Introduction
Materials and Methods
Plant Material
Seed Dormancy Classification
Germination Characteristics of Stored Seeds
Experiment Management and Statistical Analysis
Results
Morphological Characteristics of Seeds
Seed Germination ConditionsMorphological Parameters
Germination Characteristics are Affected by Storage Condition
Changes of Endogenous GAs and ABA Concentration in M. rossii Seeds
Discussion
Introduction
Mukdenia rossii (Oliv.) Koidz., of the family Saxifragaceae, is a perennial herb. In Korea, the species is known as ‘Doldanpung’ because it is usually found in the cracks of rocks, and it has 5-7 maple-shaped leaves at the end of a long petiole. The species is mostly found in Gangwon-do, Gyeonggi-do, and Pyeonganbuk-do in the Republic of Korea. Its habitat includes areas around valleys and cliffs, and cracks in stone. The plant grows up to 30 cm in length. The panicles usually form in May, with pink-accented white flowers at the end of peduncle (Lee, 2003), and the tender leaves of M. rossii are consumed as a vegetable (Food Materials Information, 2017). Furthermore, its demand as a horticultural crop has increased due to its high ornamental value.
Seed dormancy is highly related to the plant growth conditions (Geneve, 2003). Various seed dormancy classifications have been proposed; Lang (1987) simplified the types of seed dormancy to eco-dormancy, para-dormancy, and endo-dormancy based on endogenous and exogenous germination factors. However, Baskin and Baskin (2004) reported biological and morphological factors that explain types of dormancy and concluded that there are five types: physiological dormancy (PD), morphological dormancy (MD), morphophysiological dormancy (MPD), physical dormancy (PY), and combinational dormancy (PY+PD); and each type requires a different dormancy-breaking method.
Freshly matured nondormant (ND) seeds usually exhibit a germination rate of over 80% within four weeks in the proper germination condition (Baskin and Baskin, 2004). However, the long-term storage of seeds might negatively affect the rate of and energy for germination. Furthermore, the storage temperature and period required to maintain seed viability can differ by species (Tagaki, 2001a, 2001b; Chang and Lee, 2007; Lee et al., 2014). Storing seeds under conditions similar to their natural habitat might extend their dormancy and suppress germination (Lee et al., 2014).
Priming seeds can improve their rate of germination. Priming refers to submerging seeds in a solution of low water potential for a certain period before sowing to control their absorption of moisture (Akers and Holley, 1986). Further, priming reduces the number of days to germination and increases seed water content by activating enzymes involved in amino acid and mRNA synthesis, and DNA replication in the seeds (Braford et al., 1988; Norton, 1988). Priming also rejuvenates the functions of the cell membrane, which prevents stored nutrient loss and improves germination potential (Coolbear et al., 1984; Khan, 1992).
Gibberellic acid (GA) is a growth regulator that can be widely applied for breaks seed dormancy and improves germination rate, and can be used as a substitute for low temperature and priming treatments (Abe et al., 1991). Therefore, the inhibition of germination caused by low endogenous GA levels can be overcome with exogenous GA treatment. This phenomenon has been observed in Primula spp., barley, wild ornamental plants, and horseradish (Khan and Tolbert, 1969; Chavagnat and Jeudy, 1981; Fukai and Oe, 1990; Kim and Um, 1995; Kim et al., 1996). Furthermore, the relative balance in the levels of endogenous abscisic acid (ABA) (inhibitor) and GAs (promoter) in seeds is key to its germination, as this balance determines whether the seed germinates or enters dormancy (Finkelstein et al., 2008; Yamaguchi, 2008; Nambara et al., 2010; Linkies and Leubner-Metzger, 2012).
Findings in our study indicate that seed storage temperature and period are closely related to changes in the content of endogenous hormones. To date, studies of seed storage in plant native to Korea have been limited to germination rate vis-a-vis temperature and period of storage (Tagaki, 2001a, 2001b; Chang and Lee, 2007; Hwang et al., 2012). The present study aimed to establish conditions that improve germination rate and storage conditions of M. rossii seeds to maintain long-term seed vitality in ND seeds.
