Introduction
Materials and Methods
Plant Material and Experimental Setup
Weight Loss and Ethylene Production Rate
Firmness
Polygalacturonase (PG) Activity and Pectin Content
Statistical Analysis
Results and Discussion
Changes in Weight Loss
Changes in Ethylene Production Rate
Firmness
PG Activity and Pectin Content
Conclusions
Introduction
Muskmelon (Cucumis melo L. var. reticulatus) is a juicy and tasty fruit of the Cucurbitaceae family, which contains all the edible gourds, such as cucumbers, watermelons, squash, and pumpkins (Milind and Kulwant, 2011). Muskmelon is believed to have originated from East Africa and has been distributed from the country of origin to the Mediterranean region, Asia, Northern Europe, and America (Youn et al., 2009). Muskmelon is cultivated in all tropical and subtropical areas of the world for its nutritional and medicinal value. According to FAOSTAT (2016), worldwide annual production of melons including muskmelon (cantaloupes) was 31.17 million Mt from a planted area of 1.24 million ha, out of which the Republic of Korea took a share of 161,692 Mt from a planted area
of 5,273 ha. In Korea, consumption has been rising with the increase of the cultivation area and with high-quality melon production since the 1990s (Park and Moon, 2004).
In Korea, melon crops are generally grown during the winter-spring season in greenhouses where environmental conditions for the crops are precisely controlled with high energy input (Suh et al., 2012). The high production cost of melons and the consumer’s preference for high-quality fruits demand a relevant postharvest packaging system.
Muskmelon is a climacteric fruit harvested before it is fully ripe; ripening could take place during handling after harvest. The ripening process particularly occurs with climacteric fruit, whereby the ripening is assisted by respiration and is associated with ethylene production (Alexander and Grierson, 2002). Seo et al. (2018) studied the effects of ripening conditions in ‘Earl’s Talent’ cultivar and reported lower weight loss and respiration rate at 10°C than at 25°C throughout the ripening period. They also pointed out that ‘Earl’s Talent’ maintained firmness (1 cm from the peel) up to 18 d at 10°C of ripening, while those stored at 25°C started softening from 12 d of ripening.
Firmness and quality attributes related to firmness are highly desirable as consumers typically associate texture with freshness (Syahidah et al., 2015). Consumers also require persistent vine-end attachment to the fruit as an indication of freshness of the fruit. Farmers harvest vine-attached fruit commonly, and they put it in the packaging box with the vine end (stem end) facing upward in Gangwon province, Republic of Korea. However, Cantwell (1996) indicated that melons ripen from the inside and blossom end first. As the fruit start to ripen, the blossom end, which carries the weight of the fruit, softens rapidly, which in turn increases postharvest losses. Interest in reducing postharvest losses of fresh muskmelon fruit has led to an observation of how the muskmelon undergoes a longer marketability and shelf life at ambient temperature. Therefore, in this study, we investigated the effect of fruit weight and placement position in the packaging box on the fruit firmness and its related quality attributes and suggested the best options for longer marketability and shelf life of muskmelon fruit.
Materials and Methods
Plant Material and Experimental Setup
‘Earl’s Talent’ (NongWoo Bio Co., Ltd., Suwon, Korea) muskmelon cultivar, which is commonly grown by farmers in Gangwon province, Republic of Korea, was seeded on trays. After 4 weeks, seedlings were transplanted to soil in the Kangwon National University farm greenhouse. Drip fertigation and the recommended standard growing practices were implemented throughout the growing period.
Pollination was performed when the female flowers from the 11 - 13th stem node were ready for pollination. The growing tip was removed after the plants reached the 23 - 24th stem node. Fruit were harvested after 53 d of transplanting on July 17, 2017.
