivities to these ion beams, and further, that the survival curves werenonlinearly related to the LET, with the peak value of the survival curves being 113 keVㆍμm-1 (Tanaka et al., 1997).
Proton beams have long been used as low-LET ionizing radiation. The LET values for 9 GeV, 645 MeV, and 50 MeV protons are 0.23, 0.25, and 1.25 keVㆍμm-1, respectively. These values are close to the LET values of electron beams or gamma rays (value of gamma rays 0.2 keVㆍμm-1); however, they are considerably lower than those of heavy-element ion beams. Further, previous studies indicated that low-energy protons (<10 MeV, >4.6 keVㆍμm-1) have higher relative biological effectiveness (Yang, 1999). Consequently, low-energy protons can be considered as promising mutagens in plant breeding.
Ion beams induce DNA damage and genomic instability in cells by generating reactive oxidative species (Esault et al., 2010; Mughal et al., 2012), which in turn inhibit cell division and cell elongation (Vazquez-Tello, 1996) and suppress tissue regeneration and germination (Zhou et al., 2006; Wu and Yu, 2001). In the context of mutation by ion beams, it has been reported that exposure of roses, carnations, and wheat to ion beams can induce a wide variety of mutations in flower color and flower shape (Okamura et al., 2003; Wu and Yu, 2001). The mechanisms underlying the biological effects of low-energy ion beams on plants may involve a combination of energy deposition, mass deposition, and the exchange of mass and charge (Lin and Yu, 1996).
Lavender is widely grown for its essential oils in Asia and Europe. However, the oil content of lavender has gradually declined, as has the oil quality. Consequently, mutation breeding using proton ion beams has been considered a promising method to generate lines with improved properties. However, the radiation sensitivity of lavender to protons and electrons has not thus far been reported. Therefore, in the present study, we evaluated the effect of low-energy protons, electrons, and 60Co gamma rays on lavender seed germination and survival, with the aim of developing salt-resistant lavender mutants and mutants with higher oil contents.
Materials and Methods
Plant Materials
Seeds of Lavandula angustifolia purchased from BOlier (BOlier Company, the Netherlands) were stored at 4°C after irradiation. The seeds used in the present study were 2.15 mm in length and 1.10 mm in width. The thickness of the seed coat was 0.07 mm, and the weight of 1000 seeds was 1 g.
Irradiation Methods
Proton beam irradiation: The seeds were irradiated by proton beams using a tandem electrostatic accelerator operating at 2×1.7 MV (Peking University facility, Beijing, China) under vacuum. The ion dosage regulated by electrical current was set at 4, 50.5, 101, 202, 404, and 4040 Gy, in that order. Seeds of 1 g weight were used for each treatment, which was carried out thrice for each dose. The protocols used for the irradiation of seeds by proton ions have been described elsewhere (Qin et al., 2007). Seeds exposed to vacuum were used as the control.
Electron beam irradiation: The electron beams that were used to treat the seeds were generated using the BF-5 Electron Linear Accelerator at Beijing Normal University (Beijing, China). The electron beam doses used in this study were 100, 200, 300, 400, 600, 800, and 1000 Gy, in that order. The beam energy was 3 MeV with a dose rate of 100 Gyㆍmin-1. Each irradiation treatment was repeated three times, and 1.0 g of lavender seeds was irradiated each time.
Gamma-ray irradiation: Gamma rays from the 60Co gamma-ray source at Peking University were used to irradiate theseeds. Lavender seeds were exposed to gamma-ray doses of 20, 50, 100, 150, 200, 250, and 300 Gy, in that order, at a dose rate of 10 Gyㆍmin-1. The gamma rays from the 60Co source had energies of 1.17 MeV and 1.33 MeV. Each dose irradiation was repeated three times. As before, 1.0 g of lavender seeds was irradiated each time, and as mentioned previously, the control seeds were not exposed to gamma-ray irradiation.
Seed germination and in vitro culture: Three replicates were used for each irradiation dose with each replicate containing 100 seeds. After irradiation, these 100 seeds were placed on wet filter paper in a petri dish and incubated at 25°C under continuous light (3500 lux). A seed was considered to have germinated when the root was longer than 2 mm.
The embryos of irradiated seeds were transferred to MS medium not containing any hormones. Cultures were incubated at 25°C under continuous light (3500 lux). The embryo malformation and survival rates were scored after 30 days. Embryos exhibiting no intact leaves, stems, or roots and dwarfs and seedlings that did not flower in the entire growing season were defined as malformed. The survival rate was calculated based on the number of embryos that grew into seedlings with more than four leaves.