Materials and Methods
Plant Material
The seeds were collected from the dried capsules of Mukdenia rossii (Oliv.) Koidz. plants gathered from around Andong-si, Gyeongsangbuk-do, Republic of Korea in May 2014. The capsules were dried for 48 h at 23 ± 2.0°C, and then sieved (850 µm) to sort the seeds from other plant material.
Only morphologically regular seeds were used in the present study. Seed length (mm) and width (mm), and the weight of 1,000 seeds (mg) were measured using a digital vernier caliper (NA500-150S; Bluebird, China) and electronic scale (IB-610S; Innotem, Korea), respectively. Each measurement was repeated 10 times. The seed moisture content was measured as the difference in weight before and after drying the seeds at 70°C with hot air for 48 h.
Seed Dormancy Classification
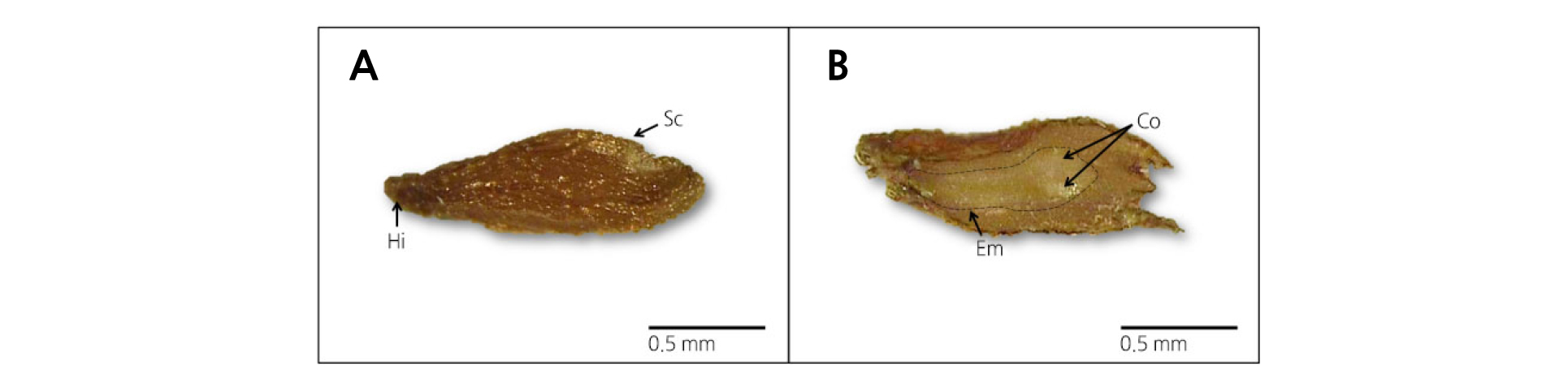
The seed dormancy type was determined according to Nicolaeva (2001), Baskin and Baskin (2004), and Cho et al. (2016, 2018) by the following three steps. First, selected seeds were cut along the major or minor axes using a stainless blade (Platinum ST-300; Dorco, Korea). The cross-section was photographed using an icamscope (ICS, Sometech inc., Korea) and then observed using IT Plus 4.0 software. Subsequently, the length of embryo was calculated as a ratio to seed length (E:S ratio, %). The embryo development status was distinguished based on criteria such as whether the cotyledon was differentiated or developed but not differentiated, and whether the embryo was immature or non-existent. Second, the selected seeds were submerged in distilled water for 24 h at 4 ± 1.0°C and then placed in Petri dishes (∅8.9 cm) lined with two sheets of filter paper (∅90 mm, Advantec, Toyo, Japan). The Petri dishes were then placed in growth chambers (LI-1400, LI-COR; Lincoln, NE, USA) set for continuous light (23 ± 0.5 µmol·m-2·s-1 PPFD) or dark conditions at a temperature of 20°C. Filter paper were wetted with sterilized water every day to avoid drying and percent germination (%) was calculated by totaling all the seeds that germinated in 30 d. Third, the selected seeds were submerged in water for 7 d and the moisture absorption ratio was recorded. The seeds were submerged in test tubes containing 15 mL distilled water and then refrigerated at 4 ± 1.0°C. Every 24 h, the seeds were taken out and the seed coat was wiped with filter paper, weighed, and then submerged in test tubes containing distilled water. The interval moisture absorption rate (%) was calculated by the accumulative moisture content. The average weight of seeds was also calculated at every 24 h.
