Introduction
Materials and Methods
Antisense Expression Construct of LsRZF1 and Transformation
Plant materials, Growth Conditions and Drought Stress Induction
Determination of Soil and Leaf Water Content
Measurement of Chlorophyll Content
Measurement of Ion Leakage, Malondialdehyde (MDA), and Pro Contents
Reverse Transcription (RT)-PCR and Quantitative Real-Time PCR (qPCR) Assay
Statistical Analysis
Results and Discussion
Amino acid Analysis of the RING C3H2C3-type LsRZF1 Family from C. lanatus
Isolation of Gongdae Ring Zinc Finger 1 (GdRZF1) from C. lanatus cv. Gongdae
Generation and Molecular Characterization of lsrzf1 Antisense Transgenic Watermelon Plants
Reduced Expression of GdRZF1 Induces Phenotypes Insensitive to Drought Stress
Reduced Expression of GdRZF1 Confers High Tolerance to Drought Stress
Physiological Analysis of lsrzf1 Transgenic Lines under Drought Donditions
Effect of Drought on Stress-related Genes
Introduction
Drought is a key environmental stress of crops, and is the main constraint of crop productivity, growth and development. Tolerance to water deficit stress is complex in nature, and comprises several biochemical and physiological processes that are activated during plant growth. Production of proline (Pro) is common in plants exposed to low temperatures, heavy metals, salt stress, pathogen infection, and drought stress (Hare and Cress, 1997; Siripornadulsil et al., 2002). Pro is an important compatible solute, and a chaperone component that stabilizes protein structure. It also acts as a source of carbon storage, and as an antioxidant to regulate free radical levels (Hare et al., 1999). Recently, Min et al. (2014) reported that ubiquitination modulates Pro accumulation during drought stress. Pro production may also be involved in environmental stress signaling to induce the adaptive process response.
Protein ubiquitination is a biochemical, post-translational process employed by animals and plants to regulate various developmental and cellular responses (Dye and Schulman, 2007). In plants, ubiquitination-related proteins are involved in responses to environmental stress, hormones, pathogens, cell differentiation and cell cycle progression (Craig et al., 2009; Santner and Estelle, 2009; Marrocco et al., 2010; Ryu et al., 2010).
Really Interesting New Gene (RING) domain-containing ubiquitin E3 ligase is one of the largest gene families in plants. The RING zinc-finger family can be divided into two types: RING-H2 (C3H2C3) and RING-HC (C3HC4), depending on the presence of either a cysteine (Cys) or histidine (His) residue, respectively, in the fifth position of the motif (Freemont, 2000). Recently reports have found that RING-type proteins are important in responses to abiotic stress. For example, they are involved in light response, senescence, defense signaling, and tolerance mechanisms against high salinity, drought, and cold stress (Craig et al., 2009; Smirnova et al., 2011; Ju et al., 2013; Min et al., 2014).
Previously, we functionally characterized C3H2C3 RING-type zinc finger proteins isolated from Arabidopsis thaliana (AtRZF1) and Lagenaria siceraria (LsRZF1) (Ju et al., 2013; Min et al., 2014). Functional studies illustrated that AtRZF1 and LsRZF1 negatively participate in Pro accumulation-mediated control of early Arabidopsis seedling growth under drought stress. Drought response assays indicated that while AtRZF1-overexpressing and LsRZF1-overexpressing Arabidopsis plants were more sensitive to water deficit conditions, the atrzf1 mutant was less sensitive. This implies that the function of LsRZF1 is similar to AtRZF1 in terms of response to dehydration stress. Our previous data also showed that AtRZF1 and LsRZF1 are functional RING-type ubiquitin E3 ligases (Ju et al., 2013; Min et al., 2014).
