Introduction
Materials and Methods
Plant Materials and Growth Conditions
Light Treatments
Measurement of Chlorophyll Fluorescence
Data Collection and Statistical Analysis
Results and Discussion
Introduction
The Phalaenopsis genus (Blume: Orchidaceae) contains approximately 66 species, and comprises the most economically important flowering potted plant around the world because of its easy culture practices, diverse flower colors, unique floral structure, and flower longevity (Christenson, 2001; Chen and Chen, 2011; De et al., 2014). The orchid is commercially cultivated in many countries, includingGermany, Japan, The Netherlands, Taiwan, and the United States of America (De et al., 2014). During commercial production, Phalaenopsis plants are grown for over 12 months to produce mature flowering plants. And, although they have a low light requirement of about 200 - 400 μmol·m-2·s-1 (Lee, 2000; Lopez and Runkle, 2005; Guo et al., 2012), a high temperature (above 28°C) is necessary to prevent the development of immature inflorescence initiation during the vegetative growth period. Heating costs are the largest expense in Phalaenopsis cultivation (Lopez et al., 2007; Pollet et al., 2011; An et al., 2013; Lee et al., 2015); therefore, new cultivation methods or technologies that shorten the cultivation period and reduce heating costs are needed to improve the economic efficiency of Phalaenopsis production.
A closed plant-production system (called a ‘closed system’) allows the precise control of environmental conditions for plant production, regardless of the weather outside (Kozai, 2007). These closed systems can be used to produce high-quality commercial crops in less time by providing optimal growth conditions. Phalaenopsis plants have short stems and can be cultivated at a high plant density during their vegetative growth period; therefore, these plants are suitable crops for multilayer cultivation in a closed system, which may reduce cultivation time and production cost.
For multi-layer cultivation in a closed system, artificial lights are commonly used in the absence of solar radiation, so it is important to determine the optimum lighting source for each crop. During the last few decades, light-emitting diodes (LEDs) has been used as an alternative artificial lighting source for plant growth (Ouzounis et al., 2015). LEDs have attractive characteristics such as wavelength specificity, high electrical efficiency, long operating life, and low heat generation (Bourget, 2008; Craig and Runkle, 2013), allowing researchers to use LEDs in close proximity to the plant canopy and to easily regulate the wavelength and intensity of light reaching the plants (Ouzounis et al., 2015).
Light is an energy source required for photosynthesis, and its characteristics (light quality, quantity, and duration) are important factors regulating plant growth and development (Dole and Wilkins, 2005). Plants perceive light signals through three major photoreceptor families; phytochromes, cryptochromes, and ultraviolet receptors (Kami et al., 2010; Craig and Runkle, 2013). Specific light wavelengths corresponding to particular colors induce different plant growth responses (Dole and Wilkins, 2005); for example, the suppression of stem growth by blue light has been reported in many crops (Casal and Smith, 1989; Runkle and Heins, 2001; Li and Kubota, 2009), while plants grown under high red light conditions showed increases in stem length and leaf size (Runkle and Heins, 2002; Dole and Wilkins, 2005; Son and Oh, 2013; Meng and Runkle, 2015).
In Phalaenopsis, some studies have examined the effects of light quality on chlorophyll fluorescence, flowering, and in vitro plant cultivation (Shin et al., 2008; Ouzounis et al., 2015; Dueck et al., 2016). Dueck et al. (2016) suggested that a high red:far-red ratio stimulates the production of multiple inflorescences. However, to our knowledge, no data has been published on the effects of light quality on Phalaenopsis cultivation in a closed system. The objective of this study was to determine the effects of different artificial lighting sources and intensities on vegetative growth of young Phalaenopsis plants, and to determine the optimal lighting source for use in a closed plant - production system for Phalaenopsis cultivation.