After harvesting, the fruit were transported directly to the laboratory and graded based on fruit fresh weight into small (1.0 - 1.4 kg), medium (1.4 - 1.8 kg), and large (>1.8 kg) fruit. After sorting, the vine was removed from the stem end and the stem scar was covered with plaster (Kumsung K & T Co., Ltd., Gwangju, Korea) to reduce water loss and to avoid contamination. Fruit were then placed in the box sideways and downward, as indicated in Fig. 1, and compared with the control (vine attached and stem end facing upward).
The graded and packed fruit were stored at 25°C and 50 ± 5% RH. Data were collected for 15 days. Five fruit were used for each sampling day. Weight loss, flesh firmness (1 cm from the peel), pectin content, and PG activity were measured.
Weight Loss and Ethylene Production Rate
Fruit were weighed at the beginning of ripening for the initial weight measurement. Weight measurements were continued during each day of observation at 3-d intervals. The loss of fruit weight was then calculated by subtracting each day’s observed weight from the initial weight.
Ethylene production rate of the muskmelon fruit was measured from fruit that were sealed in 4.65-L airtight containers and incubated for 3 hrs (Tilahun et al., 2018). A 1-mL gas sample was taken from the headspace of each container using a gas-tight syringe and injected into a gas chromatograph to measure ethylene production. The Shimadzu GC-2010 gas chromatograph (Shimadzu Corporation, Kyoto, Japan) was equipped with a BP 20 wax column (30 m × 0.25 mm × 0.25 µm; SGE Analytical Science, Melrose Park, Australia) and a flame ionization detector. The detector and injector were operated at 127°C, the oven temperature was set at 50°C, and the flow rate of the carrier gas (N2) was 0.67 mL·s-1. The result was expressed as µL C2H4 kg-1·h-1
Firmness
Sample slices were excised from the area of the fruit touching the bottom of the packaging box, and firmness measurements of flesh at 1 cm from the peel were done using a rheometer (Sun Scientific Co. Lt., model compac-100, Tokyo, Japan). The pressure was applied in the vertical direction with a maximum force of 10 kg using a 3-mm-diameter round probe with a flat end. The results were expressed in N.
Polygalacturonase (PG) Activity and Pectin Content
Enzyme Extraction and PG Activity
Distilled water was added to 5 g of frozen melon samples obtained from slices excised from the area of the fruit touching the bottom of the packaging box and homogenized for 1 min. The mixture samples were stirred for 1 h using a mechanical stirrer. After filtration of the samples using Whatman filter paper No. 2, total volume was brought to 12 mL by adding distilled water, and enzyme extraction was performed according to Seo et al. (2018).
Then, 0.2 mL of 0.5% polygalacturonase and 50 mM of sodium acetate buffer (pH 4.5) were added to 0.2 mL of enzyme solution and incubated at 37°C for 30 minutes. After adding 2 mL of 0.1 M borate buffer (pH 9.0) and 0.4 mL of 1% 2-cyanoacetamide, the mixture was vortexed and boiled at 100°C for 10 minutes (Gross, 1982). After cooling for 10 minutes, the absorbance at 276 nm was measured using a spectrophotometer (Thermo Fisher Scientific, Madison, WI, USA). The standard curve was made using polygalacturonic acid (Fisher Scientific Korea Ltd., Incheon, Korea).
Ethanol-Insoluble Solid (EIS)
EIS was extracted as described by Seo et al. (2018); 20 mL of 80% ethanol was added to 5 g of frozen melon flesh sample and homogenized for 3 minutes. The mixture sample was boiled at 100°C in a water bath for 10 minutes then cooled to room temperature. The cooled sample was filtered with Miracloth (Calbiochem, La Jolla, CA, USA), and the residue was washed with 10 mL of 80% ethanol and 10 mL of 100% acetone. The filtrate was then dried in a desiccator at 38°C and stored at room temperature for 10 minutes, following the methods of Kim et al. (2010).
Extraction of Soluble Pectin
Fourteen milliliters of distilled water was added to 0.1 g of EIS, and the mixture was incubated at 30°C for 15 minutes. After filtration, the EIS was extracted twice in succession under the same conditions. The filtrate volume was adjusted to 50 mL by distilled water to obtain water-soluble pectin (WSP).