Grafting is an important technique for cultivating fruit-bearing plants in Korea. Watermelons are grafted to increase their tolerance to drought and cold, and to improve yield by enhancing soil nutrient uptake (Oda, 1995). For these reasons, watermelons are grafted onto Citrullus lanatus cv. Gongdae, a hardier species in the soil environment, and a species that is widely used as watermelon rootstock (Park et al., 2005). In an effort to improve the tolerance of watermelon to drought stress, we investigated the role of GdRZF1, a homolog of the LsRZF1 gene in the species C. lanatus cv. Gongdae.
In this study, we demonstrate that the GdRZF1 protein functions in response to drought treatment. Disruption of GdRZF1 expression in transgenic lsrzf1 antisense watermelon plants enhances drought tolerance during seedling growth because it induces Pro accumulation, and increases the expression of stress-inducible genes. These results suggest that GdRZF1 is capable of regulating the drought response in Citrullus species.
Materials and Methods
Antisense Expression Construct of LsRZF1 and Transformation
Total RNA was isolated from L. siceraria leaves using Trizol reagent (Invitrogen, Carlsbad, CA, USA). Full-length LsRZF1 cDNA was obtained using the following primers: 5ʹ-GCGGATCCATGTCAGCTGGTCGGAACA-3ʹ (BamHI site underlined) and 5ʹ-GCGAGCTCTCATGGTGAATTAGTGTAT-3ʹ (SacI site underlined), based on the sequence information contained in a cDNA database (http://www.icugi.org). Products amplified by polymerase chain reaction (PCR) were double-digested with SacI and BamHI restriction enzymes, and directionally cloned into the plant expression vector pCAMBIA1301Z. The resulting construct was introduced into Agrobacterium tumefaciens strain LBA4404.
C. lanatus cv. Gongdae embryos were sterilized with 2% sodium hypochlorite solution for 7 min, thoroughly rinsed 10 times with distilled water, and germinated on Murashige and Skoog (MS) medium (Murashige and Skoog, 1962) under a dark cycle and 60% relative humidity, at 28°C for 7 d. After germination, the cotyledons were used for plant transformation via A. tumefaciens LBA4404 containing the CaMV35S promoter-LsRZF1 antisense cassette. Transformed calli were regenerated on MS medium containing 0.1 mg·L-1 indole-3-acetic acid (IAA), 3 mg·L-1 benzylaminopurine (BAP), 250 mg·L-1 carbenicillin, and 10 mg·L-1 hygromycine. Plantlets were acclimated in pots in a greenhouse for 2 weeks, after which they were transferred to soil.
Homozygous lines (T4 generation) from 10 independent transformants were obtained, and three lines (lsrzf14-1, 6-1, and 9-1) were selected for phenotypic analysis. Hygromycine (25 µg·mL-1) resistance of the T2 generation from these three selected lsrzf1 antisense lines were segregated as a single locus.
Plant materials, Growth Conditions and Drought Stress Induction
L. sicerari or C. lanatus cv. Gongdae (wild-type) plants were grown in a growth chamber under intense light (800 µmol·m-2 s-1 PPFD) at 28°C, 60% relative humidity, and 16-h/8-h light/dark conditions. For drought challenge, seedlings were grown in pots and watered every 4 d. After 21 d, plants were divided into two groups: one group was exposed to drought stress by withholding water for 12 d, and the control group was watered normally.
Determination of Soil and Leaf Water Content
The volumetric soil water content of each plant sample was measured with a TDR 300 soil moisture meter (FieldScout, Spectrum Technologies, Inc., USA). To determine the relative water content (RWC) of leaves, the fresh weight (FW) was recorded, then leaf samples were placed in distilled water to saturate for 4 h, then the turgid weight (TW) was recorded. The dry weight (DW) of leaves was determined after oven drying at 65°C for 48 h. RWC was calculated according to Weaherley (1950), using the following formula:
RWC (%) = [(FW – DW) / (TW – DW)] × 100.