Materials and Methods
Plant Materials and Growth Conditions
Young Phalaenopsis ‘Blanc Rouge’ and Doritaenopsis (an intergeneric hybrid between Doritis and Phalaenopsis)‘Mantefon’ plants that had been acclimated for one month in a greenhouse after deflasking, were purchased from a commercial grower (Sang Mi Orchids, Taean, Korea) on May 14, 2015. The plants were transplanted into 4 - cm transparent plastic pots containing sphagnum moss and grown in a closed plant - production system at the Experimental Farm of Seoul National University, Suwon, Korea. The mean leaf spans were 9.7 and 11.9 cm in ‘Blanc Rouge’ and ‘Mantefon’, respectively, at the start of photo - treatments. Ten plants from each cultivar were placed on growth beds for each light treatment, in a completely randomized design. The temperature and relative humidity in the plant - production system were maintained at 28 ± 1°C and 60 ± 10%, respectively. The plants were fertigated once a week with water soluble fertilizer (electrical conductivity: 1.0 mS·cm-1; Technigro 20N - 9P - 20K, Sun - Gro Horticulture, Bellevue, WA, USA) by hand - drip irrigation.
Light Treatments
The one - month - old ‘Blanc Rouge’ and ‘Mantefon’ plants were cultivated under either fluorescent lamps (FL40EX - D, Kumho Electric Inc., Seoul, Korea) at 80 μmol·m-2·s-1 (F80) or 160 μmol·m-2·s-1 (F160), cool - white LEDs (GMG Korea Inc., Busan, Korea) at 80 μmol·m-2·s-1 (C80) or 160 μmol·m-2·s-1 (C160), or warm-white LEDs (GMG Korea Inc., Busan, Korea) at 80 μmol·m-2·s-1 (W80) or 160 μmol·m-2·s-1 (W160). The relative quantum flux was measured using a spectroradiometer (StellarNet, Tampa, FL, USA), and the blue (400 - 500 nm) : green (500 - 600 nm) : red (600 - 700 nm) : far - red (700 - 800 nm) ratios of the fluorescent lamps, cool-white LEDs, and warm-white LEDs were 1 : 1.3 : 0.8 : 0.1, 1 : 1.3 : 0.6 : 0.1, and 1 : 2.7 : 2.3 : 0.4, respectively (Fig. 1). The photoperiod was set to 12 h. Each treatment was maintained for 16 weeks in a closed plant - production system.
Fig. 1.
Relative spectral distributions of the light emitted by the (A) fluorescent lamps, (B) cool - white light-emitting diodes (LEDs), and (C) warm - white LEDs used in this study.
Measurement of Chlorophyll Fluorescence
The chlorophyll fluorescence of the uppermost mature leaf was measured using a PAM chlorophyll fluorometer (PAM 2000, Heinz Walz, Effeltrich, Germany) after 16 weeks of light treatments. Six plants were randomly selected and placed in the dark for 30 min. After dark adaptation, the minimal fluorescence (Fo) with a weak red light and the maximum fluorescence (Fm) with a saturating white light at 207 μmol·m-2·s-1 of the uppermost mature leaf were measured in order to calculate the maximum quantum efficiency of photosystem II (Fv / Fm). The yield of variable fluorescence (Fv) was calculated from the equation Fv = Fm - Fo.
Data Collection and Statistical Analysis
The number of new leaves (leaves longer than 0.5 cm developed after the treatment), leaf span (the length across the plant from leaf tip to the opposite), and the length and width of the uppermost mature leaf were measured every four weeks for each plant. The fresh weight of shoots and roots was measured at the end of treatments, and the dry weight was measured after drying at 80°C for 72 hours. The relative chlorophyll content of the uppermost mature leaf was measured every four weeks for each plant using a SPAD meter (SPAD 502, Konica Minolta Sensing Inc., Sakai, Osaka, Japan). Statistical analyses were performed by ANOVA in the SAS system for Windows (version 9.3; SAS Institute Inc., Cary, NC, USA). Comparisons among treatments were performed using Duncan’s multiple range test at p ≤ 0.05. Graph module analyses were performed using SigmaPlot software (version 10.0; Systat Software, Inc., Chicago, IL, USA).