Fourteen milliliters of 0.4% ammonium oxalate solution was added to the remaining WSP extract residue, and the residue repeatedly extracted three times at 30°C. The filtrate was adjusted to 50 mL by ammonium oxalate to obtain ammonium-soluble pectin (ASP). Then, ASP extract residues were extracted twice with 20 mL of 0.05 N hydrochloric acid at 85°C for 1 hour. The filtrate was adjusted to 40 mL to obtain hydrochloric acid-soluble pectin (HSP). Finally, HSP extracts residue were extracted three times with 14 mL of 0.05 N sodium hydroxide solution at 30°C for 15 minutes. The filtrate was adjusted to 50 mL to obtain sodium hydroxide-soluble pectin (SSP) (Rouse et al., 1962; Zhang et al., 2007).
Pectinase Enzyme Extraction
After soaking 0.2 g of EIS in 95% ethanol, 40 mL of 0.5% versene solution (5 g of ethylenediamine tetraacetic acid tetrasodium salt dissolved in 1 L of distilled water) was added. The pH was adjusted to 11.5 with 1 N NaOH solution and kept at 25°C for 30 minutes. After that, the pH was maintained between 4.0 and 4.5 using acetic acid. Finally, 30 mg of pectinase was added to react for 1 hour and used as an enzyme extract (McCready and McComb, 1952).
Quantification of Pectin
One milliliter each of fraction extract and enzyme extract was added to a glass tube using the methods followed by Blumenkrantz and Asboe-Hansen (1973) and Kintner and Van Buren (1982). The mixture was cooled in an ice water bath for 5 minutes and then 6 mL of a 12.5 mM H2SO4/tetraborate solution (tetraborate dissolved in H2SO4) was added. The sample was then vortexed and boiled at 100°C for 5 minutes and immediately cooled in an ice water bath. After that, 0.1 mL of 0.5% NaOH solution was added for carbohydrate inhibition and 0.1 mL of 0.15% m-hydroxydiphenyl solution was added for color development. After 30 minutes, the absorbance was measured at 520 nm using a UV-spectrophotometer (Thermo Fisher Scientific, Madison, WI, USA). Standard curves were prepared using galacturonic acid monohydrate (Sigma-Aldrich Chemical Co. St. Louis, MO, USA).
Statistical Analysis
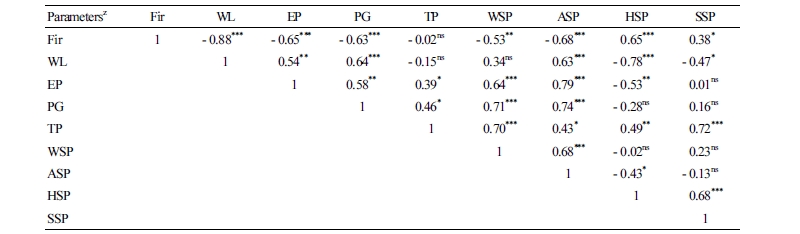
The experiment was conducted in a completely randomized design with five replicates for all parameters considered. The data were analyzed with an analysis of variance (ANOVA) at p < 0.05 using SAS statistical software (SAS/STAT ® 9.1; SAS Institute Inc., Cary, NC, USA). A Pearson correlation test was used to correlate the collected parameters.
Results and Discussion
Changes in Weight Loss
Significant differences (p < 0.05) in weight loss were observed among fruit sizes, but there were no significant differences among storage positions. Large, medium, and small fruit showed a maximum weight reduction of 8.43%, 7.60%, and 6.97%, respectively, on the 15th day of storage. In our study, large and medium fruit lost 5% more weight than after 9 days of storage, irrespective of the storage positions (Fig. 2A, B, C). Youn et al. (2009) indicated that most fruits and vegetables start to lose their marketability with a weight loss of more than 5%. However, the maximum acceptable weight loss even for tomato fruit has been reported to range from 6 to 7% (Nunes, 2008). Getinet et al. (2008) also suggested 10% physiological loss in weight to be considered as an index of termination of the shelf life of commodities. The open netted epidermis of muskmelon fruit favors high water loss and causes softening and shriveling after about 2 weeks (Krarup et al., 2009). In this study, the fruit were marketable even after 2 weeks of shelf life based on weight loss; however, the parts of the fruit that were in contact with the packaging box became soft in the fruit placed upward and sideways, and consumers did not prefer purchasing such fruit. Therefore, considering only weight loss as an indicator for marketability of muskmelon could be misleading unless assisted by other firmness-related quality attributes.