Measurement of Chlorophyll Content
Chlorophyll (Chl) a and b contents of watermelon leaves were determined by measuring the absorbance at 663 and 645 nm of extracts prepared using 95% ethanol, according to the method described by Lichtenthaler (1987).
Measurement of Ion Leakage, Malondialdehyde (MDA), and Pro Contents
The loss of cytosol solutes following drought treatment was measured using an ion conductivity meter (Orion 150 A plus, Thermo Electron Corporation, USA) over a 12-h period, and compared with the total conductivity of the solution following tissue destruction of the leaves. Ion leakage content was calculated according to the method described by Shi et al. (2014).
To determine MDA content, samples were ground in 10% (w/v) trichloroacetic acid (TCA) solution, centrifuged for 3 min at 12,000 rpm, and the supernatant was collected. Then, 1 mL of 10% (w/v) TCA containing 0.6% (w/v) thiobarbituric acid (TBA) was added to an aliquot of the supernatant. The mixture was then maintained in a boiling water bath for 15 min, and cooled on ice. The absorbance of the colored supernatant was measured at 532 nm (A532), and was corrected for non-specific absorbance at 450 nm (A450) and 600 nm (A600). The MDA content was calculated using the equation (Zhang et al., 2012):
MDA = 6.45(A532 – A600) – 0.56 × A450
Pro content was calculated according to the method described by Bates et al. (1973). Pro was extracted from each sample by homogenizing in 3% sulfosalicylic acid. The extract was reacted with ninhydrin mixture (6.8% phosphoric acid, 80% glacial acetic acid, and 70.17 mM ninhydrin) at 100°C for 60 min. The reaction mixture was extracted with toluene. Absorbance of the toluene layer was estimated at 520 nm in a spectrophotometer (JASCO, Tokyo, Japan). Pro content was calculated on a FW basis, as follows:
[(ng Pro/mL × mL extraction buffer)/115.5 ng nmol]/g material = nmol proline/g FW sample.
Reverse Transcription (RT)-PCR and Quantitative Real-Time PCR (qPCR) Assay
Total RNA was extracted from seedling plants using the Plant RNeasy extraction kit (Qiagen, Valencia, CA). The RNA concentration was measured with a spectrophotometer.
qPCR was carried out with Rotor-Gene 6000 apparatus (Corbett Research, Mortlake, NSW, Australia), and results were analyzed using RG6000 1.7 software (Corbett Research). qPCR was carried out using the SensiMix One-Step kit (Quantance, London, UK). C. lanatus cv. Gongdae Actin 7 (GdACT7) was used as the internal control. Quantitative data analysis was carried out using the delta-delta-CT method (Livak and Schmittgen, 2001). Each sample was carried out in three independent experiments. The reaction primers utilized are listed in Suppl. Table 1s.
RT-PCR measured the levels of GdRZF1 and GdACT7 expression in wild-type (WT) and transgenic plants, utilizing 300 ng of RNA; the primers utilized are listed in Suppl. Table 1s. PCR proceeded for 27 cycles, as follows: 94°C for 15 s; 57°C for 20 s; and 72°C for 1 min. PCR product was then loaded onto a 1.2% (w/v) agarose gel to visualize the amplified DNA.
Statistical Analysis
Data were statistically analyzed using Microsoft Excel and (Ver. 23, IBM, New York, USA). Analysis of variance was used to compare statistical differences, based on Student’s t-test at a significant level of 0.01 < p < 0.05 or p < 0.01.