To obtain salt-resistant mutants, salt-stress trials for lavender seeds were performed in vitro in MS medium containing 120 or 200 mmolㆍL-1 of NaCl for 30 days , followed by a 30-day recovery in regular MS medium, and then another 30 days on the high salt media. Subsequently, the surviving seedlings were scored when they grew new shoots.
The mutation rates and mutation spectra were measured in the field, and the oil contents of the two strains were measured. The oil content of the control without radiation treatments was 0.98 ± 0.12%.
Statistical Analysis
Statistical analysis was performed using the SPSS 17.0 software package. Analysis of variance (ANOVA) and least significant difference (LSD) tests were performed.
Results and Discussions
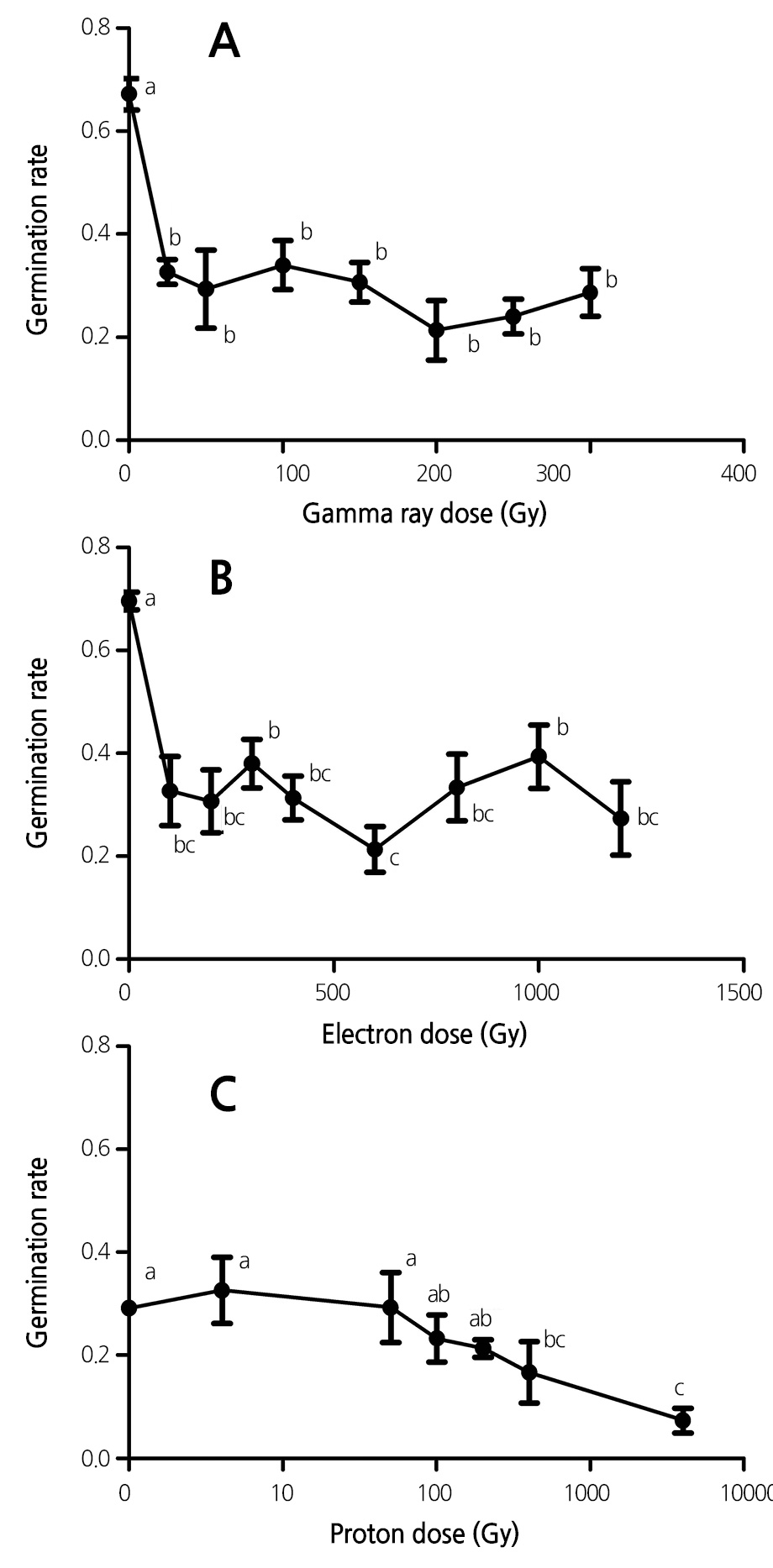
Effect of Ionizing Radiation on Seed Germination
The germination rates of lavender seeds exposed to the three types of ionizing radiation (60Co gamma rays, 3 MeV electrons, and 1.89 MeV protons) were compared. As shown in Figure 1, exposure of lavender seeds to the three ionizing radiations led to a significant reduction in the seed germination rate. Moreover, the radiation-induced reduction in seed germination rate was dependent of radiation dose used in the experiment at low doses (Fig. 1). For example, the seed germination rate was reduced from 67.2% to 29.3% after exposure to 50 Gy of 60Co gamma rays, and the seed germination rate was relatively constant when the radiation dose was further increased up to 300 Gy. A similar trend was observed for electron and proton beams (Fig. 1).