Changes in Ethylene Production Rate
Changes in ethylene production rate of large, medium, and small ‘Earl’s Talent’ muskmelon fruit placed in different positions in the packing box at RT are shown in Fig. 1. Significant differences (p < 0.05) in ethylene production rate were observed among different sized fruit placed upward in the packaging box. The concentration of C2H4 was 7.18 µL·kg-1·h-1 immediately after harvest and increased to 8.03 µL·kg-1·h-1 and 7.99 µL·kg-1·h-1 for large and medium upward-positioned fruit, respectively, on day 15. However, a low production rate of 7.68 µL·kg-1·h-1 ethylene was observed on small fruit on day 15 (Fig. 2C). Similar to our current work, Seo et al. (2018) reported a trend of lower ethylene production of ‘Earl’s Talent’ muskmelon cultivar compared with ‘Honey One’ cultivar. Based on our results, we have seen that muskmelon fruit positioned upward in the packaging box showed a much higher ethylene production rate than fruit placed downward and sideways. As observed in our study, an increased production of ethylene could promote softening and aging of muskmelon fruit starting from the blossom end. In agreement with our current results, Cantwell (1996) also indicated that melons ripen from the inside and blossom end first.
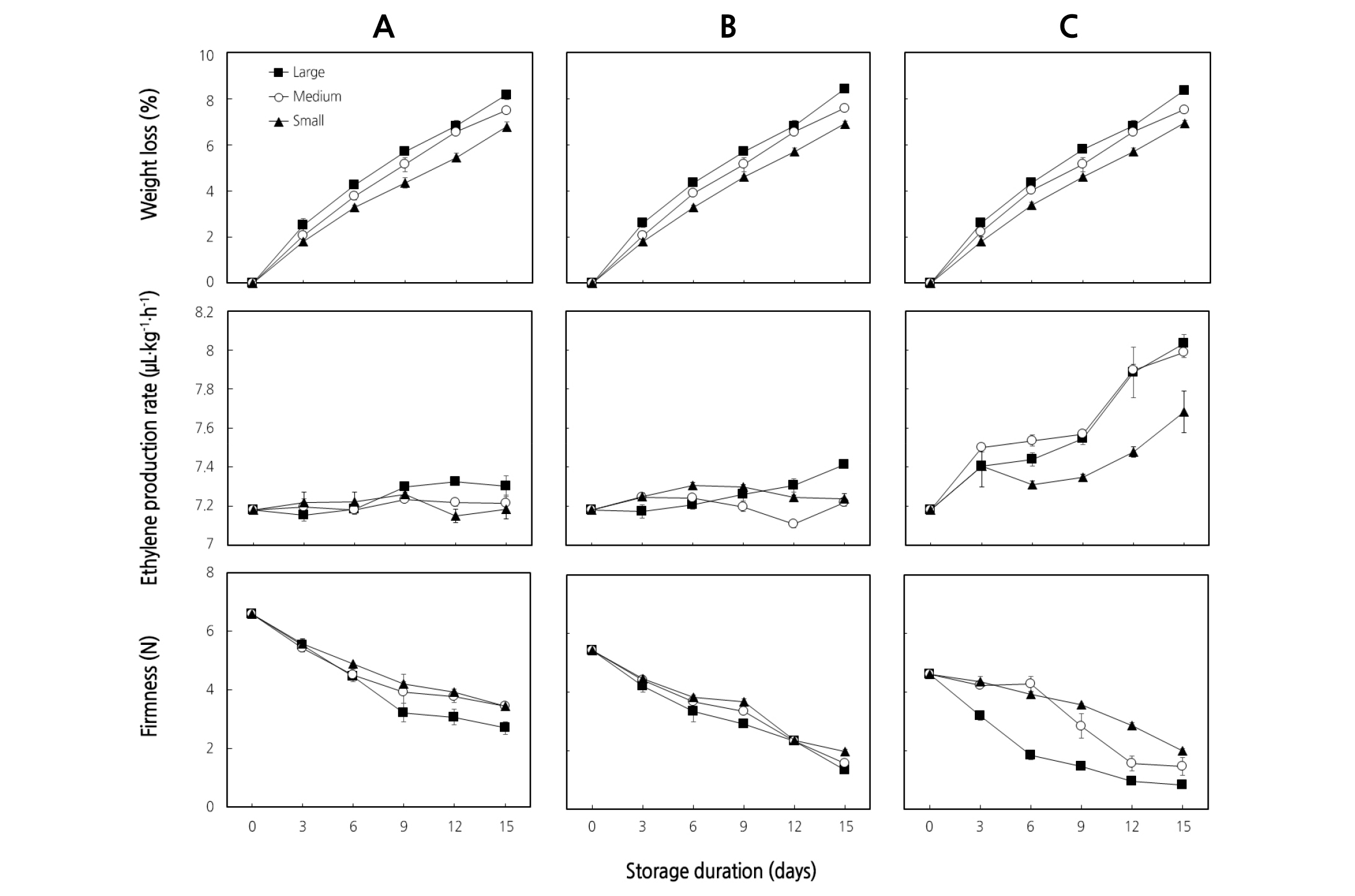
Fig. 2. Weight loss, ethylene production rate, and firmness of ‘Earl’s Talent’ muskmelon fruit placed in the packaging box downward (A), sideways (B), and upward (C) as affected by fruit size for 15 days at 25°C. Each data point is the mean of five sample replicates ± standard error where larger than the symbols.
Firmness
An important ripening and quality indicator for deciding whether to purchase a melon is firmness (Nishiyama et al., 2007). In this study, sample slices were taken from the area of the fruit touching the packaging box, and we measured the firmness of flesh at 1 cm from the peel for muskmelon fruit positioned downward, sideways, and upward. The results revealed that the firmness of large fruit positioned upward reduced significantly (p < 0.05) compared with medium and small fruit (Fig. 2C). The firmness was 4.60 N immediately after harvest at the blossom end of the fruit