Results and Discussion
Amino acid Analysis of the RING C3H2C3-type LsRZF1 Family from C. lanatus
In previous work, identification of the Arabidopsis thaliana RING Zinc Finger 1 (AtRZF1) gene allowed us to isolate the ortholog LsRZF1 from the gourd species L. siceraria (Min et al., 2014). In the watermelon genome, LsRZF1 belongs to the RING C3H2C3-type zinc finger family. Analysis of its molecular functions reveal that it is a negative E3 ubiquitin ligase involved in drought stress responses (Min et al., 2014). LsRZF1 contains a RING C3H2C3-type motif in its central region (177–217) (Fig. 1A). As shown in Fig. 1B, the amino acid sequence of the RING C3H2C3-type domain is considerably homologous with GdRZF1, and with 14 unknown members of the C3H2C3-type zinc finger family in C. lanatus. The protein harbored a predicted RING C3H2C3-type zinc finger domain, as shown by the Cucurbit Genomics Database (http://www.icugi.org) and the MyHits software program (http://myhits.isb-sib.ch). Phylogenetic analysis using MEGA7 software (http://www.megasoftware.net/) revealed that RING C3H2C3-type zinc finger members are divided into four groups (Fig. 1C), and LsRZF1 belongs to group I. The group I C3H2C3-type zinc finger proteins are structurally similar, with approximately 24.8–94.7% amino acid sequence identity.
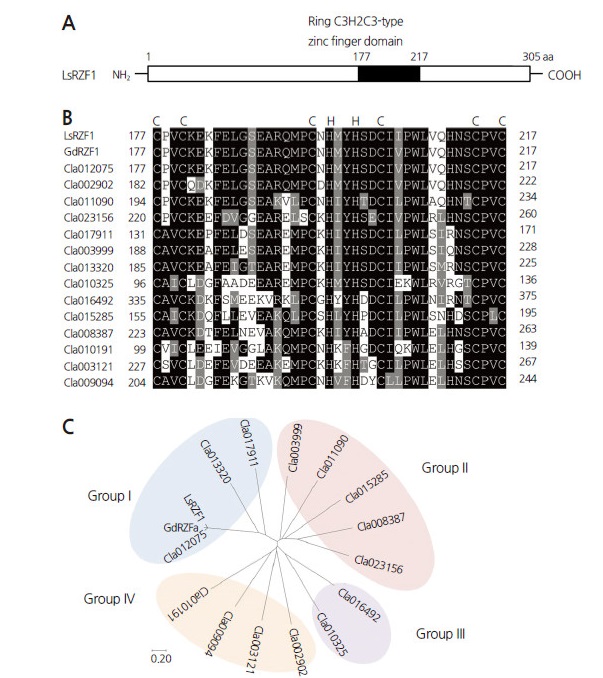
Fig. 1.
Sequence analysis of LsRZF1. (A) Schematic structure of the LsRZF1 protein. Black box represents the RING C3H2C3-type zinc finger domain (177–217). (B) Multiple sequence alignment of LsRZF1 and other RING C3H2C3-type zinc-finger homologs from Citrullus lanatus. Shown here are the sequences of LsRZF1, GdRZF1, Cla012075, Cla002902, Cla011090, Cla023156, Cla017911, Cla003999, Cla013320, Cla010325, Cla016492, Cla015285, Cla008387, Cla010191, Cla003121, and Cla009094 (http://www.icugi.org). Gray shading indicates similar amino acids; black shading indicates identical amino acids. (C) Phylogenetic relationship between LsRZF1 homologs from watermelon. LsRZF1 homologs are divided into four groups: LsRZF1, along with GdRZF1, Cla012075, Cla013320, and Cla017911 homologs, belong to group I. Bootstrap values (>70) are presented near the corresponding branch.
Isolation of Gongdae Ring Zinc Finger 1 (GdRZF1) from C. lanatus cv. Gongdae
One of the C. lanatus cv. Gongdae RING C3H2C3-type zinc finger homologs shared considerable sequence identity with LsRZF1 from a gourd (L. siceraria), and was named GdRZF1 (Gongdae Ring Zinc Finger 1). Overall homology values of 93% or 94% identity were observed between LsRZF1 and GdRZF1 cDNAs (Suppl. Fig. 1sA) or proteins (Suppl. Fig. 1sB), respectively. The coding region of GdRZF1 is 768 bp in length, encoding 255 amino acids (28.1 kDa) (Suppl. Fig. s1). GdRZF1 contains a single C3H2C3-type motif in its central region (177–217) (Suppl. Fig. 1sB). These data suggest that GdRZF1 belongs to the RING C3H2C3-type zinc finger family.