Germination Characteristics of Stored Seeds
To investigate optimal germination temperature and light conditions, M. rossii seeds that had been stored under dry conditions for 14 months at 4 ± 1.0°C were submerged in distilled water. The soaked seeds were placed on a Petri dish lined with two sheets of filter paper and placed under the following conditions: 15°C, 20°C, 25°C, or 30°C; either in the light (23 ± 0.5 µmol·m-2·s-1 PPFD, continuous light) or dark. To improve the rate of germination and to encourage uniform seedling growth, the seeds were submerged in GA3 (100, 200, and 500 mg·L-1; ≥ 90%, Acros, NJ, USA) and potassium nitrate (KNO3)(5, 10, 20, and 40 mM; 99.0%, Junsei Chemical Co., Ltd., Tokyo, Japan) solutions for 24 h. The treated seeds were washed three times in distilled water, and then their germination characteristics at 30°C in the light were analyzed.
Analysis of Endogenous GAs and ABA in Long-term Stored M. rossii Seeds
The selected seeds were placed in test tubes and sealed in zipper bags along with silica gel (Mr. keeper; Sungel, Korea). They were stored for 2, 4, and 6 months at 25, 4, -20, and -80°C. After the storage period, the germination rate (%) for each specimen was analyzed at 20°C in light conditions, following the methods similar to those used to determine germination characteristics. In addition, the seeds that had been stored for 6 months at different temperatures were used to quantify the endogenous GAs and ABA contents.
Endogenous GAs were extracted from 1.0 g freeze-dried seeds following the procedure described by Lee et al. (1998, 2015). The extracted GAs were chromatographed on a 3.9 × 300 mm Bondapak C18 column (Waters Corp., Milford, MA) and the selected fractions were methylated and subjected to gas chromatography/mass spectrometry (GC/MS) with selected ion monitoring (SIM) (6890N Network GC System and 5973 Network Mass Selective Detector; Agilent Technologies, PaloAlto, CA). In the present study, GA24, GA9, GA7, and GA 4 in the non C-13-hydroxylation pathway and GA20 and GA1 in the early C-13-hydroxylation pathway were quantified.
Endogenous ABA content in seeds was quantified using frozen samples as described by Qi et al. (1998) and Lee et al. (2015). The extracts were combined and evaporated before the residues were dissolved in dichloromethane and passed through a silica cartridge. ABA was recovered by elution with 10 mL diethyl ether:methanol. The extracts were dried and methylated by adding diazomethane for GC/MS-SIM analysis. Lab-Base (ThermoQuset, Manchester, UK) data system software was used to monitor responses to ions of m/e 162 and 190 for Me-ABA, and m/e 166 and 194 for Me-[2H6]-ABA. The concentration of ABA was quantified by the peak area of endogenous and standard ABA samples.
Experiment Management and Statistical Analysis
The filter papers containing M. rossii seeds were continuously moistened with sterilized water and any contaminated Petri dishes were replaced. Every 24 h, the percent germination (PG, %) and mean germination time (MGT, days) were calculated. A germinated seed was defined as a seed with a radicle emerged through the seed coat by at least 1 mm. In addition, the germination energy (GE, %) was calculated as the ratio of the number of seeds germinated within a certain time period after sowing to the total number of germinated seeds. Furthermore, time taken for germination rate to reach 50% (T50) was also calculated.
All germination experiments were repeated four times, with each iteration containing 100 seeds. The SAS version 9.4 (SAS institute Inc., Cary, NC, USA) was used to compute average and standard error. Factor analysis and statistical significance at p < 0.05 used Duncan’s multiple range test.