and decreased to 0.81, 1.44, and 1.99 N for large, medium, and small fruit, respectively, on day 15 for the fruit placed upward. Although the decreasing trend was observed for all sized muskmelon fruit in all tested positions as the storage days proceeded, large fruits reduced firmness faster than medium and small ones (Fig. 2A, B, C). The results indicate that, small fruit could be placed in any position in the packaging box without significant loss of firmness. However, placing the large fruit in the box downward could be beneficial to maintain firmness of muskmelon during shelf life at ambient temperature. Based on firmness, the order of importance of fruit position could be downward > sideways > upward for large, medium, and small fruit, respectively.
PG Activity and Pectin Content
The cell wall of fruits and vegetables mainly consists of cellulose, hemicellulose, pectin, and water (Sila et al., 2009). Structural changes in the cell wall polysaccharides occur chiefly in pectin content during ripening and storage (Paniagua et al., 2014). These changes in pectin content are related to modifications in the texture of fruits (Houben et al., 2011). Pectins present in plant cell walls vary in their solubility during extraction (Mierczyńska et al., 2015). Depending on the solvent used, pectin fraction is identified as a water-soluble pectin (WSP), ammonium-soluble pectin (ASP), hydrochloric acid-soluble pectin (HSP), and sodium hydroxide-soluble pectin (SSP); the amount of galacturonic acid in each fraction usually differs by plant species and physiological development stage (Gawkowska et al., 2018). Polygalacturonases, pectinmethylesterases, and lyases are the three major classes of pectin-degrading enzymes that lead to a loss of firmness and reduction of shelf life (Jayani et al. 2005; Wei et al. 2010). Polygalacturonases hydrolyze the polygalacturonic acid chain by adding water and are the most abundant among all the pectinolytic enzymes (Jayani et al. 2005; Wei et al. 2010). Hadfield et al. (1998) reported that ripening-related cell wall disassembly, which involves in pectin depolymerization and solubilization, in melon might be mediated by the activity of polygalacturonase.