Results and Discussion
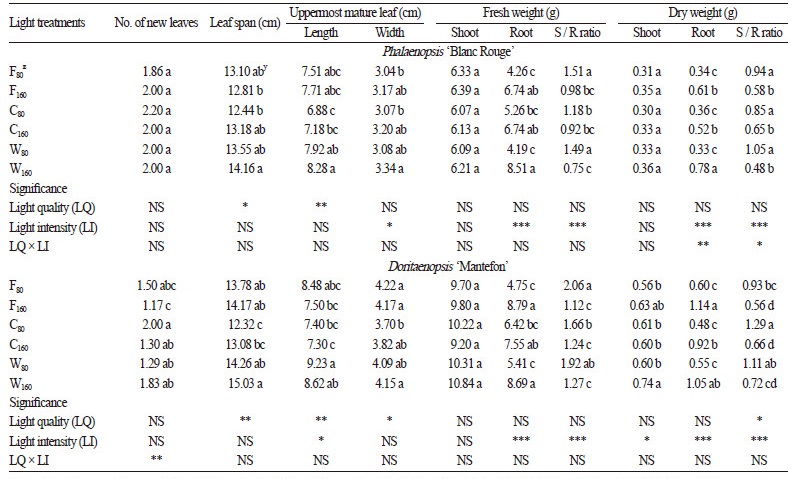
High light intensity promoted vegetative growth of both the young Phalaenopsis and Doritaenopsis plants (Table 1, Fig. 2A and B). The leaf span was slightly greater in ‘Blanc Rouge’ plants treated with 160 μmol·m-2·s-1 light, regardless of the lighting source, although these differences were not statistically significant. The width of the uppermost mature leaf in ‘Blanc Rouge’ plants was also increased under the higher light intensity ( p≤0.05), but the length was not affected by light intensity. The fresh and dry weights of the ‘Blanc Rouge’ and ‘Mantefon’ roots were significantly increased under the higher light intensity ( p≤0.001 in both cases). In the ‘Mantefon’ plants, a larger leaf span and a wider uppermost mature leaf were observed under the high-intensity light conditions, and the shoot dry weight slightly increased under the higher light intensity ( p≤0.05). These results, showing that high light intensity promotes the growth and development of Phalaenopsis plants, has also been reported by other researchers (Kubota and Yoneda, 1993; Wang, 1995; Lootens and Heursel, 1998; Lin and Hsu, 2004; Guo et al., 2012). Guo et al. (2012) reported that the nocturnal CO2 uptake rate increased with high light intensity in Phalaenopsis ‘TS97’, and Wang (1995) found that the spike development and anthesis of plants grown under 60 or 160 μmol·m-2·s-1 light conditions occurred earlier than plants grown under 0 or 8 μmol·m-2·s-1 light in ‘Joseph Hampton’. Since plants were only treated with two different light intensities in the present work, further studies using a wider variety of light intensities are still required to determine the optimum light intensity for Phalaenopsis plants.
Fig. 2.
Vegetative growth of young (A) Phalaenopsis ‘Blanc Rouge’ and (B) Doritaenopsis ‘Mantefon’ plants after 16 weeks of light treatments. F80 and F160, fluorescent lamps at 80 and 160 μmol·m-2·s-1; C80 and C160, cool - white light-emitting diodes (LEDs) at 80 and 160 μmol·m-2·s-1; W80 and W160, warm - white LEDs at 80 and 160 μmol·m-2·s-1, respectively.
The longest leaf span was observed in both cultivars when grown under warm - white LEDs (Table 1, Fig. 2A and B). The length of the uppermost mature leaf was significantly longer in both ‘Blanc Rouge’ and ‘Mantefon’ plants when treated with warm-white LEDs than other lighting sources ( p ≤ 0.01), with the shortest leaves observed in the cool - white LEDs treatment. Plants grown under fluorescent lamps showed longer leaf lengths than plants grown under cool - white LEDs, although there were no significant differences in fresh or dry weights among lighting sources. These results indicate that red light mainly contribute to the growth of these orchids, since the red light ratio was the highest in warm - white LEDs and lowest in cool - white LEDs (Fig. 1B and C) which induced more and less growth, respectively. Many studies have found that red light positively affects the growth of various crops such as chrysanthemum, impatiens, lettuce, petunia, salvia, and tomato (Khattak and Pearson, 2006; Son and Oh, 2013; Wollaeger and Runkle, 2014). In a study by Wollaeger and Runkle (2014), the leaf areas of impatiens, tomato, salvia, and petunia plants were greater under higher red light conditions. However, the biomass of shoot and root was not significantly affected by light quality in this study. These results might beattributed to the limited short growth of the Phalaenopsis plants.