Generation and Molecular Characterization of lsrzf1 Antisense Transgenic Watermelon Plants
To investigate the functional consequences of reduced GdRZF1 expression in Citrullus species, we generated transgenic watermelon plants in which the LsRZF1 gene was constitutively expressed in the antisense-direction, under the control of the CaMV35S promoter (Fig. 2A). GdRZF1 expression was detected by qPCR in three independent lsrzf1 antisense transgenic lines (lsrzf1 4-1, lsrzf1 6-1, and lsrzf1 9-1). GdRZF1 expression was knocked-down in the lsrzf1 antisense watermelon transgenic lines (Fig. 2B). To further characterize the T-DNA-associated DNA sequence, specific primers (35SF1 and LsR1; HF1 and HR1) (Supplementary Suppl. Table 1s) were used to confirm the insertion within WT and lsrzf1 antisense transgenic plant genomes (Figs. 2A and C). Using 35SF1-LsR1 and HF1-HR1 primer pairs, the lsrzf1 antisense transgenic lines (lsrzf1 4-1, lsrzf1 6-1, and lsrzf1 9-1), but not the WT plant, was detected by PCR (Fig. 2C). Genomic PCR-based genotyping analysis revealed that lsrzf1 antisense construction was linked to reduced expression of the GdRZF1 gene.
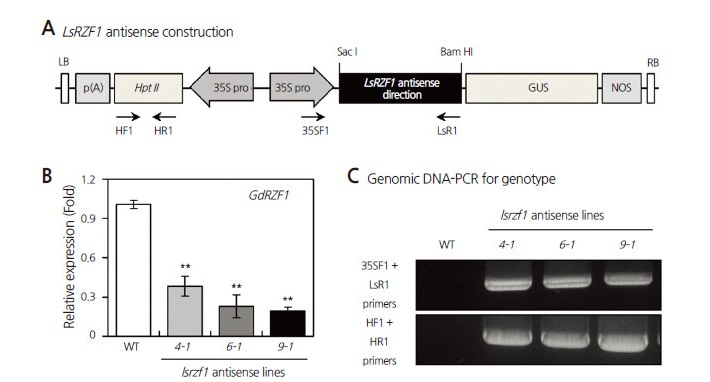
Fig. 2.
Molecular analyses of lsrzf1 antisense transgenic watermelon plants. (A) Schematic representation of the plant expression vector pCAMBIA1301Z used for LsRZF1 antisense construction under the control of the CaMV35S promoter (35S pro). LB, left border; p(A), poly (A); Hpt II, hygromycine B phosphotransferase; GUS, beta-glucuronidase; NOS, nopaline synthase terminator; RB, right border. Arrows mark the positions of specific primers for polymerase chain reaction (PCR) amplification. (B) Expression levels of GdRZF1 in wild-type (WT) and three independent lsrzf1 antisense transgenic lines (lsrzf1 4-1, lsrzf1 6-1, and lsrzf1 9-1) were determined by quantitative PCR (qPCR) analysis using total RNA extracted from 12-d-old watermelon seedlings. GdActin7 (GdACT7) was used as an internal control in qPCR. Values were normalized to the endogenous content of GdACT7 transcript. Error bars indicate standard deviations of three independent biological samples. Asterisks (**) denote a statistically significant difference compared with the WT (p < 0.01). (C) Genotype analyses of the WT and lsrzf1 antisense transgenic plant genomes. Genomic DNA-PCR using the primers 35SF1-LsR1 and HF1-HR1 detected a DNA band specific for lsrzf1 antisense transgenic lines.