Results
Morphological Characteristics of Seeds
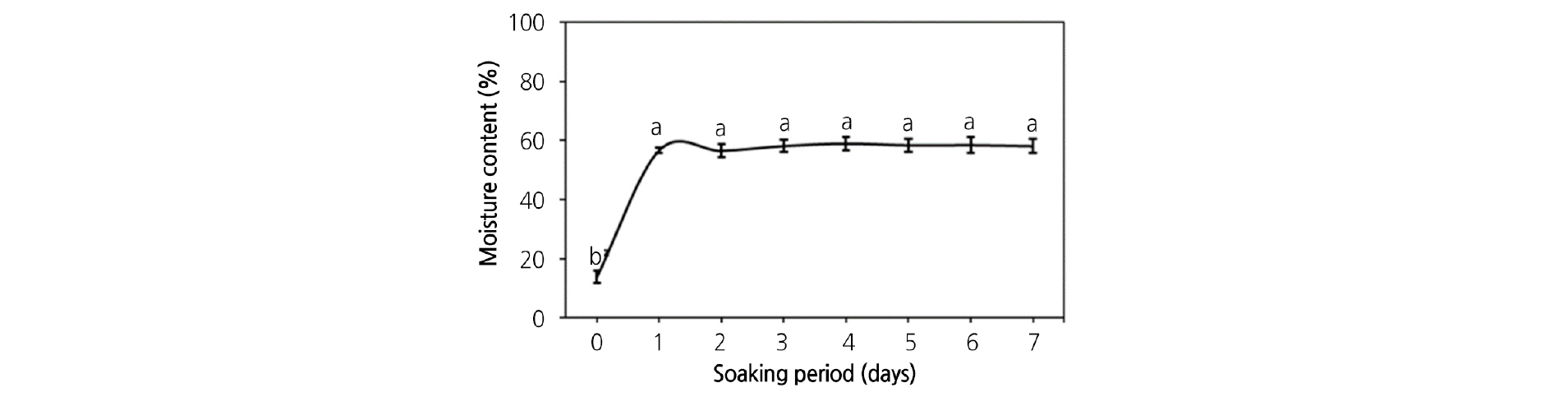
The average M. rossii seed size was 1.15 × 0.28 mm and the weight of 1,000 seeds was 46.9 mg. The seeds contained a distinguishable embryo and endosperm, and the presence of cotyledons could be observed in sufficiently developed embryos (Fig. 1). The E:S ratio of the seeds was 50.9%, and the seeds had an excellent percent of germination of over 95.5% (data not shown). The seeds achieved the maximum moisture content of 56.7% in 24 h and maintained a similar hydration level for 7 d (Fig. 2).
Seed Germination Conditions
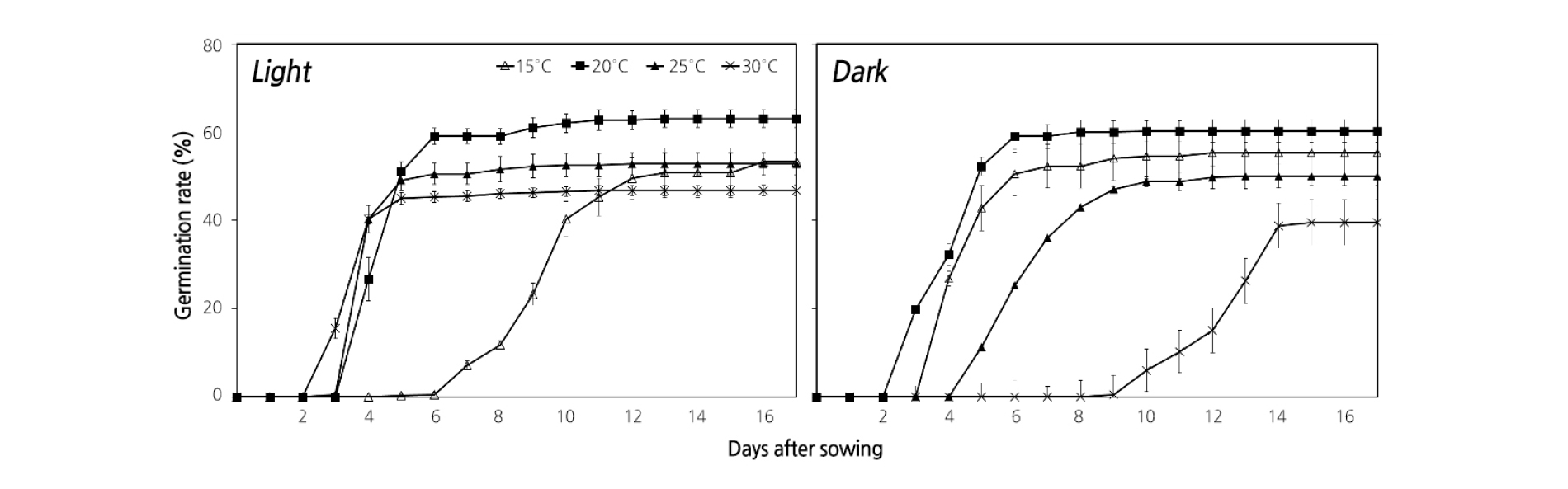
The seeds stored at a low temperature for 14 months were submerged in distilled water for 24 h and used for light and temperature experiments. The rate of seed germination under light conditions at 20°C was the highest (63.0%) (Fig. 3), while. The germination rate at the same temperature in dark conditions was 60.5%. The germination characteristics of M. rossii seeds were influenced less by light than by temperature. In particular, the onset of germination was delayed, at 15°C regardless of the presence of light, at 15°C. In all treatments, germination was completed within 16 d.
