Introduction
Materials and Methods
Plant materials
Seed characteristics
Seed water absorption
Initial germination test
Phenology experiment
Move-along experiment
Plant growth regulator treatment
Effect of stratification on dormancy breaking
Statistical analysis
Results
Seed basic information
Initial germination test
Embryo growth, germination, and shoot emergence under natural environmental conditions in the phenology experiment
Embryo growth and germination under simulated natural environmental conditions in the move-along experiment
Effect of plant growth regulator treatment on dormancy breaking
Effect of stratification at various temperatures and periods on breaking dormancy
Discussion
Introduction
The genus Corydalis in the Fumariaceae family is the largest in the order Papaverales, comprising over 470 species. Several Corydalis species, such as C. turtschaninovii, C. ternate, C. fumariifolia, and C. ambigua, are recorded in the pharmacopeias of Korea and Japan and have been used in some regions of China (Jiang et al. 2018). The main pharmacologically active compounds in Corydalis are alkaloids, with 381 types reported thus far. They are known for their clinical therapeutic effects, including inhibition of gastric juice secretion, acting as analgesics, and serving as antibacterial agents (Deng et al. 2021). C. platycarpa produces racemose flowers and is widely distributed in the north-temperate regions, including Korea, China, Myanmar, and Japan (Raman et al. 2022). Many Corydalis species have pharmacological benefits; however, information on their germination characteristics and dormancy remains limited, thus hindering their utilization.
Due to various environmental issues and global warming, ecosystems are rapidly changing, with the most significant problem being reduced plant species diversity (Walther et al. 2002). Recently, factors such as climate crisis, random harvesting, drought, and overgrazing have decreased the density and distribution of many wild plants (Jahantab et al. 2015). The availability of genetic and pharmacological resources has decreased due to biodiversity loss (Zarei-Gavkosh et al. 2022). Therefore, to restore ecosystems and increase resource availability, studying the life cycles of plants, environmental conditions, and seed dormancy is necessary.
Seed dormancy is an evolutionary adaptation that prevents germination under unfavorable conditions for plant growth. The type of dormancy varies depending on the environment and species (Leubner-Metzger 2006; Linkies et al. 2010; Gremer et al. 2020). Seed dormancy is classified into five categories: physical dormancy (PY), physiological dormancy (PD), morphological dormancy (MD), morphophysiological dormancy (MPD), and combinational dormancy (PY+PD). Each dormancy type is further divided into subclasses (Baskin and Baskin 2014). Among the five major types of dormancy, MD requires embryo growth owing to an underdeveloped embryo. Many temperate species with MD exhibit MPD, where PD interferes with embryo growth and germination (Dalziell et al. 2019). Therefore, MPD requires more than 4 weeks for embryo growth along with pretreatment (e.g., warm, cold, or warm + cold stratification) for breaking dormancy and germination (Chien et al. 2011a).
MPD is classified into nine levels based on the response to gibberellic acid (GA), the requirements for cold or warm stratification for dormancy breaking, the temperature needed for embryo growth, and the timing of shoot and root emergence (Zhang et al. 2021). Simple MPD is suitable for embryo growth in warm stratification, whereas complex MPD occurs only in cold stratification (Mohammadianfar et al. 2024). Complex MPD is divided into three levels, non-deep, intermediate, and deep, based on the seed’s response to cold or warm stratification and GA3 levels (Zardari et al. 2019). Non-deep and intermediate levels can replace low or warm temperatures with exogenous GA treatment (Doodman et al. 2023). However, the deep level does not respond to exogenous GA treatment. Therefore, to determine MPD levels, assessing the effects of various temperatures and GAs on embryo growth and germination is necessary.
When the natural environment turns favorable for breaking seed dormancy, seeds undergo physiological changes, with the primary hormones involved in dormancy and germination, GAs, and abscisic acid (ABA), coming into play (Grappin et al. 2000; Geneve 2003). A long period is required to break seed dormancy, and research should focus on shortening the dormancy-breaking period for species conservation and commercial utilization. GAs break dormancy and promote seed germination by stimulating germination metabolism, whereas ABA inhibits germination and induces dormancy. The balance between ABA and GA is crucial for regulating dormancy (Gómez-Cadenas 2001). Fluridone (Flu) prevents ABA biosynthesis, thereby inhibiting seed dormancy, while GA3 and GA4+7 promote germination (Aihua et al. 2018; Wei et al. 2010; Chen et al. 2021). Stratification effectively alleviates dormancy in temperate species and regulates seed dormancy through the interaction of various hormones, including GA and ABA (Leon et al. 2007). Utilizing various plant growth regulators and stratification that act on seed dormancy regulation can improve methods for breaking dormancy.
Therefore, the objectives of this study were two-fold: first, to classify the MPD levels of C. platycarpa seeds through responses to temperature, phenology, move-along test, and GA3; second, to establish methods for shortening the dormancy-breaking period through various treatments (plant growth regulators and stratification) to enhance seed utilization.
Materials and Methods
Plant materials
C. platycarpa seeds collected from habitats in Ulleung-gun, Gyeongsangbuk-do, South Korea, in June 2022.These seeds were provided from the National Institute of Biological Resources and only those sufficiently matured at the time of collection were harvested. The seeds collected immediately after harvest were dried in the shade, subsequently selected, and sealed in a plastic bag with silica gel, then stored at 4°C until used in the experiment.
Seed characteristics
Seed basic characteristics, including thousand-seed weight, length and width, embryo-to-seed ratio (E:S ratio), viability, and fidelity were recorded. The 1,000-seed weight was measured in 4 replicates using a precision balance (AS200, Radwag, Radom, Poland). The length and width of 10 seeds were measured using a stereo microscope (SZ61, OLYMPUS, Tokyo, Japan) equipped with a CMOS camera (eXcope F-630, Dixi Sci., Daejeon, Korea). The E:S ratio was determined by cutting the seeds transversely using a double-edged razor blade and measuring the embryo and seed lengths under the stereo microscope to calculate the average ratio. Seed viability was assessed by soaking 50 seeds in distilled water for 24 h at 20°C in the dark, followed by immersion in a 1.0% tetrazolium solution (Triphenyl tetrazolium chloride, St. Louis, MO, USA) for 24 h at 30°C in the dark. The seeds were cut transversely, and the staining pattern was evaluated to determine viability, expressed as a percentage. Seed fidelity was assessed by taking X-ray images of 50 seeds in 3 replicates using a small microfocus X-ray TV inspection device (EMT-F70, Softex, Tokyo, Japan) and calculating the percentage of seeds with fidelity.