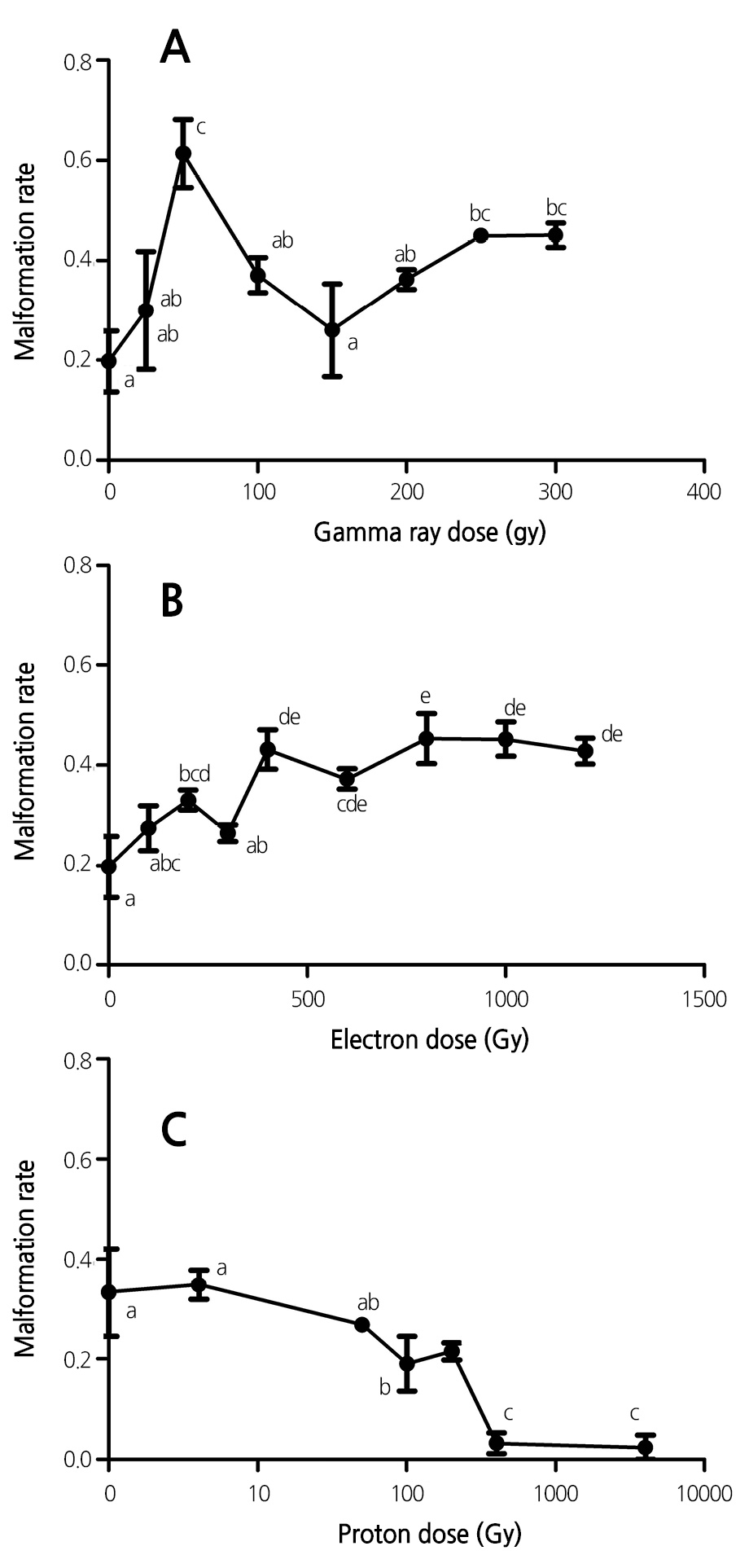
Effect of Ionizing Radiation on Embryo Malformation Rate
To examine whether the ionizing radiation affects embryo development in addition to seed germination, we next investigated the effect of the radiation on embryo development by determining the embryo malformation rate. Exposure of lavender seeds to gamma rays markedly enhanced the embryo malformation rate, and the embryo malformation rate increased with gamma-ray dose (Fig. 2). For example, the malformation rate was found to reach a peak after exposure to agamma-ray dose of 50 Gy, and a further increase in gamma-ray dose attenuated the gamma-ray-induced embryo malformation rate (Fig. 2A).