Effects of Chemical Treatments on Seed Germination
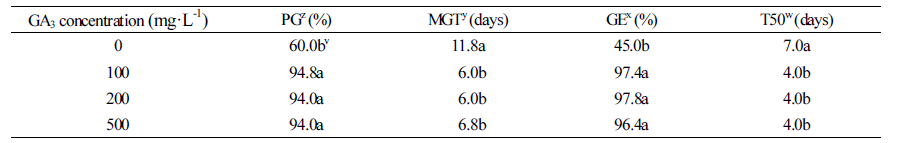
To improve the rate of germination of long-term stored M. rossii seeds, the seeds were treated with chemicals. Gibberellic acid (GA), regardless of its concentration, increased germination rates above 90% and reduced mean germination time (MGT) by at least 5 d (Table 1). Germination energy (GE) was improved by over 50% and T50 was shortened by three 3 d. Therefore, the germination improving effects of GA3 were confirmed; however, the concentration made there was little difference in to these effects due to concentration.
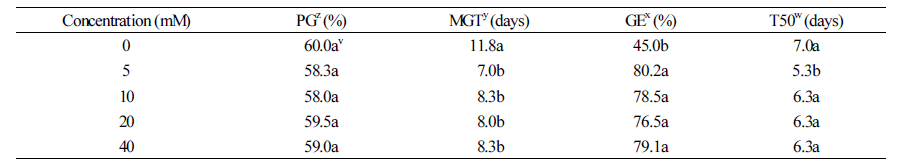
Potassium nitrate did not improve the rate of germination, regardless of its concentration (Table 2). However, MGT was reduced by at least 3 d and GE was improved by at least 30% at all concentrations. Potassium nitrate at a concentration of 5 mM also shortened the T50 by 1.7 d and advanced germination.
Germination Characteristics are Affected by Storage Condition
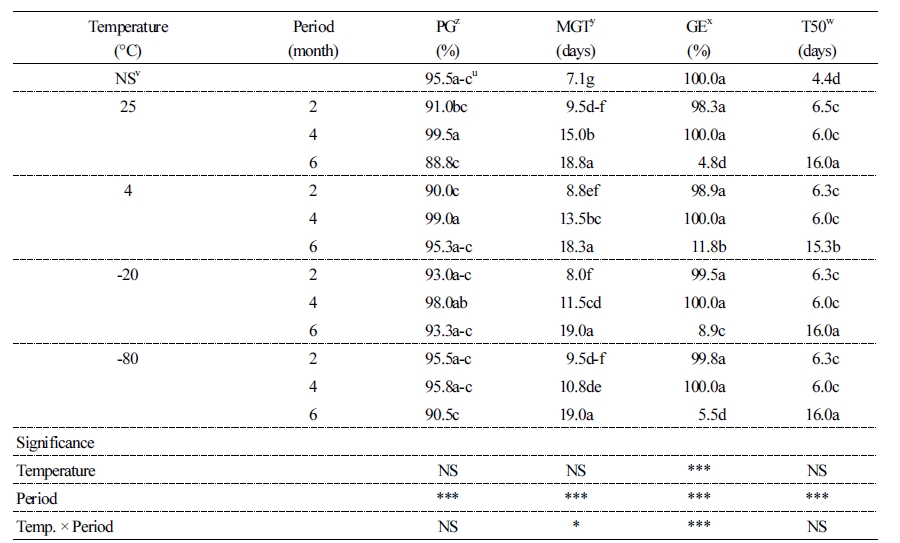
Six months of storage reduced the rate of germination, regardless of temperature. It also reduced MGT and T50 (Table 3). Furthermore, GE was reduced by at least 85% during the six months at all temperatures tested. The storage period significantly correlated with all germination characteristics (p < 0.001). MGT did not change with storage temperature (Table 3). The temperature and duration of storage, either individually or in combination, significantly affected GE (p < 0.001).