Seed water absorption
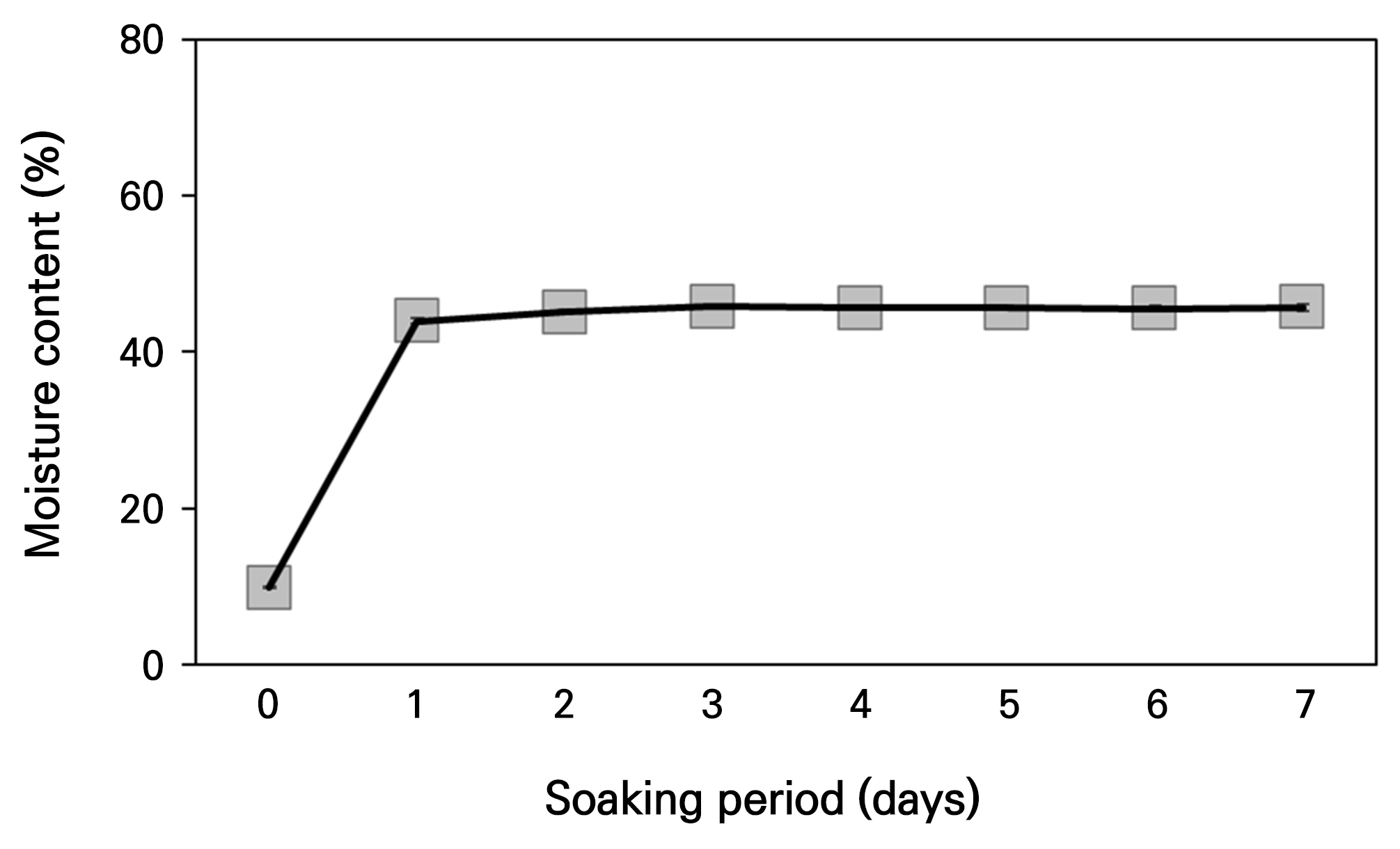
A water absorption test was conducted to measure the initial and maximum moisture content of the seeds and determine PY. The weight of 100 seeds was measured in four replicates, and the seeds were soaked in distilled water and kept in the dark at 4°C. Every 24 h, the surface moisture of the seeds was removed with a paper towel, and their weight was measured. After each measurement, the seeds were re-immersed in distilled water and this process was repeated for 7 days. At the end of the measurement period, the seeds were dried in an oven at 70°C for 48 h to determine their dry weight. The seeds’ initial and maximum moisture content were then calculated based on the dry weight.
Initial germination test
An initial germination test was conducted to observe seed germination characteristics and determine the presence of PD. Seed disinfection was performed by soaking the seeds in a 1.4% sodium hypochlorite solution for 10 min and rinsing with distilled water for more than 3 min. Subsequently, 25 seeds were placed on Petri dishes (10010, SPL Life Sciences, Pocheon, Korea) in 4 replicates lined with 2 filter papers and moistened with 4 mL of distilled water. The germination test was conducted under constant temperature conditions of 4, 15, 20, 25, and 30°C, and alternating temperature conditions of 15/6°C (14/10 h), 20/10°C (14/10 h), and 25/15°C (16/8 h). For all temperature treatments, the light environment was set at 30 µmol·m-2·s-1 PPFD using white LEDs under both light and dark conditions. Germination was observed weekly for 40 weeks, and at the end of the experiment, the viability of the ungerminated seeds was tested using a 1.0% tetrazolium solution.
Phenology experiment
A phenology experiment was conducted in the garden in front of the greenhouse at the Chungbuk National University experimental farm (36°37'49.29"N, 127°27'9.08"E) starting from June 30, 2023, to monitor the timing of embryo growth, germination, and shoot emergence in a natural environment. During the experiment, the temperature of the experimental area was measured hourly using a data logger (EL-USB-2-LCD, Lascar electronics, Whiteparish, UK), and the mean, minimum, and maximum temperatures were recorded.
Embryo growth and germination were studied by placing 120 mesh bags, each filled with 50 seeds and perlite (New Pershine No. 2, Green Fire Chemicals, Seoul, Korea), at a depth of exactly 1 cm below the soil surface in the garden. Every 2 weeks, mesh bags were randomly excavated from the soil to assess the germination of the 50 seeds, expressed as a percentage. Ten seeds were cut to measure embryo length (mm), and the average length was calculated. The embryo of C. platycarpa seeds grows in a curved shape, so its length (mm) was measured as a curve using a tool within the CMOS camera software designed to measure curves, mounted on a stereomicroscope for accurate measurement. The curve of the embryo was referenced from the cross-section of the fully developed seed to establish the embryo length measurement.
To monitor shoot emergence, 50 seeds were uniformly sown at a depth of 1 cm below the soil surface in plastic pots (15 cm) filled with soil from the garden, in three replicates. The three plastic pots were buried in the garden in front of the greenhouse at the Chungbuk National University experimental farm. Shoot emergence was observed every 2 weeks and calculated as a percentage.