Similarly, exposure of lavender seeds to electron beams also induced an increase in the embryo malformation rate (Fig. 2B). The rate of the electron-beam-induced embryo malformation was less than that for gamma rays. In addition, electronbeam- induced embryo malformation was positively dependent on the electron-beam dosage.
By contrast, exposure of seeds to proton beams significantly reduced the malformation rate, and the effect was positively correlated with the proton-beam dosage (Fig. 2C). For example, exposure of lavender seeds to proton-beam doses of 50.5 and 404 Gy reduced the malformation rate from 33.3% to 26.8% and 3.2%, respectively. One reason for this reduction was that the physiological damage caused by the proton ion beam to the plant tissue was less than that caused by gamma rays and electron beams. In addition, a slight “injury” from proton beams can activate the damage repair system of lavender, thereby further reducing the damage (De Micco et al., 2011). Furthermore, most of the kinetic energy of proton beams with dosages of 404 Gy and 4040 Gy is transformed into thermal energy, and consequently, the lavender seeds were burnt by the excess heat; these seeds did not germinate or survive for a sufficiently long period to test for malformation, particularly for damaged seedlings (Figs. 1C and 3C).
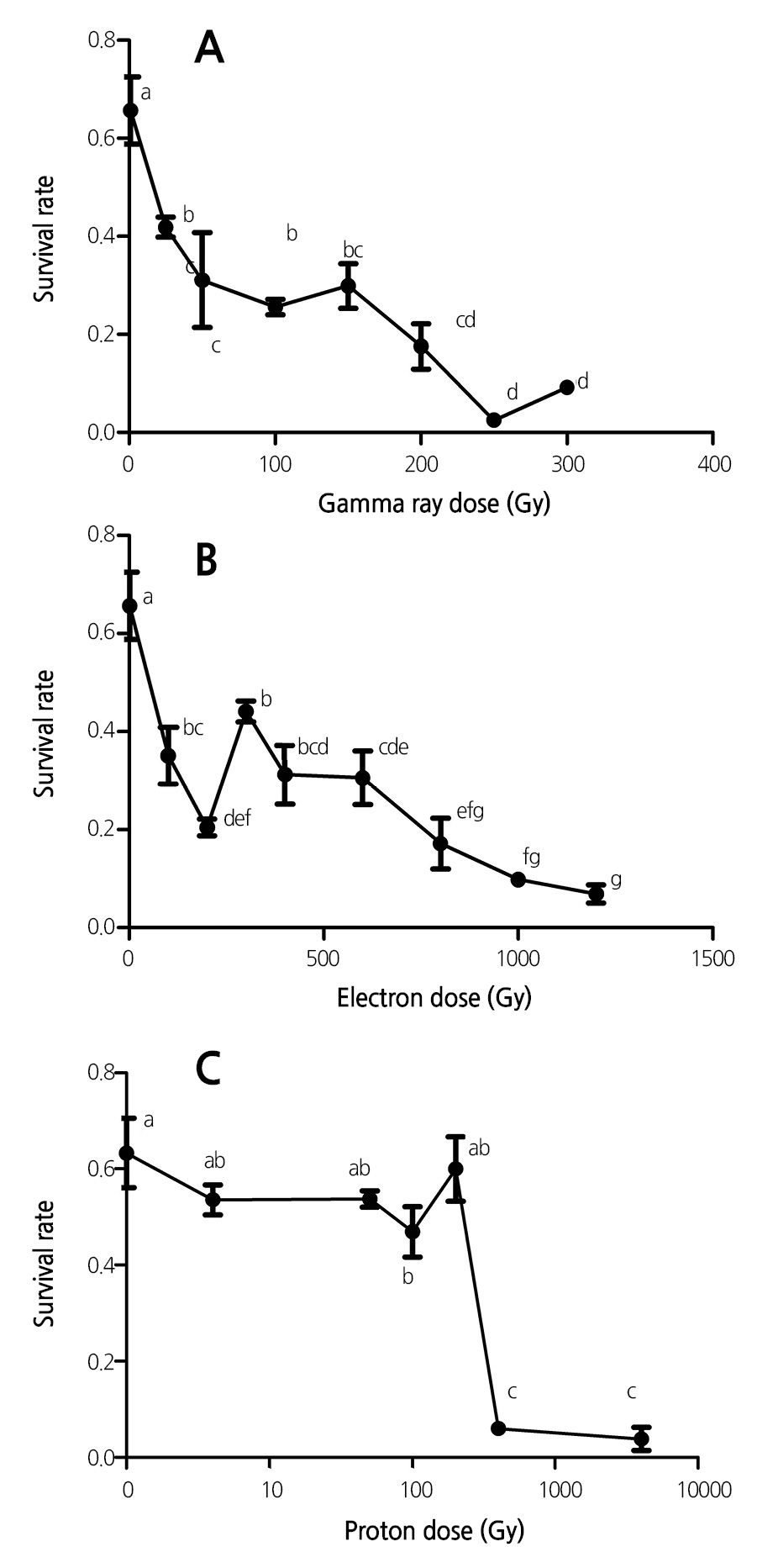
Effect of Radiation on Embryo Survival Rate
In general, exposure of lavender seeds to the three ionizing radiations led to a decrease in the embryo survival rate, and further, the reduction in survival rate was dose-dependent (Fig. 3). Moreover, the reduction in survival rate induced by gamma-ray irradiation was more than those induced by electron and proton beams. The magnitude of reduction in the embryo survival rate after low-dose proton-beam irradiation was less than those in the cases of gamma rays and electron beams. For example, irradiation with a proton-beam dose of 101 Gy reduced the survival rate by 16.5%; however, an increase in the irradiation dose to 404 Gy suppressed the survival rate to 6.0% (Figs. 3C, 7C, and 7E).