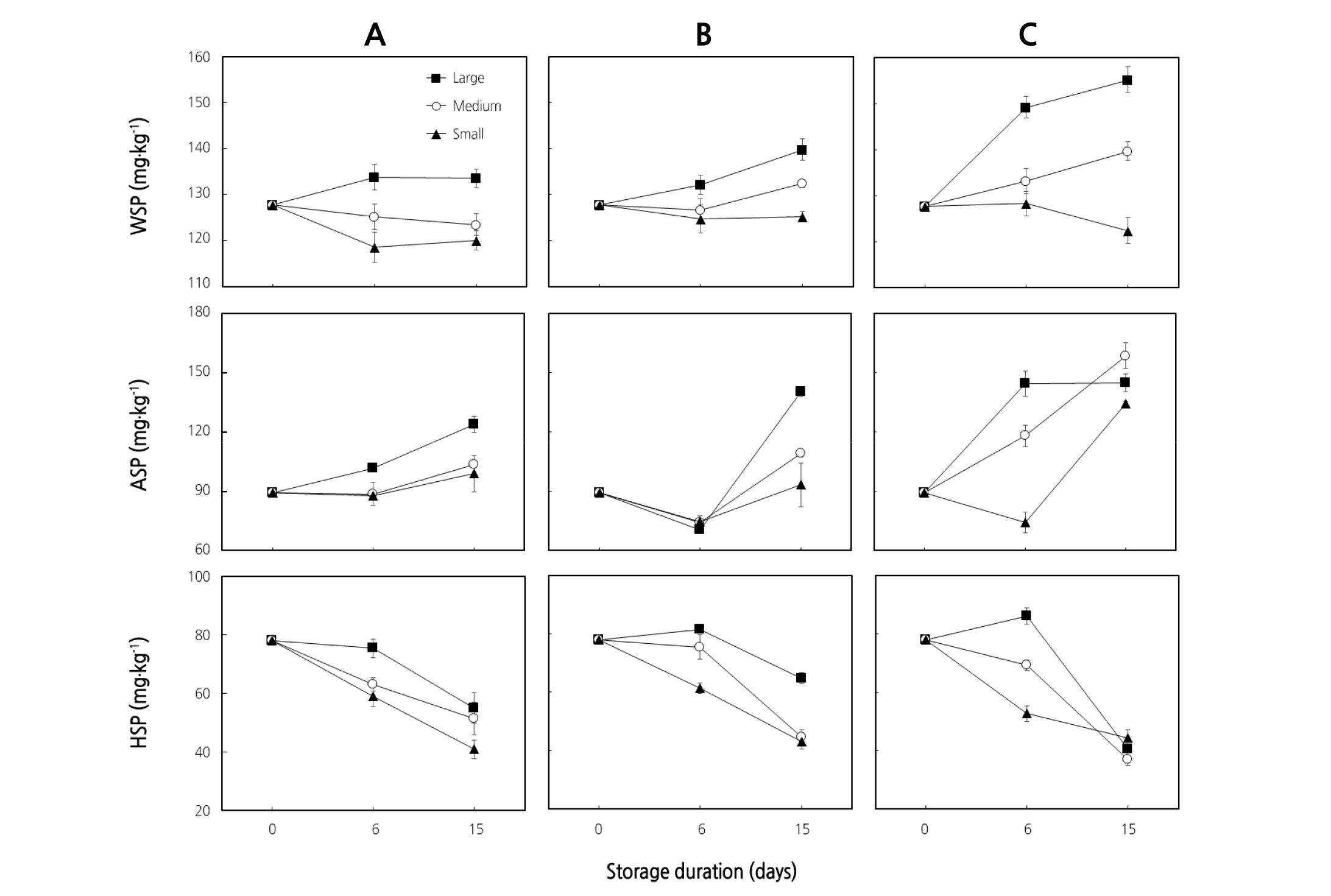
Fig. 3. Water-soluble pectin (WSP), ammonium-soluble pectin (ASP), and hydrochloric acid-soluble pectin (HSP) of ‘Earl’s Talent’ muskmelon fruit placed in the packaging box downward (A), sideways (B), and upward (C) as affected by fruit size for 15 days at 25°C. Each data point is the mean of five sample replicates ± standard error where larger than the symbols.