Move-along experiment
A move-along experiment was conducted to simulate seasonal temperature changes in the laboratory and observe embryo growth and germination, mimicking natural environmental cycles. The experiment was sequentially conducted as follows: Move A sequence: 4 (12 weeks) → 15/6 (4 weeks) → 20/10 (4 weeks) → 25/15 (12 weeks) → 20/10 (4 weeks) → 15/6°C (4 weeks); Move B sequence: 25/15 (12 weeks) → 20/10 (4 weeks) → 15/6 (4 weeks) → 4 (12 weeks) → 15/6 (4 weeks) → 20/10°C (4 weeks). For both Move A and B sequences, 50 seeds were used in 12 replicates. Every 2 weeks, the embryo length (mm) of 10 seeds was measured and categorized into < 2.8 mm and 2.8–3.3 mm; the percentages were recorded. For germination, 100 seeds in 6 replicates were sown as controls at 4, 15/6, 20/10, and 25/15°C and in both Move A and B sequences. Germination was observed every 2 weeks and calculated as a percentage.
Plant growth regulator treatment
An experiment was conducted using the plant growth regulators GAs and Flu to break dormancy. The seeds were treated with solutions of 0 (distilled water), 100, 200, and 500 mg·L-1 of GA3 (Cas No. 77-06-5; Sigma-Aldrich, St. Louis, MO, USA), GA4+7 (2:1, Cas No. 468-44-0/510-75-8, KisanBio, Seoul, Korea), and 0 (distilled water), 10, 100, 200, and 500 µM of Flu (Cas No. 59756-60-4, Sigma-Aldrich, St. Louis, MO, USA) by soaking the seeds at 4°C for 48 h in the dark. In 25 seeds with 4 replicates, the treated seeds were sown under constant temperatures of 4, 15, 20, 25, and 30°C in light conditions, and alternating temperature conditions of 15/6, 20/10, and 25/15°C. Germination was observed weekly for 20 weeks and calculated as the final germination percentage.
Effect of stratification on dormancy breaking
Stratification at temperatures higher than 4°C was performed to assess its effectiveness in breaking dormancy considering the temperatures and durations C. platycarpa seeds experience in their natural environment after dispersal. The stratification temperatures and durations included non-treatment (control), seeds stratified at 15/6, 20/10, and 25/15°C for 4 weeks, and seeds stratified in sequences of 20/10 (4 weeks) → 15/6°C (4 weeks) and 25/15 (12 weeks) → 20/10 (4 weeks) → 15/6°C (4 weeks). These stratified seeds were moved to 4°C for observation of germination. Germination was monitored weekly until no further germination was observed, and the effects of stratification were assessed accordingly.
Statistical analysis
The weight of 1,000 seeds, E:S ratio, and germination experiments are expressed as the mean and standard error (SE). Significance was assessed using analysis of variance to test the null hypothesis, followed by Duncan’s Multiple Range Test to compare the means, with a significance level of p < 0.05. Analyses were performed using the Statistical Analysis Software (SAS, Version 9.4; SAS Institute Inc., Cary, NC, USA).
Results
Seed basic information
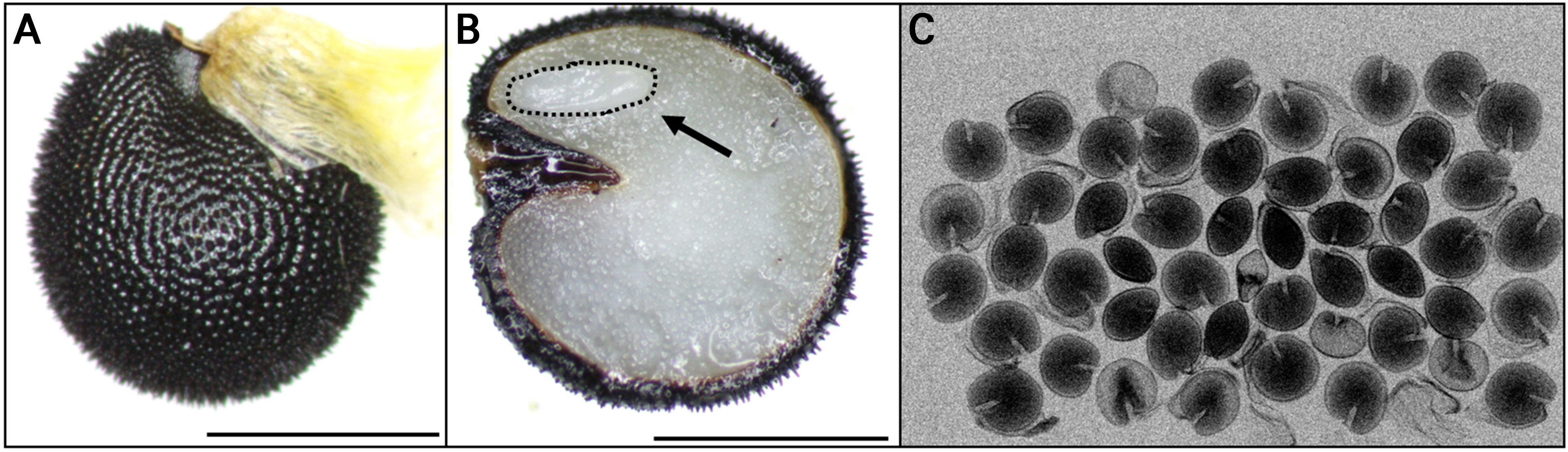
The weight of 1,000 C. platycarpa seeds was 1.31 ± 0.05 g, and the seed size was measured at 1.93 × 1.67 mm (Table 1). The E:S ratio of fresh seeds was 0.22 ± 0.01, and a seed viability test using 1.0% tetrazolium solution exhibited 96.0% viability (Fig. 1 and Table 1). X-ray analysis revealed a seed fidelity of 93.0% (Fig. 1C). The initial moisture content of C. platycarpa seeds was 8.91 ± 0.16%, reaching a maximum moisture content of 39.7% on day 2 of water absorption (Fig. 2).
Table 1.