A significant interaction between light quality and intensity was observed in the root dry weight of ‘Blanc Rouge’ plants and in the number of new leaves of ‘Mantefon’ plants (Table 1); however, the general growth responses to light quality or intensity showed little interaction between the two effects in both cultivars. Previous researches using Arabidopsis has suggested that light quality is involved in the acclimation response to light intensity (Anderson et al., 1995; Walters and Horton, 1995; Wagner et al., 2008). However; the impact of the interaction between light quality and intensity on cultivation is unclear. The shoot/root dry weight ratio ranged from 0.48 to 1.05 and from 0.56 to 1.29 in the ‘Blanc Rouge’ and ‘Mantefon’ plants, respectively (Table 1), with lower ratios in the plants treated with 160 μmol·m-2·s-1 light. In a study by Miralles et al. (2011), a decrease in light intensity by shading increased the shoot / root ratio of Rhamnus alternus. A phenomenon that could be explained by a reduction in evaporative demand under shaded conditions, affecting the water absorption needs, thereby possibly reducing the root growth (Rhie et al., 2014). It can therefore be expected that the relatively higher light intensity in our study would have influenced the water absorption demand and root growth.
Typically, the chlorophyll content is related to the rate of plant growth (Brougham, 1960; Son and Oh, 2013); however, no differences in chlorophyll content were noted among different light qualities despite the increased growth rate under high red light conditions (Table 1, Fig. 3A and B). Chlorophyll content tends to increase in more shaded plants, possibly asan adjustment to the low-light conditions (Mendes et al., 2001; Chen et al., 2005; Scuderi et al., 2012), which was also observed in the present study, where the lower light intensity slightly increased the chlorophyll content in the intergeneric hybrid Doritaenopsis. Several studies have reported that the light saturation point of Phalaenopsis plants is about 130 - 200 μmol·m-2·s-1 (Ota et al., 1991; Lootens and Heursel, 1998; Guo et al., 2012); therefore, the 80 μmol·m-2·s-1 used in this study was insufficient to meet the light requirements of the ‘Blanc Rouge’ and ‘Mantefon’ plants.
Fig. 3.
Relative chlorophyll content (SPAD value) of young (A) Phalaenopsis ‘Blanc Rouge’ and (B) Doritaenopsis ‘Mantefon’ plants after 16 weeks of light treatments. The data shown are the mean ± SE (n = 8). Differences were considered significant at p≤0.05.
The maximum quantum efficiency (Fv / Fm) of non-photoinhibited leaves appears to be about 0.8 - 0.83 (Maxwell and Johnson, 2000; Lin and Hsu, 2004). In this study, Fv / Fm ranged from 0.78 to 0.81 in ‘Blanc Rouge’ plants and from 0.77 to 0.81 in ‘Mantefon’ plants (Fig. 4), which was similar to the results of normally grown Phalaenopsis plants in other studies (Lin and Hsu, 2004; Hsu, 2007; Pollet et al., 2009). Under high blue - light conditions, Fv / Fm was slightly higher, which may be attributed to the participation of blue light in photosynthetic acclimation to environmental cues (Anderson et al., 1995; Walters, 2005). Ouzounis et al. (2015) also found that the Fv / Fm value under 100% red light was lower in Phalaenopsis plants when compared with light conditions that contained blue light; thus, we conclude that a certain amount of blue light is necessary for efficient photosynthesis in Phalaenopsis plants.
Fig. 4.
Maximum quantum efficiency of photosystem II (Fv / Fm) of the uppermost mature leaves of young (A) Phalaenopsis ‘Blanc Rouge’ and (B) Doritaenopsis ‘Mantefon’ plants after 16 weeks of light treatments. The data shown are the mean ± SE (n = 6). Differences were considered significant at p ≤ 0.05.
In conclusion, vegetative growth of Phalaenopsis plants was promoted by a higher light intensity.; however, since light requirements differ among cultivars, by plant age, or following different acclimation conditions, a more detailed study is needed to determine the optimum light intensity conditions. Warm - white LEDs that contain a high red - light ratio improved vegetative growth of ‘Blanc Rouge’ and ‘Mantefon’ plants, indicating that they are a better lighting source than cool - white LEDs or fluorescent lamps for vegetative growth of Phalaenopsis plants. The results of this study could therefore be useful in the selection of artificial lighting for use in closed plant-production systems in order to effectively promote vegetative growth of Phalaenopsis plants.