In this study, the decrease in firmness of muskmelon fruit coincided with an increasing trend of WSP and ASP, while an increasing trend up to the 6th day of storage followed by decreasing trends of HSP and SSP were observed regardless of the fruit size and placement positions in the box (Fig. 3A, B, C). Significant differences (p < 0.05) in WSP and ASP were observed among the different sized fruit, irrespective of fruit placement position in the packaging box. Both WSP and ASP increased significantly (p < 0.05) as the storage days proceeded in large and medium fruit placed sideways and upward (Fig. 3A, B, C). A decreasing trend in HSP and SSP was observed as the storage days proceeded in all sized fruit. Individual values of WSP, ASP, HSP, and SSP for each fruit size were summed up to predict total pectin in the fruit (Fig. 4). The highest total pectin content (490 mg·kg-1) was recorded from larger fruit placed upward in the packaging box on the 6th day of storage and reduced afterwards; however, the lowest total pectin content (318.08 mg·kg-1) was recorded from small fruit placed downward in the packaging box after 15 storage days. The decrease in firmness could be due to an increase of PG activity (Fig. 4A, B, C) as reported by Lee et al. (2007) and Kim et al. (2011), which can cause solubilization of pectins and may contribute to their loss from the cell wall and resulting in softening (Fig. 2A, B, C) when fruit were stored at ambient temperature (Ketsa and Daengkanit, 1999; Shewfelt, 1965).
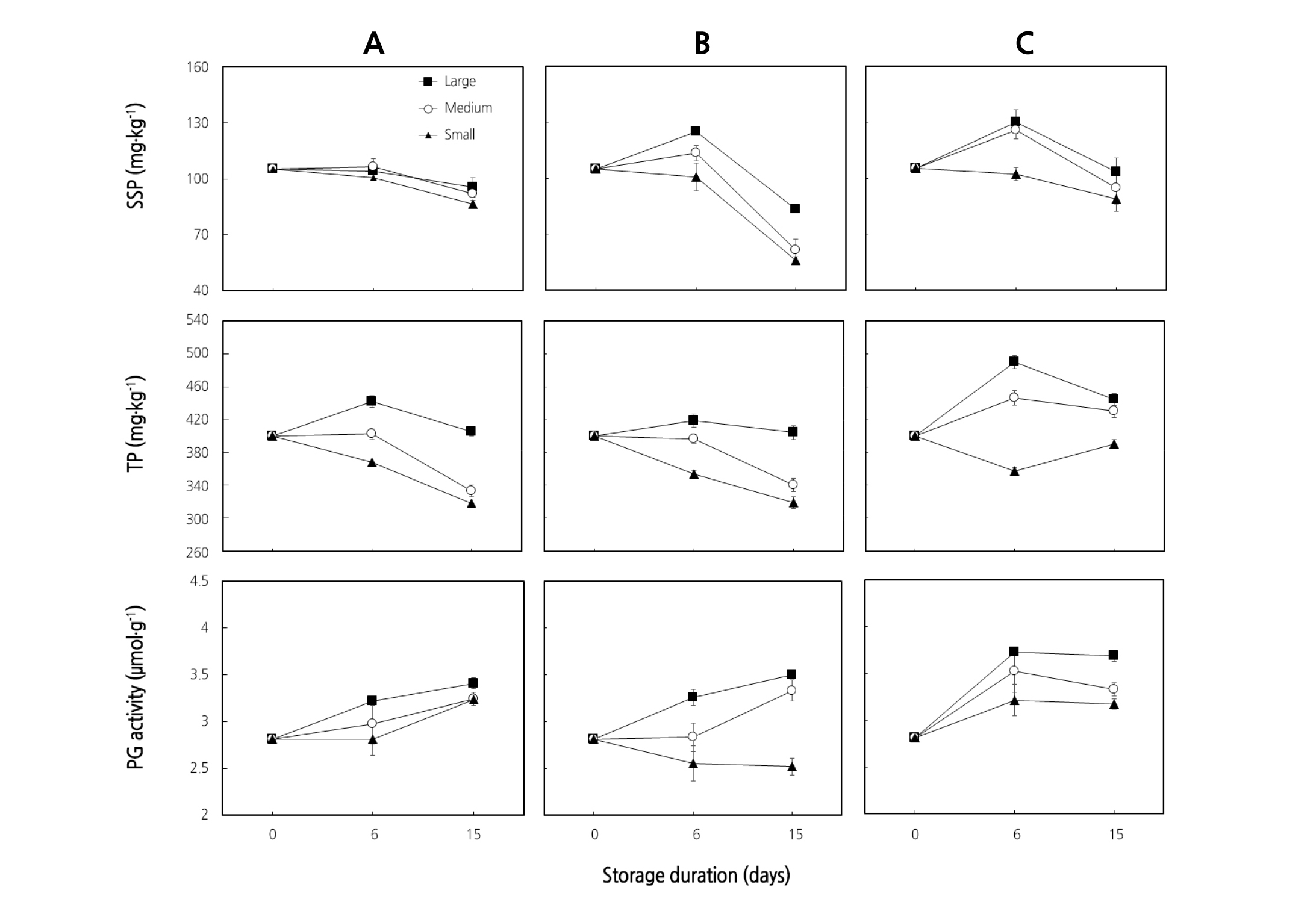