Basic information of the collected Corydalis platycarpa seeds used in this study
| Weightz (g) | Sizey (mm) | E:S ratio | Viability (%) | Fidelity (%) |
| 1.31 ± 0.05 | 1.93 × 1.67 | 0.22 ± 0.01 | 96.0 | 93.0 |
Initial germination test
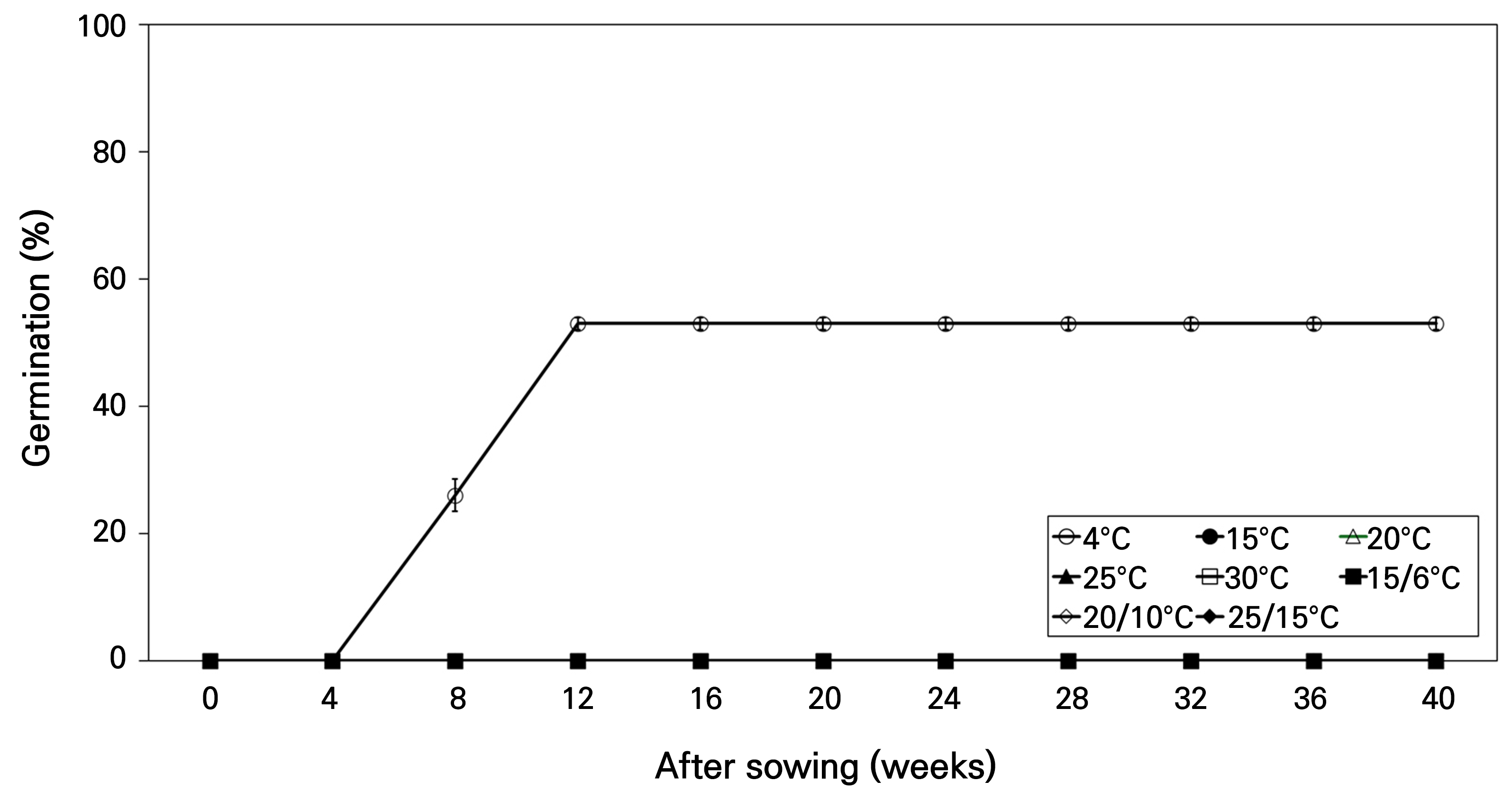
The initial germination test results exhibited no germination at any temperature except 4°C over 40 weeks (Fig. 3). Germination was observed at 4°C after 8 weeks of the test, with 53.0% germination by 12 weeks. However, no additional germination was observed after 12 weeks.
Embryo growth, germination, and shoot emergence under natural environmental conditions in the phenology experiment
At the start of the phenology experiment on June 30, 2023, the fresh seeds of C. platycarpa had an initial embryo length of 0.45 ± 0.01 mm and a critical embryo length of 3.22 ± 0.12 mm. The E:S ratio was 0.22 ± 0.01, indicating underdeveloped embryos, which were 13.5% of the critical embryo length (Figs. 4 and 5). The embryos of seeds buried in the natural environment did not grow during the summer and fall, and embryo growth was observed as winter began on November 17, 2024, with an average temperature of 2.9°C. Significant embryo growth was recorded as temperatures decreased with the onset of winter, and by February 9, 2024, most embryos were fully developed. The maximum embryo length of 3.22 ± 0.12 mm was observed on February 23, 2024. In the natural environment, germination began immediately after the embryos had fully grown by February 23, 2024, at an average temperature of 2.8°C. By March 8, 2024, more than 80.0% germination was observed, and shoot emergence was noted, with an average temperature of 3.6°C. As temperatures increased, shoot emergence was recorded, reaching 87.0% by April 19, 2024, with an average temperature of 18.3°C.
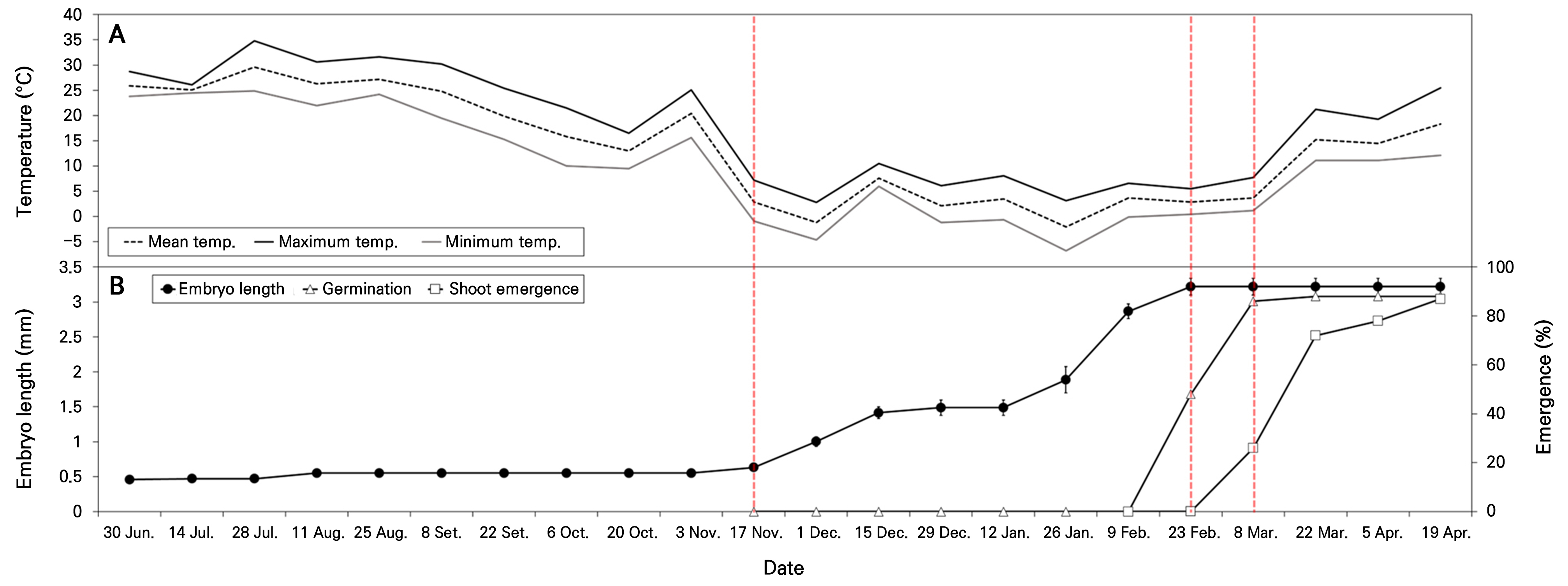
Fig. 4.