Changes of Endogenous GAs and ABA Concentration in M. rossii Seeds
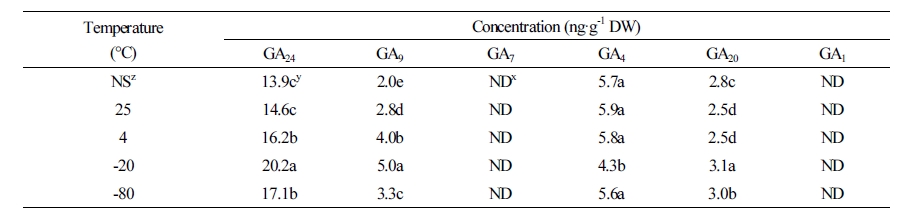
In M. rossii seeds, the content of the two hormones directly affecting germination was analyzed. The results revealed that irrespective of storage temperature, GA4, GA9, GA20, and GA24 were detected; however, GA1 and GA7 were not detected (Table 4). When seeds were stored at -20°C, the GA9, GA20, and GA24 contents were 2.99, 0.38, and 6.26 ng·g-1 DW, respectively, which were was more than those that measured in control samples. However, the GA4 content was 1.34 ng·g-1 DW less than that of the control (Table 4). Furthermore, the GA4 content increased only in seeds that had been stored at -20°C. The GA24, GA9, and GA4 contents were the same or were marginally increased at warm storage temperatures (25°C); however, the GA20 content was 0.31 ng·g-1 DW less than that of control samples (Table 4).
The endogenous ABA content of M. rossii seeds that had been dry-stored at different temperatures was analyzed. At 25°C, endogenous ABA was 7.6 ng·g-1 DW, which was more than that of non-stored seeds (Fig. 4). However, storage at -20°C, significantly decreased the ABA content, and which was 20.0 ng·g-1 DW less than that of the seeds before storage. Storing seeds at 4°C or -80°C did not change the ABA content. Furthermore, the ABA content was found to be sensitive to seed storage temperature.