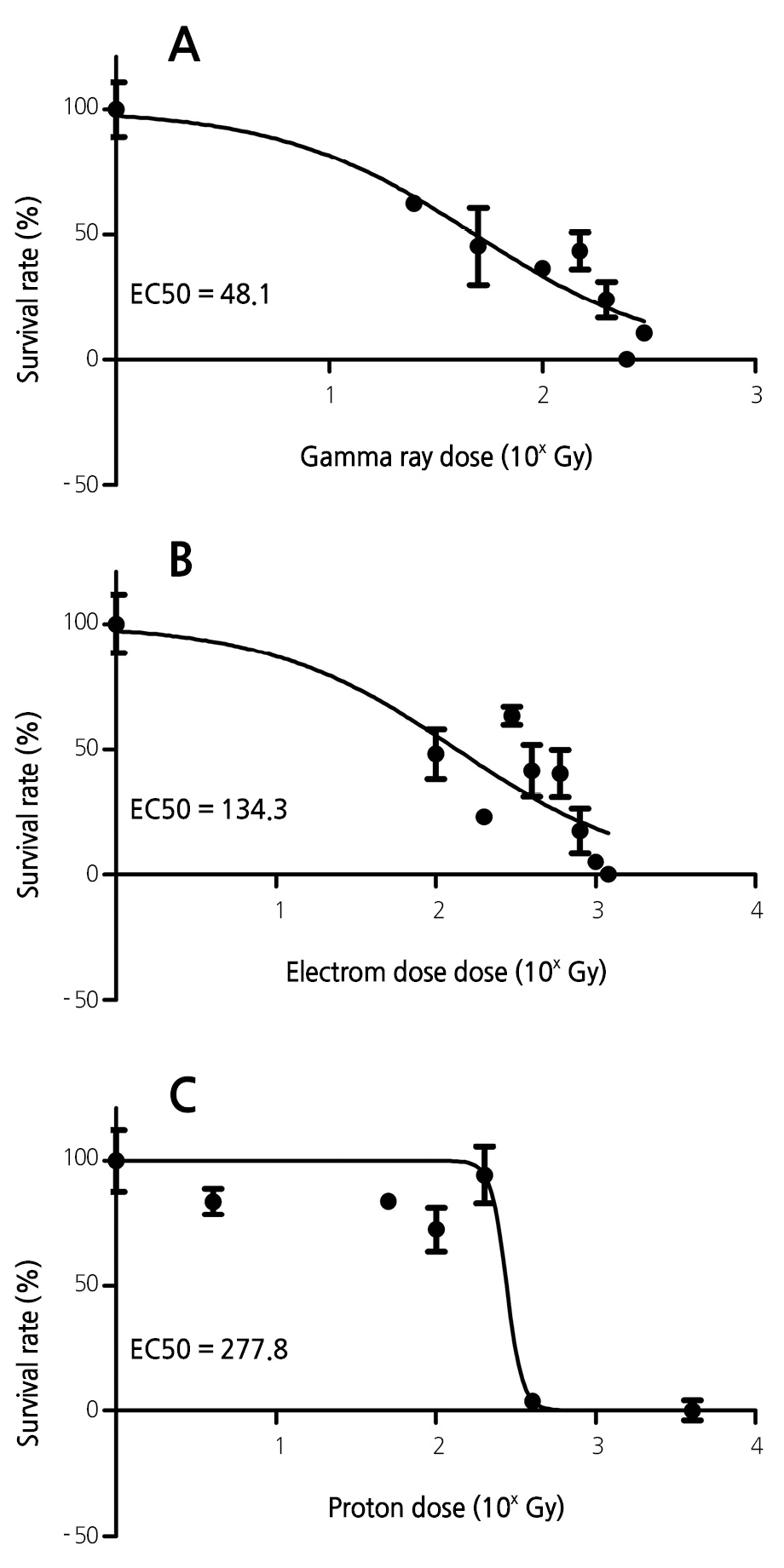
Half-lethal Dose of Radiation for Lavender Seeds
The half-lethal dose is an important parameter in mutation breeding. Different types of radiation have different half-lethal doses for a given species or organ. To compare the half-lethal doses of gamma rays, electrons, and protons, the dose data were converted to their respective logarithm values, plotted, and fitted with sigmoidal fitting curves (Fig. 4). The half-lethal dose (EC50) values obtained were 48.1 Gy, 134.3 Gy, and 277.8 Gy for gamma rays, electrons, and protons, respectively. These results reveal that gamma rays reduce lavender survival rates to a greater extent than do electron and proton beams s, as evidenced by the lower EC50 value of gamma rays.