Phenology of embryo growth, germination, and shoot emergence of Corydalis platycarpa seeds buried at a soil depth of 1 cm on June 30, 2023. A, daily mean, maximum, and minimum temperatures; B, phenology of embryo growth, germination, and shoot emergence. The red dotted lines represent the timing of embryo growth, germination, and shoot emergence. Embryo length (●), germination (△), and shoot emergence (□). Bars represent standard errors (n = 10).
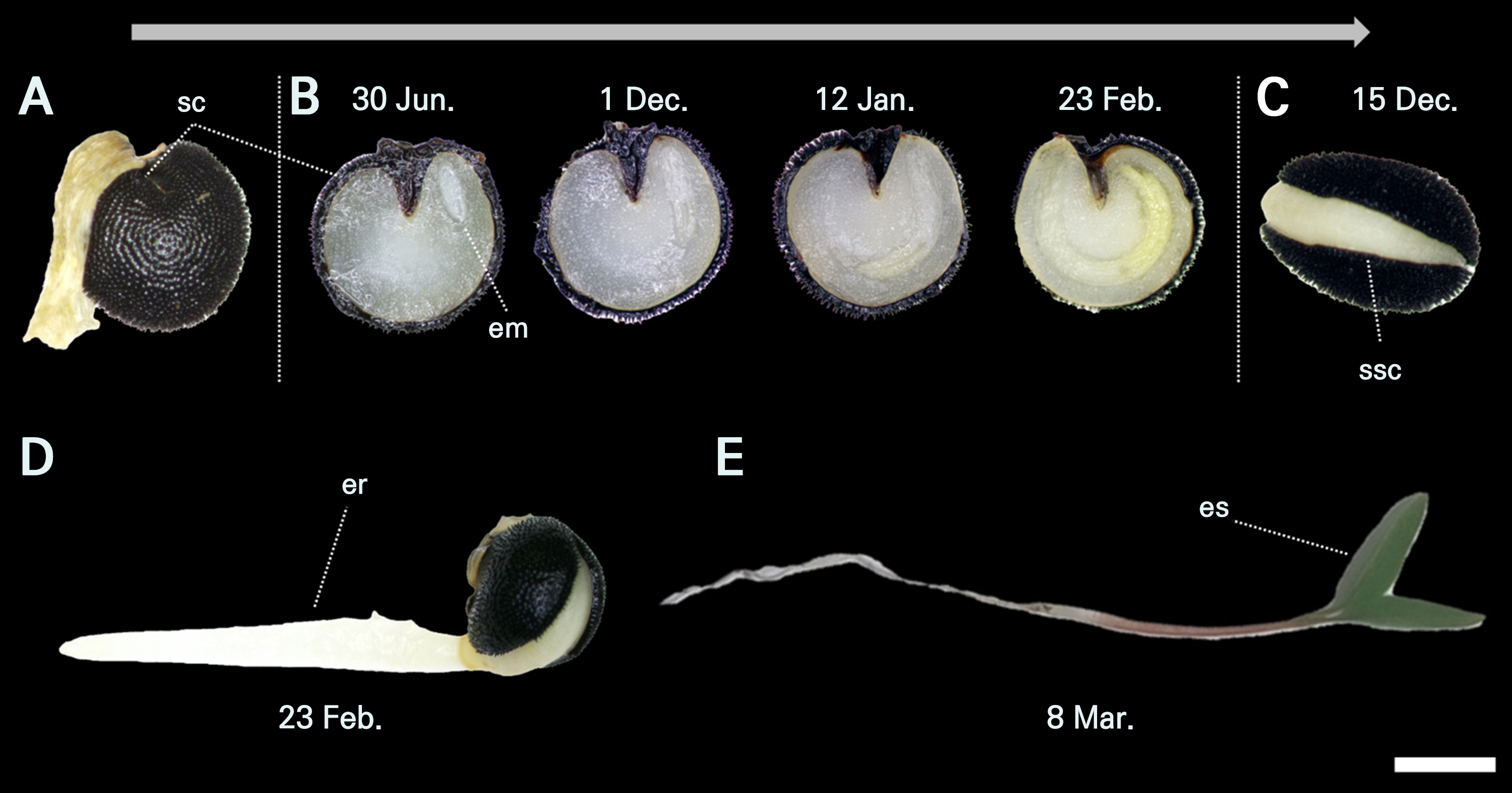
Fig. 5.
Changes in embryo morphology of Corydalis platycarpa seeds that were buried on June 30, 2023. A, Fresh seed; B, the location of the embryo in the cut seeds, the arrow refers to the growth of the undeveloped embryo until the fully developed embryo; C, split seed coat; D, seed showing germination; E, seed with emergent shoots. em, embryo; er, emerged radicle; es, emerged shoots; sc, seed coat; ssc, split seed coat. The scale bar (white horizontal line) indicates 1 mm.