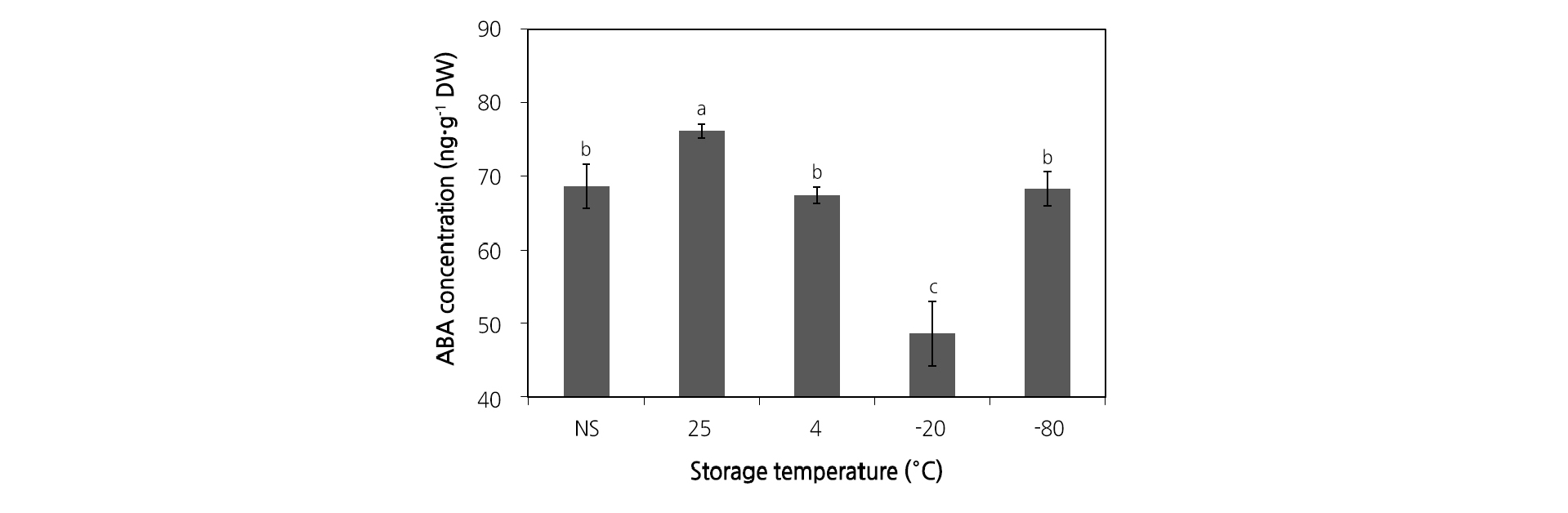
Fig. 4. Change in endogenous abscisic acid concentration in Mukdenia rossii (Oliv.) Koidz. seeds stored at different temperatures for six months. Different letters indicate significant differences based on Duncan’s multiple range tests at p < 0.05. NS mean non-stored seeds after selection. Vertical bars indicate standard error of the mean (n = 200).
Discussion
The direct sowing germination rate and embryo development are important factors that distinguish ND from MD (Baskin and Baskin, 2004; Cho et al., 2016). M. rossii seeds are dispersed with a sufficiently developed embryo. Their direct sowing germination rate was over 80%, and the seeds were permeable water; therefore, the seeds M. rossii were classified as ND. Studies on seed storage methods for maintaining high germination rate are necessary for horticultural uses.
The seeds that we dry-stored for 14 months had the highest germination rate (63.0%) at 20°C in the light, and the germination characteristics of M. rossii were temperature-dependent (Fig. 3). The optimal germination temperature of seeds varies depending on the native environment. In particular, temperatures between 24°C and 30°C are considered optimal for species native to temperate regions (Bewley and Black, 1982; Hartmann et al., 1997). Seed germination of Begonia partita depends on light (Han, 2006), and the seed germination characteristics of Cyclamen persicum differ with temperature (Oh et al., 2003); these observations are similar to the findings of the present study, which suggests that the germination conditions for M. rossii seeds are 20°C and in the light. Furthermore, the germination of M. rossii seeds was only marginally affected by light.
Germination acceleration differs not only with concentration of GA3, duration, and temperature, but also with species (Yoo et al., 1999). Gibberellic acid is a growth regulator hormone that is used as a substitute for low temperature and priming treatments that break seed dormancy and improve germination (Abe et al., 1991). The lack of germination due to low endogenous GA content can be overcome by artificial GA treatment. This has been confirmed in Primula spp. (Chavagnat and Jeudy, 1981; Fukai and Oe, 1990) and other plants, such as barley, wild ornamental plants, and horse radish (Khan and Tolbert, 1969; Kim and Um, 1995; Kim et al., 1996).
Studies have reported that non-stored M. rossii seeds have a germination rate of > 80% in direct sowing experiments (Table 3). However, in the present study, the 14-month storage period decreased the germination rate to 60.5% (Fig. 3). This decrease could be modified with a treatment of 100 mg·L-1 GA3, which increased the rate to its previous level (≥ 90.0%) (Table 1). Thus, the 14-month storage period might have decreased the endogenous GA content and inhibited the germination of M. rossii seeds and therefore, a treatment with 100 mg·L-1 of GA3 can improve the germination potential of M. rossii seeds.