Mutation Rates and Mutation Spectra of Radiations
The mutation rates induced by electron and proton beams were higher than that induced by gamma rays, and the mutation spectra of the electron and proton beams were wider than that of gamma rays. More salt-resistant mutants were generated from electron-beam and proton-beam radiation than from gamma ray radiation. Further, one clone with elevated levels of essential oils (oil content of 1.21%) was obtained from electron-beam irradiation and another (oil content of 1.35%) from proton-beam irradiation (Table 1). The types of mutants arising from lavender seeds irradiated with electron and gamma rays are shown in Fig. 8.
Discussion
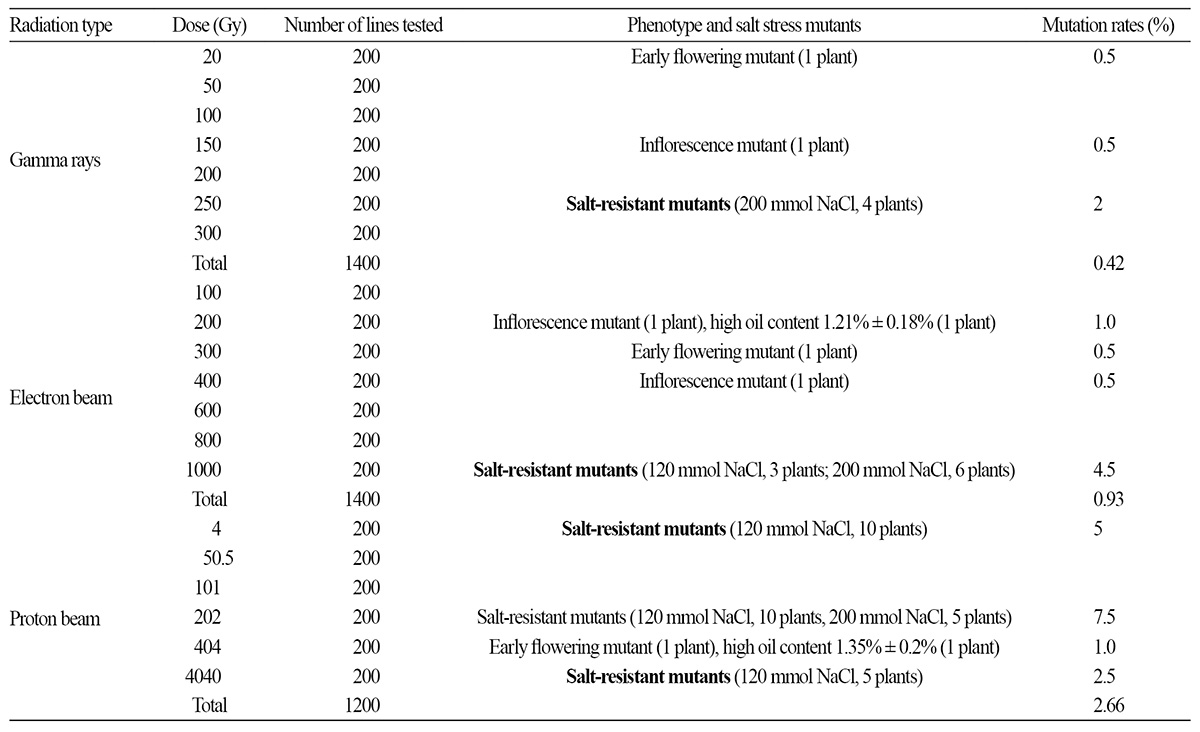
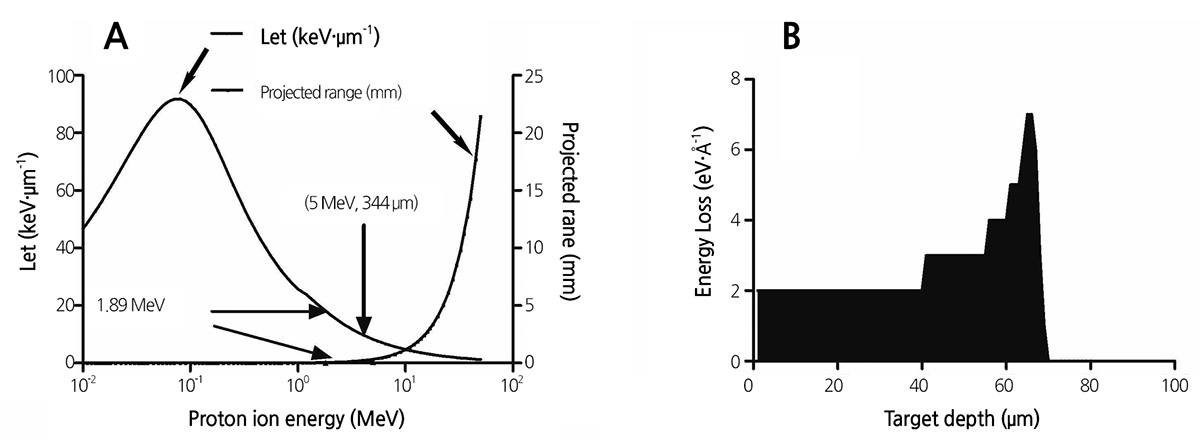
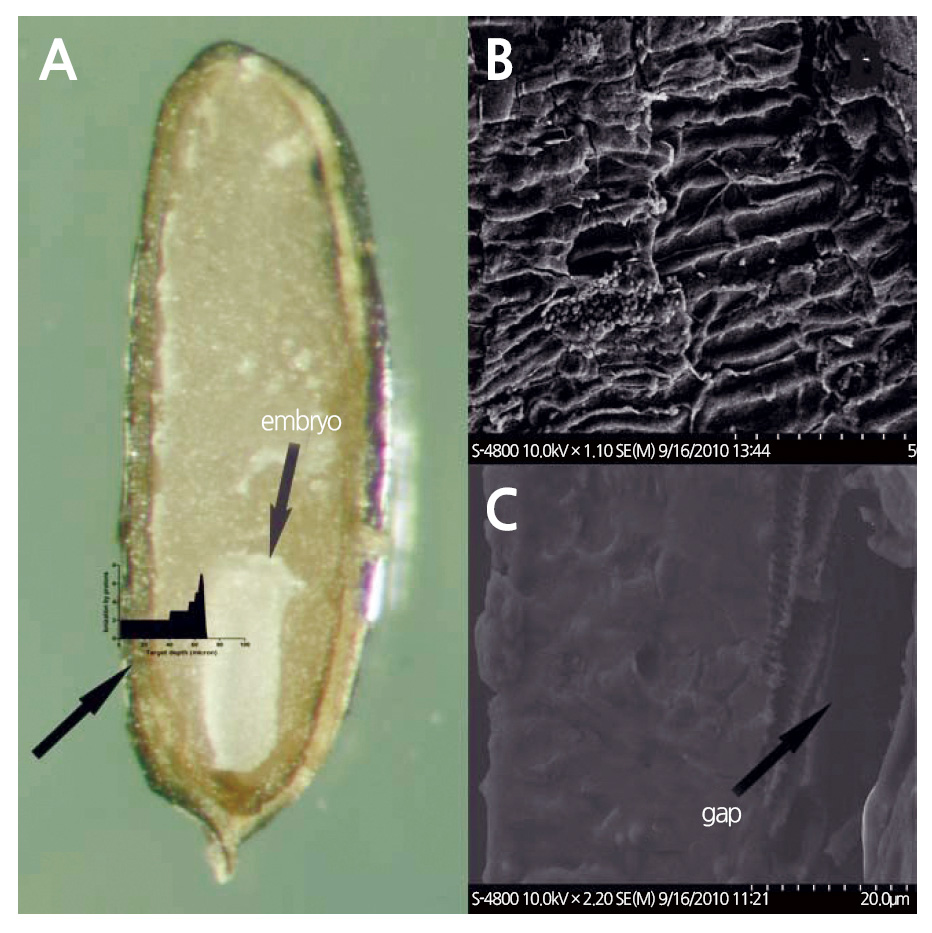
The relatively small size of the lavender seed and its thin seed coat may allow low-energy proton beams to pass through the seed coat and target the enclosed embryo. We calculated the energy range of protons targeting the embryo using SRIM (2013 version) software, and found that 1.89 MeV protons can traverse distances of 68.13 μm (Fig. 5B) in pure water and 73.80 μm in a target with a density of 0.9 gㆍcm-3 (Ziegler et al., 2010). The density value of 0.9 gㆍcm-3 is close to the specific gravity of lavender seeds with a seed coat thickness of 70 μm. There are large gaps between the testa and endotesta of lavender seeds (Figs. 6B and 6C), and the seed coat is very spongy and porous, thereby allowing proton-ion penetration. The density of the seed coat may be considerably smaller than 0.9 gㆍcm-3. Therefore, protons can penetrate the coat and reach the embryo to induce mutations.