Embryo growth and germination under simulated natural environmental conditions in the move-along experiment
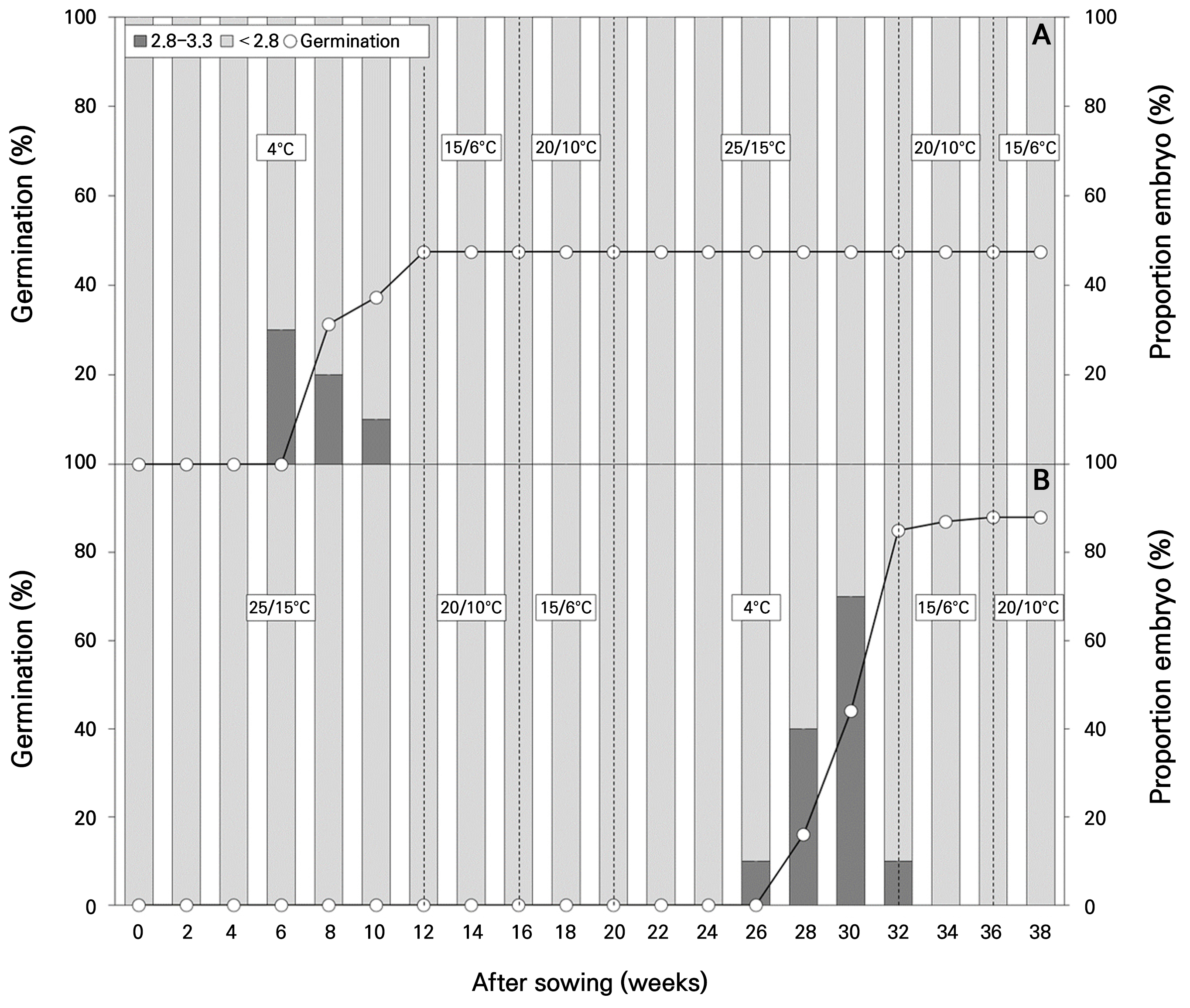
The move-along experiment, which simulated natural environmental cycles, exhibited different tendencies in the two sequences (Fig. 6). In Move A, beginning at the winter temperature of 4°C, embryo growth was observed from 6 weeks of incubation, and 47.5% germination was noted by 12 weeks, indicating germination that did not reach viability (Fig. 6A). No further embryo growth and germination were observed in the subsequent seasonal temperatures after winter temperature. In Move B, beginning at 25/15°C, no embryo growth or germination was observed during the summer and fall temperatures up to 24 weeks (Fig. 6B). However, embryo growth was noted from 26 weeks after the seeds were transferred to 4°C, and germination was observed from 28 weeks. In Move B, embryo growth continued until 32 weeks, with 85.0% germination observed, reaching germination of 88.0% by 38 weeks. The final germination at 38 weeks was 47.5% and 88.0% for Move A and Move B, respectively, with higher germination observed in Move B.
Effect of plant growth regulator treatment on dormancy breaking
Treatment with various concentrations of the plant growth regulators, GAs and Flu, the germination of C. platycarpa seeds during the 20-week germination experiment (Fig. 7). Germination was not observed at any temperature except 4°C, where germination percentages ranged from 47.0–55.0% by 20 weeks, with no significant differences between the concentrations of plant growth regulators.
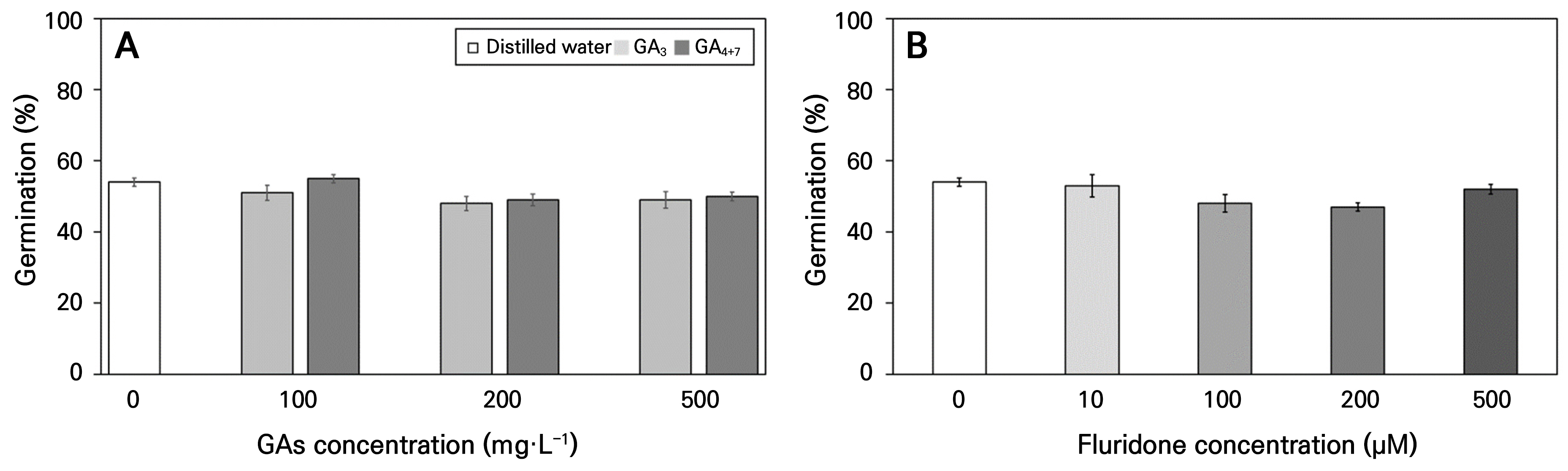
Fig. 7.