Thereafter, to assess whether the reduced expression of GdRZF1 gene is involved in fruit morphology under normal conditions, we examined several fruit parameters of WT and three lsrzf1 antisense transgenic watermelon lines (lsrzf1 4-1, lsrzf1 6-1, and lsrzf1 9-1). As shown in Supplementary (Suppl. Table 2s, fruit morphology of the WT and lsrzf1 antisense transgenic lines was not affected under normal conditions.
Reduced Expression of GdRZF1 Induces Phenotypes Insensitive to Drought Stress
We have previously demonstrated that LsRZF1 is required to modulate the tolerance of Arabidopsis to drought stress (Min et al., 2014). To investigate whether lsrzf1 antisense transgenic watermelon plants were resistant to drought stress, the drought-induced phenotype was evaluated by assessing the extent of leaf yellowing and wilting. After 7 or 12 d under drought stress, yellowing leaves were highly attenuated in lsrzf1 antisense lines. However, compared with those detected in the antisense lines, WT leaves displayed wilting and yellowing symptoms (Suppl. Fig. 2sA). These phenotypes showed that the lsrzf1 antisense lines were more likely than WT plants to be insensitive to drought stress.
To assess the effects of drought treatments on soil moisture, we also measured the volumetric soil water content (VSWC) of watermelon samples. As shown in Suppl. Fig. 2sB, 4 d after drought treatment, VSWCs was two-fold lower in all samples than in untreated pots, while VSWCs was exhausted 12 d after drought treatment. These results suggest that expression of GdRZF1 is associated with leaf wilting and greening under drought conditions.
Reduced Expression of GdRZF1 Confers High Tolerance to Drought Stress
To investigate whether lsrzf1 antisense lines affect the ability of plants to respond to drought stress, we analyzed RWC in the third, fourth and fifth leaves. As shown in Fig. 3A, RWC was higher in lsrzf1 transgenic lines (lsrzf1 4-1, lsrzf1 6-1, and lsrzf1 9-1) than in WT plants 12 d after drought treatment.
We also measured FW as a measure of drought-induced damage. There was little difference in FW between WT and transgenic plants under normal conditions. However, 12 d after drought treatment, the relative suppression in FW of the WT plant was significantly more than in lsrzf1 antisense lines (Fig. 3B).
We also measured the Chl a and b contents in the fourth or fifth leaf of 33-d-old plants in each category (Fig. 3C). After 12 d of drought treatment, Chl content in lsrzf1 transgenic lines was significantly higher than in the WT leaves. These results suggest that lsrzf1 transgenic lines are more tolerant to water deficit stress than WT plants because of reduced GdRZF1expression.
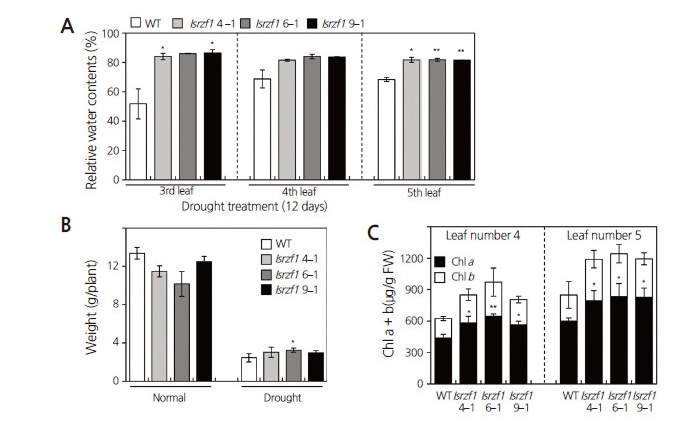
Fig. 3.