The biological effectiveness of a radiation beam is closely related to its LET (De Micco et al., 2011). The LET of protons increases with a decrease in energy (Fig. 5A) according to the stopping theory, and the LET of 1.89 MeV protons is 18 keVㆍ μm-1 (Ziegler et al., 2010). The LET values of gamma rays, electrons, 9000 MeV protons, and 10 MeV protons are 0.2, 0.2, 0.23, and 4.6 keVㆍμm-1, respectively (Yang, 1999). The LET of 1.89 MeV protons is higher than those of gamma rays and electrons, and thus, low-energy protons can induce more mutations than other low-LET ionizing radiations. Low-energy proton ion beams have previously been used to irradiate seeds of the model plant Arabidopsis thaliana, and it has been determined that 2.6 MeV ion beams are more effective than 6.5 MeV beams and that water-soaked seeds are more sensitive to proton ion beams than are dry seeds (Qin et al., 2006; Qin et al., 2007). These results suggest that ion beams absorbed by a target cell can cause more damage than ion beams that only pass through the cell. Studies of high-energy proton ion beams have shown that proton beams can generate more significant biological effects at the end of their ion paths (Yang, 1999).
It has been shown that low-energy nitrogen-ion beams with energy of 30 keV can efficiently induce mutations in wheat and rice (Wu and Yu, 2001; Yu et al., 1989). The mechanisms underlying the biological effects of low-energy ion beams on plants may involve a combination of energy deposition, mass deposition, and the exchange of mass and charge (Lin and Yu, 1996). However, low-energy ions do not reach target seeds according to calculations made using the Stopping and Range of Ions in Matter (SRIM 2013) software package (Ziegler et al., 2010). A possible explanation for the deep ionizing effect of low-energy ions on plant cells is secondary X-rays generated from the interaction between ions and the target seed atoms. The recombination of protons with electrons from the target material can lead to the emission of continuous braking X-rays. Braking X-rays can penetrate biological materials deeper than ions and electrons, thus inducing mutations. Therefore, the biological effects of ions or electron beams can be detected beyond their theoretical range of effectiveness (Huang et al., 2009). For example, Lin et al. reported that the effects of electron beams on several flowers are more significant than those of gamma rays (Lin et al., 2002).
A proton ion beam is characterized by its strong ionization (the ionization peak) at the end of the ion path (Fig. 5B). When this ionization peak is located within the embryo, it exhibits a higher LET and a more effective mutation outcome. In this study, we determined that protons penetrate the seed coat and part of the cotyledon, but do not reach the center of the embryo (Fig. 6A). This may explain why the ionizing effect of the proton beam did not increase with increasing dosage. To enhance the ionizing effect of proton beams on lavender seeds, the proton energy should be set to 5 MeV, so that the projection range of protons in lavender seeds is greater than the thickness of lavender seeds. This will distribute the ionization peak at the center of the embryo, thereby leading to more effective mutation.
Ionizing radiations have been widely used in breeding programs to induce morphological, physiological, and genetic mutations (De Micco et al., 2011). Proton-ion beams of low energy have higher LETs than electron beams and gamma rays, and they can form promising mutagens for small-seed plants, single cell algae, and bacteria. The findings presented here provide important guidelines for mutation breeding of lavender.
Conclusion
Prompted by the lack of knowledge regarding the effect of ionizing radiation on lavender, we determined the half-lethal dose of gamma rays, electrons, and protons was 48.1 Gy, 134.3 Gy, and 277.8 Gy, respectively. Our results show lavender seeds irradiated with proton ion beams had a higher survival rate and a lower malformation rate than did those irradiated with gamma rays and electron beams. The proton-ion beam yields low irradiation damage, and it induces mutations with low irradiation damage if the proton energy is enhanced to 5 MeV. In our future studies, we plan to irradiate lavender seeds with 5 MeV protons to induce mutations, with the aim of generating high quality mutants with high oil contents and resistance to salt stress. This dosage of protons can also be used to examine the mutation mechanisms in cells and tissues that lie within the Bragg peak of energetic protons.