Final germination of Corydalis platycarpa seeds treated with plant growth regulators for 20 weeks at 4°C. Different concentrations of gibberellins (GAs) (A) and fluridone (Flu) (B) treatment. There were no significant differences in concentrations of GAs and Flu at each temperature, as determined by Duncan’s multiple range test (p < 0.05). Bars represent standard errors (n = 4).
Effect of stratification at various temperatures and periods on breaking dormancy
The stratification temperature and periods exhibited different patterns in germination at 4°C (Fig. 8). Control seeds showed 54.0% germination at 4°C after 20 weeks; in comparison, stratified seeds, when moved to 4°C, exhibited significantly higher germination of 68.0–88.0%. Seeds stratified at 20/10°C (4 weeks) and 25/15°C (4 weeks) showed final germination of 68.0 and 72.0% at 4°C, respectively, not reaching the viability of 96.0%. However, seeds stratified at 15/6°C (4 weeks) and 25/15 (12 weeks) → 20/10 (4 weeks) → 15/6°C (4 weeks) showed final germination of 88.0% and 87.0% at 4°C, respectively; the results were similar to the findings on viability.
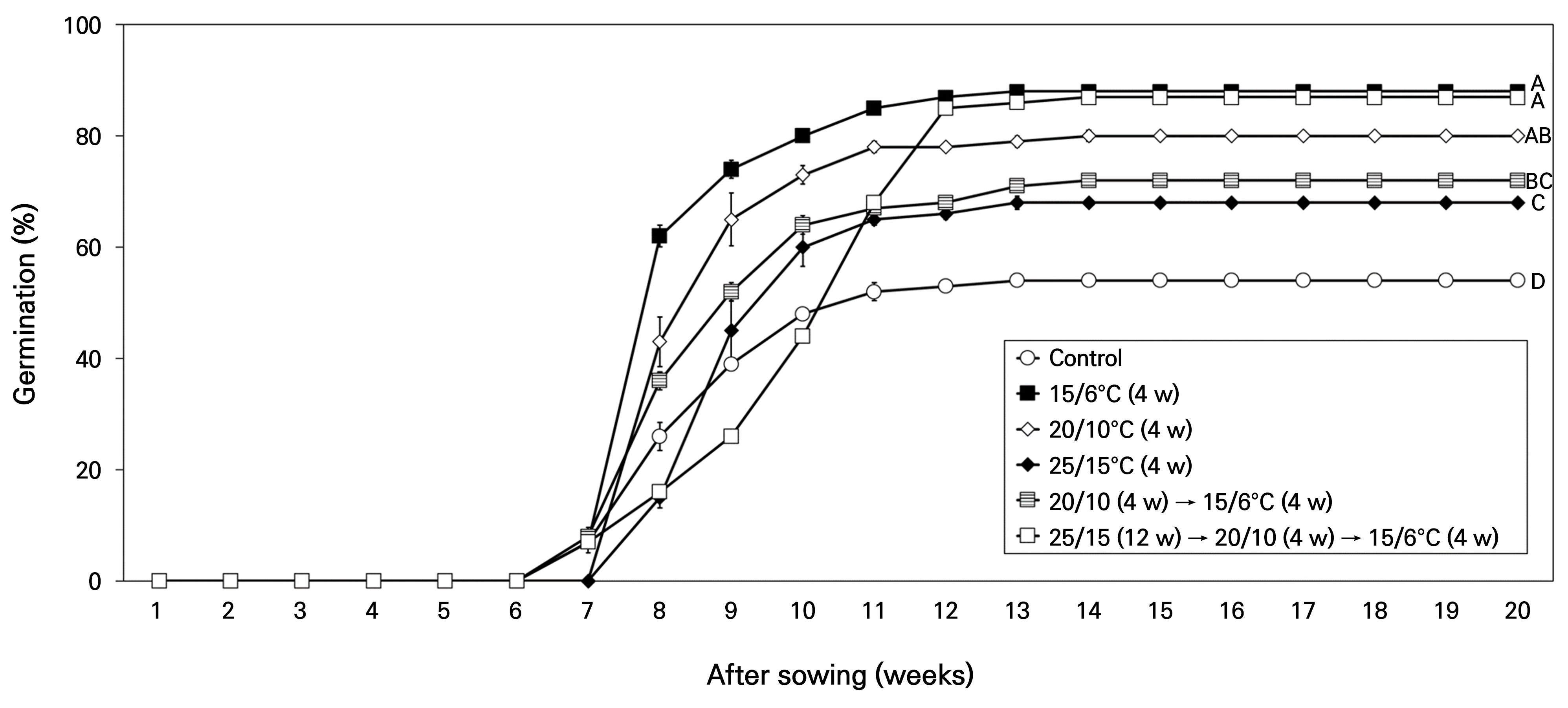
Fig. 8.
Percent germination of Corydalis platycarpa seeds for 20 weeks at 4°C after stratification at various temperatures and periods. Control, non-treatment. Different letters indicate significant differences in final germination percentage among temperatures as determined by Duncan’s multiple range test (p < 0.05). Bars represent standard errors (n = 4).
Discussion
Fresh seeds of C. platycarpa exhibit an embryo length of 0.46 mm and an E:S ratio of 0.22, indicating an underdeveloped embryo. An underdeveloped embryo must grow to a certain extent before seed germination (Zhang et al. 2023), and seeds with underdeveloped embryos that grow under appropriate conditions are categorized as exhibiting MD (Baskin and Baskin 2014). Phenology experiments in natural environments and move-along experiments simulating natural conditions showed that the embryo length of C. platycarpa seeds grew to over 2.8 mm before germination. Therefore, the fresh seeds of C. platycarpa were confirmed to show MD due to their underdeveloped embryos. PY is characterized by a seed coat that does not absorb water, comprising lignified cells and macrosclereids in the palisade layers (Turner et al. 2005). Seeds with PY are considered permeable if their weight increases by more than 20.0% after water absorption (Baskin and Baskin 2004). In the water absorption test, C. platycarpa seeds showed a maximum moisture content of 39.7% on day 2, indicating no characteristics of PY. In the study, C. platycarpa seeds were found to have a viability of 96.0% using a 1.0% tetrazolium solution test but no germination was observed at any temperature within 4 weeks. Seeds that do not germinate within 4 weeks under optimal conditions are classified as non-viable or PD (Lee et al. 2022). In MD, embryo growth and germination are completed within 4 weeks under optimal temperature, humidity, and light conditions after dispersal from the parent plant. However, seeds with an underdeveloped embryo with a specific physiological mechanism inhibiting germination are classified as showing MPD (Zarei-Gavkosh et al. 2022). Therefore, the fresh seeds of C. platycarpa are classified as exhibiting MPD due to their underdeveloped embryos and inability to germinate within 4 weeks under appropriate conditions.