Priming using inorganic substance can reduce the number of days to germination by activating enzymes involved in amino acid and mRNA synthesis, and DNA replication in the seeds (Brafordetal, 1988; Norton, 1988). Potassium nitrate, in particular, supplies essential nutrients for protein biosynthesis such as nitrogen. And it has a positive effect on moisture absorption. In the present study, various concentrations KNO3 did not improve the germination rate; however, it reduced MGT and T50, and improved GE by 35% (Table 2). The treatment with KNO3 was an effective pre-treatment that enhanced quick and consistent propagation of M. rossii seeds.
The M. rossii seeds are ND and maintain their germination rate during a storage period. However, the MGT and T50 were reduced, and GE was significantly decreased, which delayed germination as the storage period increased (Table 3). Seed germination of Cynanchum wilfordii (Hwang et al., 2012) and Astragalus membranaceus (Kim et al., 2001) is improved by storing them at -20°C. However, the seed germination rate of Trichosanthes kirilowii and T. kirilowii var. japonica decreased following storage at -20°C compared to that of seeds stored for six months at 2°C and at room temperature (Lee et al., 2014). This has been attributed to an increase in the seed dormancy under conditions similar to the environment of the species’ native habitat (Lee et al., 2014). The germination rate of Polygonatum macranthum seeds was < 20% even after storing them for 212 d at >20°C; however, its germination rate was 88%-96% after 60 d storage at low temperatures (Tagaki, 2001a). The Polygonatum odoratum seed germination energy is improved when they are stored for 94 d at 5°C (Tagaki, 2001b). Furthermore, P. odoratum var. pluriflorum seeds maintain germination rate similar to untreated seeds during two years of dry storage at 4°C (Chang and Lee, 2007). Therefore, the temperature at which seeds maintain their germination potential can vary by species.
The GA biosynthesis has two paths: early C-13 hydroxylation (ECH: GA53 → GA44 → GA19 → GA20 → GA1) that activates GA1; and non C-13 hydroxylation (NCH: GA12 → GA15 → GA24 → GA9 → GA4) that activates GA4. In many species, GA1 is the most commonly activated form of GA (Huh et al., 2006; Sponsel, 1995).
Endogenous content of GAs in M. rossii seeds was analyzed in the present study. Irrespective of storage temperature, GA4, GA9, GA20, and GA24 were detected in the seeds; however, GA1 was synthesized by ECH pathway was not observed. Furthermore, GA20 is synthesized along the same pathway but was present at a relatively low content (Table 4). GA4 is synthesized by the NCH pathway, and its content was higher than that of other GAs. These findings indicate that the pathway involved in the biosynthesis of endogenous GA in M. rossii seeds is NCH. Lilium davuricum (Eum et al., 2011) and Arabidopsis thaliana (Talon et al., 1990) have activated GA4, while Cyclamen spp. has similar GA1 and GA4 content (Oh et al., 2015). The type of activated GA may differ by plant species. The relative balance of endogenous ABA and GAs in a seed controls their germination and dormancy. When the relative content of GAs is high, the dormancy of the seeds ends and their sensitivity to GA increases (Yamaguchi, 2008; Nambara et al., 2010; Linkies and Leubner-Metzger, 2012). In contrast, dormancy persists when the ABA content is high (Finkelstein et al., 2008). In the present study, the M. rossii seeds that were stored for six months at 25°C had higher ABA content when compared with that in the control (Fig. 4); however, the change in GA content was lowest at 25°C (Table 4). Therefore, storing seeds at high temperatures might increase ABA content, which would not be appropriate for optimal storage conditions. The M. rossii seeds stored at -20°C exhibited a decrease in activated GA4 levels and increased levels of precursors of GAs, such as GA24 and GA9. Furthermore, in contrast to the increase of activated GA4, the ABA content was significantly decreased at -20°C (Fig. 4), which was conducive for seed dormancy and storage.
The results of the present study indicate that when the seeds of M. rossii are stored at a temperature of -20°C, they maintain their germination rate. Furthermore, there is an increase in their endogenous GA content and a decrease in ABA content, which increases the relative content of GA in the seeds. Therefore, -20°C is considered an effective storage temperature for effective propagation of M. rossii.