Fig. 4. Sodium hydroxide-soluble pectin (SSP), total pectin (TP), and polygalacturonase (PG) activity of ‘Earl’s Talent’ muskmelon fruit placed in the packaging box downward (A), sideways (B), and upward (C) as affected by fruit size for 15 days at 25°C. Each data point is the mean of five sample replicates ± standard error where larger than the symbols.
Low PG activity was found at the beginning of storage days for all sizes and positions (Fig. 4A, B, C), but as the storage days proceeded, there was a significant increase in polygalacturonase activity. The higher PG activity was found in large fruit regardless of fruit positions in the box and coincided with the loss of firmness. Simultaneous occurrence of an increase in PG activity and softening of fruit was also reported in durian (Ketsa and Daengkanit, 1999), mango (Ketsa et al., 1998), and tomato (Hobson, 1965).
In this study, a highly significant negative correlation (r2 = - 0.88, - 0.65, and - 0.63) was found between firmness and weight loss, ethylene production rate, and PG activity, respectively (Table 1). This could be explained by the softening of the fruit with the cumulative effect of endogenous ripening hormone (ethylene) and PG activity, which in turn lead to cell wall metabolism and weight loss.
Conclusions
This study was conducted to identify the effect of fruit weight and fruit placement position in the packaging box on firmness-related quality attributes of ‘Earl’s Talent’ muskmelon cultivar during shelf life at 25°C. Based on the results, the order of importance of fruit placement position in the packaging box could be downward > sideways > upward for large, medium, and small fruit, respectively. It is recommended to place heavier muskmelon fruit in the box downward for storage and distribution at ambient temperature.