In the initial germination test, C. platycarpa seeds exhibited germination only at 4°C, with germination of 53.0% over 40 weeks, when not attaining viability (96.0%). This suggests that while the optimal temperature for germination of C. platycarpa seeds is the low temperature of 4°C, the seeds that did not germinate may require additional temperature treatment, indicating the presence of two levels of MPD. C. platycarpa flowers from April to May in natural environments, and its seeds are dispersed from May to June (NIBR 2024). After dispersal, the seeds experience summer and autumn, with high proportions of embryo growth and germination observed in the winter when temperatures decrease. The move-along experiment simulating the natural environment showed that the Move A sequence did not achieve viability, exhibiting limited embryo growth and germination. However, in the Move B sequence, seeds experienced summer and autumn temperatures followed by winter, resulting in significant embryo growth and germination in most seeds. The key finding of this study is that the phenology and move-along experiments consistently showed high rates of embryo growth and germination following exposure to summer and autumn temperatures, followed by temperatures in winter. This indicates that C. platycarpa seeds require warm temperatures before encountering low temperatures for successful germination. Other Corydalis species required warm temperatures before low temperatures to induce germination (Vandelook and Van Assche 2009). For instance, C. pumil requires both summer and winter temperatures for germination (Stolle 2004), and C. ambigua needs summer, autumn, and winter temperatures (Kondo et al. 2005). This implies that many Corydalis species commonly need warm temperatures before low temperatures to break seed dormancy.
Seeds requiring warm temperatures of 15°C or higher for embryo growth are classified as exhibiting simple MPD, while those requiring low temperatures of 0–10°C are classified as having complex MPD (Nikolaeva 1977). Therefore, C. platycarpa seeds were classified as having complex MPD based on the phenology and move-along experiments, resulting in embryo growth at the low temperatures of winter. The MPD subclasses of simple and complex types are further divided into non-deep, intermediate, and deep, based on the degree of dormancy (Kim et al. 2021). Among them, deep MPD is characterized by the inability of GA3 to shorten dormancy and substitute for cold or warm stratification (Liu et al. 2023). In phenology and move-along experiments, germination was observed after experiencing summer and autumn temperatures followed by winter temperatures, whereas GA3 treatment did not significantly affect germination at any temperature, indicating that GA3 cannot replace cold or warm stratification, resulting in its classification under deep MPD. However, within the MPD subclasses, if autumn temperatures are required before winter temperatures for embryo growth, it is classified as non-deep complex MPD (Baskin and Baskin 2014). If winter temperatures are necessary for embryo growth and germination but GA cannot substitute warm or cold stratification, it is classified as deep complex MPD (Yaqoob and Nawchoo 2017). Thus, about 45.0% of germinable C. platycarpa seeds require warm temperatures before winter, classifying them as non-deep complex MPD. The remaining 55.0% required winter temperatures for embryo growth and germination, and as GA3 could not replace cold or warm stratification, they were classified as deep complex MPD. Therefore, C. platycarpa seeds show two levels of MPD, and such varying levels of dormancy within the same species have been observed in many plants, including Euphorbia characias and Eremopyrum distans, exhibiting racemose inflorescences (Espadaler and Gomez 2001; Wang et al. 2010).
Abscisic acid (ABA) and GAs are key plant hormones for seed germination and dormancy (Wareing et al. 1971). Flu used in the study inhibits ABA biosynthesis, alleviating seed dormancy and promoting germination (Yoshioka et al. 1998; Ali-Rachedi et al. 2004; Feurtado et al. 2007; Goggin and Powles 2014). Exogenous GA treatment is generally effective in breaking seed dormancy (Bewley and Black 1994). However, in this study, neither Flu nor GAs significantly affected the germination of C. platycarpa seeds. Seed dormancy is primarily regulated by the balance between ABA and GA signals but is also modulated through the interactions among various hormones (Shu et al. 2016). Various hormones, such as auxin and cytokinin, play roles in germination. Therefore, the dormancy may not have been broken due to high levels of ABA or other germination-inhibiting hormones (Finch-Savage and Leubner-Metzger 2006).
After dispersal, seeds naturally undergo stratification by being buried in the soil through multiple seasons, which breaks their dormancy (Chien et al. 2011b; Fernández-Pascual et al. 2015). Stratification is commonly used to break seed dormancy, and during this period, the regulation of GA and ABA in seeds leads to germination (Ma et al. 2023). In this study, C. platycarpa seeds showed germination during the winter temperatures following summer and autumn, as observed in the phenology and move-along experiments, indicating that the breaking of dormancy and germination take a long period. This poses a significant constraint on seed utilization, highlighting the need for solutions to shorten the germination period. C. platycarpa seeds required warm temperatures before winter temperatures for high germination. Thus, stratification at 25/15°C (12 weeks) → 20/10°C (4 weeks) → 15/6°C (4 weeks) showed a high germination of 87.0% but required a long stratification period of 20 weeks. Conversely, stratification at 15/6°C (4 weeks) resulted in 88.0% germination, effectively breaking dormancy within a short stratification period of 4 weeks. In this study, C. platycarpa seeds could efficiently improve dormancy breaking through 4 weeks of stratification at 15/6°C. However, a long period was still required to break dormancy in C. platycarpa seeds, indicating the need for further research to shorten the stratification period. The findings can provide solutions for ecological restoration programs and useful information for seed utilization (Hidayati et al. 2012).
In conclusion, the fresh seeds of C. platycarpa possess underdeveloped embryos and do not germinate within 4 weeks, resulting in MPD. Specifically, about 45.0% of C. platycarpa seeds require warm temperatures before exposure to low temperatures, showing non-deep complex MPD. However, the remaining about 55.0% of seeds do not respond to GA3 and only require low temperatures for germination, exhibiting deep complex MPD. Thus, C. platycarpa seeds exhibit two levels of MPD. Although dormancy breaking and germination in the natural environment take a long time, stratification at 15/6°C for 4 weeks effectively reduces the period required for dormancy breaking. Therefore, this study, which classifies dormancy types and improves dormancy-breaking methods, can serve as a foundational resource for ecological restoration and increasing the availability of resources.