Drought stress analysis of wild-type (WT) and lsrzf1 transgenic watermelon lines. (A) Relative water content (RWC) in the third, fourth or fifth leaves was quantified 12 d after samples were subjected to drought treatment. (B) Effect of drought treatment on plant fresh weight (FW). Plants were grown in soil-filled pots for 33 d, then measured for FW. (C) Chlorophyll a and b contents in the fourth or fifth leaves were measured 12 d after samples were subjected to drought treatment. Data shown are triplicates, n = 3 each. Error bars in each graph indicate standard deviations. Asterisks in each graph represent statistically significant differences compared with WT plants: *, 0.05 < p < 0.01, and **, p < 0.01.
Physiological Analysis of lsrzf1 Transgenic Lines under Drought Donditions
After 12 d of drought stress treatment, we examined ion leakage, MDA, and Pro contents as physiological parameters of drought stress (Zhang and Kirkham, 1994; Ju et al., 2013). Ion leakage and MDA levels of WT leaves were higher than those of the lsrzf1 antisense lines under drought stress conditions (Figs. 4A and 4B). This suggests that the degree of plasma membrane damage was greater in WT plants than in lsrzf1 transgenic lines under drought stress conditions. LsRZF1 was negatively involved in Pro accumulation under drought stress in Arabidopsis (Min et al., 2014); therefore, we measured Pro levels in the WT and transgenic watermelon plants. Under normal conditions, Pro levels were equally low in all plants. Under drought conditions, the lsrzf1 antisense lines accumulated more Pro than the WT plants (Fig. 4C). Thus, these results indicate that changes in drought-related physiological parameters occurred later in the lsrzf1 antisense lines than in the WT plants during drought stress conditions.
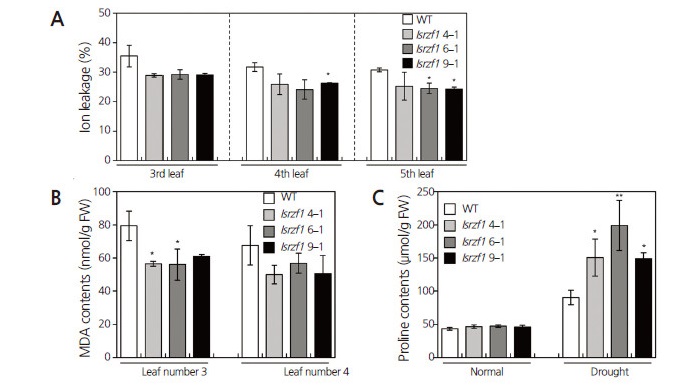
Fig. 4.
Physiological analyses of lsrzf1 transgenic watermelon lines under drought conditions. (A) Ion leakage in the third, fourth or fifth leaves of wild-type (WT) and lsrzf1 transgenic (lsrzf1 4-1, lsrzf1 6-1, and lsrzf1 9-1) plants after 12 d of drought stress treatment. (B) Malondialdehyde (MDA) contents in the third or fourth leaves of WT and lsrzf1 transgenic plants after 12 d of drought stress treatment. (C) Proline contents in WT and lsrzf1 transgenic plants. The 21-day-old plants were grown for 12 days with (drought) or without (normal) watering. Data shown are triplicates, n = 3 each. Error bars indicate standard deviations. Asterisks represent a statistically significant difference compared with WT plants: *, 0.05 < p < 0.01; **, p < 0.01.
Effect of Drought on Stress-related Genes
Expression of P5CS (for Delta 1-Pyrroline-5-Carboxylate Synthase), P5CR (for Delta 1-Pyrroline-5-Carboxylate Reductase), and RAB18 (for Responsive to ABA 18) were induced by various abiotic stresses (Ju et al., 2013; Min et al., 2014). Transcripts of the P5CS, P5CR and RAB18 genes are elevated by ABA, drought, salt, and cold stress. Suppl. Fig. 3s shows that the transcript expression levels of stress-inducible genes, including GdP5CS, GdP5CR and GdRAB18, were higher in lsrzf1 antisense transgenic lines than in WT under drought stress conditions. These data support the concept that GdRZF1 modulates the expression of these stress-related genes under drought stress conditions.


